Abstract
Rationale:
Crystalglobulinemia is a rare complication of monoclonal gammopathy wherein crystallized immunoglobulins deposit in various organs causing occlusive vasculopathy, endothelial damage, and thrombosis. It should be differentiated from light chain cast nephropathy without crystalline nephropathy through timely diagnosis with a kidney biopsy.
Presenting concerns of the patient:
We report a case of a 74-year-old female with polyarthralgia, chest pain, petechial rash, and acute kidney injury.
Diagnoses:
Kidney biopsy revealed eosinophilic casts in the tubular lumen and similar occlusive crystalline deposits within the glomerular vasculature and interlobular arteries. Bone marrow biopsy and serum electrophoresis confirmed immunoglobulin G (IgG) κ multiple myeloma.
Interventions:
Dialysis was initiated for severe oligoanuric acute kidney injury. The patient was treated with 5 sessions of plasmapheresis and 11 cycles of clone reduction chemotherapy with CyBorD (cyclophosphamide, bortezomib, and dexamethasone).
Outcomes:
This patient achieved excellent kidney recovery and is no longer dialysis dependent.
Teaching points:
Crystalglobulinemia should be suspected in patients with rapidly progressive acute kidney injury and monoclonal gammopathy. Timely investigation with kidney biopsy to differentiate this condition from light chain cast nephropathy and initiation of appropriate treatment can lead to remission of disease and excellent recovery of kidney function.
What was known before
Crystalglobulinaemia is a rare complication of multiple myeloma that is associated with high mortality and morbidity.
What this adds
Our case demonstrates that timely investigation with a kidney biopsy in a patient with rapidly progressing kidney failure and multiple myeloma to differentiate this disease entity from light chain cast nephropathy, followed by plasmapheresis and clone reduction therapy, can lead to disease remission and reduced mortality.
Introduction
Crystalglobulinemia, an unusual cause of kidney disease in dysproteinemia, can occur as the initial manifestation of multiple myeloma.1,2 It is characterized by the deposition of crystallized monoclonal paraprotein in vasculature causing endothelial injury and typically presents with acute kidney failure, polyarthralgia, and necrotising cutaneous lesions. 3 Morbidity and mortality associated with this disease is high. 4 We report a rare case of crystalglobulinemia associated with myocardial injury and severe crystalline nephropathy successfully treated with plasmapheresis and clone reduction chemotherapy with kidney and hematological recovery.
Case: Clinical Findings
A 74-year-old female was admitted to our hospital with 2 weeks of progressively severe chest and back pain, polyarthralgia, and acute kidney injury. At presentation, her serum creatinine was 368 µmol/L. Her troponin-T was 447 ng/L (normal <14 ng/L), which peaked at 1223 ng/L seven days later, without ischemic changes on the electrocardiogram. A coronary angiogram showed 60% stenosis of the first diagonal branch of the left anterior descending artery, which was medically managed with clopidogrel 75 mg daily and continued on her regular aspirin 100 mg daily. A petechial rash developed in bilateral upper and lower limbs resembling livedo reticularis. Our patient had worsening oliguric acute kidney injury and the serum creatinine peaked at 763 µmol/L on day 5 of admission. Urine microscopy revealed no evidence of hematuria with no casts, and there was moderate proteinuria (0.55 g/24 h). Hemodialysis was commenced due to medically refractory hyperkalemia to 7.3 mmol/L.
Diagnostic Focus and Assessment
Initial investigations showed hemoglobin 106 g/L (normal 115-165), platelet count 243×109/L (normal 150-450), lactate dehydrogenase (LDH) 258U/L (normal <250), reticulocytes 3.1% (normal 0.2-2.0), haptoglobin 3.0 g/L (normal 0.3-2.0), and direct antiglobulin test was negative. Other investigations were as follows: anti-myeloperoxidase antibodies, anti-proteinase 3, anti-glomerular basement membrane negative, anti-nuclear antibodies, anti-double stranded DNA negative with normal C3 1.34 g/L (normal 0.90-1.8), and low C4 0.08 g/L (normal 0.1-0.4). HIV, hepatitis B and C serology were negative. Serum protein electrophoresis revealed IgG kappa paraprotein of 25.7 g/L, free kappa light chain 309 mg/L (normal 3.3-19.4), free lambda light chain 14.17 mg/L (normal 5.71-26.30), and κ/λ ratio 21.8 (normal 0.26-1.85). Urine Bence-Jones protein was positive (0.16 g/24 h) and identified as IgG kappa on immunofixation. Serum immunoglobulin (Ig) levels demonstrated IgG 19.8 g/L (normal 7.0-16.0), IgA 0.34 g/L (normal 0.70-4.70), and IgM <0.05 g/L (normal 0.40-2.30). Serum cryoglobulin was negative. Renal ultrasound showed normal sized kidneys with normal cortical thickness and echogenicity.
Kidney biopsy was delayed until day 8 of admission due to potential bleeding risk with antiplatelet therapy. Examination of the biopsy under light microscopy (LM) showed 1 out of the 18 glomeruli was sclerosed. Crystalline eosinophilic casts were present in the lumens of many medullary tubules (Figure 1B). Similar crystalline deposits were present in a small interlobular artery with occlusion of the lumen, and focally in the intima of a large interlobular artery. Several glomeruli showed similar mainly crystalline deposits in glomerular capillary loops occluding lumens (Figure 1A). There was a focal cellular reaction to deposits including some neutrophils in glomerular capillary loops and very focal mild endocapillary proliferation. There was patchy intimal inflammation of reactive appearance in arteries adjacent to crystal deposits but no transmural vasculitis. A single possible thrombotic microangiopathy lesion was present in an afferent artery near one glomerulus. Ultra-structure showed widening of subendothelial spaces of some glomerular capillary loops with subendothelial flocculent material consistent with a thrombotic microangiopathy. Tubular crystal deposits had an organized parallel linear ultrastructure. Immunofluorescent studies were not contributory as the sample for IF was infarcted. The crystals were weakly staining for Periodic acid-Schiff (PAS) and Congo red negative. Electron microscopy (EM) confirmed glomerular changes consistent with thrombotic microangiopathy and numerous protein casts with some containing crystals (Figures 1C and 1D). The final diagnosis of light chain cast nephropathy and crystalglobulinemia was made. Bone marrow biopsy showed 38% abnormal plasma cells (phenotype CD19- and CD56++) with cytoplasmic expression of kappa cell surface immunoglobulin light chains, consistent with a diagnosis of plasma cell multiple myeloma. Abnormal high-risk cytogenic profile was evident on further analysis, including 1q21 amplication, deletion of 13q14 and 13q34 in 28% of cells, and dual fusion consistent with t(4;14) in 26% of cells. An X-ray skeletal survey was performed which showed no evidence of lytic or sclerotic bony lesions.

(A) Eosinophilic crystalline deposits in glomerular capillary loops. (B) Crystalline casts in medullary tubules. (C, D) Electron microscopy showing crystalline ultrastructure.
Therapeutic Focus and Assessment
Our patient received a total of 11 cycles of standard dosing CyBorD: Cyclophosphamide was withheld for the first cycle and then she received 500 mg with mesna cover (day 1, 8, 15), bortezomib 1.3 mg/m2 (day 1, 4, 8, 11, q21 days and then weekly from cycle 5), and oral dexamethasone 20 mg (day 1, 2, 8, 9, 11, 12). Plasmapheresis was initiated to reduce the monoclonal paraprotein burden, and a total of 5 sessions were completed over a period of 9 days. She received daily plasma exchange for the first 3 consecutive days, and subsequently on days 5 and 9.
Follow-up and Outcomes
There was immediate improvement in her symptoms following the first session of plasmapheresis; her symptoms of chest and back pain, polyarthralgia, and petechial rash rapidly improved. Punch biopsies of her rash from multiple sites obtained after the initiation of plasmapheresis showed no evidence of vasculitis, hyaline-type thrombi, or crystals within the small vessels. Our patient received 11 cycles of cyclophosphamide, bortezomib, and dexamethasone (CyBorD) clone reduction chemotherapy with complete resolution of her symptoms, normalization of the troponin levels, reduction of her paraprotein level to <1 g/L, and excellent recovery of kidney function. Two months later, she was no longer dialysis-dependent. Thirty-two months later, her latest serum creatinine is 100 µmol/L and urine albumin/creatinine ratio is 1.2 mg/mmol (Figure 2).
Timeline
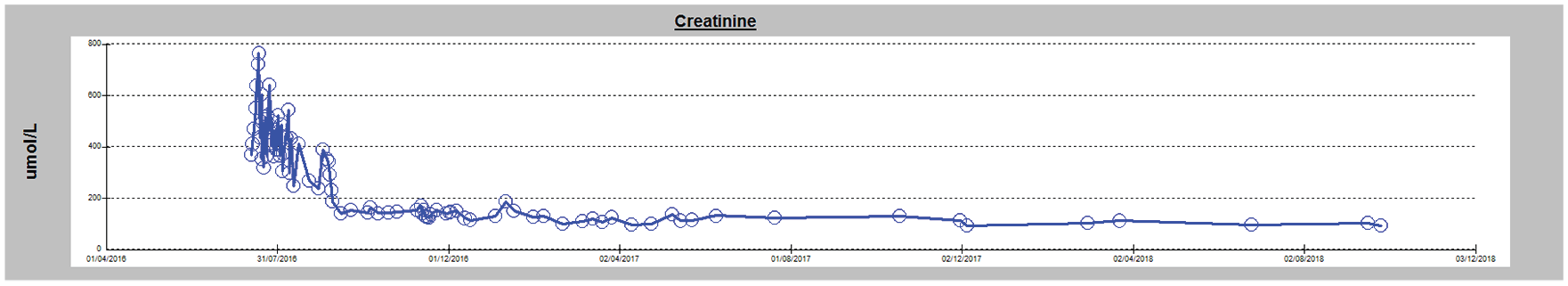
Improvement in renal function during course of treatment.
Discussion
Crystalglobulin-induced nephropathy is a rare life-threatening disease that has been described in cases of monoclonal gammopathy.3-5 Paraprotein crystalline nephropathy can be classified as intracellular (in light chain proximal tubulopathy and crystal-storing histiocytosis) or extracellular (in crystalline variant light chain cast nephropathy and crystalglobulinemia), with the extracellular variant usually manifesting as acute kidney injury and is often fatal.2,4,6 If the crystals are cryoprecipitating, the disease is termed cryocrystalglobulinemia. 7 The kidney biopsy in this case demonstrated a morphological variant of myeloma cast nephropathy wherein the tubular casts were crystalline, with further evidence of crystal deposition in the arteries and glomerular capillary lumens supporting the diagnosis of extracellular crystalglobulin-induced nephropathy.
The mechanism of crystal formation is not well understood. It is thought to be formed by Fc-Fc interactions of IgG monoclonal proteins, possibly due to abnormal glycosylation of light chains or interaction with albumin.1,8-10 The crystals precipitate in vasculature and can cause occlusive vasculopathy, endothelial damage, and thrombosis in various organs, manifesting as renal insufficiency, polyarthropathy, peripheral neuropathy, necrotizing cutaneous lesions, and gastrointestinal ulceration.4,10,11 Myocardial involvement has also been described but is not well documented. 2
Most cases described in the literature have had a fulminant and fatal clinical course with survival from time of diagnosis usually ranging between a few weeks to 12 months. 5 Relapse of the disease is not uncommon after an initial partial response to treatment and the likelihood of restoration of renal function is very low, with most patients remaining dialysis-dependent. 2 Here we report a rare case of a patient with myeloma-related crystalglobulinemia that was dialysis-dependent who not only survived, but had excellent recovery of kidney function. Other reported cases of survival in myeloma-related crystalline nephropathy have only had partial recovery of kidney function,2,5 while some patients never required dialysis.8,12 It is postulated that successful treatment depends upon the early diagnosis and aggressive treatment of the disease, even if the paraprotein levels are minimal or undetectable.4-6,8 It is a different entity to hyperviscosity syndrome associated with multiple myeloma, whereby the paraprotein increases the internal resistance or “thickness” of the blood 13 without the formation of crystalline casts. In our case, it was thought that a paraprotein level of 25.7-g/L IgG kappa would unlikely be associated with hyperviscosity, albeit a plasma viscosity level was not performed. Given that her paraprotein burden was not high, plasmapheresis was not necessarily indicated. However, it was utilized to prevent further crystalline damage until the clone reduction therapy could take effect.14,15 Crystalglobulinemia has been more recently recognized also in MGRS (monoclonal gammopathy of renal significance), a monoclonal gammopathy affecting the kidneys with minimal paraprotein levels and not yet meeting the diagnostic criteria for myeloma. 16 There have been at least 2 reported cases of crystalglobulinemia in MGRS who were treated with CyBorD with excellent renal recovery,3,5 which supports the benefit of early diagnosis and treatment.
Crystalglobulinemia can often mimic other diseases, such as vasculitis 17 or rheumatological conditions, 10 which poses an important diagnostic challenge. 18 Our patient had been diagnosed with seronegative arthritis and polymyalgia rheumatic and treated with methotrexate and steroids without any symptomatic relief. These symptoms resolved completely with plasmapheresis and clone reduction chemotherapy. Furthermore, her chest pain significantly improved with the initiation of the plasmapheresis, suggesting that the process of myocardial injury may have been secondary to the deposition of crystalglobulins, albeit a cardiac biopsy was not performed to confirm this postulation.
Conclusion
Crystalglobulinemia is a rare disease that should be suspected in patients with rapidly progressive acute kidney injury and monoclonal gammopathy. Our case demonstrates that timely investigation with kidney biopsy and appropriate treatment can lead to remission of multiple myeloma and excellent recovery of kidney function.
Footnotes
Author Contributions
A.C. contributed to writing manuscript and retrospective medical analysis. C.L. contributed to drafting manuscript. L.V. contributed to critical review and editing of manuscript. S.-J.H. contributed to critical review. F.P. contributed to critical review. S.V.B. contributed to conceptualization, critical review and editing, and supervision.
Ethics Approval and Consent to Participate
The authors have no ethical conflicts to disclose. Our patient provided informed consent for the publication of this case report.
Consent for Publication
Consent for publication has been provided by all authors.
Availability of Data and Materials
The data and materials are not available for this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
