Abstract
Objective
The scientific evidence indicates little or no difference in the effectiveness or cost of using of metered-dose inhalers (MDIs) versus nebulisation to treat acute asthma in the emergency department (ED). However, the use of MDIs raises questions of environmental impact. Our objective was to compare the ecological footprint of salbutamol administered by MDI versus nebulisation.
Design
Life cycle assessment in which we inventoried and quantified the resources extracted and pollutants emitted by each therapeutic option, from the manufacturing of medication and equipment to their disposal by incineration.
Setting
EDs of the CHU de Québec-Université Laval (Canada).
Participants
Not applicable.
Main outcome measures
Each item of life cycle inventory data was translated into CO2-equivalent emissions (CO2eq) using the IPCC2021/GWP100 method. Results were estimated for the administration of one and three treatments of 800 µg of salbutamol by MDI and 5 mg by nebulisation (standard doses for adults and children ≥ 24 kg).
Results
One and three ED-administered treatments with salbutamol emit respectively 1.9 and 4.0 kg of CO2eq via MDI versus 0.9 and 1.0 kg via nebulisation, which corresponds to 5.5 and 11.6 km and to 2.7 and 2.8 km travelled in a subcompact car. Each series of eight inhalations from an MDI releases 1.1 kg of CO2eq due to emission of the hydrofluoroalkane propellant.
Conclusions
Considering the absence or minimal difference in clinical effectiveness, this study suggests that nebulisation may be a more eco-efficient administration route than MDIs in the emergency treatment of asthma.
Introduction
Healthcare services are responsible for 4.6% of greenhouse gas emissions worldwide. 1 The USA and Canada rank first and third, respectively, in per capita healthcare-related emissions of CO2 equivalents (CO2eq). 1 Increasing emissions of greenhouse gases contribute to global warming, of which the negative impact on human health includes an increase in heat-related illnesses and respiratory problems as well as wider spread of infectious diseases. 2
The use of metered-dose inhalers (MDIs) to administer bronchodilators (e.g. salbutamol) is considered a significant source of health-service-associated greenhouse gas emissions. 3 It has been estimated that in the United Kingdom, up to 13% of such emissions result from the use of MDIs to treat asthma or chronic obstructive pulmonary disease (COPD). The main greenhouse gas emitted from these devices is the hydrofluoroalkane (HFA) propellant. Some HFAs have atmospheric warming potentials up to 3000 times that of CO2.4,5 Consequently, replacing MDIs with HFA-free administration methods is regarded as desirable whenever possible. However, MDI use has become the standard of care in many EDs.6,7 Administration of a bronchodilator in the form of fine droplets is the main therapeutic alternative in the hospital setting. This mode, known as nebulisation, is said to have clinical effectiveness comparable to MDIs for key clinical outcomes such hospital admission or Forced Expiratory Volume in One Second (FEV1), although MDIs are associated with a shorter ED length of stay and fewer tremors in pediatrics. 8 To our knowledge, the environmental impact of nebulisation in the ED setting has not been evaluated and compared with that of MDIs. 5
The objective of this study was to compare the environmental footprints of MDI and nebulisation when used to administer salbutamol to adults in EDs.
Methods
Design and setting
This is an observational study comparing MDIs and nebulisation as methods of administering salbutamol to asthmatic patients in EDs, using a life cycle assessment methodology to calculate the ‘cradle to grave’ environmental footprints (e-Figure 1). The study was conducted during fiscal year 2022–2023 at the CHU de Québec-Université Laval (hereafter the CHU), an academic institution that manages five EDs located throughout Québec City (Canada) and receiving more than 240,000 visits annually.
Life cycle assessment
We evaluated an MDI (100 µg of salbutamol/dose, 200 doses) and a nebulisation ampoule (5 mg/mL, 10 mL), both products manufactured by GSK. An MDI is for single-patient use, whereas multiple patients can be treated with a single nebulisation ampoule.
The assessment followed ISO standards 14,040 and 140449,10 and consisted of four phases:
1. Definition of the goal, scope, and functional unit
We compared one and three administrations of a standard dose of salbutamol in the ED for adults and children weighing ≥ 24 kg, namely 800 μg (eight puffs) via MDI and 5 mg (1 mL) via nebulisation (functional unit). The life cycle assessment included production, packaging, transportation, use in the ED, and incineration, assuming all material disposal takes place at the hospital. We considered MDI treatments with a reusable plastic spacer or a single-use cardboard spacer, as well as all the reusable material (nebulising cup, plastic tubing, and oxygen mask) required for nebulisation.
2. Life cycle inventory
We decomposed the life cycles of the two therapeutic options to identify all emissions and extractions within the system boundaries. In addition to the product monographs, we obtained the inventory data for all components of the MDI, plastic and cardboard spacers, nebulisation ampoule and materials using attenuated total reflectance infrared spectroscopy, which provides detailed information about the molecular composition of a product. The composition of the MDI canister gas was analysed using multinuclear nuclear magnetic resonance and gas-phase infrared spectroscopies. The methods for identifying the volatile components are described in e-Appendix 1. The production plants were located using procurement information obtained from our institution. The road distance to the biomedical waste management company used by the CHU (Stericycle) was determined. The complete life cycles were modelled using the openLCA software and the ecoinvent life cycle inventory database (version 3.9.1) and adapting some processes to the Québec context.
3. Impact assessment and outcome measures
The inventoried emissions and extractions were translated into environmental indicators in the following impact categories: (i) 4. Interpretation of results
The indicator results were interpreted relative to the functional units for one or three administrations, which are common initial prescriptions for asthma treatment in many ED protocols. Hotspots were identified, that is, the life cycle elements having the highest contribution to the indicator.
Alternative scenarios and extrapolations
Patients discharged from the CHU EDs are often allowed to keep the MDI for use at home. We considered this alternative scenario in the model by assuming that all such MDIs were emptied and ended up in the same incinerator as if they had been left in the ED.
We also estimated the annual environmental footprint of the asthma treatments based on medication consumption recorded at the CHU during 2022–2023. Since it was not possible to retrace the number of administrations and the dosages for each patient treated with an MDI, we assumed a minimum of three administrations of 800 µg (eight inhalations) for each MDI delivered by the pharmacy to the ED during the study period.
Statistical analysis
The life cycle inventory and impact assessment calculations were performed in openLCA (GreenDelta, version 2.4.1) using the ecoinvent database (v.3.9.1 cut-off by classification, published in 2023) to model the background processes (materials production, energy generation, transport and waste treatment). We report the results as point estimates and stacked bar charts, in accordance with the standards used for life cycle assessment reporting. To account for the uncertainties of the proxies used in the inventory, we conducted a Monte Carlo simulation with 1000 iterations. Uncertainty for each inventory flow was characterised using the Pedigree Matrix approach, 11 which evaluates data quality across five domains (reliability, completeness, temporal, geographical, and technological representativeness). For the foreground processes we modelled specifically for this study, the investigator-assigned ratings were converted into geometric standard deviations and applied as lognormal distributions. For the background processes, the ecoinvent datasets already included these distributions, which were then used directly in the Monte Carlo simulation. From this simulation, we calculated point estimates for each indicator, differences between salbutamol administration modes, and 95% confidence intervals. The models of the evaluated systems are available here. 12
Results
Life cycle inventories
The life cycle inventories of salbutamol treatments using MDIs and nebulisation are reported in Table 1. The HFA used in the inhaler is 1,1,1,2-tetrafluoroethane, a gas having a global warming potential 1500 times that of CO2.
Life cycle inventories of salbutamol administration in emergency departments using metered-dose inhalers and nebulization.

The distances of the production plant and the incinerator (for both salbutamol administration methods) from the CHU were respectively 1605 and 1813 km.
Impact categories
The environmental footprint of the two methods is reported per impact category for one and three administrations in Tables 2 and 3. Specifically, for the climate change indicator, one and three ED-administered salbutamol treatments emit 1.9 and 4.0 kg of CO₂eq via MDI, compared with 0.9 and 1.0 kg via nebulisation. These emissions correspond to driving 5.5 km and 11.6 km, or 2.7 km and 2.8 km in a compact car (e.g. Honda Civic, Toyota Corolla). e-Figures 2–5, presented in the supplementary material, provide a detailed breakdown of the contributions of each life-cycle stage to the different indicators for both MDI and nebulisation treatments.
Climate change and human health indicators for salbutamol administration in the emergency department.

Point estimates obtained from impact evaluation of the life cycle assessment methodology.
Medians and interquartile ranges (IQR) estimated using a Monte Carlo simulation (1000 iterations).
#admin: number of administrations in the emergency department.
MDI: metered-dose inhaler; CO2eq: CO2 equivalent emissions; DALY: disability-adjusted life-year.
Biodiversity and fossil resource use indicators for salbutamol administration in the emergency department.

Point estimates obtained from impact evaluation of the life cycle assessment methodology.
Medians and interquartile ranges (IQR) estimated using a Monte Carlo simulation (1000 iterations).
#admin: number of administrations in the emergency department.
MDI: metered-dose inhaler; PDF·m2·year: Potentially disappeared fraction of species over square meter years.
The climate change hotspot for MDIs is HFA emissions, which account for 56% and 79% of the total CO2-equivalent emissions for one and three administrations, respectively. In contrast, inhaler and spacer production contribute 33% and 16% of the total CO2-equivalent emissions for one and three administrations, respectively. Their carbon footprint of MDIs is thus linked to their use: each series of eight inhalations emits 1.1 kg of CO2eq as 1,1,1,2-tetrafluoroethane. These emissions are also a notable contributor to the other indicators (especially in the case of three series) but MDI and spacer production and disposal (of the plastic spacer more than the cardboard) are also important contributors. The results also indicate that although the single-use cardboard spacer leaves a smaller footprint than the plastic spacer after one administration, this difference diminishes or reverses in favour of the plastic spacer after three administrations, since the same cardboard can hardly be reused for more than one administration.
For nebulisation, cup and mask production and disposal are hotspots for all indicators. Cup and mask production accounts for 65% and 62% of the total CO2-equivalent emissions for one and three administrations, respectively, while their end-of-life disposal contributes 33% and 31% of the total CO2-equivalent emissions for one and three administrations, respectively. Since these devices are reusable, indicator results do not increase much with the number of administrations.
Because identical transport distances from the manufacturing plant to the user facility and from the facility to the incinerator were applied in modelling the life cycles of MDIs and nebulisation, transport does not influence the comparative assessment of their environmental footprints. In addition, the contribution of transport to the overall life cycles is marginal, accounting for only 1.9% and 3.3% of CO₂-equivalent emissions for MDIs and nebulisation, respectively.
Table 4 displays the median and 95% confidence interval of the difference between MDI and nebulisation for each impact category. The
Differences between administering salbutamol via metered-dose inhaler and nebulization in the emergency department: environmental indicators.
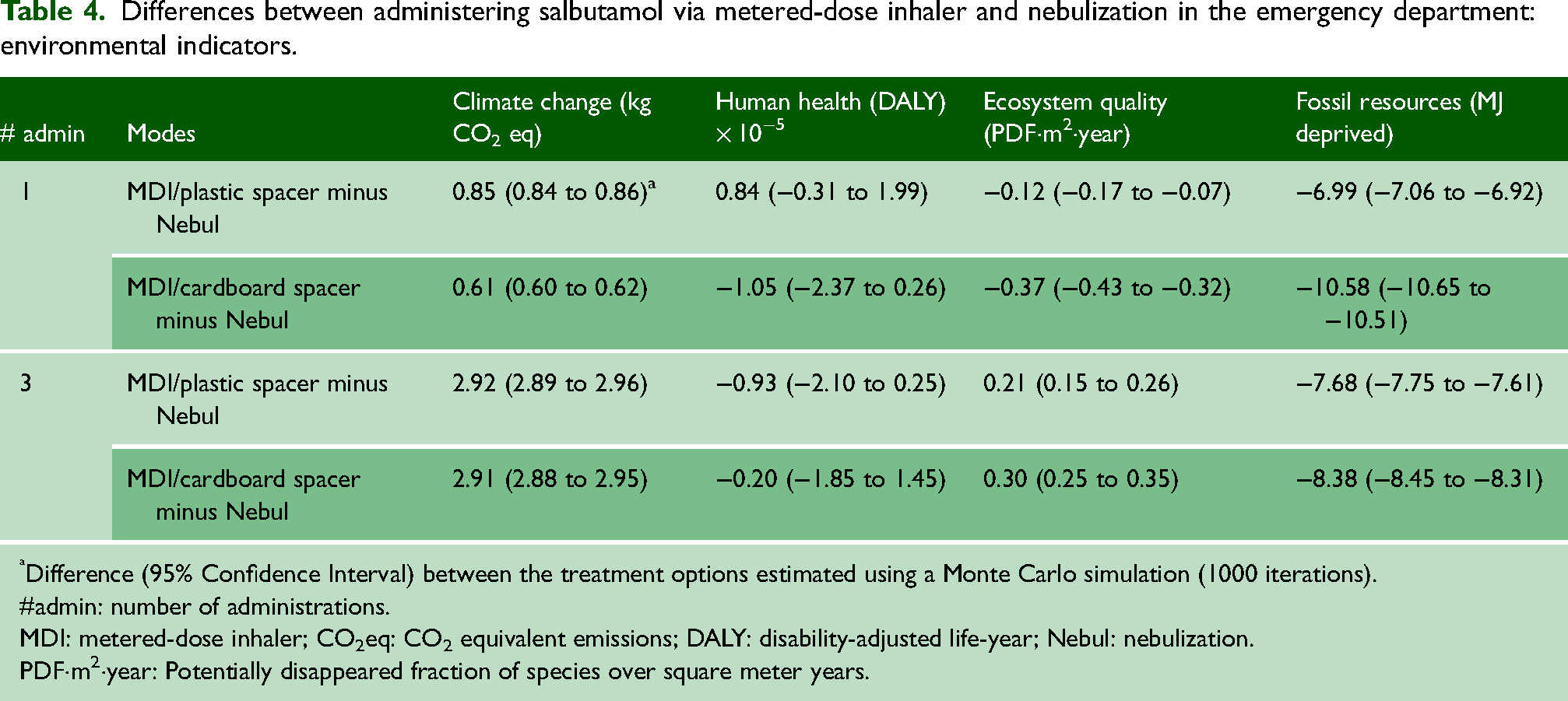
Difference (95% Confidence Interval) between the treatment options estimated using a Monte Carlo simulation (1000 iterations).
#admin: number of administrations.
MDI: metered-dose inhaler; CO2eq: CO2 equivalent emissions; DALY: disability-adjusted life-year; Nebul: nebulization.
PDF·m2·year: Potentially disappeared fraction of species over square meter years.
MDI left to the patient for home use
The environmental footprint of the scenario in which the patient upon discharge is allowed to keep the MDI for use at home is reported in e-Table 1. For all categories, the impact of this scenario is lower than disposal of the MDI at the hospital. While the contribution of HFA emission continues to increase, the contribution of MDI production and disposal is spread over 200 inhalations instead of eight or 24 (one or three administrations) per functional unit. The previously observed differences between MDIs and nebulisation for all indicators do not change significantly under this scenario. We estimated a difference of 2.5–2.6 kg CO2eq between MDI and nebulisation for three treatments, with no significant differences noted in other categories of impact.
One-year ecological footprint
In fiscal year 2022–2023, 4815 MDIs and 233 nebuliser ampoules were dispensed in the five EDs associated with the CHU. The corresponding annual environmental footprint is reported by impact category in Table 5, based on each patient receiving three administrations of salbutamol while in the ED. An emission of 19,260 kg of CO2 equivalent is attributable to the use of MDIs. If all patients had received nebulisation, this emission would have been 14,416 kg lower, and losses of 0.93 × 10−2 DALY and 1606 PDF·m2·year would have been avoided, but an additional 25,984 MJ in fossil resources would have been consumed. The 14,416 kg of CO2eq emissions is equivalent to driving 84,800 km in a compact car (e.g. Honda Civic, Toyota Corolla) or 19 trips from New York to Los Angeles.
Extrapolation of the ecological footprint of salbutamol administration via metered-dose inhaler and nebulization according to medication consumption recorded at the CHU de Québec-Université Laval in fiscal year 2022–23.

Represents the number of salbutamol metered-dose inhalers served in the emergency department in 2022–23.
Represents the number of nebulization ampoules (
MDI: metered-dose inhaler; CO2eq: CO2 equivalent emissions; DALY: disability-adjusted life year; PDF.m2.year: Potentially Disappeared Fraction of species per square meter per year.
Discussion
Statement of principal findings
In this product/process life cycle assessment, we examined ED treatment of acute asthma using salbutamol administration via MDIs versus nebulisation. Nebulisation was found to have half the carbon footprint of one MDI administration and one quarter of three MDI administrations. The two methods are indistinguishable in the
Strengths and weaknesses of the study
This study has several noteworthy strengths. First, to our knowledge, it is the first to compare the ecological footprint of MDIs and nebulisation, the two main methods of bronchodilator administration in EDs and hospital settings. In addition, it rigorously followed ISO 14040 and ISO 14044 standards governing life cycle assessment methodology. Notably, it includes a Monte Carlo simulation (1000 iterations) to account for data uncertainty and generate confidence intervals that strengthen the robustness of the comparison. Finally, it offers an extrapolation of the functional unit over a full year of activity in the participating EDs, allowing readers to better understand the potential ecological impact of their own therapeutic choices.
Among the limitations of this study, we must mention our creation of the life cycle inventories without access to industry information. However, the method used is scientifically robust and reproducible, and we are confident that the resulting analysis adequately estimates the differences in environmental footprint between MDI use and nebulisation. Our results might have differed if other inventory databases and impact assessment methods had been used. However, the ecoinvent database is one of the most widely used in the world, and processes were adapted to the Québec context when this was relevant. The CO2eq assessment method is that proposed by the Intergovernmental Panel on Climate Change, a widely recognised international organisation created under the auspices of the UN to assess risks related to climate change. We believe that this choice of tools makes our analysis as robust and credible as presently possible. Finally, to estimate the one-year environmental footprint, we assumed that patients received their three MDI treatments at 20-min intervals, since this is the protocolised prescription for asthma treatment in the CHU EDs. Since a significant number of patients end up receiving more than three treatments, our extrapolations are likely underestimates of the real impact.
Differences in results with previous studies
Goulet et al. compared HFA-based MDIs to a portable nebuliser device for use at home by patients with COPD. 13 Comparisons with their study should be made carefully since: (1) a different functional unit was used (200 µg of Albuterol by MDI and 3 mg by nebuliser); (2) the nebuliser device studied is not used in the ED; and (3) MDI administration was modelled alone without a spacer. Still, the authors calculated a carbon footprint 2‒3 times higher for one dose with an MDI compared to one dose with a nebuliser, whereas we estimated that one emergency treatment with an MDI has twice the carbon footprint of nebulisation. In absolute numbers, their CO2eq associated with MDI use appears disproportionately low (0.097 kg CO2eq for two inhalations) compared to our estimates (1.89 kg CO2eq for eight inhalations). However, the Goulet et al. model contained 6.7 g of HFA compared to 17.3 g used in our calculations. Their method of estimating the quantity of HFA is not described in detail, whereas our approach is reproducible and aligns with the quantities estimated another study that focused on gas kinetics associated with pressurised MDIs. 14
We also modelled the scenario in which the patient uses the MDI at home, which was found to lead to a lower carbon footprint. However, dry powder inhalers (DPI), which can be used by patients aged 6 and above, 15 have been found comparable in efficacy 16 and cost 17 to MDIs and to have a carbon footprint 20–30 times smaller. 18 If DPIs are in fact usable at home in the same manner as bronchodilators, then discarding MDIs after use in the ED would become the approach having the least impact as a source of greenhouse gas emission.
Implications for clinicians and future research
This study of a specific aspect of the environmental footprint of healthcare systems suggests that nebulisation is a more eco-efficient method of administering bronchodilators in EDs that receive many patients needing such treatments. However, particularly in children, this method has been associated with more side effects and longer ED stays. 8 Further research is needed to determine if the use of DPIs is effective and safe as ED treatment of mild to moderate acute asthma, which would allow nebulisation to be reserved for the most severe cases.
Conclusion
Acute asthma is one of the most frequent conditions treated in EDs. The administration of salbutamol for this purpose usually involves the use of an MDI, which has a significant environmental footprint. Our study shows that administering salbutamol by nebulisation has a smaller footprint. Given that more and more governments around the world are committed to providing healthcare services with net-zero greenhouse gas emissions over the coming decades, implementing low-emission ED treatment protocols for acute asthma should be one of many avenues to explore to achieve this goal.
Supplemental Material
sj-docx-1-shr-10.1177_20542704251396706 - Supplemental material for Ecological footprint of salbutamol administration by metered-dose inhaler versus nebulisation in acute asthma: a life-cycle assessment
Supplemental material, sj-docx-1-shr-10.1177_20542704251396706 for Ecological footprint of salbutamol administration by metered-dose inhaler versus nebulisation in acute asthma: a life-cycle assessment by Simon Berthelot, Jean-François Ménard, Guillaume Bélanger-Chabot, Gabriela Arias Garcia, Diego Mantovani, Chantale Simard, Jason R Guertin, Tania Marx and Ariane Bluteau in JRSM Open
Footnotes
Abbreviations
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Association des spécialistes en médecine d'urgence du Québec.
Ethical approval
Ethical approval was not required for this study, as it involved only a life-cycle assessment based on material analysis and non-individual, non-identifiable data.
Data availability
All life cycle assessment models are freely available on the Université Laval research data repository. 12
Author contributions
SB and JRG designed the study. SB and AB supervised its conduct. AB, TM, CS and SB collected the inventory data. SB and JFM carried out the life cycle assessment. SB drafted the initial manuscript. All authors proofread the manuscript and approved its submitted version. SB assumes responsibility for the integrity of the data used in this study and the accuracy of its analysis.
Acknowledgements
The authors thank Ms. Laurie Ouellet for granting permission to use her figure illustrating the life cycle.
Provenance
Not commissioned; peer-reviewed by Anthony David Dayan and Julie Morris
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
