Abstract
Background:
Inotropic dependence and diuretic resistance in patients with cardiorenal syndrome (CRS) lead to frequent hospitalizations and are associated with high mortality. Starting peritoneal dialysis (PD) acutely (within 2 weeks of a heart failure hospitalization) offers effective volume removal without hemodynamic compromise in this population. There is little data on this approach in the North American literature.
Objective:
To determine whether volume-overloaded patients with CRS on maximal doses of diuretic therapy had reduced hospitalization for heart failure following PD initiation.
Design:
Retrospective cohort study.
Setting:
Academic hospital network (University Health Network, Toronto, Ontario).
Patients:
Patients with CRS receiving a bedside catheter and starting PD within 2 weeks of insertion at the University Health Network from January 1, 2013, to December 31, 2018.
Methods and measurements:
Data for heart failure–related hospitalizations and length of stay 6 months before and after PD initiation were collected. Patients who died, switched to hemodialysis, or were transferred to another facility within 6 months of starting PD were excluded from the analysis.
Results:
We identified 31 patients with CRS who had a bedside PD catheter inserted. The average age of patients was 66.0 ± 13.0 years. There were 7 (22.6%) deaths and 4 (12.9%) transfers to other programs or hemodialysis within 6 months of catheter insertion. After exclusion, we analyzed hospitalization and length of stay data for 20 patients. The hospitalization rate 6 months before PD initiation was 6.9 admissions per 1000 patient-days. This decreased to 2.5 admissions per 1000 patient-days after PD initiation. In addition, there was also a significant reduction in the average length of stay per hospitalization (24.1-3.9 days; P = .001).
Limitations:
Our study did not assess the severity of heart failure symptoms using a standardized functional classification system. We did not assess quality of life and illness intrusiveness scores before and after starting dialysis, nor did we capture non–heart-failure-related hospitalizations or external admissions at other hospital sites. We limited eligibility to clinically stable patients with no prior major abdominal surgical history in a single Canadian PD program using bedside ultrasound approach for catheter insertions by experienced nephrologists and included a small number of patients.
Conclusions:
Volume-overloaded patients with CRS receiving maximal diuretic therapy have lower hospitalization rates and shorter stays after initiation of PD. The development of a bedside PD catheter insertion program and close collaboration between nephrology and cardiology services may facilitate acute start dialysis in this population.
Introduction
Heart failure (HF) is a leading cause of hospitalization among adults in North America. In the United States, approximately 6.2 million Americans are diagnosed with HF, affecting 2.2% of the population. 1 In Canada, there were more than 65 000 hospitalizations due to HF between 2017 and 2018, with an average length of stay of 8.9 days. 2 Heart failure–related hospitalizations also place a significant economic burden on the health care systems in Canada and the United States.1,3
Cardiorenal syndrome (CRS) represents a spectrum of disorders that involve both the heart and the kidneys, where acute or chronic dysfunction in one organ can lead to acute or chronic dysfunction in the other.4,5 Observational data have demonstrated that as many as 30% of patients with a diagnosis of HF can also have chronic kidney dysfunction. 6 Patients with CRS also tend to have worsening kidney function during periods of acute illness and hospital stays, leading to prolonged hospitalizations and worse clinical outcomes.5-7
There are many reasons for kidney function to worsen in patients with CRS. Patients with HF may be unable to generate forward blood flow, resulting in kidney hypoperfusion and activation of the renin-angiotensin-aldosterone system (RAAS).4,5,8 Activation of RAAS leads to further salt and water retention, increased preload, and worsening pump failure. 8 Conversely, medications used in the treatment of HF, such as diuretics and RAAS inhibitors, can also worsen kidney function, particularly in patients with an established history of kidney dysfunction. 9
Diuretic resistance is a sequela of CRS and chronic diuretic use with associated hyperplastic changes in kidney distal tubular cells, which can lead to kidney salt retention and recurrent episodes of volume overload and pulmonary edema.10,11 Many patients with CRS have diuretic-resistant HF and may require additional interventions such as inotropic support to optimize cardiac function. A randomized trial (the UNLOAD trial) demonstrated that continuous extracorporeal ultrafiltration within this population reduced rehospitalization rates and HF resource utilization. 12 However, treatment with extracorporeal ultrafiltration is expensive, carries risk of hemodynamic compromise, and is thus not a feasible approach.
Acute-start peritoneal dialysis (PD) offers effective volume removal without hemodynamic compromise in this population. There is little data on this approach in the North American literature. Acute-start PD can be initiated following the insertion of a catheter at the bedside by a nephrologist in select patients (see “Methods” section). The use of the percutaneous bedside approach is preferred in this group due to their tenuous hemodynamic status, which renders them poor candidates for operative catheter insertion using general anesthesia.
The aim of our study was to determine whether volume-overloaded patients with CRS on maximal doses of diuretic therapy have reduced HF rehospitalization rates and shorter hospital lengths of stay following acute PD initiation. We hypothesized that there would be a 50% reduction in subsequent HF-related hospitalizations in patients with CRS following acute PD initiation.
Methods
Study Design and Participants
We conducted a retrospective review of patients with a diagnosis of CRS who received a bedside PD catheter and started on PD within 2 weeks of insertion at the University Health Network from January 1, 2013, to December 31, 2018. The ARECCI (A Project Ethics Community Consensus Initiative) screening tool demonstrated that this project was consistent with quality improvement at minimal risk to participants. 13 As a result, formal research ethics board review was waived for this study.
All patients who underwent a bedside PD catheter insertion were admitted to hospital and had a diagnosis of type 2 CRS (chronic HF leading to progressive chronic kidney dysfunction) along with glomerular filtration rates of <30 mL/min/1.73m2. Patients with hyperkalemia (>5.5 mmol/L), severe metabolic acidosis (bicarbonate <10 mmol/L), and on ventilatory support were not considered for acute initiation of PD. In addition, patients with prior abdominal surgeries, redundant skin folds, and those with reduced visibility of the lower abdomen for proposed exit sites were not considered for bedside PD catheter insertion.
Intervention
Prior to 2013, volume-overloaded inpatients with CRS and diuretic resistance (receiving furosemide >120-200 mg intravenously per day) were referred to the nephrology consult service at our center for consideration of hemodialysis. Furthermore, a standardized process for assessing and managing diuretic resistance in volume-overloaded patients with CRS was lacking. Patients were often started on hemodialysis without considering PD as an alternative. In addition, diuretic-resistant patients with CRS at our center had prolonged stays in hospital, typically averaging more than 20 days per visit. Root-cause analysis revealed that many factors accounted for a prolonged stay in hospital for patients with CRS; primary driving factors were patient-specific and organizational variables (Figure 1). Implementing an acute-start PD program to facilitate ultrafiltration and improve access to care was thought to address many of these factors. Furthermore, in the presence of emerging data that showed benefit of PD in patients with HF, our institution implemented an acute-start PD program with bedside insertions performed by a nephrologist.
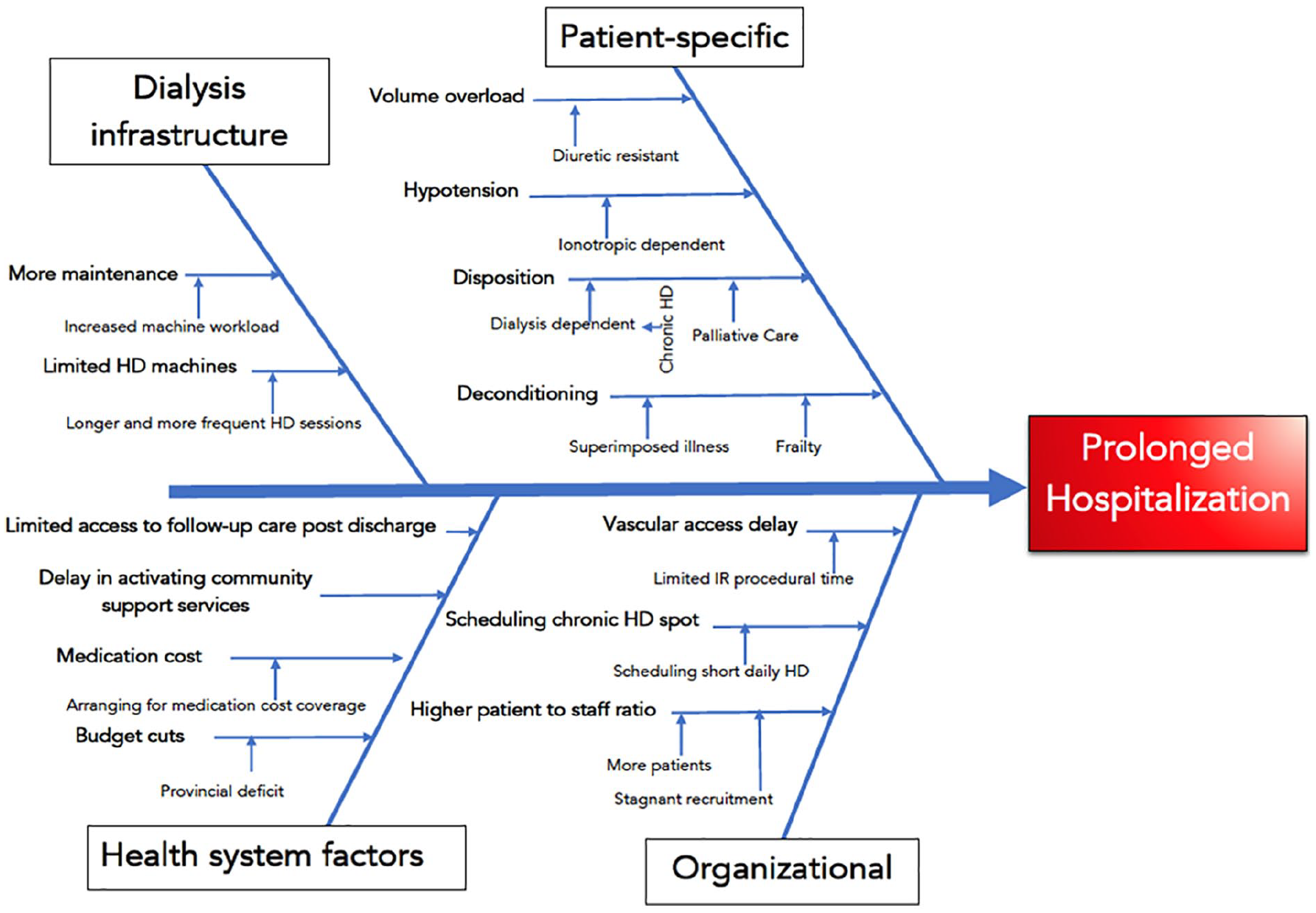
Ishikawa diagram demonstrating various underlying factors that lead to prolonged hospitalization in patients with cardiorenal syndrome at our institution.
At our institution, there is a dedicated multidisciplinary HF team that cares for inpatients with a diagnosis of CRS. This team notifies the nephrology consult team about patients with kidney dysfunction and refractory response to diuretics in the presence of volume overload. The nephrology team then evaluates whether the patient requires dialysis, based on clinical symptoms and laboratory investigations. Patients who are suitable for either PD or hemodialysis are then seen by a dialysis modality educator. If the patient expresses a preference for PD, he or she is assessed by a nephrologist to determine suitability for a bedside PD catheter insertion (Figure 2).

Process of selecting patients with cardiorenal syndrome for bedside PD catheter insertion and acute-start PD.
If the patient is an appropriate candidate, PD catheters are inserted using ultrasound guidance with the Seldinger technique under local anesthetic. A swan-neck coiled tip double-cuffed catheter is used for all insertions. All patients are started on at least 1 exchange daily with low-volume (750-1000 mL) icodextrin, mainly for ultrafiltration purposes rather than for solute clearance. With this in mind, some patients with solute clearance needs were initially started on incremental PD using low-volume glucose-containing dialysate fluid with a minimum of 2 to 3 exchanges on a cycler.
Outcomes
The primary outcome of our study was HF-related hospitalizations. We examined HF hospitalizations 6 months before and 6 months after the initiation of acute PD. We also looked at the hospital length of stay for HF-related admissions before and after PD initiation. Our secondary outcomes included all-cause mortality, PD-related complications, and early catheter failure (<3 months after insertion).
Statistical Analysis
All continuous data variables are presented as means with standard deviations, whereas categorical data are presented as absolute values and percentages within groups. Statistical tests comparing categorical variables were conducted by χ2 test and Kruskal-Wallis to compare continuous variables. Patients who died, switched to hemodialysis, or were transferred to another facility were excluded from the primary analysis. All statistical analyses were completed using SPSS statistical software (Version 25; IBM, Armonk, NY).
Results
Baseline Characteristics
Thirty-one patients with CRS underwent a bedside PD catheter insertion and were started acutely on PD. The average age of patients was 66.0 ± 13.0 years, and 27 (87.1%) patients were men. Patients were diagnosed with volume overload based on assessment conducted by clinicians in cardiology and nephrology. All patients were receiving a minimum daily dose of 180 mg of intravenous furosemide at the time of assessment for PD catheter insertion.
The leading cause of HF was ischemic cardiomyopathy (38.7%), followed by nonischemic dilated cardiomyopathy (32.3%), valvular disease (19.3%), and hypertrophic cardiomyopathy (9.7%). Eleven patients (35.5%) had diabetes, and 14 (45.2%) had a history of hypertension. The mean left ventricular ejection fraction was 36.5% ± 16.1% for 29 (93.5%) patients; 2 (6.5%) patients were on ventricular assist devices at the time of PD initiation and did not have echocardiograms performed. Eleven patients had completed 24-hour urine collections before the start of dialysis. The mean daily urine output was 1518.8 ± 662.9 mL. The mean estimated glomerular filtration rate (GFR) at the start of dialysis was 19.1 ± 7.2 mL/min/1.73m2 (Table 1).
Characteristics of Patients With Cardiorenal Syndrome Who Received Acute Peritoneal Dialysis.

Note. GFR = glomerular filtration rate was calculated using Chronic Kidney Disease Epidemiology Collaboration calculation; LVEF = left ventricular ejection fraction.
11 patients completed 24-hour urine collection before the start of dialysis.
29 patients had LVEF measurements prior to dialysis; 2 patients with ventricular assist devices did not have LVEF measurements.
There were 7 (22.6%) deaths and 4 (12.9%) transfers to other dialysis programs outside of our center within 6 months of catheter insertion (Table 2). Six patients who died were initiated on PD for palliative purposes to manage dyspnea and volume overload (Table 2). After excluding deaths and transfers to other facilities, we performed a subgroup analysis of patients (n = 20) who remained on PD 6 months after initiation to examine their hospitalization rates and length of stay data.
Patient Outcomes Within the First 6 Months of Acute-Start PD.

Note. PD = peritoneal dialysis.
Abdominal hernia was surgically repaired without catheter manipulation within 6 months of insertion.
Outcomes
In our study, HF-related hospitalization rate reduced from 5.9 to 4.6 admissions per 1000 patient-days after the initiation of PD, a 22% reduction in HF-related admission rates. There was also a statistically significant reduction in the average length of stay for all patients from 26.3 to 14 days after PD initiation (P = .048) (Figure 3). A subgroup analysis of patients who remained on PD up to 6 months after initiation also revealed a reduced HF-related hospitalization rate from 6.9 to 2.5 admissions per 1000 patient-days (Figure 4). This was a 63.7% reduction in admission rates following the insertion of PD catheter for acute-start PD. The average length of stay in hospital for HF-related admission also reduced by 83.8% from an average of 24.1 to 3.9 days (P = .001).

Hospitalization rate and average length of stay for all cardiorenal syndrome patients who underwent a bedside PD catheter insertion.

Hospitalization rate and average length of stay for patients with cardiorenal syndrome who remained in the PD program 6 months after PD initiation.
Balancing Measures and Complications
No increase in rates of PD peritonitis was observed up to 6 months after PD initiation compared with our local PD peritonitis rate. There was only 1 episode of peritonitis with Streptococcus viridans organism. This was equivalent to a peritonitis rate of 0.1 episodes per patient-year, consistent with our local peritonitis rate for all patients with PD of <0.2 episodes per patient-year. There were no exit-site infections seen in any of the patients in our study (Table 2).
The most common complication following acute-start PD initiation in our study was leak of dialysis fluid through the catheter exit site. Exit-site leaks occurred in 5 (16.1%) of all patients who received a PD catheter and were managed by reducing PD fill volumes. Among all patients, 1 (3.2%) had an epigastric abdominal wall hernia that required surgical repair within the first 6 months. No patients required surgical revision of their catheters within 6 months following initial insertion. One (3.2%) patient had bleeding from the exit site following insertion, and PD initiation was delayed by 1 week until bleeding resolved (Table 2).
Discussion
We have demonstrated that volume-overloaded patients with CRS receiving maximal diuretic therapy have lower HF hospitalization rates and shorter hospital stays after PD initiation. In select patients, bedside PD catheter insertion using ultrasound technique can be used by nephrologists to facilitate more acute PD initiation with minimal complications. The findings from our study are consistent with smaller observational studies and case reports published outside of North America, showing reductions in HF hospitalization rates after PD initiation in patients with significant kidney dysfunction and diuretic-resistant HF.14-17 In addition, recent reviews have demonstrated that PD is a safe and feasible treatment for the management of symptoms in patients with refractory HF.18,19
Patients with CRS with diuretic resistance have significant morbidity related to multiple hospital admissions from recurrent volume-overloaded states.5,6 Although extracorporeal therapies may help in alleviating the symptoms of volume overload and in turn reducing recurrent hospitalizations, they are more expensive than PD and may be associated with higher rates of hypotension.12,20 Extracorporeal treatments also typically require patients spending extended periods of time at health care facilities. As a result, PD is a more attractive means of achieving ultrafiltration at a lower cost with reduced hemodynamic compromise and the ability to access care outside health care facilities.
Economic reviews of dialysis in Canada have demonstrated that home-based therapies like PD are approximately half the annual average cost of in-center hemodialysis.20-22 Population-based data also revealed that dialysis in the home is more cost-effective and associated with better survival than in-center hemodialysis even after adjustment for baseline patient characteristics. 22 Approaches that focus on extracorporeal ultrafiltration of patients with CRS will incur a higher burden of cost to the health care system than PD. Therefore, the wider adoption of PD also offers significant economic benefits to the health care system and to patient outcomes compared with in-center hemodialysis.
Exit-site leaks were the most common complication seen in patients following catheter insertion. This is consistent with published data that demonstrate an increased risk of leak when PD is initiated within 14 days of catheter insertion. 23 Although we saw a higher rate of leaks in our study, all patients were managed by reducing their fill volumes or holding PD for at least 1 to 2 weeks. Practitioners must be aware of the increased risk of dialysate fluid leak in patients receiving acute-start PD, but this can be easily managed by reducing the fill volume or holding PD for a brief period of time. (Small solute clearance values are of secondary importance in patient with CRS, and frequently mandatory targets will be met using kidney measurements alone.) Furthermore, bedside operator adoptions of operative insertion techniques which have been shown to decrease the risk of PD fluid leak, such as rectus sheath tunneling and use of a purse string suture around the deep cuff, are likely to lower the risk further. 24 Apart from the early leaks identified in our study, PD-related infectious complications were low, comparable to local rates of infectious complications in non-CRS patients on PD.
Preliminary evidence suggests that PD with a sodium-free osmotic solution (10% dextrose) is well tolerated and produces significantly greater sodium removal than the standard PD solution. The use of low sodium PD solutions may further optimize outcomes in this patient population. 25
Our study had several important limitations. First, we did not assess the severity of patient HF symptoms using a standardized functional classification system such as the New York Heart Association. Although there may be rater-related subjective variability in applying this system, it remains a powerful predictor of hospitalizations and survival in this population.26,27
Second, we did not assess the quality of life and illness intrusiveness scores for patients before and after starting dialysis. Despite the reductions in hospitalizations seen in our patients following PD initiation, an understanding of the impact of dialysis on their quality of life would allow for a better understanding of illness intrusiveness. Recent expert opinions have highlighted the importance of adopting palliative approaches to dialysis patients with advanced illness.28,29 A palliative approach to dialysis also affords the opportunity for clinicians to engage in shared decision making with patients and have timely discussions about overall prognosis. 28 Although 6 patients were started on PD for palliative purposes to reduce symptoms of dyspnea from volume overload, the lack of objective measurements limits the interpretation of PD as an effective tool for palliative purposes in our study.
Third, although there were no hospitalizations related to PD complications in our study, we did not capture non–HF-related hospitalizations. These hospitalizations may reflect other sequelae associated with CRS or comorbid disease and may adversely impact quality of life for patients as well. While reductions in HF hospitalizations may represent regression to the mean, diuretic resistance among this subgroup of patients with HF carries a higher burden of comorbidity for patients, and so reduced hospitalization is likely to represent a true improvement related to PD initiation.
Fourth, patients undergoing bedside PD catheter insertions were selected based on clinical stability and no prior major abdominal surgical history. This selection bias may limit the generalizability of our findings to a wider subgroup of patients with CRS.
Fifth, admissions at other hospital sites not affiliated with the University Health Network were not captured in our study. We may have underestimated the frequency of hospital admissions both before and after PD catheter insertion.
Finally, our findings are limited to a single Canadian PD program using a bedside ultrasound approach for PD catheter insertions in small number of patients performed by an experienced nephrologist and may not be generalizable to other centers and populations. Expertise and resources may vary among centers; therefore, PD programs considering acute-start plans in patients with CRS must adopt practices consistent with local clinical data and quality improvement initiatives. Furthermore, the small sample size in our study is likely multifactorial as patients are unlikely to agree to an invasive procedure and start dialysis when they are elderly with good residual urine output and fluctuations in their GFR. These limitations were also highlighted as the reasons for the low recruitment numbers in the attempted prospective randomized Peritoneal Dialysis in Heart Failure (PDHF) study, 30 underscoring the challenges of conducting research with large numbers to examine benefits of ultrafiltration with PD in the HF population.
Conclusions
In summary, PD is often underused in the acute management of volume-overloaded states in patients with CRS receiving maximal diuretic therapy. The Triple Aim of health system performance as outlined by the Institute for Healthcare Improvement focuses on patient experience, patient outcomes, and health care costs. 31 Our results showed reduced HF-related hospitalizations and shorter duration of hospital stay with initiation of acute-start PD for patients with CRS.
Our findings support PD as a low-cost intervention with significant clinical benefit and minimal associated harms. Practitioners should therefore consider acute-start PD in this population. Future studies should examine other patient-important outcomes, including self-reported quality of life, with PD initiation. Care pathways with collaborative partnerships between cardiology and nephrology services that are able to facilitate PD initiation in this setting may be warranted to improve patient-important outcomes and decrease resource utilization.
Footnotes
Acknowledgements
The preliminary findings from this study were presented as an oral abstract at the American Society of Nephrology—Annual Kidney Week on November 8, 2019, in Washington DC, USA.
Ethics Approval and Consent to Participate
The authors have no ethical conflicts to disclose. This project was part of a practice audit and quality improvement initiative at our institution with minimal to no risk to patients, satisfying criteria that aligns with quality improvement and therefore formal research ethics was waived.
Consent for Publication
Consent for publication has been provided by all authors.
Availability of Data and Materials
No additional data or materials are available for this review. Please contact corresponding author with any requests.
Author Contributions
B.A. wrote the manuscript with the support of A.A., A.I., M.G., Z.A., R.M., and J.B. B.A., A.A., A.I., M.G., and Z.A. conducted analysis of the data with supervision from R.M. and J.B. R.M. developed innovative and structured assessment approach described in this study. All authors discussed the results and performed critical review in developing the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
