Abstract
Background:
Diabetic kidney disease (DKD) is a progressive kidney disease and a leading cause of end-stage renal disease (ESRD). Diabetic kidney disease has been strongly associated with increased risk of cardiovascular morbidity and mortality. Despite their susceptibility to cardiovascular diseases (CVDs), patients with DKD are less likely to receive appropriate cardiovascular risk modification as they are generally excluded from major cardiovascular trials. Awareness of vulnerability of these patients necessitates investigating potential interventions that would lessen their risk of adverse outcomes.
Objectives:
This study aimed to explore the effect of bone marrow–derived mesenchymal stem cells (MSCs) in modulating cardiovascular risk factors that develop with the progression of DKD.
Methods:
A total of 60 adult female albino rats were allocated into 3 groups: control group, untreated DKD group, and mesenchymal stem cells–treated diabetic kidney disease (MSCs-DKD) group. Blood pressure, blood glucose level, lipid profile, and atherogenic index were used to assess cardiovascular risk. All rats were killed and subjected to in vitro aortic reactivity studies 8 weeks after induction of diabetes. The MSCs-DKD rats received a single intravenous injection of MSCs 4 weeks after diabetes induction.
Results:
Mesenchymal stem cells injection significantly decreased blood pressure, atherogenic index, and blood glucose compared with untreated rats. The MSCs-DKD aorta also exhibited significant enhancement of vascular reactivity parameters despite absence of improvement in kidney function. These findings conformed to tracked MSCs, which were found residing in aortic and pancreatic tissues and absent in kidneys.
Conclusions:
Mesenchymal stem cells hold hope of improving cardiovascular risk and mortality in patients with DKD, particularly those deteriorating to ESRD.
Introduction
Diabetic kidney disease (DKD) is a chronic renal insufficiency that arises as a consequence of continuous insidious kidney damage and scarring in patients with diabetes mellitus (DM). It is a leading cause of deterioration of quality of life and premature death due to cardiovascular complications. 1
Diabetic kidney disease is considered a marker of cumulative vascular damage and can accelerate cardiovascular disease through several mechanisms, such as blood pressure dysregulation, retention of uremic toxins, anemia, and altered mineral metabolism. 2 A direct association between microalbuminuria and cardiovascular diseases (CVDs) such as coronary artery disease, peripheral arterial disease, and stroke has been established. 3 In fact, excess mortality among patients with DM appears to be largely limited to the subgroup with DKD and explained by their high burden of CVD. 4
Endothelial function is the first to react to positive or negative influences targeting the vessels. Thus, assessment of endothelial function will reveal the earliest changes. 5 Endothelial dysfunction (“the functional atherosclerosis”) precedes by decades morphological atherosclerosis and overt cardiovascular complications. 6 Therefore, detecting and treating endothelial dysfunction is a major focus in the prevention of vascular complications of DKD. 7
Hypertension is a critical risk factor for progression of DKD and one of the most prevalent comorbidities. Failure to manage hypertension properly results in an increased risk of cardiovascular events, as well as a faster progression of DKD. 8 Dyslipidemia has also been recognized to be involved in the progression of DKD, the major abnormalities being elevated triglycerides (TG) and low high-density lipoprotein (HDL) cholesterol. Modified low-density lipoprotein (LDL), such as glycated and oxidized LDL, was found capable inducing vascular and renal cellular dysfunction. 9
Individuals with DKD have increased mortality as a result of the strong association with CVDs. 10 Not only mortality that is of concern, but also morbidity that can significantly reduce quality of life and surge overall costs of care related to DKD. Patients with chronic kidney disease (CKD) have high health care costs that grow more in the presence of cardiovascular comorbidities. Health care plans targeting CKD can benefit from interventions that slow or prevent kidney disease. 11 This highlights that appropriate approach to comorbidities such as hypertension, dyslipidemia, and vascular dysfunction is important in management of DKD.
Stem cells therapy has been explored widely as a promising tool to replace or heal damaged tissues or even treat diseases. Mesenchymal stem cells (MSCs) were experimented in streptozotocin (STZ)-induced DM and succeeded in reverting hyperglycemia and preventing nephropathy in diabetic mice. 12 Interestingly, MSCs administration in renovascular hypertension model successfully prevented the progressive increase of arterial pressure by suppression of renin-angiotensin system (RAS) and decreased sympathetic hyperactivity, 13 2 mechanisms implicated in progression of DKD. Mesenchymal stem cell treatment also significantly reduced dyslipidemia and chronic inflammation in experimental model of induced atherosclerosis. 14 In the context of vascular dysfunction, MSC administration was able to enhance vascular reactivity in patients with heart failure and exhibited significant clinical potentials in other disorders associated with vascular endothelial dysfunction. 15
To conclude, MSCs have potential benefits of tackling several comorbidities that are associated and contributes to the progression of DKD. It is for these potentials, MSCs were chosen in this study to explore their ability to modulate cardiovascular risk factors that accompany DKD.
Materials and Methods
Experimental Animals
This study was done on 60 female adult albino rats, with body weight ranging from 200 to 300 g. Rats were purchased from experimental animals farm in El-Giza and maintained up to (5/cage) in the animal house of the Physiology Department, Faculty of Medicine, Ain Shams University, under standard conditions of boarding and given regular diet consisting of rat chow, milk, bread, and green vegetables. Animals had free access to water, kept at room temperature, and were maintained on a 12-hour light/dark cycle. The rats were accommodated to laboratory conditions for 1 week before starting the experiments.
Animals were not exposed to unnecessary pain or stress and animal manipulation were performed with maximal care and hygiene. This work was approved by Ain Shams Faculty of Medicine Research Committee under Federal Wide Assurance 000017585.
Rats used in this study were allocated into the following groups:
Control group (n = 20): Rats in this group received a single intraperitoneal (i.p.) injection of STZ solvent and 1 mL of citrate buffer. Rats were studied 8 weeks later.
Untreated DKD group (n = 20): Rats in this group were subjected to induction of DM by a single i.p. injection of STZ in a dose of 35 mg/kg, left untreated to develop DKD, then studied 8 weeks later.
Bone marrow–derived mesenchymal stem cells–treated diabetic kidney disease (MSCs-DKD) group (n = 20): Rats in this group were subjected to a single i.p. injection of STZ (35 mg/kg) to induce DM, left untreated to develop DKD, then 4 weeks later, the rats were treated with an intravenous injection of 1.5 × 106 bone marrow–derived MSCs. This group was studied 4 weeks after MSCs transplantation.
Mortality rates among groups were as follows: 3 deaths out of 23 control rats (0.1%), 24 deaths out of 44 rats in DKD group (mortality rate of 55%), 11 deaths out of 33 rats in MSCs-DKD group (mortality rate of 33%).
Induction of DM
Rats were rendered diabetic by a single i.p. injection of STZ (Sigma, St. Louis, MO) in a dose of 35 mg/kg body weight. The dose was modified from 40 mg/kg 16 to achieve the diabetic state with a minimal mortality rate. Streptozotocin was freshly dissolved in citrate buffer and injected to fasted rats within 20 minutes of dissolution because of its unstable nature.
The citrate buffer was prepared by adding 47 mL of 0.05 M citric acid (Morgan Specialty Chemicals, Egypt) to 53 mL of 0.05 M trisodium dihydrate (Morgan Specialty Chemicals). 17
The STZ-injected rats were allowed to drink 10% sucrose solution for 24 hours following the injection to overcome drug-induced hypoglycemia. 18 After 48 hours of STZ injection, rats having blood glucose levels ≥200 mg/dL were considered diabetics and were included in the study.
Bone Marrow–Derived MSCs Preparation
Isolation and propagation of MSCs
Bone marrow of rats was harvested by flushing the tibiae and femurs of 6-week-old male rats with Dulbecco’s Modified Eagle Medium (DMEM; GIBCO/BRL, Waltham, MA) supplemented with 13% fetal bovine serum (GIBCO/BRL). Nucleated cells were isolated with a density gradient (Ficoll/Paque [Pharmacia]) and resuspended in complete culture medium composed of 500 mL DMEM supplemented with 13% fetal bovine serum, 1.5% penicillin/streptomycin, and 0.01% fungizone (GIBCO/BRL). Cells were incubated at 37°C in 5% humidified CO2 for 12 to 14 days as primary culture. Media were changed every 2 to 3 days.
When large colonies developed (80%-90% confluence), cultures were washed twice with phosphate-buffered saline and the cells were trypsinized with 0.25% trypsin in 1 mM ethylenediaminetetraacetic acid (EDTA; GIBCO/BRL) for 5 minutes at 37°C. After centrifugation, cells were resuspended with serum-supplemented medium and incubated in 50-cm2 culture flasks. 19 On day 14, the adherent colonies of cells were trypsinized and counted (Figure 1).
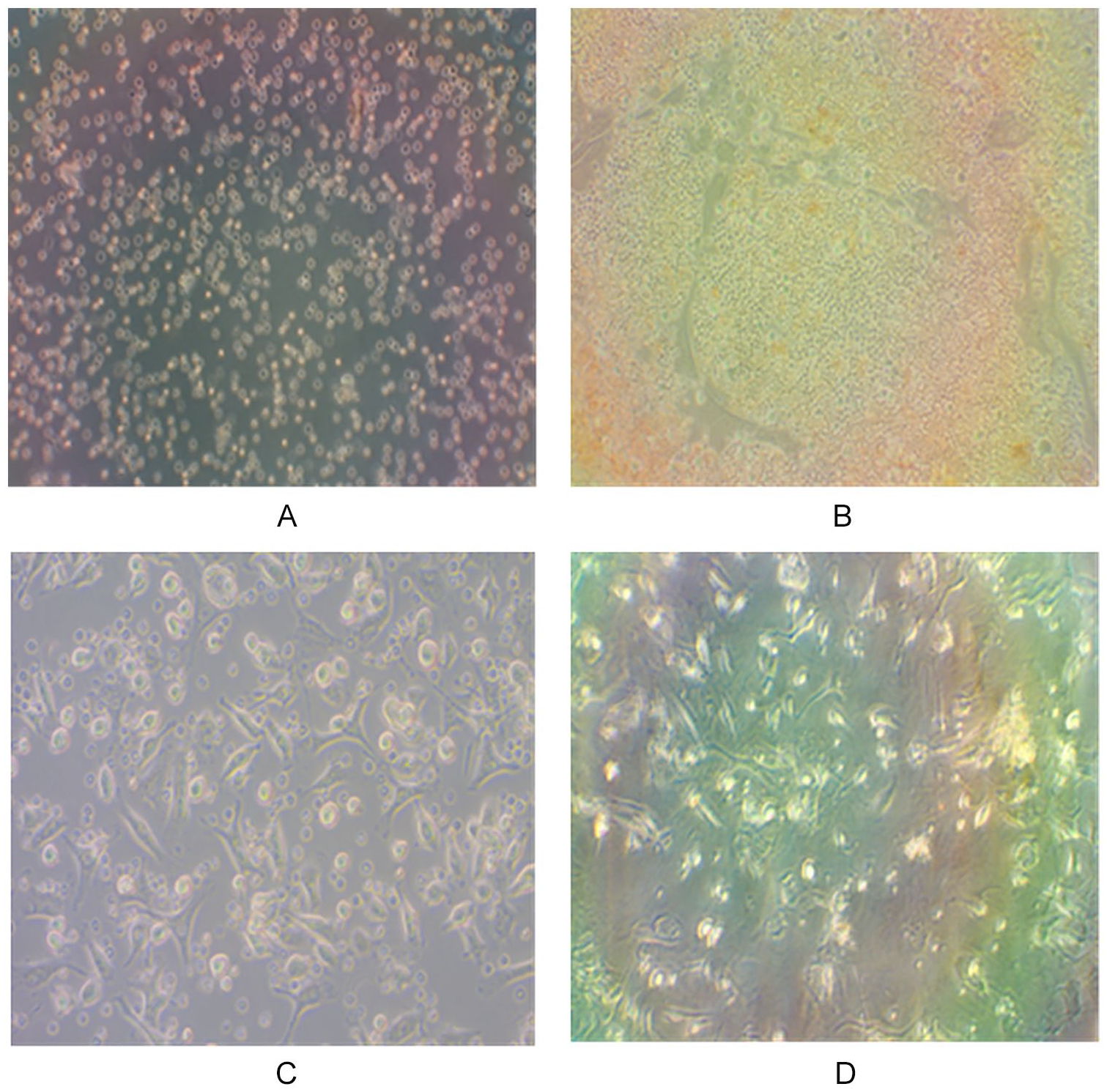
Bone marrow–derived mesenchymal stem cells at days of culturing: (A) day 1, (B) day 3, (C) day 8, and (D) 100% confluent.
Identification of BM-derived MSCs from rat
Cells were identified as being MSCs by their morphology using Giemsa stain, by flow cytometric characterization by surface marker expression. Mesenchymal stem cells were positive for CD90 and CD105 surface markers and negative for CD34 and their power to differentiate into osteocytes. 20 Differentiation into osteocytes was achieved by adding 1 to 1000 nM dexamethasone, 0.25 mM ascorbic acid, and 1 to 10 mM β-glycerophosphate to the medium. Differentiation of MSCs into osteoblasts was achieved through morphological changes—Alizarin red staining of differentiated osteoblasts (Figure 2).

Flow cytometric characterization of bone marrow–derived MSCs.
Labeling of stem cells with PKH26
PKH26 is a red fluorochrome. It has excitation (551 nm) and emission (567 nm) characteristics compatible with rhodamine or phycoerythrin detection systems. The linkers are physiologically stable and show little to no toxic side effects on cell systems. Labeled cells retain both biological and proliferating activity and are ideal for in vitro cell labeling, in vitro proliferation studies, and long-term, in vivo cell tracking.
In the current work, undifferentiated MSCs were labeled with PKH26 according to the manufacturer’s recommendations (Sigma). Cells were injected intravenously into rat tail vein in MSC-DN group, 4 weeks after STZ injection. Four weeks later, the aortic, pancreatic, and kidney tissues were examined with fluorescence microscope to detect the cells stained with PKH26. 21
Experimental Studies
In vivo studies
Arterial blood pressure was measured using noninvasive small animal tail-cuff pulse plethysmography (NIBP200A; Biopac Systems Inc., Goleta, CA) (Figure 3). Blood sugar was measured from rat tail blood samples of fasted rats, using blood glucose test strips of Contour TS blood glucose monitoring system (Bayer HealthCare, Postfach, Basel, Switzerland).

Graph showing steps of blood pressure measurement in rats of studied groups.
In vitro studies
Blood and tissue sampling
On the day of work, overnight fasted rats were weighed and injected intraperitoneally (i.p.) with 6000 IU/kg heparin sodium (Nile Co, Egypt). Half an hour later, the rats were anesthetized by i.p. injection of thiopental sodium (EPICO, Egypt) in a dose of 40 mg/kg. A negative tail pinch response ensured that the rat is at surgical plane of anesthesia. A midline abdominal incision is then made, and the abdominal aorta was exposed and cannulated with a catheter. The collected blood sample was immediately centrifuged, then plasma was stored at −80°C for subsequent determination of the plasma biochemical studies. Pancreas, kidneys, and segments of aorta were dried and kept in 10% buffered formalin solution and embedded in paraffin to be cut and examined with fluorescence microscope. Some specimens were stained with hematoxylin-eosin (H&E) for pertinent histological studies.
Aortic ring reactivity study
After aortic cannulation and blood sampling, the anterior thorax was dissected and lifted (Figure 4). The thoracic aorta was dissected out, excised, and rapidly placed in modified Krebs solution with the following composition in mmol/L (mM): 119 NaCl, 4.7 KCl, 1.6 CaCl2, 1.17 MgSO47H2O, 1.18 KH2PO4, 24.9 NaHCO3, and 11.1 glucose, 0.023 Na2EDTA. 22 Each aorta was cut into 2 to 3 rings (~5 mm in length), then 1 ring was suspended horizontally between 2 stainless-steel hooks, one of them was fixed in the bath and the other connected to fixed isometric force displacement transducer (UGO Basile, Comerio-Italy) and changes in arterial tone were recorded. The chamber bath was filled with 3-mL Krebs solution and continually oxygenated with a gas mixture of 95% O2 plus 5% CO2. Krebs solution in the chamber was maintained at 37°C. The ring was stretched until the optimal baseline tension of 1 g and then allowed to equilibrate for about 60 minutes; in the last 30 minutes of equilibration, washing of rings with 80 mM KCl was repeated 3 times every 10 minutes. 22

Graph showing steps of in vitro aortic reactivity studies in rats of studied groups.
Then, ring was allowed to contract in response to 20 mM KCl until reproducible evoked contractile response was obtained. After a wash and 10 minutes stabilization, the contractile response of aortic ring to 0.245 µM phenylephrine (PE) (Sigma) was used to assess endothelium-dependent vascular reactivity. After the plateau of PE-induced contraction was reached, the relaxant response of 165 µM ACh (Sigma) was obtained on top of PE contraction. Device calibration was done; 8 mm displacement on the curve was equivalent to 0.075 g tension. Absolute values of tension and relaxation were corrected to the weight of each aortic ring (g tension/g weight of tissue).
Determination of cardiac and kidney weights
At the day of work, hearts and both kidneys were blotted dry and weighed using Sartorius analytical balance, BL-210S (USA).
Laboratory analysis
Plasma levels of creatinine, TG, total cholesterol, and HDL cholesterol in plasma samples as well as urine protein/creatinine ratio (UP:UC) in urine samples were measured using Beckman Coulter AU480, fully automated analyzer at the Central Laboratories of El-Demerdash Educational Hospital, Ain Shams University. All laboratory analysis was done at the end of the study.
Low-density lipoprotein cholesterol level was calculated according to the work by Friedewald et al 23 using the following formula: LDL (mg/dL) = Total Cholesterol − HDL − TG/5.
The atherogenic index (AI) was calculated according to the work by Grundy et al 24 using the following formula:
Statistical analysis
IBM SPSS statistics (V. 26.0; IBM Corp., Armonk, NY, 2019) was used for data analysis. Date were expressed as mean ± SD for quantitative parametric measures.
The following tests were done:
Comparison between more than 2 study groups for parametric data using analysis of variance. The possible statistical significance between each 2 groups was done through the multiple comparison (post hoc test or least significant difference). The probability of error at .05 was considered significant, whereas at .01 and .001 are highly significant.
Stepwise logistic multiregression analysis was used to search for a panel (independent parameters) related to the dependent variable.
Results
Tables 1 and 2 and Figures 5 to 9 portray the impact of bone marrow–derived MSCs treatment on several parameters in the setting of established DKD.
ANOVA With Multiple Comparisons Among the 3 Studied Groups.

Note. ANOVA = analysis of variance; MAP = mean arterial pressure; PE = phenylephrine; LDL-C = low-density lipoprotein cholesterol; HDL-C = high-density lipoprotein cholesterol.
Multiple Regression Analysis of Data in Mesenchymal Stem Cells-DKD vs DKD.

Note. DKD = diabetic kidney disease; MAP = mean arterial pressure; PE = phenylephrine; LDL-C = low-density lipoprotein cholesterol; HDL-C = high-density lipoprotein cholesterol.

Comparison between DKD group and mesenchymal stem cells-DKD group regarding % loss of body weight.

Comparison between DKD group and mesenchymal stem cells-DKD group regarding mean blood pressure.

Comparison between DKD group and mesenchymal stem cells-DKD group regarding ACh/PE%.

Comparison between DKD group and mesenchymal stem cells-DKD group regarding blood glucose level.

Comparison between DKD group and mesenchymal stem cells-DKD group regarding HDL-cholesterol level.
At the end of the study, both DKD and MSCs-DKD groups exhibited significant reduction in body weight. However, the MSCs treatment resulted in significant decrease in the percent loss of body weight in MSCs-DKD group compared with the DKD group (Figure 5). This effect of MSCs treatment was not seen on individual organ weights. Kidney and cardiac weights in both DKD and MSCs-DKD groups were significantly higher than control.
Regarding vascular parameters, at the end of the study, mean arterial pressure (MAP) values of MSCs-DKD group has shown significant decrease compared with the DKD group (Figure 6). However, MAP remained significantly high compared with control values.
In vitro vascular reactivity studies have shown enhanced reactivity of aorta of MSCs-DKD group (Figure 7). Both endothelial-dependent contractile responses (PE-induced) and endothelial-dependent relaxation (ACh-induced) were significantly higher in MSCs-DKD group compared with both DKD and control groups, whereas direct myogenic contraction (KCl-induced) showed no statistical difference between all groups.
Biochemical studies have shown significant decrease in blood glucose in MSCs-DKD group compared with DKD group (Figure 8). However, it remained significantly higher compared with control group. Regarding lipid profile, HDL-C levels have shown significant elevation in MSCs-DKD group compared with DKD group (Figure 9), rendering HDL-C values nonsignificant from control levels. This rise of HDL led to significant decrease in AI of MSCs-DKD group compared with DKD group; however, it remained higher than control values. Other lipid profile parameters such as TG and LDL-C were not affected by MSCs treatment and remained significantly higher than control in both MSCs-DKD and DKD groups.
Parameters measuring kidney function did not improve after MSCs treatment. Plasma creatinine levels and UP/UC were significantly high in both MSCs-DKD and DKD groups compared with control group.
Three plasma samples from control group were excluded from results, due to a malfunction of the sample preservation equipment.
A stepwise regression analysis using MSCs-DKD group data vs DKD group data identified 3 independent predictors from known cardiovascular risk factors: MAP, blood glucose level, and endothelial function (ACh/ PE%) (P < .001 HS, F ratio = 25.06). Further stepwise analysis resulted in MAP and ACh/PE% being the least most sensitive predictors for changes between MSCs-DKD group and DKD group (P < .001 HS, F ratio = 35.994) which can be ascribed to MSCs treatment (Table 2).
PKH26-Labeled MSCs Detection by Fluorescent Microscopy
Mesenchymal stem cells labeled with PKH26 fluorescent dye detected in the aortic and pancreatic tissues, confirming that these cells homed into them. No fluorescent dye was detected in kidney tissues of treated rats (Figure 10).

MSCs labeled with PKH26 fluorescent dye detected in sections of aorta (A-B) and pancreas (C-D) of MSCs-DKD tissue samples confirming MSCs homing in these tissues.
Histopathological Examination
Specimens of aorta, pancreas, and kidney of all studies groups were stained with H&E ×400 magnification.
Aortic histopathological changes (Figure 11): Aortic wall layers in DKD group revealed vacuolation of tunica media (black arrow) (Figure 11B) when compared with control group aorta (Figure 11A). Mesenchymal stem cells treatment managed to minimize these tunica media vacuolations in some rats (Figure 11C) and to normalize the aortic layers appearance in several other aortic sections (Figure 11D).

Histological studies of specimens of aorta of the 3 studied groups (hematoxylin-eosin, original magnification ×400).
Kidney histopathological changes
Examined kidney sections from control group show the normal histological structure of renal parenchyma, normal glomerulus, and normal renal tubules (Figure 12A and 12B). The DKD group reveals multiple insults. Protein casts are found in the lumen of renal tubules (Figure 12C), vacuolar degeneration of the epithelial lining along with perivascular edema, and inflammatory cells infiltration (Figure 12D), atrophy of glomerular tuft and necrosis of the epithelial lining (Figure 12E), and glomerular basement membrane shows thickening and hyalinosis (Figure 12F). The MSCs-treated kidneys show congestion of glomerular tuft and vacuolar degeneration of renal tubular epithelium in some sections in the absence of signs of necrosis or atrophy (Figure 12G) and eosinophilic protein cast in the lumen of renal tubules (Figure 12H).

Histological studies of specimens of kidneys of the 3 studied groups (hematoxylin-eosin, original magnification ×400).
Pancreatic histopathological changes
Islets of Langerhans in the u-DN group reveal multiple vacuolations and pancreatic ducts are infiltrated by mononuclear inflammatory cells (Figure 13B) when compared with control group pancreatic sections (Figure 13A). Sections taken from MSCs treated rats show minimum vacuolations of cells of islets of Langerhans in some of them (Figure 13C), whereas other sections of pancreas have normal histopathological appearance (Figure 13D).

Histological studies of specimens of pancreas of the 3 studied groups (hematoxylin-eosin, original magnification ×400).
Discussion
The present study aimed to replicate the cardiovascular risk factors (hyperglycemia, hypertension, dyslipidemia, and endothelial dysfunction) that would accompany DKD. Then, it aimed to investigate the therapeutic potentials of bone marrow–derived MSCs on these risk factors in an attempt to find an effective adjuvant therapeutic agent for cardiovascular morbidity, along with the standard control of hypertension, hyperglycemia, and dyslipidemia in the setting of established DKD.
The main results obtained in this study showed that MSC therapy in DKD experimental model (1) reduced the increased MAP, (2) enhanced vascular reactivity, (3) improved dyslipidemia and AI, and (4) lowered blood glucose level. On the contrary, no effect of MSCs treatment was seen on renal function tests. Mesenchymal stem cells were detected in both aortic and pancreatic tissues and were not found in renal tissue. Major limitation of the study was lack of signaling mechanisms to support the hypothesis. The specific action of MSCs on vascular reactivity and cardiovascular risk factors carries endless possibilities of activated pathways, upregulation of vasoactive substances, and probable MSCs paracrine function that needs to be further explored in future research.
Mesenchymal stem cells are multipotent adult stem cells with the capacities to differentiate, replace damaged tissues, modulate their local environment, activate endogenous progenitor cells, and secrete various factors. 25 Homing of MSCs is a process that describes migration to damaged tissues in response to interaction between signaling molecules released from damaged tissues and receptors expressed on MSCs surface. 26 Following injury, MSCs preferentially home to inflammatory sites where they migrate across the endothelium and enter the injured tissue bed. 27 Several studies had MSCs administered directly into target damaged tissue; in the present study, they were systemically administered to evaluate whether these cells would reach other potentially damaged organs and tissues (still in a reversible stage of damage) such as vascular endothelium, a site potentially important in addressing comorbidities of DKD.
Mesenchymal stem cells treatment succeeded in significantly lowering MAP values in MSCs-DKD rats when compared with values of DKD rats. This can be partially attributed to the significant vascular reactivity enhancement that was observed in treated rats’ aorta during in vitro aortic reactivity studies. Interestingly, MSCs-DKD relaxation responses not only were significantly higher than DKD rats but also went over to surpass those of control rats. Regression analysis had strongly linked the decrease of MAP and relaxation responses (ACh/PE%) to the treatment by MSCs. Another mechanism for MSCs effect on MAP was proposed in other study with a model of renovascular hypertension. Mesenchymal stem cells prevented the progression of hypertension through normalization of the expression of RAS components and reduction of sympathetic nervous system activation. 13
Stem cells treatment succeeded in decreasing blood glucose levels of MSC-DKD rats to a statistically significant level when compared with DKD rats. The presence of labeled MSCs was noticed in the pancreatic tissue of MSCs-DKD rats. Wu and Mahato suggested that MSCs serve as a tropic mediator that can generate new β cells from preexisting β cells in a manner similar to angiogenesis. 28 Other study successfully attempted to transdifferentiate MSCs to insulin-producing cells emphasizing the therapeutic potential of bone marrow–derived MSC in β-cell replacement. 29
Our study also pointed out dyslipidemia as a prominent comorbidity of DKD. Significant elevations of TG and LDL levels were found in DKD rats. Recent epidemiological research identified dyslipidemia as a risk factor for development and progression of DKD and suggested it as a consequence of renal dysfunction and albuminuria. 30 Stem cell therapy was found beneficial to alter the dyslipidemic profile associated with DKD. While there were no significant statistical differences in both TG and LDL levels between the treated and untreated rats, there was a significant increase in HDL levels that managed to significantly decrease the AI in MSCs-DKD rats compared with DKD rats.
Assessment of lipid ratios was reported to be better than individual lipid parameters in predicting risk of atherogenicity and CVD. 31 The authors highlighted the rationale for using “atherogenic index” as risk indicators with greater predictive value than isolated parameters used independently. They also proposed that including the lipoprotein ratios in future recommendations of treatment of dyslipidemia will optimize the predictive capacity of the lipid profile in clinical practice. In the light of these data, the significant elevation of HDL level in the treated rats, along with decrease in the AI, demonstrates an actual improvement of cardiovascular risk that arises in the setting of DKD.
Regarding kidney function assessment, both DKD and MSCs-DKD rats have shown significant increases in plasma creatinine levels and spot UP/UC denoting established renal failure. Elevated creatinine level is a marker of deteriorating kidney function, whereas proteinuria can be considered an independent risk factor for end-stage renal disease (ESRD). 32 The random urine UP:UC ratio is capable of predicting amount of 24-hour urinary protein excretion with high accuracy. Hence, it serves as a faster diagnostic substitute for 24-hour urinary protein estimation. 32 High UP:UC ratio might indicate an irreversible kidney damage that could not provide homing factors to attract MSCs and it was beyond the cells’ capability of regeneration.
Failure of homing of MSCs in the damaged kidneys was observed in our study. Immunofluorescent imaging of treated rats did not show any labeled MSCs in treated rats’ kidney tissues. This is in consensus with results by Choi et al, 33 which observed unchanged levels of blood urea nitrogen and creatinine levels of rats treated with single venous injection of MSCs after 5/6 nephrectomy. The authors also noticed very scarce engraftment of MSCs in the kidneys of rats with chronic renal failure.
How should we view this somewhat disappointing observation? At some point, DKD progresses to irreversible ESRD, in which MSCs have no prospects of regenerating the kidney tissue and reverse uremia. Patients with ESRD rely on dialysis to replace kidney function. However, they are still prone to cardiovascular complications and it remains a major cause of morbidity and mortality. 34 Cardiovascular events, particularly heart failure and atherosclerotic events, have a major impact on health-related quality of life in ESRD. Health care systems actively endeavor continuous efforts to predict these events in an attempt to improve quality of life of patients with ESRD. 35
Mesenchymal stem cells have exhibited potential therapeutic effects targeting cardiovascular risk factors in DKD setting, particularly with irreversible kidney damage. Noticeable improvements of glycemic status, blood pressure, and lipid ratios were observed. Moreover, significant enhancement of vascular reactivity and reversal of endothelial dysfunction was observed in our present study despite the irreversible deterioration of the kidney function. Mesenchymal stem cells can be used as prophylactic measure to enhance vascular reactivity, prevent cardiovascular events, and consequently improve quality of life of patients of DKD and ESRD. Notably, recent clinical trial did support the safety and tolerability of bone marrow–derived MSCs in acute kidney injury, with no evidence of severe injection reactions or long-term adverse events, including infections or the de novo development of malignancies. 36
Conclusions
Tight control of glycemic status, hypertension, and dyslipidemia remains cornerstone intervention in the treatment of DKD. However, DKD remains a huge clinical problem with high chances of cardiovascular complications. Accordingly, there is an ongoing need for development of new therapeutic strategies in the treatment of established DKD to improve patients’ comorbidities. Our data support the notion that MSC treatment can be a promising strategy to treat DKD comorbidities in the near future.
Footnotes
Ethics Approval and Consent to Participate
Animals were not exposed to unnecessary pain or stress and animal manipulation was performed with maximal care and hygiene. This work was approved by Ain Shams Faculty of Medicine Research Committee under Federal Wide Assurance 000017585.
Consent for Publication
The authors have reviewed and approved all the work published, thereby granting SAGE Journal the right of publication of this article.
Availability of Data and Materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

