Abstract
Background:
Posttransplant lymphoproliferative disorder (PTLD) is a major complication following kidney transplantation.
Objective:
We undertook this study to characterize PTLD in kidney transplant patients in British Columbia with regard to incidence, patient and graft survival, histological subtypes, treatment modalities, and management of immunosuppression.
Design:
Retrospective cohort analysis.
Setting:
British Columbia.
Patients:
All adult patients who underwent kidney transplantation in British Columbia between January 1, 1996, and December 31, 2012, were included. Patients less than 18 years of age at the time of first transplant and multiple organ transplant recipients were excluded from analysis.
Measurements:
Patients with lymphoproliferative disorders that occurred subsequent to kidney transplantation were considered to have developed PTLD.
Methods:
Cases of PTLD were identified by cross-referencing data abstracted from the provincial transplant agency’s clinical database with the provincial cancer agency’s lymphoma registry. Patients were followed up for the development of PTLD until December 31, 2012, and for outcomes of death and graft failure until December 31, 2014. Data collection was completed via an electronic chart review.
Results:
Of 2217 kidney transplant recipients, 37 (1.7%) developed PTLD. Nine cases were early-onset PTLD, occurring within 1 year of transplant; of these cases, 6 were known/presumed Epstein-Barr virus mismatch, compared with only 2 of 28 late-onset cases. Patient survival for early-onset PTLD was 100% at 2 years post diagnosis. Late-onset PTLD had survival rates of 71.4% and 67.9% at 1 and 2 years, respectively. PTLD was associated with significantly decreased patient survival (
Limitations:
Limitations to this study include its retrospective nature and the unknown adherence of patients to prescribed immunosuppressant regimens. In addition, cumulative doses of immunosuppression received and the degree of immunosuppression reduction for PTLD management were not effectively captured.
Conclusions:
The incidence of PTLD in British Columbia following kidney transplantation was low and consistent with rates reported in the literature. The incidence of late-onset PTLD and its association with reduced patient and graft survival warrant further analysis of patients’ long-term immunosuppression.
What was known before
Posttransplant lymphoproliferative disorder is a complication following kidney transplantation that can seriously threaten long-term outcomes, including overall patient and graft survival.
What this adds
This research adds to the existing Canadian literature on the topic by presenting updated epidemiological data on posttransplant lymphoproliferative disorder in adult kidney transplant patients in British Columbia.
Introduction
Graft function and patient survival immediately following transplantation have improved dramatically since the early days of kidney transplantation, yet a number of complications still threaten long-term outcomes. Immunosuppression following transplantation is known to increase the risk of malignancy; posttransplant lymphoproliferative disorder (PTLD) is among the more common malignancies diagnosed.1-3 Specifically, lymphoproliferative disorders occur greater than 10 times more frequently in kidney transplant recipients than in the general population.4,5 PTLD is a heterogeneous disorder, ranging from very indolent to very aggressive.3,6 Given this diversity, reported survival rates can be highly variable. 1 However, lymphomas in the aggregate have tended to be aggressive in nature and often poorly responsive to treatment, resulting in 5-year survival rates of less than 40%. 7
Treatment options for PTLD vary based on the disease subtype and may include rituximab monotherapy, rituximab with concurrent or sequential chemoimmunotherapy, radiation, or surgery.8-11 The almost universal initial step is the reduction in immunosuppression with the reported response rates ranging from 25% to 63%. 12 Balancing mortality risk with PTLD and risk of graft rejection in the setting of reduced immunosuppression is a challenge for clinicians.13,14
In British Columbia (BC), the incidence of lymphoproliferative disorders following kidney transplant in a cohort of patients transplanted between 1986 and 1989 was last reported to be 1.26% in the late 1980s. 15 Since then, mycophenolate has largely replaced azathioprine as the antimetabolite of choice and, whenever possible, immunosuppression intensity has been reduced. Rapid steroid elimination protocols following kidney transplant have also since been incorporated into common practice. Whether these factors have any mediating effect on PTLD in this population is not known.
The purpose of this study was to characterize PTLD in kidney transplant patients in BC with regard to incidence, patient and graft survival, histological subtypes, treatment modalities, and management of immunosuppression.
Methods
Study Population and Study Design
This retrospective database study reviewed adult patients who underwent kidney transplantation in BC between 1996 and 2012. The study protocol was approved by the University of British Columbia Clinical Research Ethics Board and the Vancouver Coastal Health Research Institute. The institutional review bodies waived the need for informed consent.
A list of all patients who received a kidney transplant between January 1, 1996, and December 31, 2012, was generated from the clinical database of the provincial transplant agency (BC Transplant). The database includes transplant-related information on all renal transplant patients who have been seen by a transplant nephrologist in BC. This time period was selected to minimize confounding of PTLD incidence and survival by era of immunosuppression, as mycophenolate became the antimetabolite of choice in immunosuppressant protocols in BC starting in 1996. Patients less than 18 years of age at the time of first transplant and multiple organ transplant recipients were excluded from analysis.
To identify patients who developed PTLD, this list was cross-referenced with the British Columbia Cancer Agency (BCCA) registry of all patients with lymphoma diagnoses. The data in this registry are obtained from a population-based provincial arm of the Canadian national cancer registry, which is certified annually by the North American Association of Central Cancer Registries. Lymphoma diagnoses throughout the study period were coded according to World Health Organization (WHO) International Classification of Diseases for Oncology criteria by trained cancer registry abstractors. Data collection was completed via an electronic chart review. Patients were considered to have developed PTLD if the date of first kidney transplant preceded the date of lymphoma diagnosis. Patients were followed up for analysis of incident PTLD until December 31, 2012, and for outcomes of death and graft failure until December 31, 2014.
Statistical Analysis
Continuous variables were analyzed using the
Demographic Data of Kidney Transplant Patients Who Developed PTLD (n = 37) and Those Who Did Not (n = 2180).

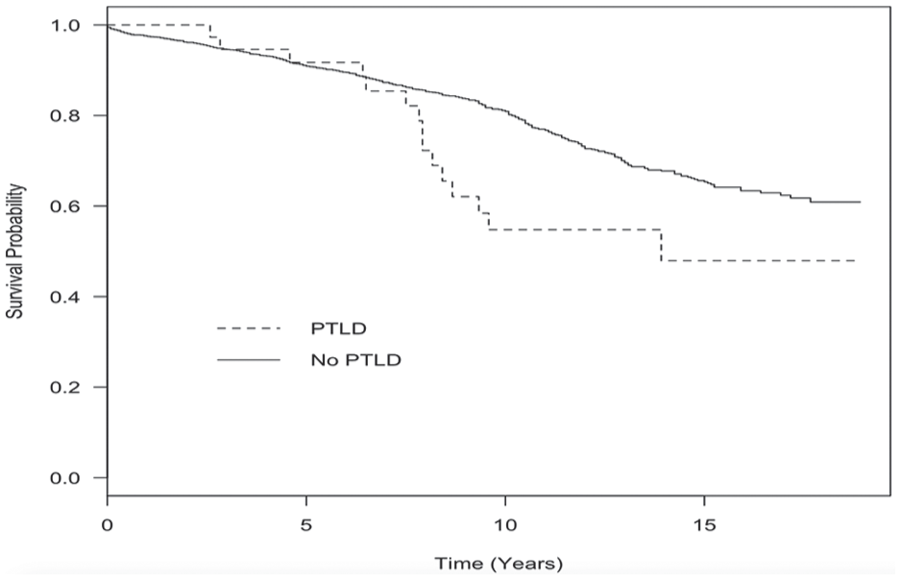
Kaplan-Meier survival analysis comparing overall survival from the time of transplant of patients who received at least 1 kidney transplant between January 1, 1996, and December 31, 2012, who went on to develop PTLD (n = 37) with patients who received a transplant during the same time period but did not develop PTLD (n = 2180).

Kaplan-Meier survival analysis comparing graft survival from the time of transplant, uncensored for death, of patients who received at least 1 kidney transplant between January 1, 1996, and December 31, 2012, who went on to develop PTLD (n = 37) with patients who received a transplant during the same time period but did not develop PTLD (n = 2180).
Results
Of the 2217 adult patients identified by BC Transplant, 45 were identified by BCCA as also having lymphoma diagnoses within this time period. Of these patients, 7 had lymphoma diagnoses that predated their kidney transplants and did not go on to develop PTLD, and 1 patient’s lymphoma diagnosis was an indolent small B-cell lymphoma. Thus, 37 of 2217 patients (1.7%) were found to have had PTLD diagnoses.
Demographic data of the 37 patients with PTLD and the 2180 patients who did not develop PTLD are summarized in Table 1. Characteristics among the PTLD patients were not statistically different from the kidney transplant patients who did not develop PTLD. Time to PTLD diagnosis ranged from 2.9 to 166 months (median 63 months) post transplant; additional characteristics of the patients who developed PTLD are presented in Table 2. Nine cases were early-onset PTLD; all others were considered late-onset cases. Of the early-onset cases, 6 were known or presumed to be EBV mismatched. Conversely, only 2 of 28 late-onset cases were EBV mismatched.
Characteristics of Kidney Transplant Patients With PTLD (n = 37).

The primary sites, treatment modalities, and outcomes of patients with diffuse large B-cell lymphoma (DLBCL), polymorphic PTLD, and multiple myeloma are described in Tables 3, 4, and 5, respectively. The same categories of information are presented for the patients with other types of PTLD in Table 6. For most patients, PTLD management also included a reduction of immunosuppression.
Primary Site, Treatment, and Outcomes of Kidney Transplant Patients With DLBCL PTLD (n = 26).

From PTLD diagnosis (days).
Primary Site, Treatment, and Outcomes of Kidney Transplant Patients With Polymorphic PTLD (n = 5).

From PTLD diagnosis (days).
Treatment and Outcomes of Patients With Multiple Myeloma Following Kidney Transplant (n = 4).

From PTLD diagnosis (days).
Primary site, Treatment, and Outcomes of Kidney Transplant Patients With PTLD, Subtype Other Than Those in Tables 1 to 3 (n = 2).

From PTLD diagnosis (days).
Twenty-six patients developed DLBCL, of which 23 patients were treated with rituximab. Of the patients treated with rituximab, 13 received rituximab monotherapy, and 10 received rituximab in combination with chemotherapy, with the most common chemotherapy regimen being CHOP-R. Among the patients who received rituximab, 16 achieved remission without recurrence (Table 3).
Among all PTLD patients, the 1- and 2-year patient survival rates were 78.4% and 75.7%, respectively. Survival was excellent among early-onset cases, with 100% surviving to 2 years post diagnosis of PTLD. Survival among late-onset PTLD patients was less favorable, with 1- and 2-year survival rates being 71.4% and 67.9%, respectively. Overall survival was significantly worse in patients with PTLD than in kidney transplant patients who did not develop PTLD (Fleming-Harrington,
Discussion
This study is the most recent update on incidence, characteristics, and outcomes of PTLD in BC kidney transplant patients since the 1980s. 15 Changes in immunosuppression protocols that have occurred since this time had the potential to alter the frequency with which PTLD was occurring.
PTLD occurred in 1.7% of patients who received kidney transplants between 1996 and 2012 in BC, which is consistent with the range of 1% to 2% that has been reported in the literature.1,7,16-18 This incidence is not appreciably different from the 6 lymphoma cases Melosky et al reported out of the 478 kidney transplant patients studied between 1986 and 1989. 15 However, Melosky et al followed patients for a mean period of only 26 months, implying a number of late-onset cases were likely not captured, and that the true frequency of PTLD may have been higher than what was reported. Other Canadian literature reports that 2.5% of kidney transplant patients during this period were affected by early-onset PTLD. 19 Considering lymphomas affect kidney transplant patients at significantly higher rates than they do the general population, the frequency of PTLD among kidney transplant patients in BC, while low, remains a concerning cause of morbidity and mortality among this population.
Several studies have identified risk factors for the development of PTLD following solid organ transplantation. Among these include EBV seronegativity of recipient, Caucasian ethnicity, male gender, and age less than 18 years.3,10,20-23 Some of these are additive, with young Caucasian males being among the highest risk of developing PTLD.20,24 Our study appears to corroborate some of this research, as 21.6% of patients with PTLD were EBV negative (documented as either EBV mismatch or, when EBV serology data were unavailable for the donor, presumed EBV mismatch) compared with only 5.7% of patients who did not develop PTLD. Although not statistically significant, there were also a greater proportion of patients who were Caucasian (73.0% vs 65.9%) and male (64.9% vs 61.0%) in the PTLD group as compared with the non-PTLD group. The majority of reported PTLD cases are associated with EBV.25,26 Therefore, the degree of immunosuppression received is an important risk factor for the development of PTLD as it influences the body’s response to EBV.21,22,27-29 In a study of over 145 000 cadaver kidney transplant recipients, OKT3 and ATG exposure were associated with a 3- to 4-fold higher incidence of PTLD, whereas anti-IL-2-receptor antibodies were not associated with an increased incidence of PTLD. 30 Interestingly, a greater proportion of PTLD patients (40.5% vs 28.1% of non-PTLD patients) in this study did not receive induction therapy with ATG, OKT3, basiliximab, or daclizumab within the first 5 days post transplant. Similarly, smaller proportions of PTLD patients received either anti-IL-2-receptor antibodies (51.4% vs 57.8% of non-PTLD patients) or ATG (5.4% vs 12.4% of non-PTLD patients) for induction therapy. As hypothesized, our PTLD population did consist of a greater proportion of patients (2.7% vs 1.8% of non-PTLD patients) who had received OKT3 induction therapy. Maintenance immunosuppression of individual patients was not examined in detail in this study; the evidence implicating individual maintenance immunosuppressant agents is conflicting and appears to suggest that the net effect of immunosuppression regimens post transplantation, not a particular individual agent, is more associated with the risk of PTLD. 31 Duration of immunosuppression therapy, a known risk factor for the development of late-onset PTLD, 10 was also not captured by these data.
Both adult kidney transplant programs in BC have utilized rapid steroid elimination protocols since 2003. It has been reported that steroid maintenance therapy is associated with a decreased risk of developing late-onset PTLD 3 ; however, other sources suggest that steroid-free protocols do not alter the frequency with which PTLD occurs.18,32 The overall 1.7% incidence of PTLD in our kidney transplant patients was consistent with other studies that did not specify the use of rapid steroid elimination protocols.7,16 Given the nature of the study design, our results permit the ability to merely speculate on the effect that rapid steroid elimination may have on the risk of PTLD. In addition, the ability to capture the degree to which, if any, steroids mitigate or propagate the risk of PTLD following kidney transplantation is challenged because of other concomitant changes to immunosuppression regimens that have likely had greater bearing on the risk of PTLD.
PTLD management varies based on the subtype; however, a nearly universal initial step is reduction in immunosuppression.1,10,11,13 The majority of our patients underwent a reduction of immunosuppression upon PTLD diagnosis; however, the degree of reduction was not studied.
In patients who fail to adequately respond to a reduction of immunosuppression, treatment options for patients with monomorphic or systemic polymorphic PTLD may include rituximab with or without chemoimmunotherapy. 11 Rituximab, an anti-CD20 monoclonal antibody not associated with treatment-related mortality, is frequently used as monotherapy after lack of response from a reduction in immunosuppression alone, or in simultaneous or sequential combination with CHOP.9,16,27,33,34 Previously, CHOP and CHOP-R were reported to have a 31% treatment-associated mortality in immunosuppressed patients. 33 However, a more recent B-cell PTLD treatment trial reported that sequential treatment with rituximab followed by CHOP chemotherapy was associated with a much lower (11%) risk of treatment-related mortality. 9
Of the 26 DLBCL patients, 23 were treated with rituximab, either alone or in combination with CHOP or CHOP-like chemotherapy. Supporting the notion that rituximab is a mainstay of therapy for a number of PTLD subtypes, outcomes were favorable for the majority of patients treated with rituximab, as 16 of the 23 patients treated with rituximab (69.6%) achieved remission without recurrence. Recurrence occurred in 4 patients, which may be a consequence of rituximab not altering cellular immune response to EBV, thereby not offering a long-term defense against EBV-positive B-cell proliferation once B-cell recovery following rituximab therapy occurs. 12 The recurrence rate seen following monotherapy with rituximab in BC is lower than that which has been observed in rituximab monotherapy prospective studies. 9
Given the heterogeneity of disorders categorized as PTLD, prognosis can be highly variable. A number of factors have been reported to negatively influence a patient’s prognosis, including late-onset PTLD, advanced age at diagnosis, central nervous system (CNS) involvement, monomorphic PTLD, T-cell PTLD, poor performance status, extranodal disease, and renal insufficiency.35-41 Our data correspond with some of these prognostic factors. Late-onset cases fared worse, with 1- and 2-year survival rates of 71.4% and 67.9% in the late-onset group compared with 100% survival at 2 years in the early-onset group. Late-onset PTLD is more likely to be EBV negative, which has been associated with worse outcomes.10,42 These findings are in contrast to results from studies reporting no difference in survival between patients with early-onset and late-onset PTLD.8,30
The overall 1- and 2-year survival rates of the 37 patients diagnosed with PTLD in BC were 78.4% and 75.7%, respectively. Our overall survival data do not appear worse than what has been reported by other retrospective database studies, with 5-year survival rates among kidney transplant patients with PTLD being between 53% and 64%.39,43 Outcomes of PTLD following kidney transplant in BC appear better in today’s era of immunosuppression than they did in the 1980s, but previous data do not specify specific survival rates. Melosky and colleagues identified 6 patients with PTLD following kidney transplant; there were no survivors among the 5 patients diagnosed with non-Hodgkin lymphoma. 15 The outcome of the sixth patient, diagnosed with Hodgkin’s lymphoma, was not specified.
The ability to accurately capture all cases of PTLD in a population is threatened by a number of factors, most notably the potential loss to follow-up of patients who develop late-onset PTLD and the possible omission of PTLD documentation in clinic records. One of this study’s strengths is that it incorporated data from both a provincial transplant database and a provincial cancer database, increasing the likelihood that all PTLD cases were captured. Limitations to this study inherent to its design include its retrospective nature, the unknown adherence to prescribed immunosuppressants, and the reduced follow-up for patients transplanted in the latter years of the time period studied. The degree of immunosuppression reduction for PTLD management was not effectively captured, nor were cumulative doses of immunosuppression, including immunosuppression used for the treatment of rejection. Furthermore, database standards were not consistent over the time period studied, limiting the quality and quantity of data that could be retrieved.
Conclusion
This study suggests the rate of PTLD in BC following kidney transplantation is low and consistent with rates reported in literature. The incidence of late-onset PTLD and its association with reduced patient and graft survival warrant further analysis of patients’ long-term immunosuppression and ongoing surveillance for the development of PTLD. Further research should focus on the treatment of PTLD, particularly given the associations between late-onset PTLD and reduced patient and graft survival.
Footnotes
Acknowledgements
The authors thank Julia Cahill and Masoud Yousefi for their support with the statistical components of this research. They also thank Yuhua Sun for her invaluable assistance with data collection. In addition, they thank Dr Laurie Sehn for her expertise and contributions to this project’s protocol development.
Ethics Approval and Consent to Participate
The study protocol was approved by the University of British Columbia Clinical Research Ethics Board and the Vancouver Coastal Health Research Institute. Participant informed consent was not required for this study.
Consent for Publication
All authors consent to this manuscript being published.
Availability of Data and Materials
Not available.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
