Abstract
Rationale:
Protocols for regional citrate anticoagulation with the hypertonic 4% trisodium citrate solution have been recently described as an anticoagulation strategy during membrane therapeutic plasma exchange (mTPE). The effect of citrate in the patient’s systemic hemostasis is negligible, thus regional citrate anticoagulation application is advantageous in circumstances in which heparin-based protocols are deemed unsafe for patients with a high risk of bleeding. The downsides of using hypertonic citrate solutions are mainly hypocalcemia and hypernatremia that ultimately can cause adverse clinical events.
Presenting concerns of the patient:
(1) A 57-year-old Caucasian female with a history of active vaginal bleeding secondary to endometrial hyperplasia. She had a history of antiphospholipid syndrome, and systemic lupus erythematosus with marked refractory autoimmune thrombocytopenia. Her platelet count was persistently below 4,000/mm3 even after different immunosuppressive regimens and daily platelet transfusions. (1) A 70-year-old Caucasian female was hospitalized presenting acute kidney injury stage 3 due to rapidly progressive antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, however without the need for renal replacement therapy. At admission, serum creatinine (sCr) was 3.56 mg/dL (normal range: 0.53-1.00 mg/dL). Her baseline sCr was 0.8 mg/dL obtained 6 months earlier. Chest tomography revealed bilateral masses compatible with granulomatous lesions and no signs of alveolar bleeding. Since severe cases of ANCA vasculitis involving the lungs may evolve with alveolar hemorrhage, heparin was avoided.
Diagnoses:
(1) Systemic lupus erythematosus-associated autoimmune thrombocytopenia and (2) ANCA-associated vasculitis with kidney and lung involvement.
Interventions:
Herein, we describe a case series of 12 consecutive mTPE treatments in 2 different patients using regional 4% trisodium citrate anticoagulation.
Outcomes:
All the sessions were uneventful, presented only minor electrolyte imbalances, and were effectively completed without early interruptions due to clotting of the plasmafilter.
Teaching points:
In our 2 cases, extracorporeal regional citrate anticoagulation was successful in optimizing plasmafilter patency without bleeding events in 2 high-risk patients using established protocols for the citrate and calcium infusions.
Introduction
Membrane therapeutic plasma exchange (mTPE) is an extracorporeal blood purification technique that enables the unselective removal of blood solutes almost up to the size of cellular components.1,2 The physical mechanism that enables mTPE is convection, in which pressure is applied to a semipermeable membrane, this causes fluid movement across the membrane and drag forces removing large molecules. 3 The indications for mTPE are mainly related to antibody-mediated diseases, in which the mechanical removal of antibodies mitigates briefly the disease process. 4 Other conditions that benefit from plasmapheresis are familial lipid disorders5,6 and acute liver failure. 7
A frequent complication of virtually all extracorporeal blood purification techniques is clotting of the circuit, which leads to blood loss and increased cost due to loss of disposables. Aiming to reduce the risk of this negative event, anticoagulation strategies are warranted. The goal of anticoagulation is to promote optimal circuit patency with minimal systemic effects on the patient. Unfractionated heparin or low-molecular-weight heparin are widely used as anticoagulants in mTPE; however, both are associated with increased risk of bleeding. 8 Importantly, pro- and anticoagulant factors are also removed by plasmapheresis, exposing the patient to an additional risk of hemorrhagic events because clotting factors, such as fibrinogen, require up to 96 h to return to baseline concentration values, while most of other factors concentration return to baseline in 24 h. 9 Recently, protocols of regional citrate anticoagulation (RCA) in mTPE are emerging,10-13 These anticoagulation strategies derive from protocols of RCA applied in continuous renal replacement therapy (CRRT). Of note, in the setting of CRRT, RCA is considered as the first choice for circuit anticoagulation 14 with a higher safety profile when compared to heparin.15,16 Nonetheless RCA is rarely used in mTPE as an anticoagulation strategy in the United States and in countries from Latin America. 1 An alternative to mTPE is centrifugal therapeutic plasma exchange (cTPE), in which the blood is diverted to a rotating chamber, and the separation of blood elements depends on the differences in density of each component. 2 One advantage of cTPE is that it can be performed with lower blood flows when compared to mTPE, 50-70 mL/min versus 120-150 mL/min, respectively. This allows cTPE to be carried out using a peripheral venous access, whereas mTPE requires a hemodialysis vascular access. However, cTPE requires a dedicated machine while mTPE uses commonly available CRRT machines; 2 therefore, the use of mTPE is becoming more common. We present a series of 2 cases in which we performed a total of 12 sessions of mTPE using RCA.
Therapeutic Focus and Assessment
We carried out 12 TPE sessions using the PrismafleX® (Baxter Healthcare Corporation, Deerfield, IL, USA) system, software version 8.1. The plasmafilter was TPE 2000® - 0,35 m2- (Gambro, Lund, Sweden). A hypertonic citrate solution, 4% trisodium citrate (citrate 136 mmol/L, Na+ 408 mmol/L) was used for RCA. All treatments had a standard prescription. Blood flow was 9.0 L/h (150 mL/min) and the desired concentration of citrate in treated blood was 3.0 mmol/L. The citrate infusion rate was 200 mL/h and was calculated according to the formula in Figure 1. Replacement flow was 2.0 L/h (33 mL/min) and the desired concentration of calcium concerning effluent generated was 3.0 mmol/L. The calcium compensation infusion was done with an external intravenous pump, the solution used was 10% calcium gluconate (223 mmol/L). The calcium compensation infusion rate was 27 mL/h and was obtained from the formula in Figure 2. Fresh frozen plasma (FFP) or 4% albumin were used as replacement fluid. Magnesium was added to FFP or 4% albumin as a solution of 10% magnesium sulfonate (405 mmol/L). For each 1000 mL of FFP or 4% albumin, 3 mL of 10% magnesium sulfonate were added to the bag, attaining a final magnesium concentration of 1.2 mmol/L.
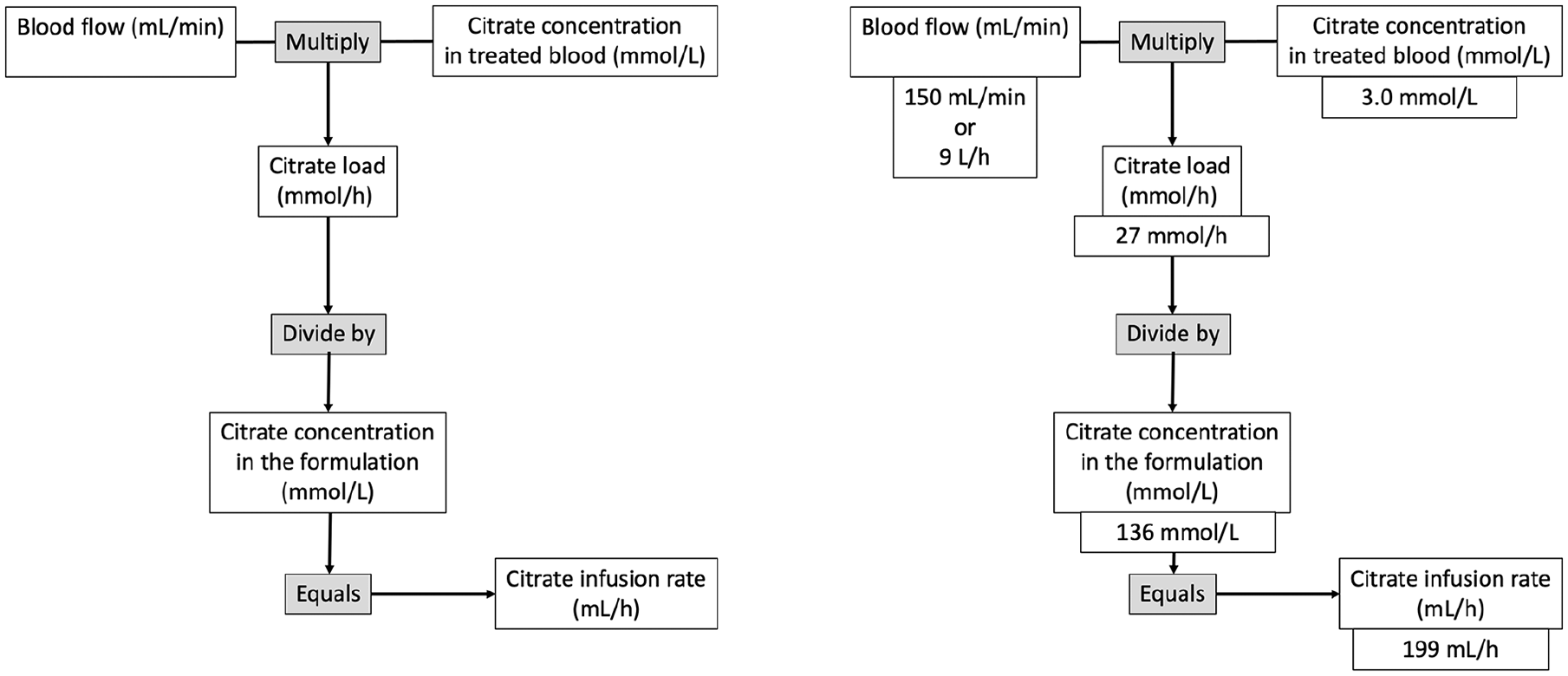
Step-by-step protocol to determine citrate infusion rate (using 4% trisodium citrate).

Step-by-step protocol to determine calcium compensation infusion rate (using 10% calcium gluconate).
Postfilter ionized calcium target was 0.20-0.45 mmol/L, and the systemic ionized calcium target was 1.00-1.35 mmol/L. The systemic magnesium target was 0.65-1.05 mmol/L. The total exchange volume per session was 1.5 times patient’s predicted plasma volume (70 mL/kg • bodyweight • [1 − hematocrit]). After 2 h of treatment initiation, systemic samples for ionized calcium and total magnesium were analyzed. In parallel, a postfilter sample for ionized calcium was obtained, and the therapy duration was around 3 h. The filtration fraction was close to 30% (filtration fraction is obtained by the sum of replacement flow plus citrate flow divided by plasma flow plus citrate flow). The sessions were carried out in alternate days. It is estimated that after each mTPE session exchanging 1.5 times patient’s predicted plasma volume, the plasma concentration of IgG is reduced by 80%. After each session, a redistribution of IgG occurs, named rebound phenomenon, that is, the shift from antibodies from the interstitial compartment toward the blood compartment. After 48 h from the session, the amount of serum IgG available for removal is almost 2-fold higher than after 24 h, justifying the sessions to be scheduled on alternate days. 1
This study was approved by the ethics committee of the Faculty of Medicine, University of Brasília (CAAE 44469620.3.0000.5558). Informed consent was waived because of the retrospective nature of the study. The treatments occurred in 2019.
Case Description
Case 1
A 57-year-old Caucasian female was hospitalized with a history of active vaginal bleeding secondary to endometrial hyperplasia. She had a history of obesity, hypertension, antiphospholipid syndrome, and systemic lupus erythematosus with marked refractory autoimmune thrombocytopenia. Her platelet count was persistently below 4,000/mm3 even after different immunosuppressive regimens and daily platelet transfusions. Plasmapheresis was proposed as a rescue treatment. Fresh frozen plasma was selected as the replacement solution because of the active vaginal bleeding. The vascular access was a nontunneled 11.5 Fr dual-lumen catheter, 20 cm, inserted in the left femoral vein. The catheter was placed in the femoral site, to the detriment of the jugular site, to enable effective manual compression in case of active bleeding to achieve hemostasis. The insertion was ultrasound-guided with simultaneous platelet transfusion and was uneventful. She carried out 5 sessions of TPE without episodes of filter clotting, tetany, arrhythmias, nor worsening in vaginal bleeding. In the last TPE session, the platelet count escalated to 73,000/mm3 and 3 days later attained a normal value (164,000/mm3, reference range: 150,000-400,000/mm3). She further developed nosocomial pyelonephritis, septic shock, and died after 7 days.
Case 2
A 70-year-old Caucasian female was hospitalized presenting acute kidney injury stage 3 due to rapidly progressive antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, however without the need for renal replacement therapy. At admission, serum creatinine (sCr) was 315 µmol/L (3.56 mg/dL), (normal range: 47-88 µmol/L [0.53-1.00 mg/dL]). Her baseline sCr was 71 µmol/L (0.8 mg/dL) obtained 6 months earlier. Chest tomography revealed a mass of 5.2 × 4.9 × 2.8 cm in the right inferior pulmonary lobe, a nodule of 0.8 cm in the right inferior pulmonary lobe and a nodule of 1.1 cm in the left inferior pulmonary lobe. The mass was biopsied showing a nonspecific inflammatory infiltrate. No signs of alveolar bleeding were reported. The patient was stable and carried out 7 sessions of TPE in a noncritical care setting. It was opted to use 4% albumin solution as replacement fluid because the patient did not have active bleeding or would not undergo any invasive procedure (the usual indications for FFP). No episodes of bleeding, tetany, arrhythmias, or filter clotting were reported. The vascular access was a nontunneled 11.5 Fr dual-lumen catheter, 20 cm, and the insertion site was the right internal jugular vein. Before the initiation of TPE, fibrinogen concentration was 255 mg/dL (normal range: 180-393 mg/dL). In between the sessions, the lowest concentration of fibrinogen was 84 mg/dL. The therapy was postponed if fibrinogen was below 150 mg/dL and reassessed daily. After each session, she received 6 units of cryoprecipitate. In addition, on the day of admission, she received intravenous methylprednisolone 250 mg. The same dose was repeated in the 2 consecutive days, followed by daily oral prednisone 1 mg/kg. Oral cyclophosphamide adjusted for the patient’s age and renal function was also initiated. After 23 days since admission, the patient was discharged with an sCr of 156 µmol/L (1.77 mg/dL).
Follow-Up and Outcomes
Including laboratory results of both patients during 12 mTPE sessions, in only one occasion, systemic ionized calcium was below the lower limit, and in 2 occasions postfilter ionized calcium was above the upper limit. Hypernatremia did not occur during the period of treatment, and on 2 out of 12 measurements, total magnesium concentration was outside the range of normality (Table 1). No episodes of bleeding or filter clotting were reported.
Laboratory Parameters From All Treatments.

Note. FFP = fresh frozen plasma. Bold digits correspond to values outside the reference range.
Discussion
We describe 2 cases of mTPE in which an anticoagulation regimen with heparin was deemed unsafe and RCA was used. Clinical expected adverse effects of RCA are mainly manifestations of hypocalcemia. The absence of episodes of bleeding, tetany, or arrhythmias indicate the clinical safety of this strategy. Citrate acts as an indirect anticoagulant as it binds to ionized calcium and reduces its plasma concentration. 17 Ionized calcium is essential in many steps of the anticoagulation cascade, and when its concentration falls below 0.3 mmol/L, clotting time tends to infinite. 18 For this reason, we targeted a postfilter ionized calcium level of 0.2 to 0.45 mmol/L. Usually to achieve this adequate range of ionized calcium, the citrate concentration in treated blood is between 3 and 4 mmol/L. 19 As such, active bleeding is not a contraindication for RCA since citrate’s impact on systemic hemostasis would only occur with systemic ionized calcium concentrations not compatible with life. During our treatments, the lowest systemic ionized calcium observed was 0.99 mmol/L (normal range: 1.00-1.35 mmol/L). Importantly, the efficacy of the RCA strategy was also demonstrated because no episodes of clotting in the extracorporeal circuit occurred. Betz et al demonstrated the feasibility of this protocol in their prior study. 11 Importantly, this protocol needs to be compared to established protocols that are used in CRRT. Here, we applied the protocol to 2 patients to further demonstrate efficacy and safety. The rationale for analyzing systemic ionized calcium and postfilter ionized calcium is to adjust pretreatment settings for the next session. Membrane therapeutic plasma exchange sessions takes around 2 h to be completed, usually when calcium results are available, the session is already completed. This differs from the practice in CRRT, where citrate and calcium infusions are adjusted according to the calcium results every 6 h. Another difference between RCA in CRRT and in mTPE refers to the calcium compensation. In CRRT, usually the target of systemic ionized calcium is achieved with a calcium concentration of 2.0 mmol/L of effluent generated. In mTPE, when blood components are used as replacement solution, they represent an additional source of citrate, present in the bags of blood derivates. When albumin is used as replacement solution, free bindings sites for calcium are present in the infused albumin molecules, reducing the amount available of ionized calcium. For these 2 reasons, in mTPE, usually the target systemic ionized is achieved only with a higher proposed calcium concentration in respect to the effluent flow, that is, 3.0 mmol/L of effluent generated, 50% higher than what is required in CRRT.
Citrate also chelates with magnesium and one pitfall in clinical practice is that only total magnesium is measured; however, exclusively ionized magnesium has significant biological activity. 20 Therefore, even when total magnesium concentration is in the normal range, the ionized fraction is likely to be reduced during RCA. This highlights the importance of magnesium compensation to avoid adverse events related to hypomagnesemia such as arrhythmias. In our series, 2 episodes of hypomagnesemia occurred without apparent clinical manifestations.
Finally, being a hypertonic solution (Na+ 408 mmol/L; 554 mOsmol/kg H2O), 4% trisodium citrate can cause hypernatremia. 21 We did not observe hypernatremia in our patients. One possible reason is the short duration of the treatments and consequently, the small amount of the solution delivered to the patient, as the infusion rate of citrate was 200 mL/h, so that after 3 h, 600 mL was infused into the circuit. Moreover, the filtration fraction was around 30%, which implies that a third of the solutes infused is removed in the plasmafilter. Therefore, roughly only 400 mL of the hypertonic solution gained access to the patient’s intravascular compartment in each session.
Our case series has several limitations: (1) in Case 1, fibrinogen concentration after mTPE sessions was not measured, and it was not possible to assess the influence of mTPE on its plasma concentration; (2) for both cases, blood gas analysis was not performed after the treatments, impeding the detection of metabolic alkalosis, which is an expected complication when RCA is used.
Teaching Points
Our case series with 12 consecutive mTPE sessions pointed out that RCA is a feasible strategy to avoid premature extracorporeal circuit clotting without adverse clinical manifestations or significant electrolyte imbalances. We believe this strategy might be particularly suitable for patients with thrombocytopenia, hypofibrinogenemia, active bleeding, in the postoperative course, or immediately after central intravenous access placement where the use of heparin is prohibitive.
Footnotes
List of Abbreviations
ANCA, antineutrophil cytoplasmic antibody; CRRT, continuous renal replacement therapy; cTPE, centrifugal therapeutic plasma exchange; FFP, fresh frozen plasma; mTPE, membrane therapeutic plasma exchange; RCA, regional citrate anticoagulation; sCr, serum creatinine.
Ethics Approval and Consent to Participate
This study was approved by the ethics committee of the Faculty of Medicine—University of Brasília (CAAE 44469620.3.0000.5558).
Consent for Publication
Informed consent was waived because of the retrospective nature of the study.
Availability of Data and Materials
Not applicable.
Author Contributions
T.R. drafted the report. All authors contributed equally in writing and revising the manuscript. All authors were responsible for critical revision of the manuscript and approved the final version before submission.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.R. has received funding for lectures, been consultant of advisory board member for ASAHI, Baxter, bioMérieux, B. Braun, CytoSorbents, ESTOR, Fresenius Medical Care, General Electric, Medtronic, and Toray in the past 3 years. T.R. has received funding for lectures, been consultant or advisory board member for AstraZeneca, Baxter, B. Braun, Contatti Medical (CytoSorbents), Eurofarma, and Jafron in the past 3 years.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Article processing charge was covered by IRRIV Foundation.
