Abstract
Background:
Pain is common in patients with chronic kidney disease (CKD). Analgesics may be appropriate for some CKD patients.
Objectives:
To determine the prevalence of overall analgesic use and the use of different types of analgesics including acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs), adjuvants, and opioids in patients with CKD.
Design:
Systematic review and meta-analysis.
Setting:
Interventional and observational studies presenting data from 2000 or later. Exclusion criteria included acute kidney injury or studies that limited the study population to a specific cause, symptom, and/or comorbidity.
Patients:
Adults with stage 3-5 CKD including dialysis patients and those managed conservatively without dialysis.
Measurements:
Data extracted included title, first author, design, country, year of data collection, publication year, mean age, stage of CKD, prevalence of analgesic use, and the types of analgesics prescribed.
Methods:
Databases searched included MEDLINE, CINAHL, EMBASE, and Cochrane Library. Two reviewers independently screened all titles and abstracts, assessed potentially relevant articles, and extracted data. We estimated pooled prevalence of analgesic use and the I2 statistic was computed to measure heterogeneity. Random-effects models were used to account for variations in study design and sample populations, and a double arcsine transformation of the prevalence variables was used to accommodate potential overweighting of studies with very large or very small prevalence measurements. Sensitivity analyses were performed to determine the magnitude of publication bias and assess possible sources of heterogeneity.
Results:
Forty studies were included in the analysis. The prevalence of overall analgesic use in the random-effects model was 50.8%. The prevalence of acetaminophen, NSAIDs, and adjuvant use was 27.5%, 17.2%, and 23.4%, respectively, while the prevalence of opioid use was 23.8%. Due to the possibility of publication bias, the actual prevalence of acetaminophen use in patients with advanced CKD may be substantially lower than this meta-analysis indicates. A trim-and-fill analysis decreased the pooled prevalence estimate of acetaminophen use to 5.4%. The prevalence rate for opioid use was highly influenced by 2 large US studies. When these were removed, the estimated prevalence decreased to 17.3%.
Limitations:
There was a lack of detailed information regarding the analgesic regimen (such as specific analgesics used within each class and inconsistent accounting for patients on multiple drugs and the use of over-the-counter analgesics such as acetaminophen and NSAIDs), patient characteristics, type of pain being treated, and the outcomes of treatment. Data on adjuvant use were very limited. These results, therefore, must be interpreted with caution.
Conclusions:
There was tremendous variability in the prescribing patterns of both nonopioid and opioid analgesics within and between countries suggesting widespread uncertainty about the optimal pharmacological approach to treating pain. Further research that incorporates robust reporting of analgesic regimens and links prescribing patterns to clinical outcomes is needed to guide optimal clinical practice.
What was known before
Pain is common in patients with chronic kidney disease (CKD), and analgesics may be appropriate for some CKD patients to promote patient-centered care and improve patient outcomes. Very little is known about current analgesic prescribing for patients with CKD.
What this adds
There is tremendous variability in the prescribing of analgesics suggesting widespread uncertainty about the optimal pharmacological approach to treating pain.
Introduction
Pain is common in patients with chronic kidney disease (CKD). It is associated with considerable disability and lower health-related quality of life (QOL) and is a significant burden on the healthcare system.1,2 The treatment for chronic nonmalignant pain may include nonopioids such as acetaminophen, adjuvants (drugs such as gabapentin and amitriptyline that are not primarily indicated to control pain but can be used for this purpose), as well as opioids.3,4
The international nephrology community advocates for routine screening and management of pain as a way to promote patient-centered and outcome-oriented healthcare.5,6 Analgesics may be appropriate for some CKD patients. However, their use, especially that of opioids for chronic pain, is accompanied by significant risks. Patients with advanced CKD are also at added risk of drug-related adverse effects and toxicity due to the altered pharmacokinetics and pharmacodynamics in kidney failure. 7 Given the limited availability of high-quality clinical trials in chronic pain for patients with CKD, current recommendations are based on guidelines for the general population, clinical experience, and best opinion.
A better understanding of the analgesic prescribing patterns for patients with advanced CKD is required to understand care gaps and optimize pain management strategies. The objective of this systematic review was to determine the prevalence of analgesic use in patients with CKD and to determine the types of analgesics being prescribed for these patients.
Methods
Search Strategy and Eligibility Criteria
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines for systematic reviews were used. The literature search was developed and conducted by an experienced librarian. The detailed protocol is outlined in Supplemental Table S1. The predetermined inclusion and exclusion criteria are outlined in Table 1. We included all interventional and observational studies that presented original data of the use of analgesics (including nonsteroidal anti-inflammatory drugs [NSAIDs], acetaminophen, opioids, and adjuvants) in adult patients with glomerular filtration rate (GFR) category (G) 3-5 CKD. Kidney transplant patients were included if they also had reduced kidney function presenting as G3-5 CKD. We included studies presenting data from 2000 or later, given the change in clinical practice around pain management with increased focus on prescribing analgesics since that time. Single-case studies or case series were excluded, as were studies that were presented only as abstracts, posters, or Letters to the Editor. Articles published in a language other than English were translated and included. An online Neural Machine Translation tool was utilized to provide a general translation of the non-English articles and native speakers were consulted as needed. Studies that only enrolled patients with a primary diagnosis of acute kidney injury or kidney transplant patients with preserved kidney graft function were excluded as were studies that limited the study population to a specific cause, symptom, and/or comorbidity (with the exception of chronic pain) of CKD as these studies were outside the scope of our study objectives. Other exclusion criteria included studies that were limited to acute or intradialytic pain.
Inclusion and Exclusion Criteria.

Note. CKD = chronic kidney disease; CKM = conservative kidney management; NSAID = nonsteroidal anti-inflammatory drug; eGFR = estimated glomerular filtration rate; ESKD = end stage kidney disease; PICOs = population, outcome, study design, and exclusion criteria.
Information Sources
Information sources included electronic databases, reference lists of relevant literature, and websites of relevant networks, organizations, and societies. The electronic databases searched included MEDLINE, CINAHL, EMBASE, and Cochrane Library databases. These were last searched on February 19, 2019.
Study Selection and Data Collection
Two reviewers independently screened all titles and abstracts to identify potentially relevant articles. Full texts of potentially relevant articles were retrieved and assessed independently by two reviewers for possible inclusion based on the predetermined selection criteria. The reference lists of reviews, systematic reviews, and guidelines were also reviewed to ensure all relevant studies were identified. The two reviewers compared individually recorded decisions for inclusion and exclusion and any disagreements were resolved based on discussion and consensus with a third reviewer.
Data Items
The outcomes of interest were the prevalence of analgesic use in patients with G 3-5 CKD and the types of analgesics prescribed. The research team developed a standardized data extraction table using Microsoft Excel. The two reviewers populated the table independently from the selected full text articles. The information collected included study location (geographical area and time), study objectives, study design, population demographics, analgesics used, and numerical data on the prevalence of analgesic use. The two data extraction tables were subsequently compared and cross-checked for accuracy and then merged into a single unified table for data analysis and presentation in the article.
Statistical Methods
All analyses were conducted using Microsoft R Open version 3.4.1 using R package meta.8,9 A meta-analysis was conducted to estimate pooled prevalence of analgesic use. Random-effects models were used to account for variations in study design and sample populations. The results were plotted using forest plots. A double arcsine transformation of the prevalence variables was used in the model to accommodate possible issues with overweighting studies with either very large or very small prevalence measurements. 10
The I2 statistic was computed to measure heterogeneity. 11 The I2 value is the percentage of total observed variation across studies due to real heterogeneity rather than chance; a value of greater than 75% is indicative of high heterogeneity. To assess the possibility of publication bias, a meta-regression testing funnel plot asymmetry was conducted using the Peters’ method.8,9,12 Sensitivity analyses using trim and fill algorithms were performed to determine the magnitude of publication bias. 13 Meta-regressions of various continuous and categorical grouping variables on prevalence were conducted to assess possible sources of heterogeneity. Bubble plots were used to illustrate the regression of transformed prevalence onto continuous covariates, and stratified forest plots were constructed to visualize the effects of grouping categorical covariates on both random-effects estimates and heterogeneity.
Results
The literature review yielded 3055 citations of which 117 were deemed eligible for full text review. Of these, 40 studies were included in the analysis. The flow chart in Figure 1 outlines this process, including reasons for exclusion. Supplemental Table S2 provides a list of excluded studies with reasons for exclusion.

Analgesic prevalence systematic review Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
Details of Included Studies
Details of the 40 included studies14-53 are reported in Table 2 and include data from 963 269 patients from 21 countries. Out of the 40 studies, 17 included a prevalence measure of overall analgesic use,14,16-18,20,23,25,29,33,36,37,43,45,46,48,49,52 33 included a prevalence measure of one or more specific analgesic drug class, defined as opioid, NSAID, adjuvant, and nonopioid,14-24,26-28,30-47,50 and 18 measured the prevalence of one or more specific analgesic drug, including acetaminophen.14-16,19,20,22,30,33,36-40,43,48,50,51,53 Three studies14,38,44 contained repeated measures; the final prevalence data were used in these cases.
Characteristics and Results of Included Studies.

Note. HD = hemodialysis; DOPPS = Dialysis Outcomes and Practice Patterns Study; COX-2 = cyclooxygenase-2; NSAID = nonsteroidal anti-inflammatory drug; CCR = creatinine clearance; FREEDOM = Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease; CKD = chronic kidney disease; GFR = glomerular filtration rate; CKM = conservative kidney management; PD = peritoneal dialysis; ICD = International Statistical Classification of Diseases and Related Health Problems; AKI = acute kidney injury.
Three prevalence groupings were extracted that had sufficient data to complete full meta-analyses: overall analgesic use prevalence (17 studies),14,16-18,20,23,25,29,33,36,37,43,45,46,48,49,52 opioid use prevalence (13 studies),14-16,18,19,21,24,26,31,37,38,40,44 and NSAID use prevalence (19 studies).14,15,19,20,22,26-28,30,32,34,35,37,39-42,47,50
A meta-analysis was also done for acetaminophen use, which included 8 studies,14,15,19,20,33,36,37,43 and adjuvant use (5 studies).19,20,26,36,43 Analyses were also conducted using data from the studies that characterized analgesics as nonopioids (8 studies),16-18,20,23,37,38,46 weak opioids (8 studies),17,20,23,26,36,43,45,46 and strong opioids (6 studies).20,26,33,36,45,46
Figure 2 displays the results from random-effects meta-analyses on the 3 main prevalence groupings. The prevalence of overall analgesic use was 50.8% (38.8%-63.3%). The prevalence for use of opioids and NSAIDs was 23.8% (15.2%-33.7%) and 17.2% (12.6%-22.3%), respectively. In all cases, heterogeneity was extremely high (I2 > 98%). The prevalence rate for opioid use was highly influenced by 2 large US studies.31,38,44 When these were removed, the estimated prevalence decreased to 17.3% (13.0%-22.7%), although the heterogeneity remained high.

Forest plot of random-effects model with pooled estimate and 95% confidence interval on (A) overall analgesic use prevalence, (B) opioid use prevalence, and (C) NSAID use prevalence.
Prevalence categories with number of articles, estimated pooled prevalence, and I2 are shown in Table 3, with a further breakdown by specific analgesic in each analgesic category in Supplemental Table S3. The prevalence for use of acetaminophen and adjuvants was 27.5% (17.6%-38.5%) and 23.4% (16.5%-31.0%), respectively. In studies where analgesics were characterized as either nonopioid, weak opioid, or strong opioid, the prevalence for use was 26.8% (19.2%-35.2%), 17.1% (10.8%-24.5%), and 6.7% (3.2%-11.2%), respectively. Heterogeneity was moderate in reported adjuvant use (I2 = 58.4%), high in strong opioid use (I2 = 74.8%), and extremely high (I2 > 98%) in acetaminophen, nonopioid, and weak opioid use.
Prevalence Categories With Number of Articles, Estimated Pooled Prevalence, and I. 2

Note. CI = confidence interval; NSAID = nonsteroidal anti-inflammatory drug.
Weak opioids are typically codeine or tramadol. All other opioids are considered strong opioids.
Many studies limited analgesic information to analgesic class without specifying the specific drug used. However, in studies where specifics were provided, diclofenac and ibuprofen were the most commonly reported NSAIDs, with 622,30,33,37,39,50 and 522,30,37,39,50 studies giving prevalence information, respectively (Supplemental Table S3). The most commonly reported weak opioid was tramadol16,19,33,37,38,43,48; the most commonly reported strong opioid was oxycodone.16,20,37,38,48 In the United States, however, hydrocodone was the most commonly reported opioid used.
Peters’ regression testing funnel plot asymmetry did not give evidence for publication bias in overall analgesic, opioid, or NSAID prevalence reporting (P = .42, P = .54, and P = .55, respectively). A similar analysis of the additional groupings suggested possible bias leading to over reporting in the literature for acetaminophen (P = .02) and nonopioid (P = .03) use. A trim-and-fill analysis decreased the pooled prevalence estimates to 5.4% (1.1%-12.2%) for acetaminophen use and 15.0% (9.4%-21.8%) for nonopioid use. However, the I2 value was not improved in either case.
Seven covariates were tested in the meta-analyses: sample size, publication year, study region, patient population, whether or not the study used prescription/insurance data vs patient reported analgesic use, and whether or not analgesics were the primary focus of the study. Results of the meta-regression with respect to overall analgesic use returned no evidence for a relationship between use and any of the covariates tested. In testing overall opioid use, the meta-regression found evidence for a relationship between prevalence and both publication year (P < .001) and sample size (P < .001); in both cases, bubble plots show an increase in prevalence reporting (Figure 3).
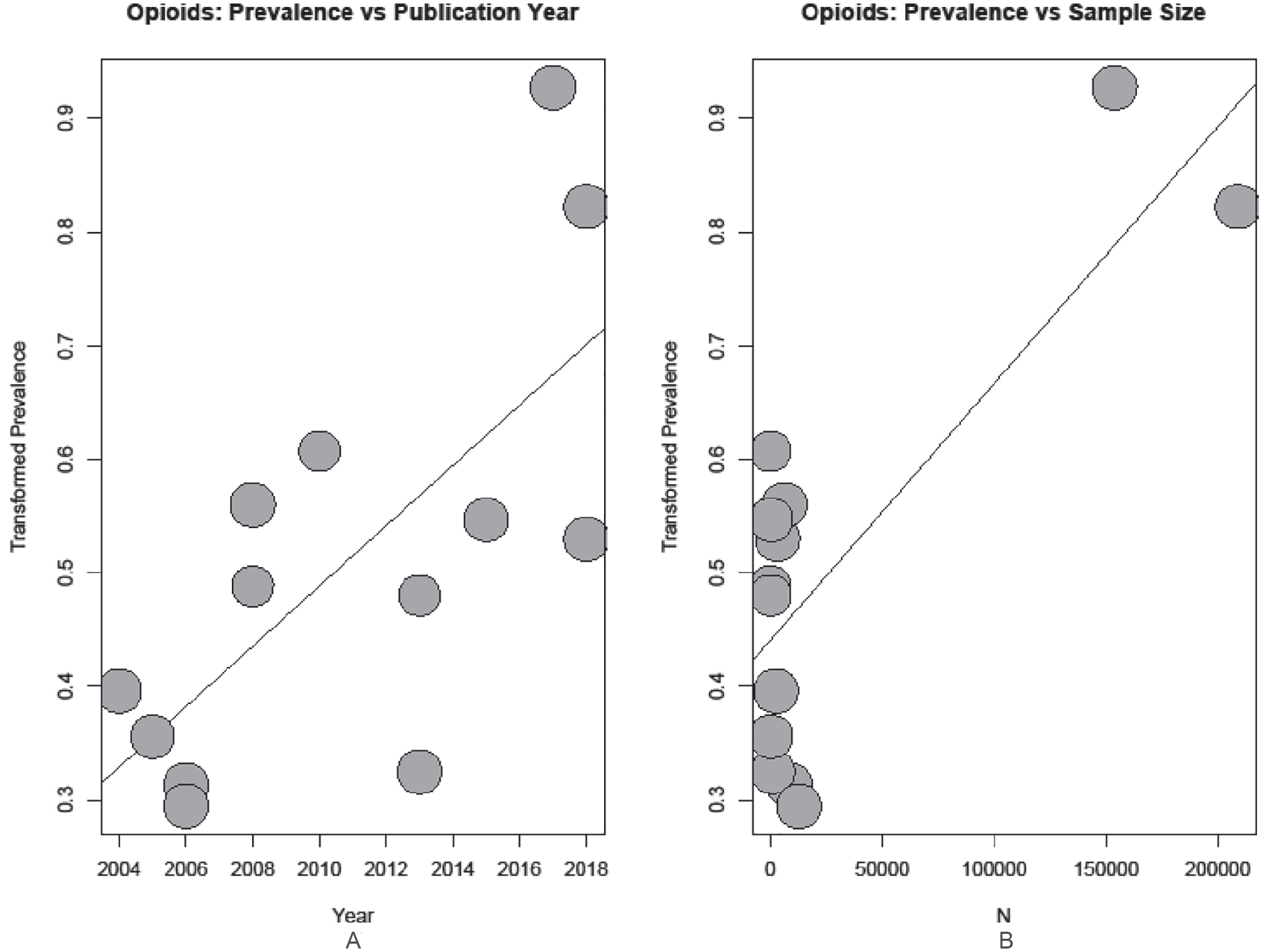
Bubble plot of reported opioid use prevalence by (A) publication year and (B) sample size.
In the case of sample size, this appears to be the result of 2 very large studies heavily influencing the results.31,44 For NSAID use, study year and region were found to be significant (P = .004 and P = .01, respectively); a bubble plot of the study year regression shows an increase in prevalence reporting by year (Figure 4). A stratified analysis of NSAID use prevalence returned 2 large regional groupings which appear to differ in reported prevalence: North America with a pooled prevalence of 10.5% (4.4%-18.8%), and Europe with a pooled prevalence estimate of 29.2% (21.5%-37.5%) (Supplemental Figure S1). Three additional regions were present (Africa, Asia, and 1 multiregion group), with pooled prevalence estimates of 8.1% (4.5%-12.8%), 22.0% (21.7%-22.4%), and 5.3% (4.8%-5.8%), respectively. However, in all cases heterogeneity remained extremely high.

Bubble plot of reported NSAID use prevalence by publication year.
Due to the smaller number of studies available to analyze acetaminophen and adjuvant use, as well as the data characterized as nonopioid, weak opioid, or strong opioid, many covariates left the data too sparse to properly interpret using regression. However, a decrease in heterogeneity was present in some models. Heterogeneity in strong opioid use decreased to I2 = 19.5% when stratified by whether prescription/insurance data were used versus self-report, with prescription data showing higher prevalence use. Stratification by region also decreased heterogeneity in strong opioid use to I2 = 63.0%, and adjuvant use to I2 = 13.7%. The European studies report lower use of strong opioids compared with North American and a single Asian study. The high heterogeneity for adjuvant use was driven by a single Asian study with low prevalence use of 11%. Reported adjuvant use in the 2 European studies of 18% to 34% was similar to the reported use in the 2 North American studies of 19% to 36%.
Discussion
The prevalence of overall analgesic use was 50.8%. Our findings highlight tremendous variability in the prescribing of both nonopioid and opioid analgesics for patients with advanced CKD within and between countries, even with the removal of some highly influential studies. This variability in the context of consistently high pain prevalence across international studies suggests that factors other than patient characteristics account for differences and that there is widespread uncertainty regarding the optimal pharmacologic management of pain in patients with CKD. The prevalence of acetaminophen use was 27.5%, but was extremely low at 5.4% once adjusted for publication bias. NSAID and adjuvant use were 17.2% and 23.4%, respectively. Opioid use was 23.8%, although decreased to 17.3% when 2 highly influential US studies were removed. 31,38,44
Pain is experienced by approximately 60% of patients with advanced CKD whether they are treated with dialysis or managed conservatively2,54 These patients have serious medical illness with complex comorbidities that present numerous potential etiologies for ongoing pain. Pain is a highly complex, multidimensional phenomenon with physical and psychosocial components; a simple, 1-dimensional approach to pain management, especially one that relies exclusively on analgesics, is unlikely to be successful. This is particularly relevant for patients with chronic pain. A multimodal therapy approach that integrates nonpharmacological therapies is considered vital for successfully managing chronic pain. Analgesics, however, play an important role in the management of chronic nonmalignant pain management for some patients. 3 The overall prevalence of analgesic use of 50.8% suggests that pain is being addressed pharmacologically in a large proportion of patients experiencing pain. Unfortunately, existing data for optimizing pharmacological approaches to chronic nonmalignant pain are highly variable. While there is evidence that long-term opioid use may be beneficial for some patients in terms of improving pain control, functional status, and QOL, 55 and there appears to be low incidences of substance abuse and serious adverse effects when analgesic doses are titrated slowly and carefully against pain, 55 the increase in opioid prescribing over the last 10 years has been accompanied by significant risks, including addiction and opioid-related hospitalizations and deaths. Quality clinical trials for pain management in patients with CKD are extremely limited and predate the opioid crisis. 56 Current recommendations for analgesic use in CKD are based on recommendations for the general population considering pharmacologic data in CKD, clinical experience, and best opinion. These recommendations have been reviewed recently elsewhere. 7 The variability in the prevalence of analgesic use and the types of analgesics prescribed likely reflects uncertainty about how best to manage pain in CKD patients given the lack of evidence.
Current recommendations advocate for the judicious use of adjuvant and nonopioid analgesics.3,7 Acetaminophen is the mainstay of treatment for mild to moderate pain in patients with CKD. The range of acetaminophen use from 4% to 6% in 2 large cohorts from the international Dialysis Outcomes and Practice Patterns Study (DOPPS)14,15 to 50% to 66% in 2 small European studies33,36 is difficult to explain outside of differing approaches to pain management. The pooled prevalence of acetaminophen use of 27.5% may represent underuse of acetaminophen. The meta-analysis was potentially subject to publication bias; the adjusted prevalence was extremely low at 5.4% indicating actual rates of acetaminophen use may be substantially lower than our pooled results indicate. Conversely, our findings may underestimate the actual use of acetaminophen as data on over-the-counter medications were limited and it is unclear in many studies whether patients had trialed acetaminophen before proceeding to another analgesic.
The prevalence for NSAIDs use was 17.2%. As with overall analgesic and acetaminophen use, NSAID use in patients with CKD was higher in European studies compared with North America. The prevalence of NSAID use appeared to increase with publication year, particularly after 2010. We speculate this might reflect the desire of care providers to avoid using opioids for pain management. However, NSAIDs are also associated with higher risks for death and hospitalization in patients with advanced CKD. 57 While NSAIDs have a role for specific indications of acute pain, their use in patients with CKD should be limited to the lowest effective dose and shortest duration, especially in the elderly.3,7
Many pains experienced by patients with CKD have a neuropathic component that is poorly responsive to NSAIDs and opioids. For pain that has a neuropathic component, adjuvant therapy such as gabapentin is typically recommended to prevent inappropriate opioid use. Data on adjuvant use were limited but with a reported use of 23.4%. Unfortunately, several studies combined all nonopioids into a single category. Some of these studies specified this to be acetaminophen and NSAIDs, while others gave no further information and theoretically could have included adjuvants. However, use of “nonopioids” appeared similar with a pooled prevalence of 26.8% and like acetaminophen, use may have been subjected to publication bias, with actual rates as low as 15%.
Current guidelines for chronic pain management only recommend opioid therapy when nonpharmacologic therapies and nonopioid analgesics have failed to control pain adequately. Recommendations suggest that opioids should be added to acetaminophen and/or the adjuvant, rather than being prescribed alone. The prevalence of opioid use in this meta-analysis was 23.8%. However, our findings indicate the potential for both publication and sample size biases; prevalence rates were influenced by 2 large studies and our sensitivity analysis indicated that actual prevalence rates might be substantially lower if there are missing studies due to publication bias. When the 2 highly influential US studies were removed,31,38,44 the estimated prevalence decreased to 17.3%. Regardless, variability in opioid use remained high, again suggesting clinical uncertainty around opioid use. While this may not represent an excessively high rate of opioid use, there are exceptions such as the large Kimmel et al 38 study that reported that 64% of 153 758 dialysis patients were prescribed an opioid in 2010 and that 23% of those patients were using opioids long term (defined as receiving an opioid prescription for ≥90 days duration). Again, there was tremendous regional variation with chronic opioid prescription rates ranging 9.5% in Hawaii to 40.6% in West Virginia. This study was one of the few that provided information regarding the specific analgesics prescribed. Opioids were prescribed without the concurrent use of an adjuvant and/or nonopioid in over 51% of patients. In addition, only 1.9% of patients received a prescription for an opioid that is considered safer for use in patients with advanced CKD, raising concerns about inappropriate opioid use.
There are several limitations that need to be considered when interpreting these results. Data available for meta-analyses were limited by inconsistent characterization of analgesics. There were very few data regarding the specific analgesics used within each class and poor characterization of over-the-counter analgesic use including acetaminophen and NSAIDs, potentially explaining some of the heterogeneity in the meta-regression and underestimating their actual use. Studies that reported use of more than 1 specific drug or class of drug often presented results grouped by the various categories without providing a measurement for overall opioid or analgesic use to account for patients on multiple drugs. If research in pain management and analgesic use is to be advanced, greater attention to the characterization of the analgesic regimen is required. In addition, publication bias cannot be ruled out, particularly for nonopioid and acetaminophen use. Furthermore, given the lack of clinical data regarding indications for analgesic use, pain characteristics (severity and nature, eg, neuropathic or nociceptive), dosing or duration of use, efficacy, or safety, we can only speculate regarding appropriate use. This is an area that requires greater clinical focus and research if we are to change clinical practice and improve patient outcomes.
Our results suggest widespread uncertainty regarding the optimal pharmacologic management of pain in patients with CKD. Safe and effective pain management that includes adequate prescribing and oversight of treatment requires a targeted clinical focus. Clinical care would benefit from increased evidence and education with collaboration across specialties such as nephrology, pain medicine, and palliative care.
Supplemental Material
Figure-S1-NSAID-Region-080120 – Supplemental material for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, Figure-S1-NSAID-Region-080120 for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Chelsy George, Syed T. Hussain, Kate Grundy and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
TableS1_Search_Strategy_211019 – Supplemental material for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, TableS1_Search_Strategy_211019 for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Chelsy George, Syed T. Hussain, Kate Grundy and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
Table_S2_Excluded_Studies_170519 – Supplemental material for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, Table_S2_Excluded_Studies_170519 for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Chelsy George, Syed T. Hussain, Kate Grundy and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
Table_S3-Detailed_Prevalence_Revised_191219 – Supplemental material for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, Table_S3-Detailed_Prevalence_Revised_191219 for Analgesic Use in Patients With Advanced Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Chelsy George, Syed T. Hussain, Kate Grundy and Liz Dennett in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
All authors consent to the publication of this manuscript.
Availability of Data and Materials
All data available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
