Abstract
Background:
Patients with chronic kidney disease may not be receiving recommended primary renal care.
Objective:
To use recently established primary care quality indicators for chronic kidney disease to determine the proportion of patients receiving recommended renal care.
Design:
Retrospective cohort study using administrative data with linked laboratory information.
Setting:
The study was conducted in Ontario, Canada, from 2006 to 2012.
Patients:
Patients over 40 years with chronic kidney disease or abnormal kidney function in primary care were included.
Measurements:
In total, 11 quality indicators were assessed for chronic kidney disease identified through a Delphi panel in areas of screening, monitoring, drug prescribing, and laboratory monitoring after initiating an angiotensin converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB).
Methods:
We calculated the proportion and cumulative incidence at the end of follow-up of patients meeting each indicator and stratified results by age, sex, cohort entry, and chronic kidney disease stage.
Results:
Less than half of patients received follow-up tests after an initial abnormal kidney function result. Most patients with chronic kidney disease received regular monitoring of serum creatinine (91%), but urine albumin-to-creatinine monitoring was lower (70%). A total of 84% of patients age 66 and older did not receive a non-steroidal anti-inflammatory drug prescription of at least 2-week duration. Three quarters of patients age 66 and older were on an ACE inhibitor or ARB, and 96% did not receive an ACE inhibitor and ARB concurrently. Among patients 66 to 80 years of age with chronic kidney disease, 65% were on a statin. One quarter of patients age 66 and older who initiated an ACE inhibitor or ARB had their serum creatinine and potassium monitored within 7 to 30 days.
Limitations:
This study was limited to people in Ontario with linked laboratory information.
Conclusions:
There was generally strong performance across many of the quality of care indicators. Areas where more attention may be needed are laboratory testing to confirm initial abnormal kidney function test results and monitoring serum creatinine and potassium after initiating a new ACE inhibitor or ARB.
What was known before
Primary care providers are not necessarily targeted by guidelines for chronic kidney disease, and therefore may not be aware of care recommendations for patients with chronic kidney disease. This may have resulted in care gaps for patients with chronic kidney disease in Ontario.
What this adds
Ontario patients with chronic kidney disease in the primary care setting are generally receiving appropriate care. Areas for improvement include recognition of chronic kidney disease, and consistent serum creatinine and potassium monitoring after initiating an angiotensin converting enzyme inhibitor or angiotensin receptor blocker.
Background
Currently, 2.9 million Canadians are living with chronic kidney disease. 1 Chronic kidney disease can progress to end-stage kidney disease (approximately 42 000 Canadians in 2013), 2 which has a worse prognosis than most cancers. 3 Early detection and prevention of kidney disease progression is a clinical and research priority in many jurisdictions worldwide, including the province of Ontario, Canada. 4
Most patients with early stage chronic kidney disease (ie, stages 1-3b) are managed in the primary care setting and are referred to nephrologists if they have advanced disease or are at increased risk of progression. National and international guidelines recommend that patients with abnormal kidney function markers (estimated glomerular filtration rate [eGFR] <60 mL/min/1.73 m2 or urine albumin-to-creatinine ratios [ACR] >3 mg/mmol) receive follow-up tests within 3 months to establish the diagnosis.5,6 Guidelines also recommend that patients with chronic kidney disease should receive ongoing kidney function monitoring, achieve optimal blood pressure control, and reduce cardiovascular risk factors.5,6 Primary care providers can meet these care indicators by prescribing statins7-9 and blood pressure–lowering medications including angiotensin converting enzyme (ACE) inhibitors/angiotensin receptor blockers (ARBs) 10 (but avoiding co-prescription of ACE inhibitors and ARBs),10-12 performing serum creatinine and potassium tests shortly after initiating an ACE inhibitor or ARB, not prescribing non-steroidal anti-inflammatory drugs (NSAIDs) for prolonged periods of time or with high doses,13,14 and discussing lifestyle modifications, such as healthy eating, regular physical activity, and smoking cessation. 8 Unfortunately, many primary care providers do not always recognize patients with chronic kidney disease,15,16 or they are unaware of guidelines for chronic kidney disease care, as primary care providers are not necessarily targeted by guidelines.15,17,18 This means that these recommendations are frequently not followed in routine practice.19-21
Care gaps for patients with chronic kidney disease in the primary care setting exist across different international settings (see Supplementary Material 1). Two previous studies have assessed the quality of care for patients with chronic kidney disease in Ontario. One study focused on a group of patients at risk of cardiovascular disease; therefore, they only collected information on one quality indicator for a subset of patients with chronic kidney disease in Eastern Ontario. 22 The other study was restricted to primary care physicians involved in an electronic medical record research initiative.23,24 These physicians have been involved in related quality of care improvement initiatives, such as diabetes and hypertension care—two known risk factors for chronic kidney disease, so they may be providing higher levels of chronic kidney disease care on average than other Ontario physicians. The purpose of this study was to use recently established quality indicators for chronic kidney disease in the primary care setting to describe the proportion of patients receiving recommended care in Ontario.
Methods
Study Design and Research Setting
In Ontario, access to primary health care and laboratory tests are covered under the Ontario Health Insurance Plan program; however, outpatient medications are only funded for patients aged 65 years or older, and certain people who are eligible for drug benefit programs. 25 These health care encounters are recorded in large administrative health care databases, which are linked using unique, encoded identifiers and held at the Institute for Clinical Evaluative Sciences (ICES).
We conducted a retrospective cohort study using the administrative data available at ICES. This study was conducted through the ICES Kidney, Dialysis and Transplantation research program and all analyses were performed at the ICES Western site in London, Ontario. This study was approved by the Sunnybrook Health Sciences Centre Research Ethics Board in Toronto, Ontario. We followed the reporting guidelines for observational studies using the REporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement (see Supplementary Material 2). 26
Data Sources
We used linked outpatient laboratory data to identify patients with markers for chronic kidney disease (reduced eGFR and elevated ACR). These data included an electronic network of all 12 hospitals in Southwestern Ontario (Cerner) and all outpatient Dynacare laboratories in Ontario. We used 7 other linked databases held at ICES to ascertain information on hospitalizations (Canadian Institute for Health Information’s Discharge Abstract Database [CIHI-DAD]); emergency department visits (CIHI’s National Ambulatory Care Reporting System [CIHI-NACRS]); physician billings for health care procedures, specialist visits, and laboratory tests (Ontario Health Insurance Plan claims database and the ICES Physician database); prescription drug information for individuals over 65 years old (Ontario Drug Benefits database); information on patients with end-stage kidney disease or previous kidney transplants (the Ontario portion of the Canadian Organ Replacement Register); and vital status information such as birth and death data (Registered Persons Database).
From 2002 onward, the 10th edition of the Canadian Modified International Classification of Disease system (ICD-10) was used to record all diagnostic codes in CIHI-DAD and CIHI-NACRS, and the Canadian Classification for Health Interventions was used to record all procedural codes.
Patients
To assess the performance of the quality of care indicators, we created 3 cohorts of patients aged 40 years or older accrued between April 1, 2006, and September 30, 2011, based on their kidney function: (1) patients with an initial eGFR value less than 60 mL/min/1.73 m2 (eGFR screening cohort), (2) patients with an initial urine ACR concentration more than 3 mg/mmol (ACR screening cohort), and (3) patients with 2 eGFR values less than 60 mL/min/1.73 m2 separated by at least 3 months but less than 18 months (chronic kidney disease cohort). Individuals could be in more than 1 cohort. The eGFR values were calculated based on serum creatinine using the Chronic Kidney Disease Epidemiology Collaboration formula (CKD-EPI). 27 As we had no data available on race, all patients were assumed to be non-black in the CKD-EPI equation (a reasonable assumption as less than 5% of the Ontario population is of black race). 28 The date of the laboratory test used to define each cohort was considered the cohort entry date; for the chronic kidney disease cohort, this was the date of the second serum creatinine test. We conducted our study from 2006 onward, as eGFR reporting for laboratories in Ontario began in February 2006.
For the eGFR and ACR screening cohorts, we excluded anyone with evidence of chronic kidney disease in the 5 years prior to cohort entry (based on codes and nephrology referrals), or any prior evidence of end-stage kidney disease (receipt of either chronic dialysis or a kidney transplant). Similarly, for the chronic kidney disease cohort, we excluded patients with prior evidence of end-stage kidney disease. We did not use urine ACR in combination with eGFR to define our chronic kidney disease cohort, as urine ACR values were not available for most patients. 8
Development of Quality of Care Benchmarks for Chronic Kidney Disease
A modified Delphi panel funded by the Ontario Renal Network was completed to develop a consensus set of quality primary care indicators for chronic kidney disease. 23 This technique has been used previously to identify quality indicators for cardiac care.29,30 The modified Delphi process ensured anonymity and iterative feedback from the group. The panel consisted of stakeholders across Canada including primary care physicians, nephrologists, and nursing and patient representatives. From over 150 initial quality indicators, the panel considered the evidence and clinical importance of each indicator and agreed on 17 final quality indicators. Based on the data available in our data sources, we were able to measure 10 of these quality indicators for the current study, with the addition of 1 other indicator. See Table 1 for the definitions of the 11 quality indicators used in this study.
Definitions of Quality of Care Indicators for CKD.
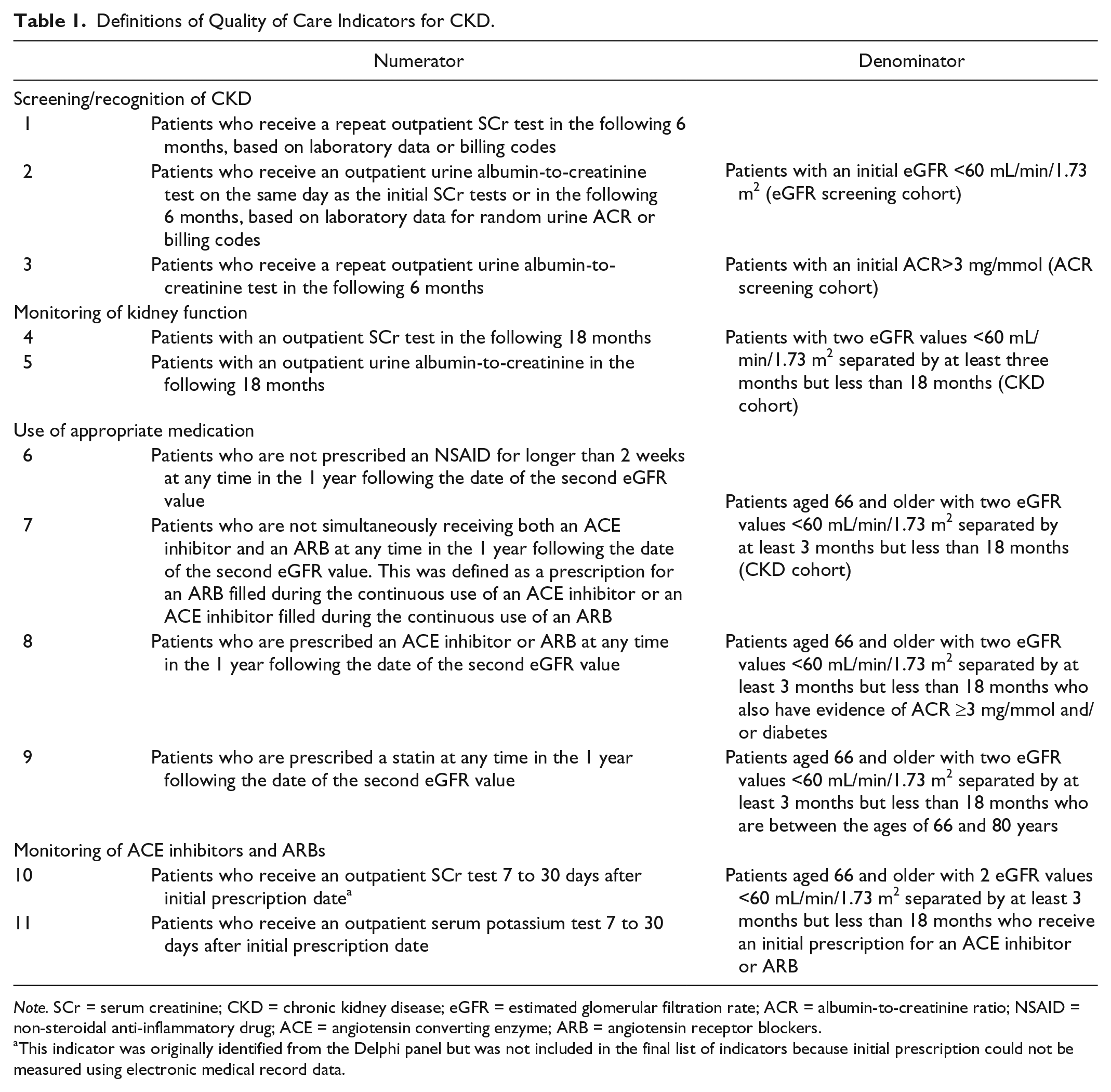
Note. SCr = serum creatinine; CKD = chronic kidney disease; eGFR = estimated glomerular filtration rate; ACR = albumin-to-creatinine ratio; NSAID = non-steroidal anti-inflammatory drug; ACE = angiotensin converting enzyme; ARB = angiotensin receptor blockers.
This indicator was originally identified from the Delphi panel but was not included in the final list of indicators because initial prescription could not be measured using electronic medical record data.
Definitions of Quality Indicators
Patients were followed forward in the data sets from 30 days to 18 months after their index date depending on the quality indicator. The first three quality indicators looked at screening or recognition of chronic kidney disease among the eGFR and ACR screening cohorts. Indicators 4 and 5 looked at monitoring of kidney function with serum creatinine and urine albumin-to-creatinine measures at least once in the 18 months following evidence of chronic kidney disease. The screening and monitoring indicators used physician billing codes to ascertain receipt of laboratory tests. Indicators 6 to 9 assessed appropriate use of medications among patients with chronic kidney disease in the 1 year following evidence of chronic kidney disease (prescribing ACE inhibitors or ARBs for patients with diabetes and/or ACR > 3 mg/mmol, avoiding co-prescription of ACE inhibitors and ARBs, prescribing statins, and avoiding prolonged use of prescription NSAIDs). The last two quality indicators looked at monitoring serum creatinine and serum potassium levels (based on physician billing codes) in the 7 to 30 days after patients were initiated on an ACE inhibitor or ARB. For the medication indicators, we excluded patients less than 66 years of age, which allowed for 1 full year of baseline medications for review (as previously mentioned, outpatient drug coverage is a universal benefit for persons 65 years and older living in Ontario). We were not able to capture over-the-counter NSAID use. See Supplementary Material 3a and 3b for administrative codes and drug names used to define indicators.
Statistical Analyses
All analyses were conducted using SAS version 9.4 (SAS Institute, Cary, North Carolina). We calculated the percentage of patients meeting each indicator based on the definitions for each numerator and denominator. Prior to calculating these percentages, we excluded patients who died during the follow-up period to ascertain each indicator. As a secondary analysis, we calculated the cumulative incidence function censored for death of each indicator at end of follow-up estimated using the exponential equation: 1 − e(−IR*T), where e is a mathematical constant, IR is the incidence rate or number of people with an event over person-time at risk, and T is the time period of interest.
We stratified the percentage of patients meeting each indicator by age (40 to <65, 65 to <80, and ≥80 years), sex, cohort entry period (April 2006 to December 2008 and January 2009 to September 2011), and baseline eGFR levels (≥60 mL/min/1.73 m2, 44-59 mL/min/1.73 m2, and <44 mL/min/1.73 m2). Note that indicator 3, assessing repeat ACR values, was the only indicator that included patients with the eGFR value more than or equal to 60 mL/min/1.73 m2; based on the cohort definitions, only patients with the eGFR value less than 60 mL/min/1.73 m2 were included in the assessment of the other indicators. We focused on variation across these variables because age and gender disparities in quality of care have been described previously for chronic kidney disease and other related conditions.31-35 We were also interested to see if there were changes over time. Finally, quality of care indicators for chronic kidney disease have been shown to vary based on chronic kidney disease stage.36-41
Baseline Characteristics
We examined the baseline characteristics of the 3 different cohorts. These characteristics included demographics (age, sex, rural or urban residence, and income quintile), baseline kidney function (serum creatinine, eGFR, and urine ACR), health care use in the past year (number of hospitalizations, emergency room visits, primary care visits, general internist visits, and nephrology visits), Johns Hopkins Adjusted Clinical Groups to estimate intensity of health care resource use in the past year, 42 comorbidities based on Johns Hopkins Expanded Diagnostic Clusters in the past year (ischemic heart disease, congestive heart failure, cardiac arrhythmia, acute myocardial infarction, cardiac arrest, hypertension, diabetes, chronic liver disease, malignant neoplasms, cerebrovascular disease, and peripheral vascular disease), and prescription drugs filled in the previous 120 days by patients over the age of 65 years (ACE inhibitors, ARBs, statins, and diabetes medications).
We summarized binary and categorical characteristics by proportions and continuous characteristics by means, standard deviations, medians, and interquartile ranges (IQRs, ie, 25th and 75th percentiles).
Results
Patients
See Figure 1 for the flow diagram of patients included in the 3 cohorts. There were 223 994 patients in the eGFR screening cohort, 132 442 patients in the ACR screening cohort, and 184 557 patients in the chronic kidney disease cohort. The total number of unique patients included in the study was 410 409. There were 28 442 patients in both the eGFR screening and ACR screening cohorts, 25 935 patients in both the ACR screening and chronic kidney disease cohorts, 92 688 patients in both the eGFR screening and chronic kidney disease cohorts, and 16 521 patients included in all 3 cohorts.
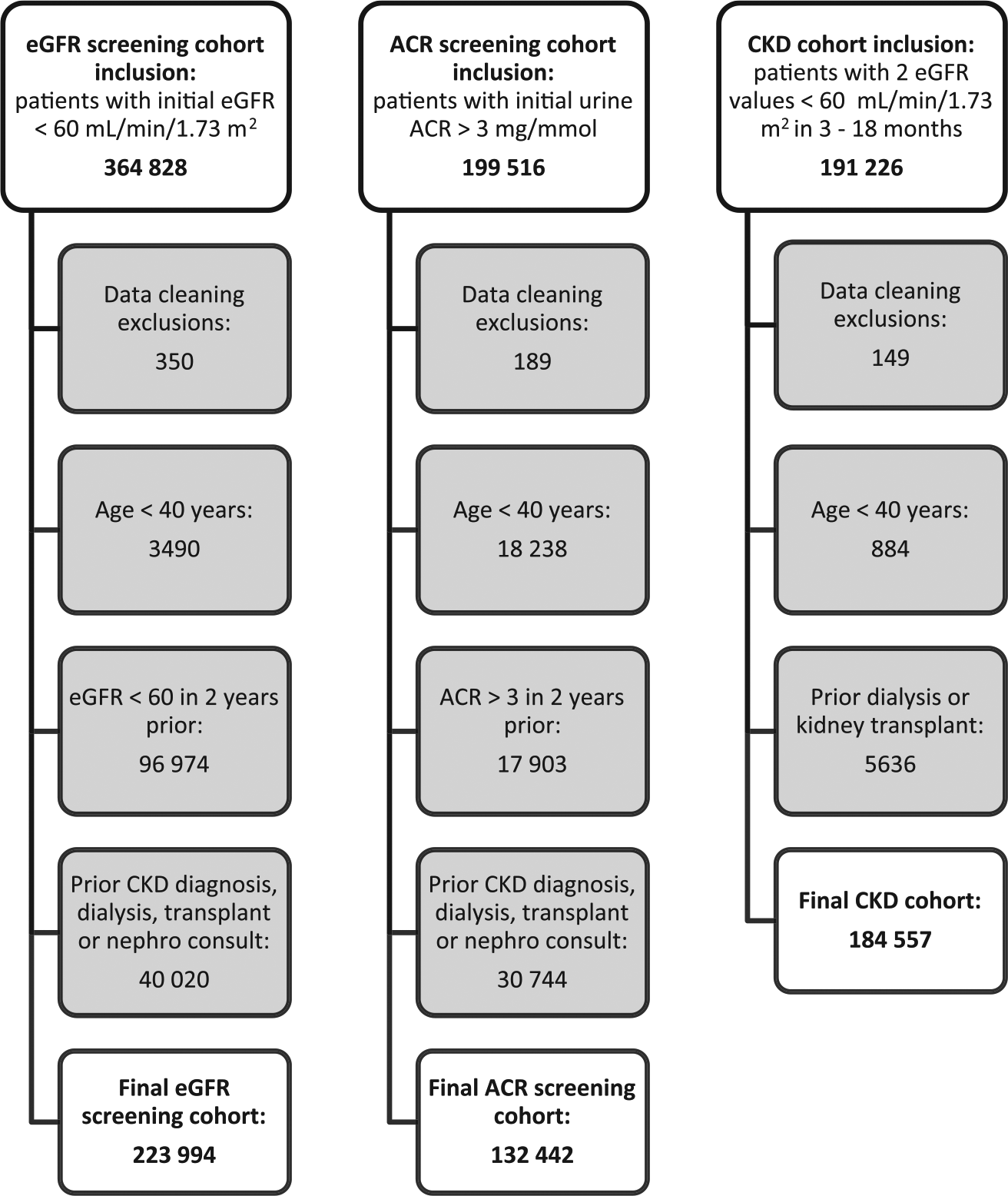
Flow diagrams of participant selection.
Baseline Characteristics
See Table 2 for the baseline characteristics of the three cohorts. The median (IQR) age of patients in the ACR screening cohort was 64 (IQR 54-74), whereas the median (IQR) age in the eGFR screening and chronic kidney disease cohorts was 74 (IQR 66-81) and 77 (IQR 70-83), respectively. Half of the patients in the ACR screening cohort were female compared with approximately 60% of the patients in the eGFR screening and chronic kidney disease cohorts. Among patients in the ACR cohort with available serum creatinine tests (89%) in the previous year, approximately 80% had an eGFR level higher than or equal to 60 mL/min/1.73 m2. Only 19% and 31% of the screening and chronic kidney disease cohorts, respectively, had urine ACR values available in the previous year. Among these patients, 68% in the screening cohort and 58% in the chronic kidney disease cohort had a urine ACR value less than 3 mg/mmol. Approximately 54% of patients in the ACR screening cohort, 23% of patients in the eGFR screening cohort, and 31% of patients in the chronic kidney disease cohort had diabetes.
Baseline Characteristics for the 3 Study Cohorts.

Note. eGFR = estimated glomerular filtration rate; ACR = albumin-to-creatinine ratio; CKD = chronic kidney disease; SD = standard deviation; IQR = interquartile range; SCr = serum creatinine; ADG = aggregated diagnosis group; ACE = angiotensin converting enzyme; ARB = angiotensin receptor blockers.
Diabetes drugs included insulin and oral anti-glycemic medications.
Quality Indicator Performance
See Table 3 for the number, proportion, and cumulative incidence of patients meeting each of the quality indicators. Median follow-up times ranged from 158 to 395 days depending on the indicator (see Supplementary Material 4). Proportions and cumulative incidence at follow-up for each indicator were similar for most indicators. Overall, screening or recognition of chronic kidney disease was around 50% (ranged from 42% for repeat urine ACR testing following an initial ACR >3 mg/mmol to 55% for urine ACR testing done on the same day [36%] or in the following 6 months after an initial eGFR value <60 mL/min/1.73 m2). The 6-month cumulative incidence for this latter indicator was 65%. Regular monitoring of kidney function was high for serum creatinine tests (91%) but was lower for urine albumin-to-creatinine tests (70%). Most (84%) patients with chronic kidney disease and 66 years or older were not receiving an NSAID prescription for 2 weeks or more in the 1 year following their chronic kidney disease date. The majority of patients 66 years or older with chronic kidney disease who also had diabetes and/or proteinuria were receiving an ACE inhibitor or ARB (75%) in the 1 year following their chronic kidney disease date, and 96% were not receiving an ACE inhibitor and an ARB concurrently. Among patients between ages 66 and 80 years, 65% received a statin. Monitoring of serum creatinine and serum potassium in the 7 to 30 days after initiating an ACE inhibitor or ARB for patients aged 66 years or older with chronic kidney disease was around 25%.
Number and Proportion of Patients Meeting Quality of Care Indicators.
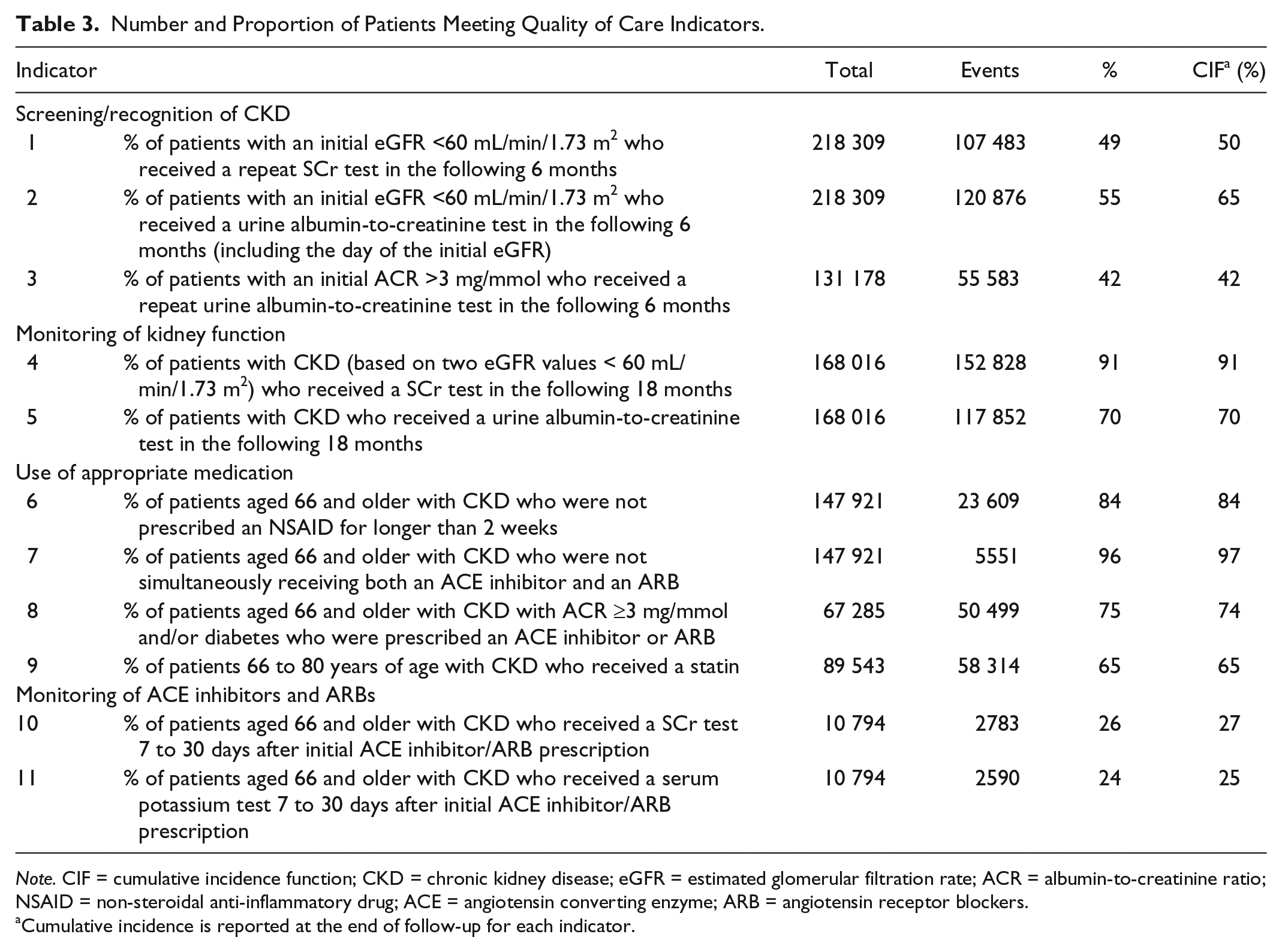
Note. CIF = cumulative incidence function; CKD = chronic kidney disease; eGFR = estimated glomerular filtration rate; ACR = albumin-to-creatinine ratio; NSAID = non-steroidal anti-inflammatory drug; ACE = angiotensin converting enzyme; ARB = angiotensin receptor blockers.
Cumulative incidence is reported at the end of follow-up for each indicator.
The percentage of patients meeting each quality indicator when stratified by age, sex, cohort entry period, and baseline eGFR is shown in Supplementary Material 5a to d. Among most quality indicators, proportions were higher for males compared with females, with the exception of the indicators for repeat urine albumin-to-creatinine test following an abnormal ACR result (44% for females and 40% for males). The screening indicators generally showed a decreasing trend with age and an increasing trend with severity of chronic kidney disease, except for the repeat urine albumin-to-creatinine screening indicator. Serum creatinine monitoring among patients with chronic kidney disease was similar when stratified by all 4 variables, where urine albumin-to-creatinine monitoring decreased with age (>80 vs 65 to <80 years) from 75% to 64%. Among the prescription indicators for patients aged 66 and older, not prescribing ACE inhibitors and ARBs concurrently increased over time (95% to 98%) and the frequency of prolonged use of prescription NSAIDs did not change over time. Prescriptions for ACE inhibitors or ARBs increased over time (74% to 78%) and decreased with age (77% to 72%). Prescriptions for statins increased with severity of chronic kidney disease (64% to 68%) and decreased with age (65% to 62%). Finally, for the monitoring of ACE inhibitor and ARB indicators, monitoring was higher for patients with the eGFR value less than 44 mL/min/1.73 m2 compared with patients with the eGFR value 44 to 59 mL/min/1.73 m2 (32% vs 22% for serum creatinine and 30% vs 21% for serum potassium).
Discussion
This is the largest and most comprehensive population-based study to assess the quality of renal care among patients being screened for, or who have, chronic kidney disease in the primary care setting in Ontario, Canada.
Overall, we found that most quality of care indicators were met by primary care providers. For instance, it was reassuring that the majority of patients with chronic kidney disease and proteinuria/diabetes were receiving ACE inhibitors or ARBs, and that patients were not being prescribed NSAIDs for prolonged use or simultaneously receiving ACE inhibitors and ARBs.
We found that around half of the patients with an initial abnormal eGFR or ACR did not receive follow-up tests to confirm whether chronic kidney disease was present or not. This is consistent with the previous literature on chronic kidney disease recognition. Another Ontario cohort study, which included physicians enrolled in an electronic medical record research initiative, found that 48% and 16% of patients with initial abnormal eGFR received repeat serum creatinine and follow-up albumin-to-creatinine testing, respectively. 23 Similarly, a large retrospective cohort study in the United Kingdom showed that only 25% of patients with incident chronic kidney disease based on laboratory values were registered as having chronic kidney disease, and only 36% of patients had an ACR test completed over the study period. 40 This lack of confirmatory tests may be partially explained by primary care physicians’ concerns for over-diagnosis. For example, a survey of primary care providers in the United States found that 30% of physicians would not classify patients as having chronic kidney disease if their eGFR was between 45 and 59 mL/min/1.73 m2 and 55% would not diagnose patients with chronic kidney disease if they had an eGFR value higher than 60 mL/min/1.73 m2 with microalbuminuria. 18 An alternative explanation could be that patients are not receiving follow-up confirmatory tests against the advice of their primary care providers. Furthermore, primary care physicians may view low eGFR as part of the normal aging process rather than as a disease. 43 Our results showed a decreasing trend of screening with age. Primary care physicians may be less likely to screen older individuals with reduced life expectancies, as they are not likely to benefit from chronic kidney disease care management.
After the presence of chronic kidney disease was established in our study (ie, 2 eGFR values <60 mL/min/1.73 m2 between 3-18 months apart), patients were found to be receiving adequate monitoring of kidney function in the following 18 months; however, this was higher for serum creatinine (91%) compared with urine albumin-to-creatinine (70%) monitoring. The lower adherence to urine albumin-to-creatinine monitoring is consistent with another Ontario cohort study, which found that only 52% of patients with chronic kidney disease received a urine albumin-to-creatinine test over a 12-month period among 84 primary care practices in Eastern Ontario. 22 A province-wide report in Alberta also showed lower adherence to albumin-to-creatinine test monitoring of 32% in the previous 2 years. 44 Furthermore, a cohort study in the United States found an even larger discrepancy between annual monitoring for serum creatinine and urine albumin-to-creatinine among patients with chronic kidney disease: 86% and 30%, respectively. 45 Primary care physicians are generally in agreement about the importance of regular serum creatinine testing for patients with chronic kidney disease, but they are less in agreement about the importance of regular urine albumin-to-creatinine testing for chronic kidney disease in the absence of diabetes.18,34 For instance, the American College of Physicians released guidelines in 2013, which recommended against urine albumin-to-creatinine monitoring among patients who are taking an ACE inhibitor or ARB; however, this was a weak recommendation based on low-quality evidence. 46 Some reported barriers include assumptions that the urine albumin-to-creatinine test does not affect patient management, concerns that there are more pressing issues for patient care or not enough time, and the belief that urine albumin-to-creatinine monitoring is not recommended for patients with chronic kidney disease in the absence of diabetes. 18
In regard to appropriate prescribing indicators, we found that the majority of patients aged 66 years and older with chronic kidney disease, diabetes and/or ACR > 3 mg/mmol were receiving an ACE inhibitor or an ARB (74%). It is unlikely that much improvement can be made for this indicator, as some of the patients have contra-indications including a history of prior adverse events with these drugs. Another Ontario cohort study also found that 75% of patients with diabetes and albuminuria were on an ACE inhibitor or ARB. 23 A province-wide report on chronic kidney disease care in Manitoba also reported high rates of ACE inhibitor and ARB use (up to 80%), especially among patients with diabetes and at high risk of chronic kidney disease progression. 47
It is reassuring that only a small proportion of patients aged 66 years and older were receiving ACE inhibitors and ARBs concurrently and that this decreased slightly over time. This decrease over time from 2006-2008 to 2009-2011 coincides with the timing of the press release from the Heart and Stroke Foundation in 2009. 12 This press release warned against co-prescribing of ACE inhibitors and ARBs, which was based on evidence from a large international clinical trial. 11 We used a conservative definition to capture ACE inhibitor and ARB co-prescribing to avoid misclassifying patients who switched from one drug to the other, so we may have missed some cases. However, our results are consistent with 2 previous studies: a large cohort study in the United Kingdom and a Dutch study focusing on patients with diabetes and chronic kidney disease, which found that 98% and 96% of patients, respectively, were not taking an ACE inhibitor and ARB concurrently.41,48
Many patients in our study aged 66 to 80 years with non-dialysis–dependent chronic kidney disease received a statin (65%); although there is room for improvement. A province-wide study in Alberta found similar results among patients with chronic kidney disease and diabetes. 44 Our results are higher than the statin-prescribing proportions observed by previous studies in the United States (ranging from 16% to 57%).39,45,49-51 Our results are more consistent with studies in Australia and Asia (ranging from 59% to 87%) and a study in the Netherlands (74%).36,48,52,53 We only looked at statin prescribing up until 2012; guidelines recommending statin use for patients with chronic kidney disease were released in 2013, 54 so there may be improvement in more recent years.
Only 16% of patients aged 66 and older with chronic kidney disease in our study were receiving an NSAID prescription for longer than 2 weeks. However, we could not capture use of non-prescription NSAIDs. Prolonged cumulative NSAID use or high dose of NSAIDs (vs low dose) among patients with reduced kidney function has been shown to be associated with accelerated kidney function decline.13,14 Our findings align with previous studies from Canada, the United States, the United Kingdom, and Australia where NSAID prescribing among patients with chronic kidney disease ranged from 1% to 16%.23,37,38,45,55,56 In a qualitative study of primary care physicians’ attitudes and knowledge about chronic kidney disease, it was described that physicians are aware that NSAIDs should be avoided in patients with chronic kidney disease but they generally prescribe NSAIDs to patients with chronic kidney disease who they deem to be at low risk of complications. 17
Our results showed that there is poor laboratory monitoring among patients aged 66 and older with chronic kidney disease who were initially prescribed an ACE inhibitor or an ARB—over three quarters of patients did not have their serum creatinine and potassium monitored in the month following their initial prescription. This may be concerning as ACE inhibitor and ARB use in patients with chronic kidney disease is associated with increased short-term elevation of serum creatinine and potassium.57,58 With adequate monitoring of patients’ serum creatinine and potassium levels, the long-term benefits of these prescriptions outweigh these short-term risks. 57 Our results are consistent with a Dutch study that also found only 34% and 28% of patients (not all with chronic kidney disease) received serum creatinine and serum potassium monitoring, respectively, within 3 weeks of initiating an ACE inhibitor or ARB. 59 Another study reported about 50% serum creatinine and potassium monitoring after initiating an ACE inhibitor among patients with hypertension, but they allowed a 6-month follow-up period in which tests could be performed. 60
Strengths and Limitations
This is the largest population-based study conducted on the quality of primary care for patients with chronic kidney disease. In addition to calculating the percentage of people meeting each indicator, we also provided the cumulative incidence at the end of follow-up, censoring for death. This provides a more accurate estimate by allowing patients who died during the follow-up period to still be eligible to meet each indicator in the period prior to their death date. However, given the small number of patients who died during follow-up for each indicator, the cumulative incidence estimates are very similar to the percentages.
Our study has some limitations. The laboratory data used to define our cohorts were from one of Ontario’s 3 largest commercial laboratories and has wide coverage across Ontario, but likely only includes approximately 20% of Ontario’s chronic kidney disease population. As such, our results may not be representative of all patients at risk of, or with, chronic kidney disease in Ontario. However, physician billing codes were used to ascertain outpatient tests completed at all Ontario commercial laboratories for the screening, monitoring, and ACE inhibitor/ARB follow-up test indicators. As such, this does not affect the internal validity of our indicator calculations. The Ontario Laboratory Information System is an electronic database capturing all outpatient laboratories in Ontario. The data from this are in the process of being linked to the ICES data holdings, so in future studies we will be able to provide more generalizable Ontario-wide reports on quality indicators for chronic kidney disease.
Our study was also limited by other health care data available in our data sources. Through the modified Delphi panel process, 17 quality of care indicators were identified but only 11 indicators could be assessed using health care administrative data. For example, we did not have information on blood pressure to determine whether targets were being met. Our drug indicators were also limited to patients 65 years or older, so we cannot make any observations on appropriate prescribing for patients with chronic kidney disease under 65 years. Furthermore, NSAIDs are also available over the counter, but we could only capture prescription NSAIDs with our data sources.
It is also important to note that our study did not capture people with chronic kidney disease who went untested (unidentified) in routine care. As such, our screening indicators only apply to patients who have received at least 1 abnormal test in our data sources during the study period. We did not assess screening in patients with risk factors for chronic kidney disease (eg, cardiovascular disease, diabetes, or hypertension).
Conclusions
Overall, we found high proportions of patients meeting most of the quality of care indicators including regular chronic kidney disease monitoring and ACE inhibitor/ARB use; however, improvement is still needed for other care indicators such as screening and recognition of chronic kidney disease, and follow-up monitoring of serum potassium and serum creatinine for new ACE inhibitor or ARB users. Future population-based studies are needed to confirm these findings, as well as studies to determine the potential impact on patient outcomes of not meeting these indicators. Future qualitative studies exploring the barriers and facilitators to the implementation of the chronic kidney disease guidelines in Ontario primary care are warranted.
Footnotes
Acknowledgements
We thank Dynacare Laboratories for providing us access to their data, and we thank the team at London Health Sciences Centre, St. Joseph’s Health Care, and the Thames Valley Hospitals for providing access to the Cerner laboratory data.
List of Abbreviations
ACE, angiotensin converting enzyme; ACR, albumin-to-creatinine ratio; ADG, aggregated diagnosis group; ARB, angiotensin receptor blocker; CCI, Canadian Classification for Health Interventions; CIHI, Canadian Institute for Health Information; CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration formula; DAD, Discharge Abstract Database; eGFR, estimated glomerular filtration rate; ICD-10, 10th edition of the Canadian Modified International Classification of Disease system; ICES, Institute for Clinical Evaluative Sciences; IQR, interquartile range; NACRS, National Ambulatory Care Reporting System; NSAID, non-steroidal anti-inflammatory drugs SCr, serum creatinine; SD, standard deviation.
Ethics Approval and Consent to Participate
This study was approved by the Sunnybrook Health Sciences Centre Research Ethics Board in Toronto, Ontario. Participant consent for this study was waived.
Consent for Publication
Not applicable.
Availability of Data and Materials
We cannot share the data used for this project due to privacy requirements at Institute for Clinical Evaluative Sciences. Only aggregated data as presented in this manuscript can be shared.
Authors’ Note
Parts of this material are based on data and information compiled and provided by Canadian Institute for Health Information. The opinions, results, and conclusions reported in this article are those of the authors and are independent from the funding and data sources. No endorsement by Institute for Clinical Evaluative Sciences, Canadian Institute for Health Information, or the Ontario Ministry of Health and Long-Term Care is intended or should be inferred.
Author Contributions
D.M.N. and A.X.G. developed the initial study plan. K.T. developed the initial literature review for chronic kidney disease care indicators and led the Delphi panel. S.B., M.M.R., and E.M. provided input and approved of the study and analysis plan. D.N. completed all analyses with the assistance of E.M. All authors interpreted the results. D.N. drafted the initial manuscript, and all other authors critically reviewed and revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.X.G. was supported by the Dr. Adam Linton Chair in Kidney Health Analytics. A.X.G., S.B., G.E.N., and A.G. are Provincial Medical Leads for the Ontario Renal Network. All other authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Danielle Nash’s training is supported by a Canadian Institutes of Health Research Doctoral Scholarship. Dr. Amit Garg was supported by the Dr. Adam Linton Chair in Kidney Health Analytics. This study was conducted by the Institute for Clinical Evaluative Sciences (ICES) Western Site through the ICES Kidney, Dialysis and Transplantation Research Program. ICES is funded by an annual grant from the Ontario Ministry of Health and Long-Term Care. ICES Western is funded by an operating grant from the Academic Medical Organization of Southwestern Ontario.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
