Abstract
Background:
There is no consensus on intravenous (IV) iron supplement dose, schedule, and serum ferritin target in functional iron deficiency anemia to maintain optimum target levels of iron stores by several guidelines.
Objective:
To examine the effect of IV iron supplementation to different targets of serum ferritin on erythropoietin dose and inflammatory markers in chronic hemodialysis (HD) patients with functional iron deficiency anemia.
Design:
A multicenter, randomized, open-label study.
Setting:
In a developing country, Thailand.
Patients:
Chronic HD patients with functional iron deficiency anemia.
Measurements:
Erythropoietin resistance index, high-sensitivity C-reactive protein, and fibroblast growth factor 23.
Methods:
Two hundred adult chronic HD patients with transferrin saturation less than 30% and serum ferritin of 200 to 400 ng/mL were randomized 1:1 to maintain serum ferritin 200 to 400 ng/mL (low-serum ferritin group, N = 100) or 600 to 700 ng/mL (high-serum ferritin group, N = 100). During a 6-week titration period, participants randomized to the high-serum ferritin group initially received 600 mg IV iron (100 mg every week), while the participants in the low-serum ferritin group did not receive IV iron. During the 6-month follow-up period, the dose of IV iron was adjusted by protocol.
Results:
The mean dose of IV iron was 108.3 ± 28.2 mg/month in the low-serum ferritin group and 192.3 ± 36.2 mg/month in the high-serum ferritin group. The mean serum ferritin was 367.0 ± 224.9 ng/mL in the low ferritin group and 619.6 ± 265.2 ng/mL in the high ferritin group. The erythropoietin resistance index was significantly decreased in the high-serum ferritin group compared to the low-serum ferritin group after receiving IV iron in the 6-week titration period (mean difference: −113.43 ± 189.14 vs 41.08 ± 207.38 unit/week/g/dL; P < .001) and 3-month follow-up period (mean differences: −88.88 ± 234.43 vs −10.48 ± 217.75 unit/week/g/dL; P = .02).
Limitations:
Short follow-up period.
Conclusion:
Maintaining a serum ferritin level of 600 to 700 ng/mL by IV iron administration of approximately 200 mg per month as a maintenance protocol can decrease erythropoietin dose requirements in chronic HD patients with functional iron deficiency anemia.
Trials registration:
The study was registered with the Thai Clinical Trials Registry TCTR20180903003.
What was known before
Intravenous (IV) iron is a standard treatment for hemodialysis patients. However, excessive use of IV iron products can potentially exacerbate adverse events. The data for determining the appropriate regimen of IV iron to maintain the optimum target of serum ferritin is required.
What this adds
We provide the maintenance protocol of IV iron treatment that can reduce the erythropoietin resistance index without serious unfavorable outcomes in chronic hemodialysis patients with functional iron deficiency anemia.
Introduction
Appropriate anemia management for hemodialysis (HD) patients is challenging. Although erythropoiesis-stimulating agents (ESAs) are the mainstay of therapy, the prescribed ESA doses in HD patients at the global level have decreased because of the addition of a black box warning that higher ESA doses might be associated with elevated cardiovascular morbidity.1,2 To reduce ESA dosing requirement, the 2012 Kidney Disease–Improving Global Outcomes (KDIGO) Clinical Practice Guideline for Anemia in Chronic Kidney Disease suggested a trial of intravenous (IV) iron if transferrin saturation (TSAT) is ≤30% and ferritin is ≤500 ng/mL for adult chronic kidney disease (CKD) patients with anemia who are not on iron or ESAs. 3 In this regard, several meta-analyses have demonstrated a superior benefit of IV over oral iron therapy in raising hemoglobin levels and decreasing requirement for ESAs in dialysis patients.4-7 Nonetheless, the potential benefits of a more liberal use of IV iron in HD patients should be weighed against risks. Excessive use of IV iron products in HD patients can potentially exacerbate oxidative stress, inflammation, and endothelial dysfunction and may increase the risk of bacterial infections and cardiovascular events.8-10
Currently available guidelines, including the 2012 KDIGO, 3 the European Renal Best Practice (ERBP), 11 the National Institute for Health and Clinical Excellence (NICE), 12 and the Caring Australasians with Renal Impairment (CARI) 13 have suggested that caution be exercised and the dose of IV iron reviewed when serum ferritin levels exceed 500 ng/mL. According to the NICE guideline, the serum ferritin level should not rise above 800 ng/mL. 12 The discrepancies in recommendations among these guidelines are likely caused by limited data. In addition, there were no recommendations of the IV iron dose or dose schedules to maintain optimum targets of iron indices following the guidelines. Most current evidence was observational, based on a small number of patients and a short follow-up period,5,14,15 all of which resulted in conflicting outcomes regarding the safety of IV iron therapy.
Therefore, we conducted a multi-center, randomized, open-label study to determine the appropriate dosage of IV iron to maintain the optimum target of serum ferritin. We also explored the effects of IV iron on erythropoietin dose as a primary outcome and the effects of IV iron on inflammation markers and serum fibroblast growth factor 23 (FGF23) as secondary outcomes in the different serum ferritin levels in chronic HD patients with functional iron deficiency anemia.
Methods
Study Population and Design
This prospective, multicenter, open-label, randomized, parallel controlled trial was conducted to examine 200 chronic HD patients at the King Chulalongkorn Memorial hospital, The Kidney Foundation of Thailand HD center, Srirattana-Kosin HD Center, Charoenkrung Pracharak hospital, Boonyavej hospital, Priest hospital, Hua Chiew hospital, and Mahesak hospital, Bangkok, Thailand. The study was registered with the Thai Clinical Trials Registry (www.clinicaltrials.in.th) with identifier number TCTR20180903003, and each participant signed a consent form.
The inclusion criteria were as follows: age ≥18 years at screening, undergoing maintenance HD 3 times a week for ≥3 months prior to screening, compliance with HD treatments, and having hemoglobin 8 to 12 g/dL, serum ferritin 200 to 400 ng/mL, and TSAT < 30% and ability to give informed consent. The exclusion criteria were as follows: active gastrointestinal bleeding, active systemic infection, and a history of iron allergy.
The included patients were randomly assigned 1:1 to maintain serum ferritin 200 to 400 ng/mL (low serum ferritin group, N = 100) or serum ferritin 600 to 700 ng/mL (high serum ferritin group, N = 100). The randomization was performed by using the envelope method (block of 4 randomization). Although the participants and treating nephrologists were not blinded to the treatment, all measurements were performed under blinded conditions (the examiner did not know whether the participants were in the low or high serum ferritin group). All patients were advised to continue their regular medications, such as antihypertensive drugs, phosphate binders, and lipid-lowering agents during the study period.
HD and Standard Medication in Dialysis Units
All the patients underwent conventional HD using high-flux dialyzers at a blood flow rate of 300 mL/min and a dialysate flow rate of 500 mL/min. The HD frequency was thrice weekly (4 hours per session). All the patients maintained spKt/V values >1.2 per HD session.
Intravenous Iron Treatment Protocol
The designed IV iron dose and schedule in the present study stem from the following. (1) From a preliminary study in our HD center, which showed that IV iron 100 mg increases serum ferritin level by 50 ng/mL at 2 weeks. (2) A previous study that illustrated iron loss via HD is approximately 100 mg/month. 16 The details of IV iron protocols are shown in the Supplemental Appendix. With these IV iron protocols, the patients in the preliminary study could maintain serum ferritin in the range assigned for each group. In the present study, participants in the low serum ferritin group did not obtain initial IV iron treatment, whereas participants randomized to the high serum ferritin group received a total IV iron dose of 600 mg (100 mg every week) during the initial period. Blood samples were obtained at the end of week 8 (as baseline).
During the follow-up period, serum ferritin, serum iron, and total iron-binding capacity (TIBC) were measured at randomization, the end of titration period, and at 3 and 6 months. If the serum ferritin fell below the target of 200 to 400 ng/mL in the low serum ferritin group or 600 to 700 ng/mL in the high serum ferritin group, IV iron was prescribed to maintain the targets for each group (as described in the Supplemental Appendix).
ESAs Supplement Protocol
The dose of ESAs was adjusted by the treating clinicians who were also site investigators to maintain hemoglobin levels in the range of 10 to 12 g/dL following the KDIGO guideline.
Measurements Obtained Before and During Treatment
The demographic data, cause of end-stage renal disease (ESRD), dialysis vintage, type of vascular access, frequency of HD, brand and dose of ESAs, route of ESAs prescription, and medications were recorded. The iron status including serum ferritin, serum iron, TIBC, anemia parameters (hemoglobin and hematocrit), and erythropoietin resistance index which was calculated from erythropoietin dose (unit) per week divided by hemoglobin level (g/dL), inflammatory markers (high-sensitivity C-reactive protein [hs-CRP]), and FGF23 were measured every 3 months until the end-of-the study (6 months).
Anemia parameters were determined at the Central Laboratory, Chulalongkorn Biochemical Research Laboratory, Bangkok, Thailand. Ferritin was determined by electrochemiluminescence immunoassay (The Roche ECLIA®).
High-sensitivity C-reactive protein was measured by latex agglutination. FGF-23 was assessed using a Human intact FGF-23 ELISA kit (Millipore Corporation, Billerica, MA, USA).
Statistical Analysis
The data were presented as the mean ± SD unless specified otherwise. The difference between the baseline mean of the 2 groups was analyzed using the Student t test. In the case of non-normally distributed data, the Mann-Whitney U test and Wilcoxon signed-rank test were applied. The categorical variables were compared using a chi-square test or Fischer exact test. For the hs-CRP and FGF23 analysis, we used paired t test to compare the natural logarithm (ln) transformed hs-CRP and FGF23 at baseline and at the end of the treatment in both groups and unpaired t test to compare between groups. The Statistical Package for the Social Sciences (SPSS) version 17.0 was used for all statistical analyses. A two-sided P value of <.05 was considered to be statistically significant.
Results
Study Population and Clinical Data
At the randomization period, a total of 704 patients were assessed for eligibility in this study. Of these, 504 were excluded because of the inclusion and exclusion criteria. The remaining 200 HD patients were enrolled in this trial and were randomly allocated to the low (n = 100) or high serum ferritin groups (n = 100; Figure 1). Fourteen patients in the low serum ferritin group and 14 patients in the high serum ferritin group were withdrawn from the study (Figure 1). There were no significant differences in baseline demographics between the 2 groups except hemoglobin levels (Table 1).

CONSORT flow diagram of this study.
Baseline Characteristics of Both Groups.

The Mean Iron Dose of 2 Groups
In the low serum ferritin group, the mean dose of IV iron was 29.43 ± 25.82 mg/week in the first 3 months and 28.03 ± 8.31 mg/week in the last 3 months. On the contrary, the mean IV iron dose was 54.70 ± 80.71 in the first 3 months and 51.98 ± 16.66 mg/week in the last 3 months in the high ferritin group (Figure 2A).

(A) Iron dose per week during follow-up in both groups. Statistically significant difference between groups: *P value < .05. (B) Serum ferritin levels during follow-up in both groups. Statistically significant difference between groups: *P value < .05. (C) Percentage of transferrin saturation during follow-up in both groups. Statistically significant difference between groups: *P value < .05. Statistically significant difference versus randomization: #P value < .05.
Effect on Serum Ferritin Between 2 Groups
The mean serum ferritin was 338.81 ± 96.38 ng/mL at randomization, 297.73 ± 211.12 ng/mL at baseline, 331.82 ± 185.65 ng/mL at 3 months, and 367.02 ± 224.93 ng/mL at 6 months in the low serum ferritin group (Figure 2B). By contrast, the mean serum ferritin was significantly increased from 333.70 ± 96.47 ng/mL at randomization to 547.50 ± 243.92 ng/mL at baseline, 591.44 ± 224.56 ng/mL at 3 months, and 619.60 ± 265.15 ng/mL at 6 months in the high serum ferritin group.
Effect on Serum TSAT Between 2 Groups
The mean serum TSAT was 23.50% ± 6.15% at randomization, 24.39% ± 12.20% at baseline, 24.07% ± 9.73% at 3 months, and 24.38% ± 8.12% at 6 months in the low serum ferritin group (Figure 2C). In the high serum ferritin group, the mean serum TSAT was significantly increased from 25.11% ± 6.53% at randomization to 28.92% ± 8.86% at baseline, 27.71% ± 9.30% at 3 months, and 28.02% ± 8.37% at 6 months.
Primary Outcome: Effect on Erythropoietin Dose Between 2 Groups
The erythropoietin resistance index was significantly decreased in the high serum ferritin group when compared to the low serum ferritin group after the 6-week titration period (mean difference: −113.43 ± 189.14 vs 41.08 ± 207.38 unit/week/g/dL; P < .001), and 3-month follow-up (mean difference: −88.88 ± 234.43 vs −10.48 ± 217.75 unit/week/g/dL; P = .02). At 6-month follow-up, a significant decrease of erythropoietin resistance index from randomization was observed in the high serum ferritin group (853.96 ± 371.72 to 765.34 ± 367.98 unit/week/g/dL, P = .001; Figure 3A).

(A) Erythropoietin resistance index during follow-up in both groups and (B) the change of erythropoietin resistance index from randomization in both groups.
At 6 months, the mean differences of erythropoietin resistance index from randomization were −62.06 ± 276.28 in the low ferritin group and −107.58 ± 271.14 unit/week/g/dL in the high ferritin group, respectively. However, there was no significant difference between the 2 groups (P = .09; Figure 3B).
Secondary Outcomes: Effect on Serum hs-CRP Between 2 Groups
A significant increase of log hs-CRP was observed in both groups during follow-up: 1.92 ± 0.67 to 2.55 ± 0.99 mg/L (P < .01) in the low serum ferritin group and 1.88 ± 0.82 to 2.82 ± 1.11 mg/L (P < .01) in the high serum ferritin group. However, there were no significant differences between the 2 groups (Table 2). There was no significant change in serum FGF23 in both groups during follow-up. In addition, there was no significant difference between the 2 groups (Table 2).
The Baseline and Follow-Up of FGF-23 and High-Sensitivity C-Reactive Protein in Both Groups.
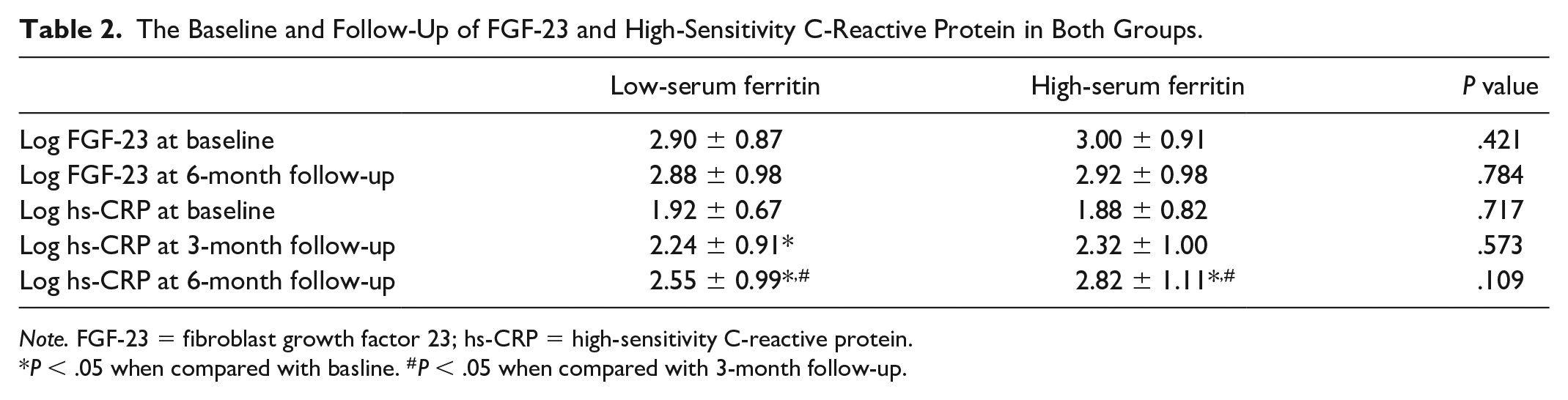
Note. FGF-23 = fibroblast growth factor 23; hs-CRP = high-sensitivity C-reactive protein.
P < .05 when compared with basline. #P < .05 when compared with 3-month follow-up.
Adverse Effects
During the follow-up period, there were 7 patients in each group, who died mainly from cardiovascular disease (Figure 1). The causes of death in the low serum ferritin group were sudden cardiac arrest (4 patients) and congestive heart failure (3 patients) and in the high serum ferritin group sudden cardiac arrest (5 patients) and congestive heart failure (2 patients). The mean serum ferritin level at mortality incidence was 315.3 ng/mL in the low serum ferritin group and 828.86 ng/mL in the high serum ferritin group. The mean erythropoietin dose at mortality incidence was 7,083.25 unit/week in the low serum ferritin group and 6,906.85 unit/week in the high serum ferritin group.
Discussion
This is a multicenter, randomized, open-label study to determine the optimum dosage of IV iron supplement to maintain an optimum target of serum ferritin. We demonstrated that the approximately 100 and 200 mg per month of IV iron supplement could maintain the serum ferritin between 200 to 400 and 600 to 700 ng/mL, respectively. In addition, after receiving IV iron supplement in a 6-week initiation period and 3-month follow-up, the erythropoietin resistance index was significantly decreased in the high-serum ferritin group when compared with the low-serum ferritin group. After 6-month follow-up, a significant decrease of erythropoietin resistance index from randomization was observed in the high serum ferritin group (Figure 2). A significantly increased hs-CRP was noted in both groups during follow-up without significant differences between the 2 groups. Serum FGF23 levels were not significantly altered in both groups during follow-up and did not significantly differ between the 2 groups (Table 2).
IV iron therapy is increasingly integral to the management of iron deficiency anemia and the maintenance of normal iron stores in CKD and dialysis patients. The major concern of IV iron therapy is the toxicity from iron overdose, particularly when the serum ferritin level is higher than 500 ng/mL, and it is generally unacceptable for the serum ferritin level to reach 800 ng/mL. In non-dialysis CKD patients, earlier studies yielded conflicting results. Although there were 2 recent randomized controlled trials (RCTs) conducted to address this issue, the outcomes were disparate.
Ferinject® assessment in patients with iron deficiency anemia and Non-Dialysis-dependent Chronic Kidney Disease (FIND-CKD) was an open-label, multicenter, prospective randomized study of non-dialysis CKD patients with anemia and iron deficiency not receiving ESAs. 17 The results showed that IV ferric carboxymaltose (FCM) targeting a serum ferritin of 400 to 600 µg/L quickly accomplished and maintained an adequate Hb level and delayed and/or reduced the need for other anemia management including ESAs, when compared to oral iron. 17 On the contrary, another RCT of IV and oral iron in CKD (REVOKE Trial) assigned patients with stage 3 and 4 CKD and iron deficiency anemia to either open-label oral ferrous sulfate or IV iron sucrose (200 mg every 2 weeks, total 1 g). 18 Unfortunately, the REVOKE trial was terminated early on the recommendation of an independent data and safety monitoring board based on little chance of finding differences in measured glomerular filtration rate (GFR) slopes, but a higher risk of serious adverse events including those from cardiovascular causes and infectious diseases in the IV iron treatment group. 18
As serum ferritin is also a marker of inflammation, the serum ferritin level in HD patients can assess inflammation occurring in HD patients as it could be elevated immediately after receiving IV iron supplements, especially in a high bolus dose regimen. The upper level of serum ferritin in HD patients is recommended to be lower than 800 ng/mL in clinical practice, but the optimum ceiling level of serum ferritin remains an enigma. A previous study in U.S. patients receiving IV iron showed increased median serum ferritin levels of 500 to 600 ng/mL in one-third of patients due to no recommendations regarding the optimum dose and protocol in several guidelines. 19 This trend has been reported in many industrialized countries 20 and has been the reason for concern regarding iron toxicity. 21
Recently, the PIVOTAL study 22 was conducted as a multicenter, open-label randomized trial in HD patients to explore the benefit of receiving either high-dose IV iron sucrose, in a proactive fashion (400 mg monthly, unless the ferritin concentration was >700 ng/mL or the TSAT was ≥40%), or low-dose iron sucrose, administered intravenously in a reactive fashion (0-400 mg monthly, with a ferritin concentration of <200 ng/mL or a TSAT of <20% triggering iron administration) on the primary endpoint including the composite of nonfatal myocardial infarction, nonfatal stroke, hospitalization for heart failure, or death, assessed in a time-to-first-event analysis. After approximately 2-years follow-up, the high-dose group had a significantly lower risk of primary end-point event when compared with the low-dose group (hazard ratio: 0.85; 95% confidence interval [CI]: 0.73-1.00; P < .001 for non-inferiority; P = .04 for superiority). In addition, the high-dose IV iron regimen resulted in significantly lower doses of ESAs when compared with low dose. 22
Of interest, patients in the high-dose group of the PIVOTAL study received a median monthly iron dose of 264 mg (interquartile range: 200-336), as compared with 145 mg (interquartile range: 100-90) in the low-dose group, 22 which was not different from our study (190 mg/month in high serum ferritin group and 110 mg/month in low serum ferritin group). Both our study and PIVOTAL did not show significant serious adverse events. Although the result in this study was similar to PIVOTAL, the targeted serum ferritin in the low iron group was different. In the PIVOTAL trial, patients were received IV iron only if the ferritin level was less than 200 ng/mL or %TSAT <20%. The mean of ferritin level was less than 200 ng/mL at the end of the study. In contrast, our study targeted a higher ferritin level in the low serum ferritin group as 200 to 400 ng/mL and the mean ferritin level of the patients was 367 ng/mL at the end of study. The present study emphasized the benefit of IV iron in patients with functional iron deficiency anemia, whereas the PIVOTAL study showed the benefit of IV iron in absolute iron deficiency anemia. Furthermore, the PIVOTAL study prescribed IV iron as a bolus regimen, so IV iron had to be stopped in some patients due to a very high serum ferritin level. On the contrary, in the present study, IV iron was given as a maintenance protocol to avoid overshooting of serum ferritin and subsequent iron toxicity.
Besides the total dose of IV iron, the schedule of IV iron treatment is also a concerning issue. In a previous study, 23 comparison of patients receiving bolus (at least 100 mg iron during at least 2 consecutive dialysis sessions) versus maintenance therapy showed that a bolus dose regimen increased risk of infection-related hospitalization (risk difference [RD], 25 additional events/1000 patient-years; 95% CI: 16-33) during follow-up in 776 203 exposure/follow-up pairs. In the present study, the IV iron supplement was scheduled as maintenance protocol as twice a month and once a month (Supplemental Appendix). This may explain why no significantly serious adverse events were reported in our study. Fourteen patients died during follow-up. The major causes of death were cardiovascular disease that might due to the baseline cardiovascular status. In this group, the mean age was 59 years old and the dialysis vintage was about 4 years. Moreover, 75% of patients had diabetes and hypertension which represented high atherosclerotic risk status. Also, the number of fatal cases was similar between the low and high serum ferritin groups. Therefore, the different dose and target of iron supplement were not the contributing factor to patient mortality.
In a previous meta-analysis, IV iron treatment had some effects on the oxidative stress and inflammation markers in cohort studies but had no significant effects in analyses of the RCTs. 4 The present study used a low monthly iron dose and administered IV iron as maintenance regimen, both of which induce less iron toxicity. Despite having no obvious serious adverse outcomes, both low and high serum ferritin groups showed significant and comparable increases of hs-CRP after receiving IV iron treatment (Table 2). There were no available data regarding inflammatory markers in the PIVOTAL study. Therefore, the detection of adverse subclinical outcomes by periodically monitoring inflammation markers is also important in IV iron-treated patients.
Iron deficiency promotes production and cleavage of intact FGF23 into C-terminal FGF23, elevated levels of which were also prospectively associated with mortality. 24 In terms of effect on serum FGF-23 level, IV FCM caused a significant decrease in FGF23 levels without changes to other bone metabolism parameters. 25 Oral ferric citrate also reduced serum FGF23 concentrations in non-dialysis CKD 26 and dialysis patients. 27 However, there were no significant changes in serum FGF23 in the present study, possibly caused by the very high levels of serum FGF23 in our population.
Conclusions
Maintaining a serum ferritin level of 600 to 700 ng/mL by IV iron treatment with an approximate total dose of 200 mg per month as a maintenance protocol can reduce the erythropoietin resistance index in chronic HD patients with functional iron deficiency anemia.
Supplemental Material
Appendix – Supplemental material for Effect of Maintenance Intravenous Iron Treatment on Erythropoietin Dose in Chronic Hemodialysis Patients: A Multicenter Randomized Controlled Trial
Supplemental material, Appendix for Effect of Maintenance Intravenous Iron Treatment on Erythropoietin Dose in Chronic Hemodialysis Patients: A Multicenter Randomized Controlled Trial by Paweena Susantitaphong, Monchai Siribumrungwong, Kullaya Takkavatakarn, Kamonrat Chongthanakorn, Songkiat Lieusuwan, Pisut Katavetin, Khajohn Tiranathanagul, Sookruetai Lekhyananda, Kriang Tungsanga, Supat Vanichakarn, Somchai Eiam-Ong and Kearkiat Praditpornsilpa in Canadian Journal of Kidney Health and Disease
Supplemental Material
CJKHD-20-0010.R1_Translation – Supplemental material for Effect of Maintenance Intravenous Iron Treatment on Erythropoietin Dose in Chronic Hemodialysis Patients: A Multicenter Randomized Controlled Trial
Supplemental material, CJKHD-20-0010.R1_Translation for Effect of Maintenance Intravenous Iron Treatment on Erythropoietin Dose in Chronic Hemodialysis Patients: A Multicenter Randomized Controlled Trial by Paweena Susantitaphong, Monchai Siribumrungwong, Kullaya Takkavatakarn, Kamonrat Chongthanakorn, Songkiat Lieusuwan, Pisut Katavetin, Khajohn Tiranathanagul, Sookruetai Lekhyananda, Kriang Tungsanga, Supat Vanichakarn, Somchai Eiam-Ong and Kearkiat Praditpornsilpa in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We would like to thank all the staff of the participating HD centers for helping us in this project.
Ethics Approval and Consent to Participate
The study was approved by the Institutional Review Board at Chulalongkorn University in accordance with the Declaration of Helsinki.
Consent for Publication
Not applicable.
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ Note
A portion of this work was presented as oral communication in the ASN annual meeting during November 5-10, 2019 in Washington, D.C, USA.
Author Contributions
P.S. and K.P. involved in conception and design of this study. Analysis and interpretation of the data were done by P.S., K.T., P.K., S.E., and K.P. Drafting of the article was carried out by P.S., K.T., and S.E. Critical revision of the article for important intellectual content was done by P.S., M.S., K.T., P.K., S.E., and K.P. Final approval of the article was done by P.S., M.S., K.T., K.C., S.L., P.K., K.T., S.L., K.T., S.V., S.E., K.T., and K.P. Provision of study materials or patients was provided by P.S., M.S., K.C., S.L., K.T., S.L., K.T., and S.E. Statistical expertise was provided by P.S. and P.K. Administrative, technical, or logistic support was provided by P.S., M.S., K.C., S.L., K.T., S.L., K.T., and S.E. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Kidney Foundation of Thailand, and Health Systems Research Institute, Thailand. Iron sucrose was received from Fresenius Medical Care, Thailand.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
