Abstract
Background:
Glomerulonephritis (GN) consists of a group of kidney diseases that are categorized based on shared histopathological features. The current classifications for GN make it difficult to distinguish the individual variability in presentation, disease progression, and response to treatment. GN is a significant cause of end-stage renal disease (ESRD), and improved therapies are desperately needed because current immunosuppressive therapies sometimes lack efficacy and can lead to significant toxicities. In recent years, the combination of high-throughput genetic approaches and technological advances has identified important regulators contributing to GN.
Objectives:
In this review, we summarize recent findings in podocyte biology and advances in experimental approaches that have opened the possibility of precision medicine in GN treatment. We provide an integrative basic science and clinical overview of new developments in GN research and the discovery of potential candidates for targeted therapies in GN.
Findings:
Advances in podocyte biology have identified many candidates for therapeutic targets and potential biomarkers of glomerular disease. The goal of precision medicine in GN is now being pursued with recent technological improvements in genetics, accessibility of biologic and clinical information with tissue biobanks, high-throughput analysis of large-scale data sets, and new human model systems such as kidney organoids.
Conclusion:
With advances in data collection, technologies, and experimental model systems, we now have vast tools available to pursue precision medicine in GN. We anticipate a growing number of studies integrating data from high-throughput analysis with the development of diagnostic tools and targeted therapies for GN in the near future.
What was known before
Recent discoveries in podocyte biology have made significant advancement in our understanding of the molecular defects for many glomerular diseases.
What this adds
Our review article highlights the ongoing efforts of implementing precision medicine for understanding the biology and treatment of glomerulonephritis.
Introduction
Glomerulonephritis (GN) is characterized by glomerular inflammation and classified based on histopathology and clinical presentation. It encompasses a spectrum of kidney diseases that collectively are the third leading cause of end-stage renal disease (ESRD). 1 The incidence of primary GN varies between 0.2 and 2.5 per 100 000 per year. 2 The risk of progression to ESRD is between 20% and 50%. 3 The pathogenesis of GN is complex with numerous factors that can trigger and contribute to the progression of glomerular injury. These include, but are not limited to, genetic predisposition, autoimmunity, malignancy, infections, and exposure to drugs.1,4,5 GN can be categorized into renal disorders with the presence of proliferative changes (including IgA nephropathy [IgAN], lupus nephritis [LN], ANCA-associated vasculitis [AAV], idiopathic membranoproliferative GN [MPGN], antiglomerular basement membrane GN, and postinfectious GN) or absence of proliferative changes (including minimal change disease [MCD], focal and segmental glomerulosclerosis [FSGS], idiopathic membranous nephropathy [MGN], and steroid-responsive and steroid-resistant nephrotic syndrome). 6 The overarching goals in GN treatment are to improve quality of life for patients, prevent ESRD, and reduce burden to our health care system. However, treatment for glomerular diseases has been hampered by the nonspecific actions of immunosuppressive agents that often have no clear therapeutic target. Furthermore, immunosuppressive treatments for GN are sometimes ineffective and often lead to adverse drug toxicities including increased risk of opportunistic infections and irreversible side effects such as avascular necrosis. Given the abundant causes and heterogeneity of GNs, there is an urgent need for a better understanding of the pathogenesis of GNs at the molecular level, and to develop effective, targeted, and ideally personalized therapy for patients with GN.
In this review, we provide an overview of the (1) biology of podocytes, (2) treatments targeting the podocyte, (3) precision medicine in GN, (4) high-throughput technologies used for GN research, and (5) models used to study GN. Our goal here is to highlight examples of recent studies characterizing the mechanisms regulating glomerular disease, and how these discoveries may improve treatment of GN.
Biology of Podocytes
Glomeruli consist of many cell types, but much interest has been in podocyte biology as it is common to many forms of glomerular disease including MCD, FSGS, and MGN (see reviews7,8). Podocytes are highly specialized, terminally differentiated epithelial cells that are located adjacent to the glomerular capillaries, where they form part of the glomerular filtration barrier. Functionally, podocytes communicate with their environment and transmit cell signals through various intracellular pathways, such as receptor tyrosine kinases, G protein–coupled receptors, nuclear receptors, and integrins (see reviews9-11). Furthermore, as key components of the glomerular filtration barrier, podocytes adhere tightly to the glomerular basement membrane (GBM) to withstand the transcapillary filtration pressure. 12 The podocyte has a complex cellular architecture composed of a parachute-like cell body that attaches to the basement membrane through primary foot processes. Structurally, podocytes have microtubule-based cellular extensions called primary processes, and actin-based membrane extensions called foot processes (Figure 1). Podocyte foot process effacement, a hallmark feature of diseases leading to nephrotic syndrome, involves disruption of the actin cytoskeleton leading to compromise of the slit diaphragm and subsequent proteinuria. 12
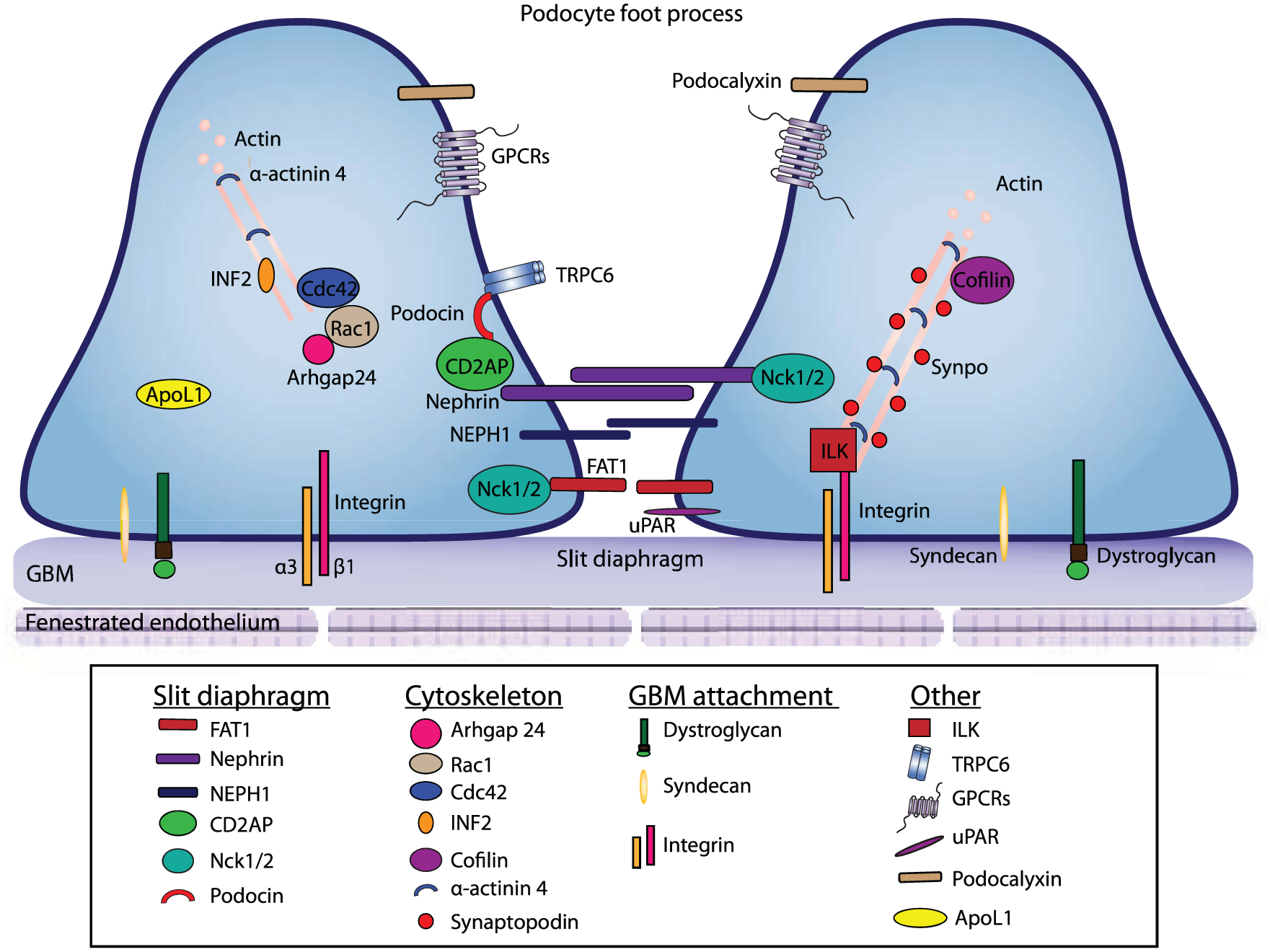
Schematic representation of podocyte foot processes.
Our understanding of podocyte biology has emerged primarily from human genetic studies which have identified several genetic mutations that encode proteins important in podocyte structure and function. Proteins involved in podocyte biology and structure can be broadly categorized into regulators of (1) slit diaphragm, (2) cytoskeleton, or (3) involvement in GBM attachment (Figure 1). Many of these proteins are implicated in podocyte injury and nephrotic syndrome.
Slit diaphragms are the cell-to-cell junctions formed between adjacent podocytes, which are crucial for proper podocyte function. 12 Mutations in 2 key slit diaphragm proteins, nephrin (a transmembrane protein in the slit-diaphragm) and podocin (a cell junction protein), result in actin cytoskeleton dysregulation and nephrotic syndrome.13-15 A recent study by New et al showed that phosphorylation of nephrin is required to stabilize and restore podocyte morphology. 16 CD2AP (CD2-associated protein) is another gene that is mutated in human glomerular disease. 17 CD2AP, a scaffolding protein, NEPH1, a member of the nephrin-like protein family,18,19 and protocadherin FAT1, an adherent junction protein 20,21 all interact with nephrin to form slit diaphragm junctions. 22 Nck1/2 are adaptor proteins that serve to link nephrin to the actin cytoskeleton of the cell.23,24 Genetically, loss of NEPH1 in mice resulted in proteinuria and perinatal lethality, 25 whereas loss of FAT1 resulted in slit junction defects. 21 Loss of Nck1/2 in mouse podocytes resulted in proteinuria and defects in foot processes morphology. 26 Together, podocin, nephrin, and their associated proteins function to connect membrane junction proteins to the actin cytoskeleton of the cell. Mutations to any of these proteins can disrupt slit diaphragm’s function and integrity, resulting in glomerular disease.
Regulators of the podocyte actin cytoskeleton are important in podocyte function; disruption of components of the cytoskeleton can results in podocyte damage. For example, Rho GTPases (Cdc42, RhoA, and Rac1) have roles in maintaining the actin-based cytoskeleton in podocytes. Specifically, experiments suggest that Cdc42 is critical for the maintenance of the glomerular filtration barrier,27,28 whereas the role of RhoA in maintenance of podocyte actin cytoskeleton is less clearly defined but may involve the stabilization of RhoA by the binding of synaptopodin, a proline-rich actin-associated protein that is involved in stress-fiber formation in podocytes.27,29 Abnormal activation of Rac1 has been shown to lead to proteinuria with foot process effacement. 30 In addition, hyperactivation of Rac1 by 3 mutant forms of Rho-GDP dissociation inhibitor-α was also shown to impair actin polymerization and slowed motility in podocytes. 31 The balance between RhoA and Rac1 signaling might be regulated in podocytes by Rho-GAP24 or Arhgap24. 32 Studies in the area of Rho-GTPase are still on-going, the relationship between RhoA, Rac1 and cytoskeletal integrity might be more dynamic than previously thought. For an in-depth review on Rho GTPase see Mouawad et al. 33 In addition, actin associated proteins are known to contribute to the development of FSGS. INF2, a formin family of actin-regulating protein plays a role in actin polymerization, and human mutations in INF2 are found in patients with FSGS. 34 Similarly, mutations in α-actinin 4, an actin-filament cross-linking protein, have also been identified in patients with FSGS. 35 The mouse model of α-actinin 4 recapitulates FSGS defects. 36 Synaptopodin is another actin-associated protein that is known to interact with α-actinin 4, and functions to regulate RhoA signalling. 27 Mice without synaptopodin have normal podocytes but exhibit slow recovery from chemically induced podocyte injury. 29 Other genes that likely have a regulatory role in the actin cytoskeleton are cofilin and anilin. Genetic deletion of known actin binding protein cofilin (actin-depolymerizing factor) resulted in proteinuria in mice, and mutations in anilin, an F-actin binding protein, were present in patients with FSGS. These studies illustrate the importance of cytoskeletal proteins in podocyte functions.37,38 Working together, these proteins function to maintain actin-cytoskeleton structural integrity of podocyte foot processes, which is crucial to podocyte function.
Podocyte attachment to the GBM is crucial for its function. Podocyte utilizes various anchor proteins to attach to the GBM. Podocytes express several integrin receptors, including the major cell-matrix adhesion receptor, integrin α3β1. In mice, loss of α3β1 weakens the podocyte-GBM interaction, and causes poor organization of laminin 511/521 into a functional GBM.39-42 Other cell-matrix adhesion receptors expressed by podocytes include the integrins α2β1 and αvβ3, and type XVII collagen.12,43 In addition, podocytes express α-dystroglycan, a transmembrane adhesion complex, that serves to attach podocytes to GBM. Researchers have found that dystroglycan levels are lowered in Minimal Change Disease. 44 Syndecan4, a heparin sulfate proteoglycan that functions in matrix adhesion through its interaction with extracellular matrix, is also involved in attachment of podocytes to GBM. In mice without Ndst1, the enzyme responsible for N-sulfation of heparan sulfate chains, there is a failure to assemble heparan sulfate glycosaminoglycan chains, resulting in disrupted localization of Syndecan4, and foot process effacement.45,46 These studies highlight the importance of proper podocyte attachment to GBM in GN.
Last, other podocyte genes implicated in nephrotic syndrome have been identified; these include ApoL1, 47 ILK, 48 TRPC6, 49 podocalyxin, 41 GPCRs, 50 and suPAR51,52 (Figure 1). Ongoing studies are dissecting the functions of these proteins. With improved understanding of their molecular mechanisms, we may be able to identify new therapeutic targets for precision medicine as well for biomarker discovery.
Treatments Targeting Podocytes
Early podocyte injury may be reversible if the actin cytoskeleton can be repaired. However, sustained glomerular injury can result in permanent podocyte damage leading to podocyte hypertrophy, cell death, and eventual kidney failure. 53 Podocytopathies including membranous nephropathy, FSGS, and MCD have had variable responses to immunosuppressive therapy. Interestingly, the beneficial actions of immunosuppressive agents may be mediated by mechanisms not directly related to immunosuppression. Some mechanisms of podocyte injury include changes to the slit diaphragm structure and/or function, dysfunction of the actin cytoskeleton, podocyte interaction with the GBM, alterations in the transcriptional regulation of podocytes, calcium homeostasis dysregulation, upregulation of the innate immune system, and upregulation of cathepsin L–mediated proteolysis. 54 Given that podocyte injury leads to a number of GN, podocytes are attractive therapeutic targets for GN. For example, activation of integrins αvβ3 by the urokinase receptor (uPAR) causes foot process effacement, proteinuria, and FSGS in mice and humans. By blocking αvβ3 with an anti-β3 antibody or the small-molecule inhibitor cilengitide, it is possible to reduce proteinuria induced by uPAR.51,52 Recently, Hayek et al showed that suPAR-activated αvβ3 integrin can bind to ApoL1 risk variants with higher affinity than wild-type ApoL1 causing more proteinuria in mice. 55 Future studies will be required to validate and further expand on these results to determine whether these identified targets are able to be therapeutic targets in human disease. Characterizations of the molecular mechanisms regulating podocyte actin cytoskeleton stabilization have provided insight to design more targeted therapies. Faul et al demonstrated that the antiproteinuric effect of cyclosporine A may not be fully explained by its immunosuppressive activity for the inhibition of nuclear factor of activated T cells (NFAT) signaling in T cells. Instead, cyclosporine appears to stabilize the actin cytoskeleton in podocytes by blocking the calcineurin-mediated dephosphorylation of synaptopodin. 56 Similarly, rituximab may stabilize the actin cytoskeleton and prevent podocyte apoptosis independent of its well characterized activity as a monoclonal antibody for CD20 on B lymphocytes. Fornoni et al found that rituximab treatment was associated with a lower incidence of posttransplant proteinuria and better preservation of glomerular filtration rate. Interestingly, when podocytes were induced by sera from patients with recurrent FSGS, rituximab or overexpression of sphingomyelin phosphodiesterase acid-like 3b (SMPDL-3b) was able to prevent disruption of the actin cytoskeleton and apoptosis of podocytes. 57 The researchers concluded that rituximab may be able to be used in high-risk patients undergoing kidney transplant to prevent FSGS.
Clinically, rituximab has been shown to have good efficacy for MGN, idiopathic nephrotic syndrome, MCD, and FSGS.58-61 Recently, Yu et al showed that abatacept (cytotoxic T lymphocyte associated antigen 4 immunoglobulin fusion protein), a costimulatory inhibitor that targets B7-1/CD80, reduced proteinuria in patients with FSGS. 62 These findings will need to be replicated in larger cohorts but suggest that therapies targeting podocytes may be feasible in the future with a better understanding of the molecular disruptions that lead to podocyte injury.
Precision Medicine in GN
The histopathological classification of GN has helped to categorize different GN with some features helpful for the diagnosis and prognostication of disease. For example, histopathological classification of ANCA 63 and IgAN (Oxford Classification 64 ) has shown prognostic value in clinical settings, and may have potential for better predicting therapeutic outcomes. Nevertheless, our current histopathological classifications cannot accurately determine which immunosuppressive agents can best treat certain GN, making it difficult to select the appropriate type and duration of treatment. For decades, the main treatments for GN have been adopted from other fields of medicine and have remained largely unchanged. Technological advancements have improved the understanding of molecular mechanisms underlying glomerular disease, and provided opportunities to develop targeted therapies. It is possible that patients are genetically predisposed to have a better response to certain therapies. Some questions that have not been answered include “Can the dose and/or duration of immunosuppressive medications be determined prior to administration of immunosuppressive agents? “Is it possible to determine the best immunosuppressive agent for a particular patient?”
Precision medicine, combining modern technologies with molecular and genetic information to identify mechanisms of diseases for targeted treatment and disease prevention, may be coming of age in nephrology. 65 Precision medicine for GN research will likely be necessary due the heterogeneity of glomerular diseases. The goal for precision medicine is now being considered by many groups who have set up biobanks for the purpose of identifying biomarkers and pathways for potential therapeutic interventions. Several large tissue banks which have been established include the European Renal cDNA Bank (ERCB), the Nephrotic Syndrome Study Network (NEPTUNE), and the Clinical Phenotyping and Resource Biobank Core (C-PROBE). 65 In Canada, the Biobank for the Molecular Classification of Kidney Disease in Calgary, Alberta, has amassed thousands of patient samples. 66 In the Muruve lab, we demonstrated that correlating clinical outcome with RNA expression profiles from kidney biopsies obtained from patient affected with GN is a promising approach to identify biomarkers.67,68 We anticipate that the biobanked samples will serve as an immense repository for research projects that can target specific conditions, and facilitate the recruitment of patient and samples that will be large enough to produce statistically meaningful data. It will be critical to have tissue banks with identical protocols to ensure reproducible handling, storage and molecular analysis of tissue (biopsy samples), and biological fluids (blood, urine, and saliva). Just as important, the tissue bank will need to be combined with large, well-defined patient databases using software tools for analyzing phenotypic information of patients (eg, PhenoTips 69 ). The ability to test and validate the predictability of novel biomarkers highlights the importance of long-term longitudinal follow-up as a component of tissue banks.
Precision medicine is in its infancy in utilizing molecular and genetic information obtained from genome-wide association studies (GWAS), exome sequencing, and whole-genome sequencing with next-generation sequencing (NGS) and VAAST (Variant Annotation, Analysis, and Search Tool). 70 The ultimate goal for precision medicine in GN research will be to use small quantities of biological fluid for large-scale analytic tools. The availability of clinical data to correlate findings with experimental results from large data sets is permitting the analysis of specific individuals and paving the way for precision medicine. One excellent example for precision medicine in GN diagnosis and treatment is the discovery of PLA2R (phospholipase A2 receptor) as the antigen involved in the pathogenesis of membranous nephropathy. Salant’s group identified PLA2R as the autoantigen in human membranous nephropathy.71-73 Since its discovery in 2009, PLA2R is being used as a biomarker for membranous nephropathy. Currently, both immunofluorescence and ELISA (eg, EUROIMMUN Anti-PLA2R ELISA (IgG)) are available for testing the presence of PLA2R in clinics. For example, in Calgary, Alberta, we have used Mitogen Advanced Diagnostics Laboratory to analyze patient blood samples for the presence of anti-PLA2R (http://mitogen.ca/requisition-form/). Anti-PLA2R is helpful to categorize MGN as primary or secondary as autoantibodies are present in approximately 70% of patients with primary MGN. 73 Furthermore, there is evidence that anti-PLA2R antibodies may be used to monitor the activity of membranous nephropathy. 74 Future studies to characterize the mechanisms by which PLA2R leads to MGN will allow the development of more specific therapeutic approaches, including antibody inhibition therapy and immunoadsorption of circulating autoantibodies. 75 Other promising biomarkers such as circulating cell-free DNA (cfDNA) has been reported to be elevated in SLE. 76 cfDNAs are promising biomarkers for the diagnosis and classifications of GNs. In addition, microRNAs are known to play multiple roles in kidney development, physiology, and pathology (see review 77 ). A major goal for precision medicine will be to develop GN biomarker panels that readily available in clinics and hospitals to accurately diagnose and guide treatment for GN.
High-Throughput Technologies in GN Research
Since the completion of the human genome project on April 14, 2003, genome-based research has led to new insights into disease mechanisms and potential targeted treatments. New approaches to better understand GN involve large-scale research by genomics (studying the genome sequencing and analysis), transcriptomics (the study of RNA transcripts that are produced by the genome), proteomics (the large-scale study of proteins), and metabolomics (the study of chemical processes involving small metabolites). For an excellent overview of definitions, limitations, and integrative approaches in glomerular disease, please refer to Mariani et al’s recent review. 78 In the next section, we will provide specific examples of recent studies that have utilized genomics, transcriptomics, proteomics, and metabolomics to study GN.
Genomics
Glomerular diseases have been studied using GWAS to compute hundreds of thousands of single-nucleotide polymorphisms (SNPs) in a genome to identify genetic associations in GN. Using GWAS, many genetic associations for MGN, 79 IgAN,80-83 LN, 84 and FSGS 85 have been established. One example of how GWAS was used to identify causal variants for nondiabetic ESRD and FSGS in African American was demonstrated by the Pollak lab for the discovery of APOL1. 47 Initially, admixture mapping studies identified MYH9 as the gene associated with FSGS, HIV-associated nephropathy, nondiabetic ESRD, and hypertensive associated ESRD.86,87 However, fine mapping and resequencing did not identify any causal variants for MYH9.88,89 Instead, using conditional GWAS, the major source of genetic risk for nondiabetic ESRD and FSGS in African Americans was localized to APOL1. 47
Recently, Hildebrandt’s group applied a powerful approach using microfluidic multiplex PCR and NGS to detect a single-gene cause in about 30% of families with steroid-resistant nephrotic syndrome. 90 Using whole-exome sequencing and high-throughput exon sequencing, they were able to identify mutations in 3 nuclear pore genes that cause steroid-resistant nephrotic syndrome. 91 The work by Hildebrandt’s group highlights the rapid changes in genomic tools available for sequencing patient samples and that the field is moving toward a more personalized approach to understand disease phenotype at the genetics level. Current genetic approaches have been instrumental in identifying risk alleles, genetic variants, or genetic modifiers of genes associated with GN. It is foreseeable in the near future that more researchers will use NGS and whole-genome sequencing to study GN as the costs of these approaches continue to decrease.
Transcriptomics
Transcriptomics studies can use a single cell or tissue to generate high-throughput RNA expression profiles. 92 Transcriptomics, a widely used high-throughput technology, 93 is being used to study glomerular diseases including IgAN,94,95 FSGS, 96 and lupus nephritis.97-99 Recently, Banchereau et al performed personalized immunomonitoring using the blood transcriptome to identify molecular networks that enable stratification of lupus nephritis patients into 7 groups. 99 Transcriptomic analysis performed using glomeruli dissected from patient biopsy samples have also been used to validate previous experimental data from mouse model of lupus nephritis.100,101 In the study by Hodgin et al, researchers used traditional microarray to analyze biopsies from patients with subtypes of FSGS. They found that genes involved in slit diaphragm function were differentially upregulated, and further showed different molecular signature involved in different glomerular injuries. 96 Similarly, Peterson et al performed cDNA microarray analysis on biopsies from patients with lupus nephritis. From the transcriptome profiling, 4 clusters of genes were identified that represent lupus nephritis molecular signatures. 97 For ANCA-associated vasculitis (AAV), Brix et al used microarray analyses of renal biopsy samples from patients with AAV crescentic GN. Interestingly, the CC chemokine ligand 18 was found to be upregulated in patients with AAV crescentic GN and proposed to be biomarker for disease activity and relapse. 102 Overall, these studies illustrate the strength of using transcriptomics to characterize molecular defects in GN. It is now possible to use small samples from kidney biopsies to perform transcriptomic analysis as Drop-seq has the capability to profiling thousands of individual cells by separating them into nanoliter sized aqueous droplets. 92 One can envision that kidney biopsies obtained from biobanks will be used to study every type of GN in the future.
Proteomics
Proteomics has been the most extensively researched “omics” in GN. Many researchers use proteomic tools to identify biomarkers for the diagnosis of glomerular diseases without the need for an invasive tissue biopsy. Numerous studies have implemented different protocols for mass spectrometric analysis such as capillary electrophoresis, liquid chromatography (LC-MS), and matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) using various biological fluids. 103 Using urine, serum, or tissue, many groups have employed proteomic techniques for biomarker discovery in GNs including ANCA-associated vasculitis, IgAN, MN, MCD, and FSGS which has been recently reviewed by L’Imperio et al. 103 The latest technologies in proteomics that have been applied to GN research are MALDI-MSI, laser capture microdissection (LCM), and SOMAscan. MALDI-MSI enables the analysis of protein expression data with tissue molecular images in formalin-fixed paraffin-embedded tissue which allows identification of tissue morphology. 104 LCM allows the selective isolation of glomeruli from kidney tissue to avoid contamination by protein or genetic material from interstitium or tubules. 105 LCM of kidney tissue has been successfully used for MPGN, amyloidosis, cryoglobulinemic GN, fibrillary GN, and immunotactoid GN.106,107 Jain et al used LCM and MS on kidney biopsy of a 58-year-old asymptomatic woman with MPGN suspected secondary to monoclonal gammopathy but an unclear diagnosis as there was a mismatch between the patient’s serum monoclonal protein (IgG kappa) and immunofluorescence staining pattern (nonspecific IgM, absence of light chain restriction). By using LCM and MS performed on the kidney biopsy tissue, deposits of monoclonal IgG kappa were detected which helped to make the diagnosis of monoclonal gammopathy-associated MPGN. Sethi et al used LCM followed by MS to compare and distinguish the constituents of the deposits from patients with amyloidosis, fibrillary GN, and immunotactoid GN to cryoglobulinemic GN. Remarkably, by using advanced proteomic methods, these researchers were able to identify different protein signatures for each disease. For example, serum amyloid P and apolipoprotein E were present in large spectra numbers in amyloidosis but not in cryoglobulinemic GN, and Ig gamma-1 chain C region in immunotactoid glomerulopathy compared with fibrillary and cryoglobulinemic GN. 107 LCM combined with different MS protocols will serve as an important platform to identify molecular differences between forms of GN and help make diagnoses for challenging cases.
SOMAscan is a new technology by SomaLogic that uses a novel targeted proteomic technique based on aptamer technology, which can currently measure 1310 protein analytes from blood. 108 This new technology allows the detection of hundreds of molecules from small quantities of substrate. In 2010, Gold et al applied SOMAscan as proteomics biomarker discovery technology to a clinical study of chronic kidney disease (CKD) and identified 2 known CKD biomarkers (cystatin C and β2-microglobulin) and an additional 58 potential CKD biomarkers. 109 These potential CKD biomarkers have yet to be validated, but SOMAscan appears to be a promising proteomic technique that could be applied to biomarker identification in glomerular disease.
Metabolomics
A metabolomics approach to study GN has gained scientific interest with recent studies showing promising results, particularly in diagnosis. The use of metabolite composition in the urine as a diagnostic tool is an attractive noninvasive alternative to renal biopsy. For example, solid-phase microextraction-chromatography-mass spectrometry (SPME-GC-MS) was used by Wang et al to study urinary volatile organic compounds, as a marker for pathological changes in the kidney. 110 Five urinary metabolites (tartronic acid, carbamic acid, sulfide, allyl methyl, hydrogen azide, and benzeneethanamine,N-[(pentafluorophenyl)methylene]-.beta.,4-bis[(trimethylsilyl)oxy]-), were identified and significantly increased in IgAN compared with MPGN patients. These results suggest urinary metabolites may be used as biomarkers to differentiate between different forms of GN. With the generation of large “omic” datasets, an integrative approach (data sharing and team work) will be the key to fully utilize this information not only for precision medicine in GN but for understanding molecular signatures of diseases as well. 78
Models in GN Research
Experimental Animal Models
Animal models, primarily rat and mouse models, have been extensively used to improve our understanding of GN. Detailed information on animal models in kidney disease and GN can be found in other reviews.111,112 Some advantages of rodent models include their small size making it economical to house animals and the ability to generate genetically defined strains making it possible to design spontaneous disease models. We have included a table for examples of available animal models that have been used for most types of GN with some of their strengths and limitations (Table 1).
Animal Models of GN.

Note. GN = Glomerulonephritis; MCD = minimal change disease; PAN = puromycin aminonucleoside; FSGS = focal segmental glomerulosclerosis; MGN = membranous glomerulopathy; IgAN = IgA nephropathy; LN = lupus Nephritis; SLE = systematic lupus erythematosus; MPO = myeloperoxidase; AAV = ANCA-associated vasculitis; GBM = glomerular basement membrane; CrGN = crescentic GN; MsPGN = mesangial proliferative GN.
The investigations using animal models are well illustrated by animal models for Heymann nephritis, ddY mouse, and the anti-GBM mouse model for MGN, IgAN, and anti-GBM respectively. The Heymann nephritis model has improved our understanding of membranous nephropathy by immunizing rats with tubular brush border antigens, but the target antigen in the human glomerulus is different from megalin-involved Heymann nephritis. 116 Megalin is not expressed in human podocytes and therefore not pathogenic in humans. Instead, PLA2R appears to be a major target but is not expressed in rodents or rabbits. 111 Transgenic animal models can circumvent the problem of genes with the caveat of potentially altering the genome. Next, there have been many models used to replicate aspects of IgAN, but each model only partially models the disease. For example, the ddY mouse model has increased circulating IgA and IgA mesangial deposits but only minimal proteinuria and no hematuria. 120 In comparison, the Thy.1 nephritis model which requires an injection of an antibody to Thy.1 (antigen found on mesangial cells) can result in mesangiolysis and mesangial cell proliferation but does not lead to IgA deposits. Last, the many animal models for anti-GBM have established the importance of anti-GBM in the pathophysiology of disease providing the rationale for using immunosuppression and plasma exchange, but limited information has been obtained with respect to the mechanisms of injury following induction with limited efficacy of therapies introduced after induction.
Differences between species limit our ability to extrapolate findings in animal models to physiological and disease conditions in the human body. Many genes and proteins that are expressed and regulated are often very different between species even when comparing mice with rats or humans with other primates. For example, results from rat models often cannot be replicated in mice. 111 Also, mouse models are particularly difficult to use for experiments modeling GN as many mouse strains are resistant to glomerulosclerosis and immune-mediated mechanisms. 111 Furthermore, mice often have alternate genotypes with additional copies of genes (eg, renin) or the absence of genes (eg, APOL1).111,128 Bearing this in mind, we and other have resorted to modeling glomerular disease using human inducible pluripotent stem cells (iPSC). Human stem cells have the advantage of having the appropriate genes and regulatory proteins animal models may lack increasing the probability of determining mechanisms regulating human biological processes and disease.
Kidney Organoids
Gene editing in human stem cells and kidney organoids (“mini-kidneys”) are invaluable tools to further develop the field of precision medicine in GN. With limited samples to process from precious kidney biopsy samples, one may envision recreating a model from individual patients using stem cells to better study disease and to serve as a platform for drug testing. Several groups have how established protocols to generate nephron progenitor cells and kidney organoids from human induced pluripotent stem cells.129-131 Recently, gene editing by the clustered regularly interspaced short palindromic repeats (CRISPR)-Cas9 system has demonstrated that podocyte genes can be knocked out in nephron progenitor cells (NPC) 131 and kidney organoids. 132 For proof of concept, Li et al used the CRISPR-Cas9 to knockout nephrin (NPHS1) in cultured NPC and were able to successfully deliver gene targeting constructs to efficiently create NPHS1 knockout nephron organoids. 131 Freedman et al demonstrated that CRISPR-Cas9 knockout of PKD1/PKD2 and podocalyxin in human stem cells and kidney organoids may be a useful approach to model polycystic kidney disease and glomerular disease, respectively. Impressively, knockout of the polycystic kidney disease genes PKD1 or PKD2 induced cyst-like structures from kidney tubules, 132 while podocalyxin knockout using CRISPR-Cas9 in stem cells and derived podocytes resulted in defective microvilli assembly and failed junctional migration. 133 Together, these studies demonstrate the ability to effectively delete genes in kidney organoids, which can result in the expected features of glomerular and tubular kidney diseases. It is foreseeable in the future that nephron progenitor cells and kidney organoids will be further enhanced to generate knockout and knockin disease models of GN using CRISPR-Cas9. Such human models will be promising platforms for disease-modeling, diagnostics and drug screening.
Future Outlook
The current classification of GN based on histopathology reflects our limited understanding of the basic molecular mechanisms that lead to disease. Not surprisingly, current treatments for GN remain nonspecific with approaches relying almost exclusively on immunosuppression with limited knowledge of the molecular targets. Immunosuppression remains the mainstay treatment for GN which has been effective for immune-mediated diseases but less effective for monogenic diseases and often leads to complications related to medication toxicities. Nonetheless, recent studies using genetic and cell biology have identified molecular defects and regulators of podocyte injury in GN, opening the possibility of developing specific or targeted therapies. In addition, new technologies in genome sequencing combined with large scale proteomics and sequencing databases will lead to the discovery of new genes to contribute to our understanding of podocyte function. We anticipate that in the near future, there will be validated biomarkers to useful to distinguish between different causes of GN. The advances in biotechnology are rapidly paving the way for translation research and personalized medicine which will certainly contribute to the development of more effective and safer therapies. With precision medicine for GN treatment on the horizon, we must be mindful of the challenges in bridging basic research to clinical practices, referred to as “Death Valleys” by the Canadian Institutes for Health Research (http://www.cihr-irsc.gc.ca/e/44000.html). Death Valley 1 is the challenges in translation of basic research, basic podocyte biology, to clinic in the form of new therapy for patients (Figure 2). Valley 2 refers to the difficulty in implementing changes in health care decision making, hence utilizing the newly developed therapeutics for GN patients.134,135 It is likely that the cost associated with new tests and technologies will be a major barrier for implementing new diagnostic tests and treatments. Both Valleys need to be overcome to truly improve patient care and quality of life.

Overview of the patient-oriented, precision medicine research scheme.
Footnotes
Acknowledgements
The authors thank Drs K. Burns and A. Levin for critical reading of the manuscript.
Ethics Approval and Consent to Participate
No ethics approval or consent to participate was required for this publication.
Consent for Publication
All authors have read and approved the final version of this manuscript.
Availability of Data and Materials
No primary data is presented in this publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Y.C.L is supported by the KRESCENT postdoctoral fellowship. J.C. is supported by an Alberta Innovates Health Solutions Clinician Fellowship and is a KRESCENT postdoctoral fellow.
