Abstract
Background:
Transcatheter aortic valve replacement (TAVR), although associated with an increased risk for acute kidney injury (AKI), may also result in improvement in renal function.
Objective:
The aim of this study is to evaluate the magnitude of kidney function improvement (KFI) after TAVR and to assess its significance on long-term mortality.
Design:
This is a prospective single center study.
Setting:
The study was conducted in cardiology department, interventional unit, in a tertiary hospital.
Patients:
The cohort included 1321 patients who underwent TAVR.
Measurements:
Serum creatinine level was measured at baseline, before the procedure, and over the next 7 days or until discharge.
Methods:
Kidney function improvement was defined as the mirror image of AKI, a reduction in pre-procedural to post-procedural minimal creatinine of more than 0.3 mg/dL, or a ratio of post-procedural minimal creatinine to pre-procedural creatinine of less than 0.66, up to 7 days after the procedure. Patients were categorized and compared for clinical endpoints according to post-procedural renal function change into 3 groups: KFI, AKI, or preserved kidney function (PKF). The primary endpoint was long-term all-cause mortality.
Results:
The incidence of KFI was 5%. In 55 out of 66 patients patients, the improvement in kidney function was minor and of unclear clinical significance. Acute kidney injury occurred in 19.1%. Estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2 was a predictor of KFI after multivariable analysis (odds ratio = 0.93 to develop KFI; confidence interval [95% CI]: 0.91-0.95, P < .001). Patients in the KFI group had a higher Society of Thoracic Surgery (STS) score than other groups. Mortality rate did not differ between KFI group and PKF group (43.9% in KFI group and 33.8% in PKF group) but was significantly higher in the AKI group (60.7%, P < .001).
Limitations:
The following are the limitations: heterozygous definitions of KFI within different studies and a single center study. Although data were collected prospectively, analysis plan was defined after data collection.
Conclusions:
Improvement in kidney function following TAVR was not a common phenomenon in our cohort and did not reduce overall mortality rate.
Introduction
Transcatheter aortic valve replacement (TAVR), first limited to high-risk patients with severe symptomatic aortic stenosis who were poor surgical candidates, is now more widely used even in lower risk patients.1-4
Acute kidney injury (AKI) is a frequent post-procedural complication seen in 8.3% to 58% of procedures, depending on the diagnostic criteria used to define AKI and various factors, including patient selection and characteristics, operator’s experience, and peri-procedural complications.5-8 Acute kidney injury complicating TAVR is associated with a higher mortality risk during short- and long-term follow-up.9-11 In most patients who develop AKI, kidney function recovers partially or completely. Nonetheless, mortality risk remains high. Two-year follow-up in patients after TAVR demonstrated 80% survival in patients who did not develop AKI, compared with 66% in patients with AKI who had complete recovery and 25% when there was no recovery in kidney function. 12
In contrast to the harmful consequences of AKI, some studies have noted a partial improvement in kidney functions after TAVR and suggested there was a positive effect on survival in these patients. They referred to this as acute kidney recovery (AKR).13-15 Several studies assessed the incidence and predictors of AKR using diverse definition criteria. As AKR has only recently been described, there is no standardized definition for the phenomenon. Some defined AKR as kidney function improvement (KFI) 48 hours after the procedure, while others extended the definition to hospital discharge or 30 days post-procedure. The criteria for estimated glomerular filtration rate (eGFR) improvement varied widely among researchers, ranging from 10% to 25% increase.13-15 As the definitions were different, the incidence of AKR ranged from 17.8% to 52%, and the impact of AKR on clinical outcomes differed broadly.15-18
The aim of the current study was to evaluate the incidence of KFI in a large cohort of patients undergoing TAVR according to different definitions, and to evaluate long-term outcomes among patients according to post-procedural kidney function.
Methods
Study Population and Data Collection
The present cohort included patients with severe symptomatic aortic stenosis that underwent TAVR at the Tel Aviv Sourasky Medical Center, Israel. Participants were enrolled to our registry between July 2009 and November 2018.19,20
Among 1530 patients enrolled, we excluded 31 patients treated with maintenance dialysis. Another 178 patients were excluded due to incomplete data. The final cohort included 1321 patients. The study was approved by the institutional ethical committee, and all patients included gave informed consent.
Severe aortic stenosis was diagnosed based on clinical, echocardiographic, and hemodynamic criteria, and suitability for TAVR was determined by a dedicated heart team. 2 Data collected included demographic and clinical characteristics, laboratory and echocardiographic data, as well as peri-procedural complications.
TAVR Procedure
All patients underwent elective pre-procedural transthoracic echocardiography, coronary and peripheral angiography, and cardiac computed tomography angiography. All the procedures were carried out by a dedicated team. The prosthetic valves used for the procedure were either Edwards SAPIEN XT, Edwards S3 prostheses (Edwards Lifesciences, Irvine, CA, USA), CoreValve, or Evolute R aortic valve prostheses (Medtronic, Minneapolis, MN, USA). The prosthetic valve was delivered through the femoral approach. The contrast media used was iodixanol (Visipaque, GE Healthcare), an iso-osmolar contrast medium.
Unless contraindicated, patients received intravenous normal saline at a rate of 100 mL/h 12 hours before procedure, that was continued 12 hours afterward, to reduce the risk of contrast nephropathy.
Laboratory Analysis and Definitions
Serum creatinine level was measured at baseline, before the procedure, and over the next 7 days or until discharge. The highest post-procedural serum creatinine value was included in the analysis for AKI, and the lowest creatinine value was included in the analysis of KFI.
Anemia was defined as hemoglobin level below 13 g/dL in men and below 12 g/dL in women.
Society of Thoracic Surgery (STS) score, a risk-prediction model validated in TAVR patients, was calculated for all patients. 21
Frailty was assessed based on Katz index. 22
Kidney Injury/Recovery Definitions
Chronic kidney disease (CKD) was defined as baseline estimated GFR (eGFR) <60 mL/min/1.73 m2, estimated from the CKD Epidemiology Collaboration (CKD-EPI) equation. 23
We defined AKI according to Valve Academic Research Consortium-2 (VARC-2) AKI classification based solely on creatinine measurements. The VARC-2 defines AKI based on the acute kidney injury network (AKIN) system but extends the timing for diagnosis of AKI to 7 days post-procedure. 24
Kidney function improvement was diagnosed only in patients who did not develop AKI, and was defined as the mirror image of AKI, a reduction in pre-procedural creatinine to post-procedural minimal creatinine of more than 0.3 mg/dL, or a ratio of post-procedural minimal creatinine to pre-procedural creatinine of less than 0.66. The last creatinine measurement was taken on the day of discharge or 7 days after the procedure, whatever came first.
The patients were grouped according to the change in renal function post-procedure into 3 groups: KFI, AKI, or preserved kidney function (PKF).
Follow-Up
Clinical and laboratory data were collected until hospital discharge. The primary endpoint was long-term all-cause mortality. Data on mortality post-discharge were retrieved from the computerized hospital record system, linked to the National Social Security database.
Statistical Analysis
All data for parametric continuous variables are presented as mean (±SD); for nonparametric continuous variables, median (Q1, Q3) is presented; and for categorical variables, a number (percentage) is presented. Continuous parametric variables (eg, age) were compared using an independent-samples one-way analysis of variance (ANOVA) test. Continuous nonparametric variables (eg, STS score) were compared using independent-samples Kruskal-Wallis test. Categorical variables were compared using a chi-square test. Multivariable Cox regressions were used to evaluate associations with mortality; parameters found to be significant with mortality in the univariable analysis were included in the Cox regression. Multivariable logistic regression was used to assess independent predictors of AKI versus PKF combined and KFI versus PKF combined. Parameters found to be significant in the univariable analysis were included in the multivariable analysis. Therefore, KFI multivariable analysis included eGFR, dyslipidemia, left ventricular ejection fraction (LVEF) at hospitalization, the change in LVEF before and after procedure (delta LVEF), aortic valve area before procedure, and STS score. Parameters included in AKI multivariable analysis were age, eGFR, diabetes mellitus, hypertension, atrial fibrillation, hemoglobin level before procedure, and STS score. To create all multivariable models, a stepwise forward regression was used to select the significant covariates for the final models. To minimize the effects of potential confounding factors, one-to-one propensity score matching was performed using the “Matchit” R package (Version 1.2.5033) with a caliper width of 0.1. We included STS score in the propensity score model in an effort to inform the propensity of the dependent variable.
A 2-tailed P value less than .05 was considered statistically significant. The statistical analyses were performed using IBM SPSS Statistics 25.0 software.
Results
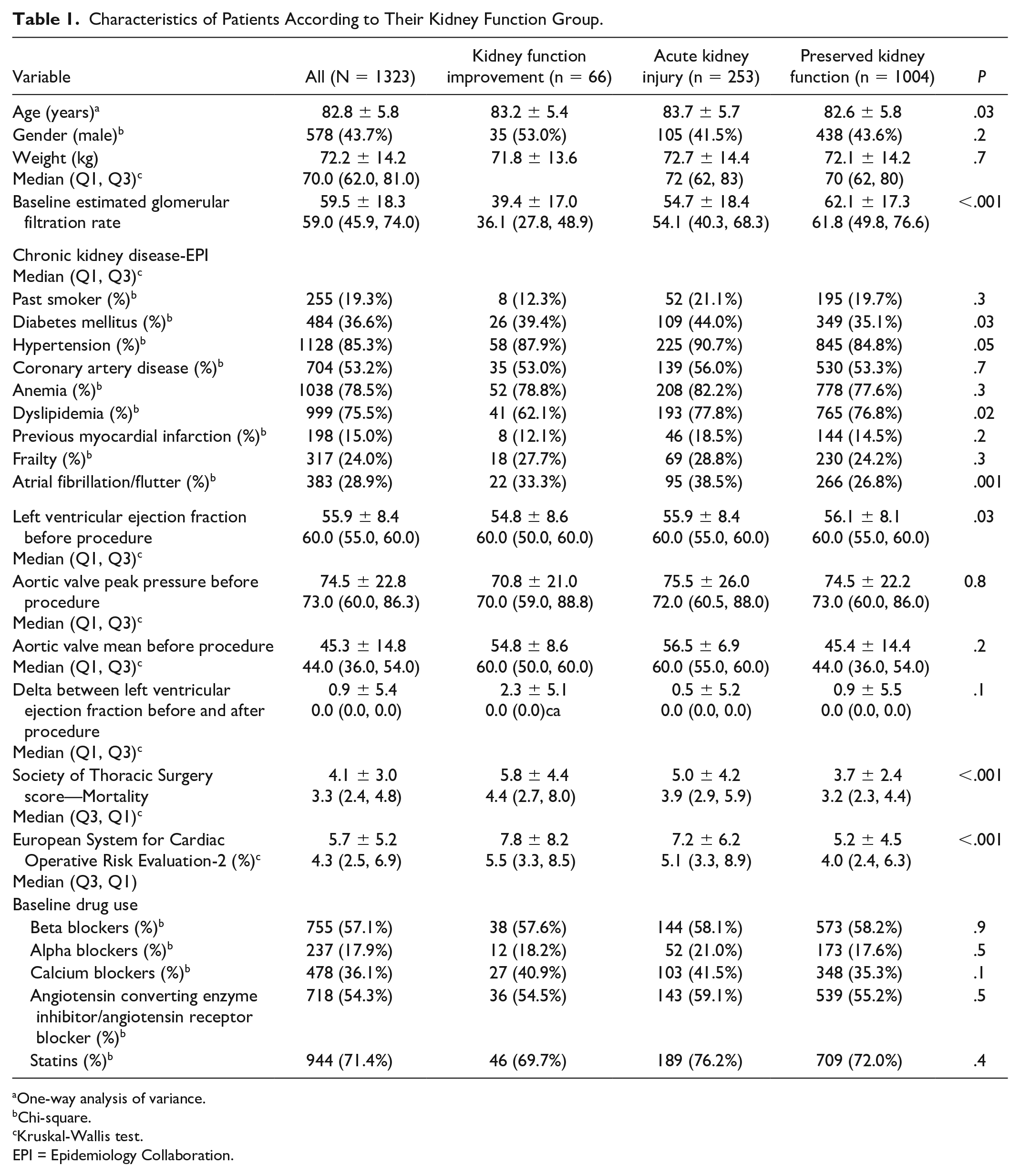
The present study included 1321 patients who underwent transfemoral TAVR. Their mean age was 82.8 ± 5.8, and 43.7% were males. The mean pre-procedural eGFR was 59.5 ± 18.3 ml/min/1.73 m2. Median follow-up period was 1099 days (ranging from 1 to 3648 days). The baseline characteristics of the cohort, divided into kidney function groups, are presented in Table 1.
Characteristics of Patients According to Their Kidney Function Group.
One-way analysis of variance.
Chi-square.
Kruskal-Wallis test.
EPI = Epidemiology Collaboration.
The incidence of KFI was 5% (66 patients), compared with 253 patients who developed AKI (19.1%) and 1003 patients with PKF (75.9%). The improvement in kidney function in 55 out of 66 patients was minor and of unclear clinical significance. Patients in the KFI group had a higher STS score than other groups (mean STS score 5.8 ± 4.4 for KFI group, 5.0 ± 4.2 for AKI group, and 3.7 ± 2.4 for PKF group; P < .001) and lower ejection fraction. Patients in the AKI group were older and had a higher prevalence of diabetes mellitus, dyslipidemia, and atrial fibrillation. In addition, baseline eGFR in the KFI group was lower than other groups (39.4 ± 17.0 in KFI group, 54.7 ± 18.4 in AKI group, and 62.1 ± 17.3 mL/min/1.73 m2 in PKF group; P < .001).
Baseline CKD was present in 647 patients: 593 with CKD 3 (eGFR 30-59 mL/min/1.73 m2), 51 patients with CKD 4 (eGFR 15-29 mL/min/1.73 m2), and 3 patients with CKD 5 nondialysis (eGFR <15 mL/min/1.73 m2). Among patients with baseline CKD, a change in kidney status was more probable: 23.3% developed AKI and 8.7% had KFI.
After a multivariable analysis, factors associated with KFI in the whole cohort were low baseline eGFR (odds ratio [OR] = 0.93, confidence interval [CI] = 0.91-0.95, P < .001) and presence of dyslipidemia (OR = 0.28, CI = 0.15-0.53, P < .001).
Predictors of AKI were low baseline eGFR, atrial fibrillation, and a higher STS score (Table 2).
Factors Associated With Kidney Function Improvement and AKI—Multivariate Analysis.

Note. AKI = acute kidney injury; eGFR = estimated glomerular filtration rate; CKD = chronic kidney disease; STS = Society of Thoracic Surgery; LVEF = left ventricular ejection fraction; CKD-EPI = CKD Epidemiology Collaboration.
Variables included in kidney function improvement prediction were eGFR, dyslipidemia, LVEF at hospitalization, delta LVEF, mean aortic valve area before procedure, and STS score.
Variables included in AKI prediction were age, eGFR, diabetes mellitus, hypertension, hemoglobin, atrial fibrillation, and STS score.
Pre-procedural LVEF was lower in the KFI group (54.8 ± 8.6, 55.9 ± 8.4, and 56.1 ± 8.1 for KFI compared with AKI and PKF, respectively, P = .03). LVEF improved significantly after procedure compared with pre-procedural values in the KFI group (mean difference = 2.3 ± 5.1, P = .003) and PKF group (mean difference = 0.9 ± 5.5, P < .001), but did not change in the AKI group (mean difference = 0.5 ± 5.2, P = .2).
During follow-up, 521 patients died: 29/66 (43.9%) in the KFI group, 153/252 (60.7%) in the AKI group, and 339/1003 (33.8%) in the PKF group (P < .001). Mortality rate was significantly higher in the AKI group, but did not differ between KFI group and PKF group after 1, 5, and 10 years of follow-up (Table 3). Patients who developed AKI had a hazard ratio for mortality of 2.89 (95% CI = 1.81-4.61), 1.87 (95% CI = 1.41-2.47), and 1.94 (95% CI = 1.49-2.52), at 1, 5, and 10 years, respectively (Figure 1). Analysis of the subgroup of patients with eGFR <60 mL/min/1.73 m2 (CKD group) compared with patients with eGFR >60 mL/min/1.73 m2 demonstrated no change in 1-year mortality even after stratification to AKI/KFI/PKF.
Association Between Kidney Function Groups and Death in 1, 5, and 10 Years.
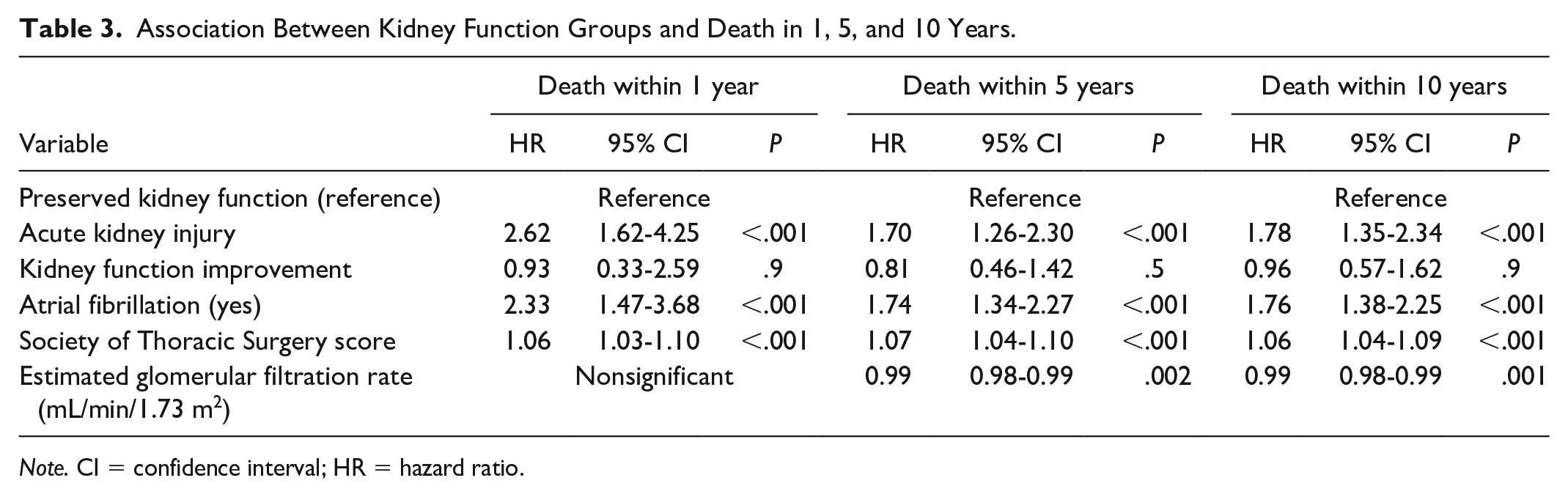
Note. CI = confidence interval; HR = hazard ratio.
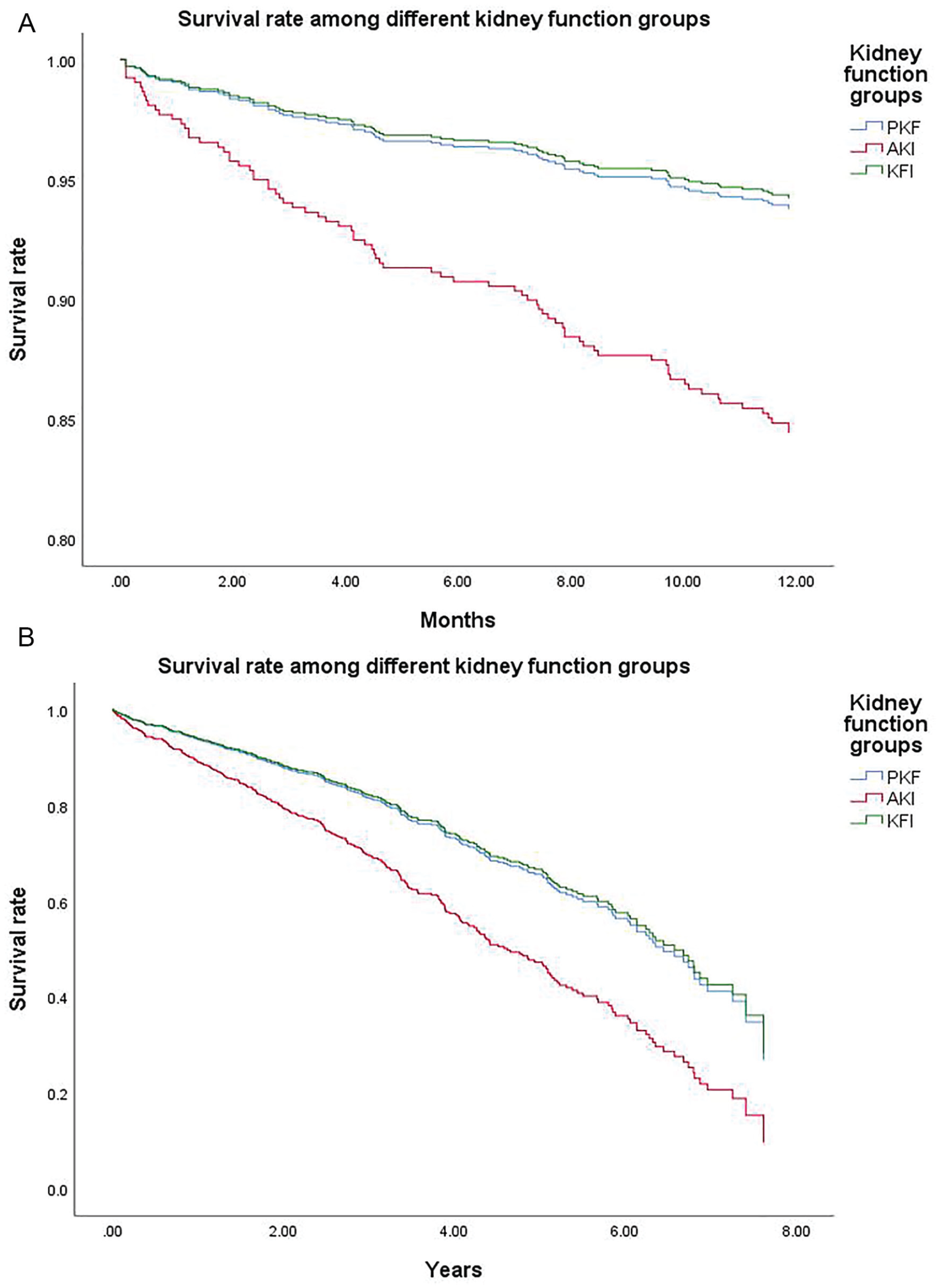
Survival rates among kidney groups—(A) 1-year survival among different kidney groups: between AKI and PKF groups, P< .001, and for KFI and PKF groups, P = .885, and (B) long-term survival among different kidney groups: between AKI and PKF groups, P < .001, and for KFI and PKF groups, P = .886.
Sensitivity Analysis
As previous studies used different definitions for AKR, which we referred to as KFI,14,16 we have run our analysis again using eGFR instead of creatinine and examined various cutoff values to define KFI. Compared with 5% KFI incidence in our original analysis, a definition of 25% improvement in eGFR resulted in KFI incidence of 10.4% (137 patients). Nevertheless, the different definition did not influence patient outcomes. Hazard ratio (HR) for 1-year mortality was 0.927 in creatinine-based KFI (95% CI = 0.33-2.59) and 0.648 in 25% eGFR improvement (95% CI = 0.26-1.64). The differences in HR were not statistically significant.
Discussion
In the current study, we found that 5% of patients demonstrated improvement in kidney function based on creatinine measurement following TAVR. Although patients in the KFI group were predisposed to develop peri-procedural complications and a higher mortality rate as reflected by a higher STS score and a lower baseline eGFR, they exhibited outcomes similar to lower risk patients.
While improvement in renal function after TAVR was reported earlier, 13 Azarbal et al were first to define this phenomenon as AKR in 2018. 15 Initially defined as an increase by more than 25% in eGFR, 48 hours after the procedure, AKR incidence was 32.5%. Thereafter, analyzing multicenter data, the AKR definition was modified and was characterized as an increase in eGFR above 25% upon hospital discharge. Using the new definition, the AKR incidence was reduced to 17.8%. 14 In a substudy of the Placement of Aortic Transcatheter Valves trial (PARTNER)-1 trial, assessing TAVR outcomes specifically in CKD patients, KFI, characterized as improvement in eGFR above 10% thirty days post-procedure was observed in 42%. 16 As there is no agreement on the definition of improvement in kidney function, we defined KFI based on the exact opposite of the VARC-2 definition of AKI. According to the VARC-2 definition, 48 hours are not enough to assess post-procedural improvement in kidney function, because peri-procedural hydration may result in false-positive cases. Creatinine measurement is preferred over eGFR as patients’ kidney function was not in steady state, making the eGFR equations less reliable. As the distinction between KFI incidences among the trials partially results from the differences in AKR or KFI characterization, it may explain the lower KFI incidence in our cohort. Indeed, we demonstrated different KFI rates when using diverse criteria, which highlight the need for a uniform classification, similar to AKI, to correctly assess the magnitude of KFI phenomenon.
Predictors of improvement in kidney function in previous studies were the presence of CKD, a lower cardiac index, previous aortic valve surgery, and avoidance of chronic beta-blocker use. A higher preoperative trans-aortic pressure gradient was also suggested as a predictor. While one study found male gender as a predictor for KFI, another study found more KFI in women. Factors associated with a reduced likelihood to develop AKR were diabetes mellitus, anemia, and a high STS score.14-16,25 In the current study, we demonstrated that KFI predictors were baseline reduced eGFR and the presence of dyslipidemia.
In a previous report, KFI did not result in improved outcome compared with no change in kidney function. Thirty days and 1-year survival, as well as repeat hospitalizations, did not differ between groups. However, KFI resulted in a significant survival advantage compared with patients with AKI. 16 Patients who developed KFI in our cohort had a higher STS score than other groups, although it was not found to be a strong predictor of KFI in a multivariable analysis. However, the tendency for a higher STS score in the KFI group may be clinically important. Although a higher STS score makes them prone to develop peri-procedural complications and higher mortality, 26 there was no increased mortality rate in the KFI group compared with patients with no change in kidney function, who had a lower baseline STS score.
Several studies assessed KFI specifically in CKD patients. They found a higher rate of KFI after TAVR in these patients. Lower baseline eGFR was associated with a tendency for greater eGFR increase after the procedure,18,25 We evaluated the implications of improved kidney function in all eGFR ranges, with specific attention to CKD patients. As previously reported, a decrease in eGFR even within the normal or mildly reduced range is associated with increased mortality. 27 Hence, our assumption that increase in eGFR even within this normal range may be of clinical significance. In our cohort, patients with reduced baseline eGFR had a higher likelihood of a change in kidney function, either KFI or AKI. Baseline eGFR was significantly lower in the KFI group compared with stable kidney function or patients who developed AKI. While 5% of patients had KFI among the whole cohort, when evaluating only the patients with baseline eGFR <60 mL/min/1.73 m2, 8.7% improved kidney function. There are important bidirectional interactions between the heart and kidneys, leading to the cardiorenal syndrome. 28 Due to these interactions, it is plausible to presume that CKD in patients with severe aortic stenosis is partially attributed to type 2 cardiorenal syndrome, where chronic cardiac dysfunction results in persistently reduced kidney function. Therefore, a probable pathophysiologic explanation for the improvement in kidney function after TAVR is that improvement in cardiac output contributes to a rise in renal plasma flow, reduction in renal venous congestion, and glomerular filtration fraction. Previous reports observed improvement in LVEF as soon as 48 hours post-TAVR. 29 In our study, although LVEF was not a predictor of KFI, patients with KFI demonstrated a statistically significant improvement in LVEF post-procedure compared with patients who developed AKI. This finding strengthens the importance of the reversal of the pathologic cardiorenal interaction in patients with severe aortic stenosis in maintaining PKF.
The study has several limitations. As KFI post-TAVR is not characterized as well as AKI, we used a new definition based on the reversal of VARC-2 definition for AKI. As patients were not in steady state, we decided to use creatinine level instead of eGFR, unlike previous studies. This definition is not identical to the one used in previous studies, and therefore it is difficult to assess the correct incidence of post-TAVR KFI. To overcome this, we examined different definitions in our cohort that led to similar outcomes. In addition, being a single center study, it is subjected to potential bias. The data were collected prospectively; however, analysis plan was defined after data collection and is subjected to possible confounders.
Conclusions
Improvement of kidney function following TAVR was not commonly observed in our large cohort in contrast to previously described data, although incidence varies with different definitions. Improvement in kidney function was not associated with reduced mortality when compared with PKF, not in the whole cohort nor in CKD patients.
Footnotes
Ethics Approval and Consent to Participate
The study was approved by local ethics committee. Informed consent was obtained from all individual participants included in the study.
Consent for Publication
There’s no patients’ identifying data in the article.
Availability of Data and Materials
The data will be available to researchers. Please contact the primary investigator.
Author Contributions
Yaron Arbel and Orit Kliuk-Ben Bassat contributed to study design and data analysis, all authors contributed to data acquisition, and Ariel Finkelstein and Shmuel Banai helped in supervision and mentorship. Each author contributed important intellectual content during manuscript drafting or revision, accepts personal accountability for the author’s own contributions, and agrees to ensure that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ariel Finkelstein is a consultant for Medtronic and Edwards Lifesciences. Amir Halkin is a consultant for Abbott Laboratories and Boston Scientific. All other authors declare having no potential conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance With Ethical Standards
The study was approved by the institutional ethical committee and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from all individual participants included in the study.
