Abstract
Background:
Loin Pain Hematuria Syndrome (LPHS) is a poorly understood clinical condition characterized by severe pain localized to the kidney but in the absence of identifiable urinary tract disease. There is no consensus on optimal treatment strategies for LPHS. Case reports and series have shown renal denervation via catheter-based radiofrequency ablation to be an effective therapeutic option for the treatment of LPHS. To determine whether catheter-based renal denervation is a meaningful addition to the treatment options in these often-difficult-to-treat LPHS patients, a randomized clinical trial is needed. Prior to conducting a definitive trial that focuses on patient outcomes, ensuring the feasibility of undertaking such a trial is required. As such, we will conduct a single-center randomized control feasibility trial designed to determine viability and provide framework and direction for a larger trial.
Objective:
The objective of the study is to determine whether conducting a randomized trial of renal denervation versus sham procedure is feasible in terms of recruitment and eligibility, and adequacy of follow-up in LPHS patients.
Design:
Single-center double-blinded, parallel-group, partial crossover, sham-controlled, randomized feasibility trial of 10 LPHS patients.
Setting:
Regina General Hospital in Regina, Saskatchewan, Canada.
Patients:
Ten LPHS patients who require opioid therapy.
Measurements:
The main feasibility outcome measures include proportion of target patients who undergo the procedure (treatment or sham) within 6 months; proportion of randomized participants (treatment or control) who entirely complete the follow-up measures at 6 weeks, 3 and 6 months; proportion of the participants who were randomized to control group, cross over after 6 months and opt-in renal denervation treatment; proportion of the crossover participants who complete the follow-up measures at 6 weeks, 3 and 6 months. Pain will be assessed using Brief Pain Inventory Score, McGill Pain Questionnaire, and a pain diary. Mood, disability, and quality of life will be measured by Center for Epidemiologic Studies Depression Scale, Oswestry Disability Index, EuroQol-5D, and Short Form Health Survey Questionnaire, respectively.
Methods:
Eligible participants will be randomized into either renal denervation (treatment group) or a sham treatment (control group). Data (pain, quality of life, mood, disability) will be collected from both groups at baseline, 6 weeks, 3 and 6 months after the intervention. After the initial 6-month follow-up is over, the participants who received the sham procedure will cross over into the treatment group and will be followed for an additional 6 months in the same manner as the treatment group. Descriptive statistics will be used to report outcomes for all patients.
Limitations:
Single-center study, small sample size.
Conclusions:
The lessons learnt from this trial will lay the framework and direction for conducting a multisite randomized controlled trial involving a larger cohort of patients.
Trial registration:
ClinicalTrials.gov (NCT04332731).
What was known before
Case reports and series have shown renal denervation via catheter-based radiofrequency ablation to be an effective therapeutic option for the treatment of Loin Pain Hematuria Syndrome (LPHS).
What this adds
This is the first double-blinded randomized controlled trial designed to determine the feasibility of a larger definitive trial to evaluate the effectiveness of renal denervation on pain relief in Loin Pain Hematuria Syndrome (LPHS) patients in a safe manner. While the main purpose of this study is to determine the feasibility of a definitive multicenter trial, it eventually has the potential to establish the efficacy of catheter-based renal denervation as a mainstream treatment option in LPHS patients.
Introduction
Problem to Be Addressed
Loin Pain Hematuria Syndrome (LPHS) is a rare clinical disorder with a reported prevalence of 0.012% 1 and typically impacts younger women. Since its initial description in 1967, 2 it remains a poorly understood clinical condition characterized by severe, unilateral, or bilateral pain localized to the kidney but in the absence of identifiable urinary tract disease. The natural history is of recurrent episodes of debilitating pain refractory to conventional pain medications. The diagnosis continues to be one of exclusion. The disease imposes a significant health and economic impact in terms of loss of productivity and quality of life in a young population as they are shuffled between numerous health care providers. 3
Multiple visits to the emergency rooms add to the significant burden of investigations and consultations. The debilitating pain leads to increased absenteeism; fragments of interpersonal relationships increase the risk of mood disorders and severely interrupt the quality of life. Adequate pain relief remains the goal but is rarely achieved. Interdisciplinary pain management clinics focusing on drugs (opioids, non-opioid analgesics, antiepileptic drugs, antidepressants, and muscle relaxants), physical, and/or behavioral medicine interventions have been disappointing with more than half of the patients experiencing no improvement in pain. 4 Innovative attempts to modulate the nerve pathways (transcutaneous electrical nerve stimulation, 5 dorsal rhizotomy, 5 capsaicin instillation in the renal pelvis,6,7 renal capsulectomy, 8 and thoracolumbar sympathectomy) 9 to achieve pain control have been associated with variable outcomes. Laparoscopic renal denervation and auto-transplantation as a means of interrupting the pathways have been associated with better pain relief, however, are associated with increased risks of arterial thrombosis and postoperative pain and morbidity. 7
Minimally invasive treatment options are needed for LPHS patients to decrease pain and positively impact the quality of life and level of functioning. The presence of pain-carrying fibers in the renal arterial adventitia presents an opportunity to interrupt the pathways by using radiofrequency nerve ablation as a minimally invasive alternative to surgical options (auto-transplantation and nephrectomy). Unlike the heterogeneous outcomes associated with renal denervation in resistant hypertension, its use in LPHS has been associated with consistently promising results. Case reports and series from our group and others have shown renal denervation via catheter-based radiofrequency ablation to be an effective therapeutic option for the treatment of LPHS.10-14 Randomized clinical trial studies are now needed to establish the efficacy of catheter-based renal denervation as a mainstream treatment option in LPHS patients.
Present Need for a Trial
Novel, innovative, effective, and less invasive approach to alleviate the suffering and disability of LPHS patients is desperately needed. Percutaneous catheter-based radiofrequency ablation of the renal sympathetic nerve fibers that travel in the renal adventitia was clinically introduced in 2009 for the treatment of drug-resistant hypertension. Unlike the well-intended surgical methods to renal denervation, this approach is minimally invasive and targets nociceptive impulses via endovascular delivery of thermal energy in a circumferential manner under local anesthesia, with negligible peri-procedural risk. There are case reports and small case series that suggest a beneficial effect of catheter-based renal denervation in LPHS.10-14 De Jager et al (n = 6 LPHS patients) and a case report from Italy have shown that catheter-based renal denervation has a positive impact on pain and decreases the use of analgesic medications.10,12
Our team has published 2 manuscripts regarding the impact of catheter-based renal denervation on LPHS patients. The first article outlined 4 successful cases, where “50% experienced complete pain relief post-procedure, whereas the other two patients had a 75% improvement in their frequency of analgesic use.” 13 We had further success in a larger 12-person cohort with LPHS, where pain, mood, disability, and quality of life improved significantly postprocedure. 14 However, these previous studies were limited, as they lacked a control group. To determine whether catheter-based renal denervation is a meaningful addition to the treatment options in these often-difficult-to-treat LPHS patients, a randomized, sham-controlled clinical trial is needed. Prior to conducting a definitive trial that focuses on patient outcomes, ensuring the feasibility of undertaking such a trial is required. As such, we propose a single-center randomized control feasibility trial designed to determine viability and provide framework and direction for a larger trial. This trial is a crucial precursor to a larger multicenter randomized controlled trial that will establish the efficacy of catheter-based renal denervation with an acceptable safety profile as a mainstream treatment option in LPHS patients.
Systematic Reviews/Observational Studies
There are no systematic reviews yet on the use of catheter-based renal denervation in LPHS patients. A recent narrative review on pain management strategies in LPHS indicates that catheter-based renal denervation seems to be a promising approach for the treatment of LPHS patients. 3 Surgical renal denervation has traditionally been a potential last line pain management strategy in LPHS cases refractory to noninvasive modalities. Surgical denervation via laparoscopic intervention performed in 9 patients over 10 years revealed favorable data with a curative intervention rate of 44% (median follow-up of 70.5 months).6,15
Individual case reports have suggested successful sympathetic renal nerve interruption via radiofrequency nerve ablation as an alternative to nephrectomy and auto-transplantation in LPHS patients. Gambaro et al first described endovascular renal denervation via catheter-based method, using radiofrequency waves for pain control in LPHS in 2013. 10 Their group reported an LPHS patient with hypertension who underwent renal denervation with successful pain relief and blood pressure reduction at 6 months.
We recently reported on the successful use of renal denervation in 12 patients to achieve pain relief. Fifty percent of the patients were completely off opiates 6 months after the procedure. The rest had relapse of pain within 6 months but not to preprocedural levels and were only taking 25% of the previously prescribed dose. The reduction in pain was accompanied by considerable improvement in functionality, mood, and quality of life. 14 De Jager et al also reported on successful outcomes in 11 patients (6 patients with LPHS and 5 patients with autosomal dominant polycystic kidney disease). A sustained pain reduction effect (up to 12 months) and a significant reduction in medication use were reported in this study. 12 These studies suggest that endovascular delivery of thermal energy could prove to be an effective percutaneously administered minimally invasive therapeutic alternative for the treatment of LPHS. A multicenter double-blind, randomized controlled trial with a sham arm is needed to establish conclusively that renal denervation safely leads to adequate pain relief.
Potential Risks to the Participants
The primary risks of the procedure are similar to the risks of all diagnostic procedures requiring catheterization of the arteries. However, the overall safety analysis of renal denervation has been very positive. Even though the endothelium of the vasculature is exposed to thermal energy, there appears to be no injury sustained as evidenced by postprocedure follow-up angiograms. We did not identify any complications in 60 patients who underwent the procedure (for hypertension and renal denervation) over the last 4 years at our center. Of the 16 LPHS patients who have experienced the procedure so far, 6 of 16 patients underwent the procedure twice and 1 of 16 underwent the procedure on 3 occasions for pain relief. These follow-up procedures allowed us to interrogate the patency of the renal artery for any luminal complications while undergoing renal angiogram. We did not identify any adverse endothelial impact related to prior procedures in our cohort. Moreover, our patients are young, physically well with excellent premorbid status, which placed them at a low risk for adverse events.
The main aim of the study is to determine whether conducting a randomized trial of renal denervation versus sham procedure is feasible in terms of recruitment and eligibility, and adequacy of follow-up in LPHS patients who require opioid therapy.
Primary Objectives
We will consider the trial to be feasible if the following criteria are met:
Eighty percent of the target population (10 patients) undergo procedure (treatment or sham) within 6 months.
Eighty percent of randomized participants (treatment or control) remain in the trial and entirely complete the follow-up measures at 6 weeks, 3 and 6 months.
Secondary Objectives
To assess the proportion of the participants after initial screening who have eligible renal artery anatomy (on angiogram).
To assess the proportion of the participants randomized to the control group who cross over after 6 months and opt-in for renal denervation treatment.
To assess the proportion of the cross over participants who complete the follow-up measures at 6 weeks, 3 and 6 months.
To examine clinical outcomes, including the potential of endothelial injury postprocedure.
To assess outcome measures including morphine-equivalent use (daily dosage), number and frequency of pain medication, type of medication, pain scores, quality of life, disability, and mood at baseline, 6 weeks, 3 and 6 months (Figure 1).

Flow chart and event schedule.
Methods
Trial Design
This trial is a double-blinded, parallel-group, partial crossover, sham-controlled, randomized feasibility trial of 10 LPHS patients recruited from a single center in Regina, Saskatchewan. Participants will be required to complete written consent forms for trial participation. Participants meeting the inclusion criteria will be randomized 1:1 into 2 groups: renal denervation (treatment group) or a sham treatment (control group). All the procedures will take place by a single interventional radiologist at the Regina General Hospital, which is the treatment site for renal denervation for Saskatchewan. The study was approved by the Research Ethics Board of the former Regina Qu’Appelle Health Region (REB-17-88). We have received approval from Health Canada to proceed with the feasibility trial.
Number of Subjects
The sample size of 10 patients (5 per group) has been chosen to assess the feasibility outcomes of this trial. We can realistically recruit and follow 10 patients (15 procedures) at a small single-center site over 24 months.
Duration of Study Period for Each Participant
All the participants will be followed up for 6 months. After this period, the participants who received the sham procedure will have the option of crossing over into the treatment group and will be followed for an additional 6 months in the same manner as the treatment group (Figure 2).
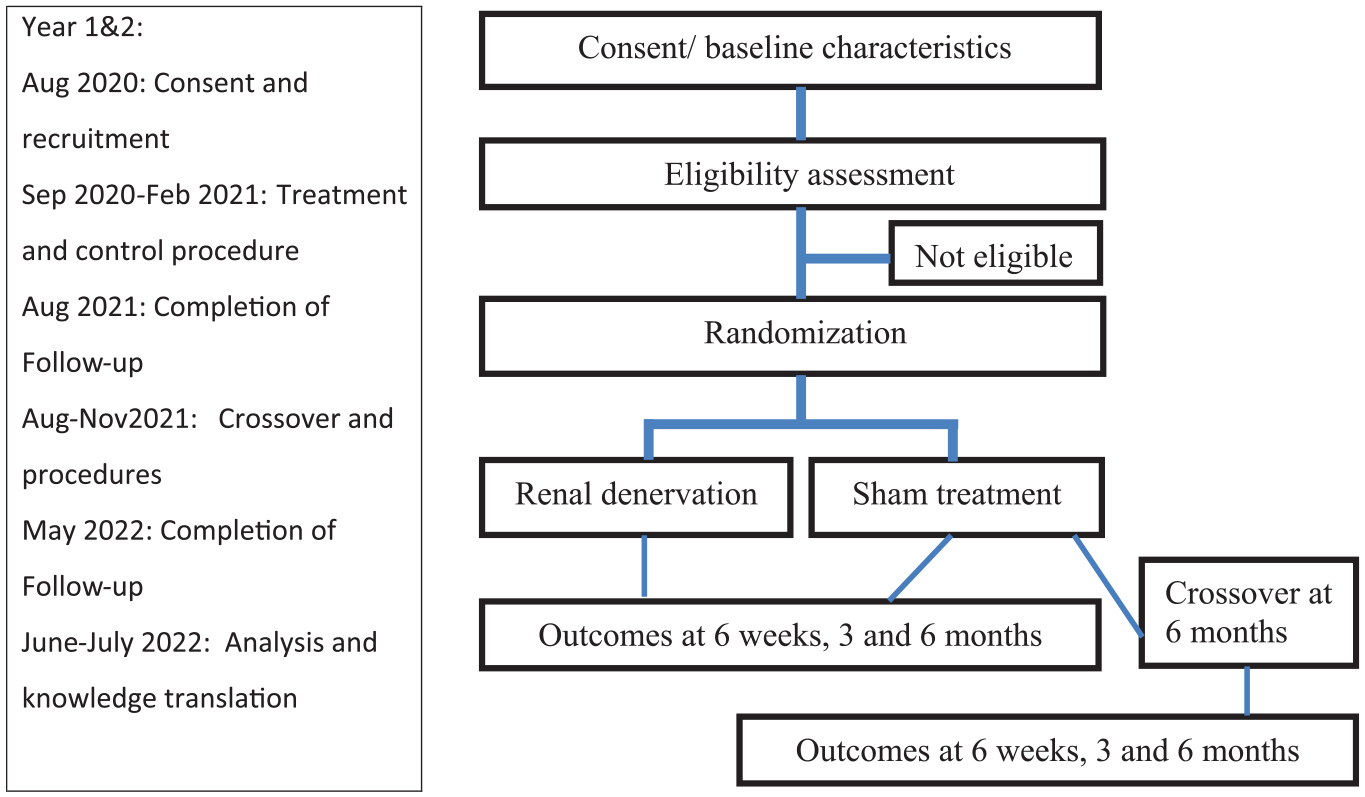
Study scheme and timeline.
Participant Selection and Informed Consent
Ten potentially eligible patients based in Canada have been identified by the principal investigator (PI) during earlier clinical visits. A trained research coordinator will approach potential participants during their subsequent clinic visit to the physician’s office. Adequate time will be given to patients to understand the trial prior to taking consent. Once a participant has consented to the trial, they will undergo screening to ensure that they meet all of the eligibility criteria based on the following inclusion/exclusion criteria.
Inclusion criteria include the following:
≥18 years of age;
Diagnosed with LPHS by a nephrologist, in consultation with a urologist (normal computed tomographic scan with contrast, MAG 3 scan, and negative cystoscopy);
Current use of opiate pain medication for LPHS treatment for >3 months;
Renal arteries with a diameter between 3 and 8 mm (on a formal renal angiogram as the Symplicity Spyral catheter can only be used in diameters between 3 and 8 mm).
Exclusion criteria include the following:
History of kidney auto-transplantation;
Prior history of renal denervation for pain relief and participation in previous renal denervation studies;
Estimated glomerular filtration rate (eGFR) <45 mL/min/1.73 m2 due to the potential of further renal injury caused by contrast exposure;
Confirmed pregnancy;
Need chronic oxygen support or mechanical ventilation via tracheostomy, continuous positive airway pressure, or bilevel positive airway pressure;
Renovascular abnormalities, prior renal angioplasty, indwelling renal stents, and/or aortic stent grafts;
Evidence of a somatoform disorder as per the SCID-5.
The initial screening will be conducted by the PI, based on the inclusion and exclusion criteria for participation in the study (except renal angiogram). The final step of the screening process is a renal angiogram which takes place on the randomization/trial intervention day. Once identified on a formal angiogram to have a diameter of >3 mm, participants will be randomized to the sham or renal denervation arm.
Baseline Data Collection
The following information will be obtained from each study participant by study coordinator and recorded in the case report forms:
Patient characteristics: sex, age, height, weight, ethnicity/race, occupation, education, location of pain (bilateral vs. unilateral, left vs. right), and comorbid conditions.
Laboratory values: complete blood count, serum electrolytes, serum urea/creatinine, urine analysis, β-hCG in women of childbearing age, and eGFR.
Patient outcomes: daily pain medication dosage (primarily opioids using morphine equivalent) and frequency of use (using a daily pain diary), pain (using Brief Pain Inventory score and McGill pain score); quality of life (using EQ-5D and Short Form Health Survey Questionnaire), self-reported disability (using Oswestry Disability Index), and mood (using Center for Epidemiologic Studies Depression Scale) (please see Supplemental Appendix “questionnaires and forms”).
Randomization
The LPHS patients who meet all criteria after the initial screening period will undergo a renal artery angiogram, at the Interventional Radiology suite, Regina General Hospital, to evaluate renal artery anatomy (arteries with a diameter between 3 and 8 mm are suitable). Only subjects with eligible renal artery anatomy will be randomized to 1 of 2 treatment arms: (1) renal denervation (treatment group), (2) sham treatment (control group) immediately following the renal angiogram. If the renal angiogram shows that renal artery anatomy is not eligible for the study, patients will be excluded from the study. A web-based randomization system (REDCap) will be used by the manager of the Interventional Radiology suite, who is not involved with clinical care, to randomly assign patients to either the treatment or control groups. All patients will have an equal chance to be assigned either treatment group with a 1:1 treatment allocation design being used.
Trial Interventions
All patients will receive local anesthesia at the Interventional Radiology suite; they will subsequently undergo a renal angiogram. If they meet the criteria for the diameter (3-8 mm), they will receive general anesthesia. It is required for denervating renal arteries as the procedure was perceived to be painful by the patients in our previous studies.
Sham Procedure
The sham procedure for participants in the control group will consist of only a renal angiogram. Participants in the control group will undergo a diagnostic renal angiogram but will not receive any therapeutic endovascular treatment. The diagnostic catheter will be kept in situ, and a dummy radiograph scan will be performed for another 10 to 15 minutes before removing the femoral sheath from the sedated patient. Participants will remain on the procedure table for at least 20 minutes after the angiogram to prevent possible unblinding of randomization allocation.
Renal Denervation Procedure
For participants in the treatment group, 3000 IU of heparin and 50 μg of Nitrocine will be administered in each renal artery. The interventional radiologist will gain percutaneous femoral access to introduce the 7Fr Terumo destination sheath under aseptic technique. A 0.36-mm-diameter guidewire will be introduced via the arterial puncture. It will be followed by the insertion of a 6Fr Symplicity Spyral catheter (Medtronic, Minneapolis, MN), which is a redesigned catheter with reduced procedural time, enhanced ease of use, and improved safety and efficacy measures. Once electrodes are well apposed angiographically, and impedance values and tracings are stable, radiofrequency energy will be delivered to the treatment site. The 4 electrodes simultaneously deliver radiofrequency energy for 60 seconds. A final renal angiogram will be obtained to check the integrity of the renal artery. Closure device will be used for all patients to allow early ambulation. After the procedures, participants in both groups will be observed closely for 4 hours in Medical Diagnostic Recovery Unit prior to discharge from the hospital.
Methods to Protect Against Bias
This study is a double-blinded study, which will remove potential bias that may affect the perceived effectiveness of catheter-based renal denervation on LPHS patients. The study subjects, PI, study coordinator, and research analyst will not be aware of the study group allocation. Bias is being controlled by completely blinding those who would provide follow-up care to the patient undergoing both the experimental and sham procedures. Only the interventional radiologist and his designated study staff will be aware at the time of the procedure as to the group assignment. The radiologist will not be involved with the clinical care following the procedure, and his knowledge of treatment allocation will not impact the scheduled follow-up allocation. Unblinding will occur after 6-month of follow-up assessment. The patient would be informed after 6 months if they were allocated to the sham arm to undergo the renal denervation procedure if they so choose.
Follow-Up Data Collection
After the renal denervation or sham procedure, patients will be followed for 6 months. At 6 weeks, 3- and 6-month posttreatment, the study coordinator will conduct a clinical or telephone follow-up to assess the number and frequency of pain medications (using a daily pain diary), pain, quality of life, mood, disability, and clinical outcomes including potential of endothelial injury postprocedure. Laboratory data will also be collected at follow-up intervals (Figure 1).
Crossover
Unblinding will occur after the 6-month of follow-up assessment. After this period, the participants who received the sham procedure will have the option of crossing over into the treatment group and will be followed for an additional 6 months in the same manner as the treatment group.
Primary and Secondary Outcomes
Primary outcomes are as follows: Proportion of randomized participants (treatment or control) who entirely complete the follow-up measures at 6 weeks, 3 and 6 months.
Secondary outcomes are as follows:
Proportion of the participants after initial screening who have eligible renal artery anatomy (on angiogram).
Proportion of the participants who maintain a pain diary.
Proportion of the participants who were randomized to control group that cross over after 6 months and opt-in renal denervation treatment.
Proportion of the cross over participants who entirely complete the follow-up measures at 6 weeks, 3 and 6 months.
Daily pain medication dosage and frequency of use, pain, quality of life, self-reported disability, and mood at baseline, 6 weeks, 3 and 6 months.
Data Analysis
As this trial is being performed with the primary intention of demonstrating the feasibility of trial, its goal is descriptive, not inferential. Descriptive statistics will be used to report baseline characteristics and outcomes for all patients. The proportion of patients meeting each of the feasibility endpoints will be presented. Variables such as medication dosage, pain, quality of life, disability, and mood will be reported as medians with the corresponding interquartile range.
We will use inferential statistical tests for clinical outcomes for the definitive large-scale trial as follows: we will compare McGill pain scores (scores 0-78) of the renal denervation treatment group to the sham control procedure group 6 months postprocedure using analysis of covariance to adjust for baseline McGill pain scores as a covariate. We will adjust with patient baseline pain scores because they will likely explain at least a moderate amount of variation in pain score. A linear model adjusted with baseline McGill pain scores with restricted cubic spline functions using 3 knots to flexibly allow for a smooth nonlinear relationship. McGill pain scores will be also compared longitudinally for treatment groups at baseline, 6 weeks, 3, and 6 months posttreatment using general estimating equations. We will compare amount of pain medications used by participants to manage LPHS pain at 6 months postprocedure, adjusting for the amount of pain medications taken at baseline.
Study Timeline
We have identified the potential participants. We plan to recruit patients from August to September 2020, with the treatment and control procedures conducted by February 2021. This study involves a follow-up of 6 months from the treatment and control procedure by August 2021. After this point, participants in the control group will go for the treatment (November 2021) and will be followed for a 6-month postprocedure (May 2022). All data will be gathered by June 2022, after which analysis and knowledge translation activities will occur (July 2022) (Figure 2).
Trial Management
Trial team
The trial will be managed by a small working group, which will include a nephrologist (PI), an interventional radiologist, a urologist, and the study staff (research scientist, research associate, research analyst). Research scientist involved in the study will be responsible for coordination. Confirmation of LPHS diagnosis and eligibility screening will be done by PI. The urologist with experience in diagnosing and treating LPHS patients provides diagnostic expertise needed for LPHS. He will work closely with the research team in the identification and recruitment of LPHS patients. The interventional radiologist has extensive experience with renal denervation procedure, and excellent operational skills will have a beneficial effect on efficiency and success rate of the proposed study. He has successfully performed most renal denervation procedures in North America. The research scientist will be responsible for recruitment (in conjunction with PI), protocol compliance followed by accurate and complete data collection. Baseline and follow-up data collection and data entry will be done by research associate and research scientist. In-kind research support from the former Regina Qu’Appelle Health Region will provide coordination and analysis services throughout the duration of the study.
Steering Committee and Data Safety and Monitoring Committee
The steering committee will be responsible for the progress of the proposed trial. The steering committee will include the trial team and a patient representative. The steering committee will meet monthly in person or through conference call to ensure that all aspects of protocol implementation are done correctly and will provide overall supervision of the trial. The steering committee will consider recommendations of the Data Safety and Monitoring Committee (DSMC) and relevant ethics committees. It will review at regular intervals relevant information arising from other sources and make decisions regarding trial presentation/publication of interim and final results.
An independent DSMC will be formed to formatively assess risks to patient safety throughout the study and to help assure the scientific validity and integrity of the trial. The committee will consist of physicians, researchers, statisticians, and administrators who are not involved in the study and have extensive nephrology and/or research experience. The committee will meet before recruitment, every week during the randomization and procedure period, and every 4 weeks postprocedure. The DSMC will review safety data bimonthly and will provide feedback to PI. The DSMC will make recommendations regarding trial continuation or protocol modification relative to patient safety and outcomes to the steering committee. Should an emergency occur, the research scientist will have access to the master list and give the PI the name, ID, and treatment allocation of the patient involved in the emergency. The PI will decide if there is an emergency with his experience as a physician.
Results
Not applicable (as trial recruitment has not been initiated).
Discussion
This trial is a vital precursor to a large-scale multicenter trial that will evaluate the efficacy of renal denervation in LPHS patients. This trial will lay the framework and direction for conducting a multisite randomized controlled trial involving a larger cohort of patients. This project will provide rich and real-time data critical for a more extensive future study and improving our team’s ability to deal with issues involving recruitment to analysis. The lessons learnt will be incorporated in our future proposal for additional funding. We intend to use the results of the study to apply to multiple grant agencies, including the Canadian Institute for Health Research for a nationwide multicenter trial.
Supplemental Material
Questionnaires_and_Forms_LPHS_Feasibility-word – Supplemental material for Feasibility Study of a Randomized Controlled Trial Investigating Renal Denervation as a Possible Treatment Option in Patients With Loin Pain Hematuria Syndrome
Supplemental material, Questionnaires_and_Forms_LPHS_Feasibility-word for Feasibility Study of a Randomized Controlled Trial Investigating Renal Denervation as a Possible Treatment Option in Patients With Loin Pain Hematuria Syndrome by Bhanu Prasad, Maryam Jafari, Kaval Kour, Kunal Goyal and Francisco Garcia in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors wish to acknowledge the former Regina Qu’Appelle Health Region, Research and Performance Support for assisting with this study.
Ethics Approval and Consent to Participate
The study was approved by the Research Ethics Board of the former Regina Qu’Appelle Health Region (REB-17-88).
Consent for Publication
Not applicable as there is no patient identifying information in this article.
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
B.P. conceived and designed the study. He also edited the final manuscript. M.J. wrote the initial draft. K.K., K.G., and F.G. assisted with the drafts. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
