Abstract
Objectives
To investigate the drugs and indications that have passed through the UK's Early Access to Medicines Scheme (EAMS) to date, the type of evidence the regulator considers when accepting a drug into the EAMS, and potential risks to patients.
Design
Analysis of publicly available data: MHRA Public Assessment Reports; Electronic Medicines Compendium database; interactive Drug Analysis Profiles database; Eudravigilance database.
Setting
United Kingdom.
Participants
The 51 ‘scientific opinions’ available on the MHRA website in June 2024.
Main Outcome Measures
Public Assessment Reports, pharmacovigilance data.
Results
After exclusions, there were 48 EAMS submissions, consisting of 48 indications and 32 drugs. 60% of indications were for cancer. Only 7% of EAMS submissions were based on double-blind, placebo-controlled randomised trials. The average sample size of studies conducted for the EAMS was 654. Most studies used surrogate (76%) and/or survival (57%) outcomes. Only 17% used subjective outcomes. For 17% of the indications, no ongoing studies were being conducted. Animal studies were conducted preclinically for all drugs and 35% also conducted in vitro studies. 47% of the drugs had elevated rates of suspected adverse reaction reports according to pharmacovigilance data.
Conclusions
We recommend that the EAMS drugs with elevated reporting rates are reviewed, that future studies of EAMS drugs use patient-centred outcomes, that preclinical studies make greater use of human biology-based approaches, that post-approval trials are conducted, and that future reviews of the EAMS centre the experience of patients.
Introduction
In April 2014, the Medicines and Healthcare products Regulatory Agency (MHRA) introduced its Early Access to Medicines Scheme (EAMS). The scheme aims to provide access to medicines that do not yet have a marketing authorisation for UK patients with life-threatening or seriously debilitating conditions if there is a clear unmet medical need. The medicine should be likely to offer a significant advantage over existing approaches and its benefits should outweigh its potential adverse effects. 1 If a drug meets the EAMS criteria, 2 a ‘positive scientific opinion’ is issued which describes the balance of risks and benefits based on the data available. The MHRA then publishes a Public Assessment Report (PAR) which summarises the clinical evidence, risks, benefits and uncertainties and supplies information about ongoing clinical studies and risk management measures. The EAMS period then starts.
The EAMS was the first UK fast-track scheme to be developed. Others (150-day National Assessment, Rolling Review, Innovative Licensing and Access Pathway, International Recognition Procedure and Project Orbis) emerged in the wake of the UK's departure from the EU and are perhaps intended to signal that the UK remains ‘open for business’. They offer various routes to faster licensing depending on the nature of the application or market readiness of the drug in question. There are no recent figures, but in 2020, the MHRA approved 36% of new drugs through accelerated pathways. 3 The US has fast-tracked drugs since 1988, and between 1992 and 2021, the FDA approved 278 drugs under accelerated approval. Initially, most were for infectious diseases but an increasing number of cancer drugs began to be fast-tracked, such that 83% between 2012 and 2021 were for oncology indications. 4 However, many expedited cancer drugs fail to demonstrate benefit in confirmatory trials5–7 and while – shockingly – some of these remain on the market, 7 a substantial number has been withdrawn in the United States of America due to a lack of overall survival benefit. 8 This clearly demonstrates the importance of confirmatory trials, 9 especially since pre-approval studies are often exploratory, using non-randomised, uncontrolled designs and surrogate outcomes. 8 Despite the obvious need for post-approval studies, however, there is little incentive for companies to conduct them; recruitment may be difficult, they risk financial loss if the results are negative, not all regulators require them and those that do fail to impose penalties for missing deadlines. Consequently, some expedited drugs remain on the market for years without good evidence of efficacy.10–12
Expedited drugs also raise safety concerns. In the United States of America, almost 57% received a black box warning (the FDA's highest safety-related warning) 13 and compared with regular drugs, they have a 48% higher rate of changes to boxed warnings and contraindications. 14 Drugs in a Canadian fast-track scheme were significantly more likely than those following a standard pathway to receive a serious safety warning and/or be removed from the market. 15 Several fast-tracked drugs have been withdrawn following deaths and severe adverse events9,13 including rofecoxib which was approved in the United States of America in 1999. In the 5 years until its withdrawal, there were an estimated 88,000–140,000 excess cases of serious coronary artery disease, 16 many of which were fatal.
Clearly then, patients taking expedited drugs face significant risks and uncertain benefits. For pharmaceutical companies, however, the advantages are manifold. Having drugs in fast-track schemes can generate early demand and help companies prepare for the global launch of their product.
17
EAMS drugs are more likely to receive a positive recommendation from the National Institute for Health and Care Excellence (NICE) than those going through standard pathways (58% vs. 43%, respectively).
18
Reviews of the EAMS19–24 all emphasise the potential gains to industry, including signalling to investors that therapy is on the ‘right track’, early advice and guidance from the regulator, access to patient populations and expert clinicians, early interaction with the NHS and NICE, a controlled environment for delivering treatments, the ability to collect real-world data, and a prioritised NICE appraisal. As for economic benefits, the same reviews note that the EAMS signals that the UK remains the ‘go to location’ for companies to develop and market their innovative therapies. When it comes to advantages for patients, however, most reviews simply
Ten years have passed since the introduction of EAMS in the UK. We examine how many drugs have passed through the scheme to date and for which indications, consider the quality of data upon which the MHRA based its decisions to expedite the drugs, and investigate their safety.
Methods
We examined all 51 scientific opinions available on the MHRA website as of 11 June 2024. These are ‘expired' opinions, that is, opinions on drugs that have gone through the EAMS and (typically) received market authorisation. 25 We extracted data on the drugs, companies and indications. From the PARs, we extracted data on pre-approval studies, including design, sample size and study outcomes, as well as ongoing studies and risk monitoring.
Preclinical studies
Information about preclinical studies is not included in the PAR so we searched for this on the Electronic Medicines Compendium (eMC). 26 This database holds up-to-date information about medicines licensed for UK use. Searches were conducted on 29–30 August 2024.
Safety
We searched i. the MHRA's interactive Drug Analysis Profiles (iDAPs) database for serious adverse reactions reported to the Yellow Card scheme and ii. the EMA's Eudravigilance database for reports of suspected adverse reactions, for each EAMS drug. Searches were conducted on 29–30 August 2024. Eudravigilance records suspected adverse reactions to medicines that have been authorised or are being studied in clinical trials in the European Economic Area. Since Brexit, it is no longer standard practice for the UK to submit data to Eudravigilance but we searched this database because many EAMS drug approvals predate Brexit and because it provides additional data about the EAMS drugs. We use the reports to provide an indicator of potential safety concerns about the EAMS drugs, not the specific EAMS indications. To enable comparisons between drugs, we divided the number of Yellow Card and Eudravigilance reports by the number of months since the drug was approved for the EAMS, and by the number of indications (
The MHRA lacks a system equivalent to the FDA's ‘boxed warnings’ that highlight potential safety concerns, 27 but it does require ‘additional Risk Minimisation Measures’ (aRRMs) for drugs associated with important risks (i.e. extra information for patients and health professionals).28,29 Another UK risk indicator is the Black Triangle, denoting that a drug's safety is not yet well established and that intensive monitoring is required, usually because the drug is new. Using the eMC, we investigated how many EAMS drugs had aRMMs or Black Triangle status (searches conducted on 22 August 2024).
Data extraction
Initially, the three authors extracted data independently but realised this was unnecessary as no judgements or interpretations were involved (data were simply cut and pasted). Consequently, one person (PP) conducted data extraction.
Results
The 51 scientific opinions relate to 51 different disease indications and 34 individual drugs. All drugs received marketing authorisation for their indications, with the exception of three withdrawn by the manufacturer at the time of analysis (raxone, cenegermin and nivolumab for advanced or recurrent gastric or gastroesophageal junction cancer after two or more systemic therapies). The analysis therefore focuses on 48 EAMS submissions relating to 48 indications and 32 individual drugs (nivolumab was approved for eight other indications).
The majority of indications were cancers (60%, 29), followed by severe atopic dermatitis (4), Pompe Disease (2), transthyretin amyloidosis (2) and 11 other diseases (Crohn's, myasthenia gravis, sickle cell, angioedema, spinal muscular atrophy, hyperoxaluria, Sars Cov-2, familial chylomicronemia, haemophilia A, hepatitis C, heart failure)
Drugs submitted to EAMS.

Studied in combination with another drug or drugs.
Information recorded in the PAR
At the time of the searches, PARs were missing for two indications (dupilumab for children aged 6–11 with atopic dermatitis; cipaglucosidase alpha for late-onset Pompe disease), leaving 46 PARs relating to 46 indications.
Study design
The design of the clinical studies conducted for the EAMS was not always clearly described in the PARs and the clinical data presented by companies varied in form and quantity, suggesting the MHRA does not demand this information in a consistent format. According to the PARs, only 3 (7%) of the 46 submissions were based on double-blind, placebo-controlled randomised trials. Eighteen PARs (39%) reported studies comparing the experimental drug with usual or another treatment (4 of which mentioned randomisation); 10 (22%) reported studies comparing the experimental drug with placebo; 8 (17%) reported studies in which no comparison was made; 5 (11%) reported ‘open label’ trials; 1 reported a study comparing different doses of the experimental drug and 1 reported a study comparing the experimental drug with no treatment. The average sample size was 654 (OSF File 1 https://osf.io/3wvb2?view_only=8aa44000a7414c999df977344f23ac35).
Study outcomes
Thirty-five studies (76%) used surrogate outcomes such as improvements in various scores, reductions in illness episodes, or changes in tumour growth, the latter being most common (

Studies using survival, surrogate, and patient-reported outcomes for 46 indications (missing data,
Ongoing studies and monitoring
Eight (17%) of the PARs clearly stated that there were no ongoing studies for the specific EAMS indication. For the remaining 38 (83%), companies stated that some kind of study or studies were ongoing, but it was not always clear whether these related to the specific EAMS indication (OSF File 3 https://osf.io/5ukj2?view_only=8aa44000a7414c999df977344f23ac35). All PARs included a risk management plan stating that healthcare professionals and patients would be given information about the medicine, that healthcare professionals would receive specific training and be asked to report adverse effects, and that patients would receive a card summarising important risks. The amount of detail provided by companies varied.
Preclinical studies
Animal studies were conducted for 31 drugs (preclinical data were missing from the eMC for isatuximab) and in vitro studies were also conducted for 11 (35%), mainly to test for genotoxicity (OSF File 4 https://osf.io/k7fuj?view_only=8aa44000a7414c999df977344f23ac35).
Safety
The drugs with the most Eudravigilance suspected adverse reaction reports were voxelotor (192 reports per month), sacubitril/valsartan (183), pembrolizumab (136), dupilumab (115), risankizumab (94), osimertinib (86), remdesivir (48), nivolumab (39) and alectinib (33). The remaining drugs had reports of 30 per month or less (Table 2, Figure 2). The drugs with the most Yellow Card reports for serious adverse drug reactions were sacubitril/valsartan (8 reports per month), venetoclax (5), rizankizumab (3.5), pembrolizumab (3), dupilumab (3), patisiran-NLP (3), remdesivir (2), avelumab (1.6), dostarlimab (1.5), polatuzamab vedotin (1.4) and emicizumab (1.4). Yellow Card reports were missing for voxelotor. The remaining drugs had reports of 1 per month or less (Table 2, Figure 3). In terms of fatalities reported to the Yellow Card system, venetoclax had the highest rate at 1.4 per month. Reports of fatalities for the remaining drugs were less than 1, although alectinib stands out at 0.9 per month (Table 2, Figure 3). Twenty (62%) of the drugs had Black Triangle Status and 15 (47%) had aRMMs in place. Taking into account both the Eudravigilance and Yellow Card schemes, 15 (47%) of the drugs raise safety concerns (Table 2, bold text) of which 6 (sacubitril/valsartan, pembrolizumab, dupilumab, risankizumab, remdesivir, alectinib) had elevated reporting rates under both schemes.
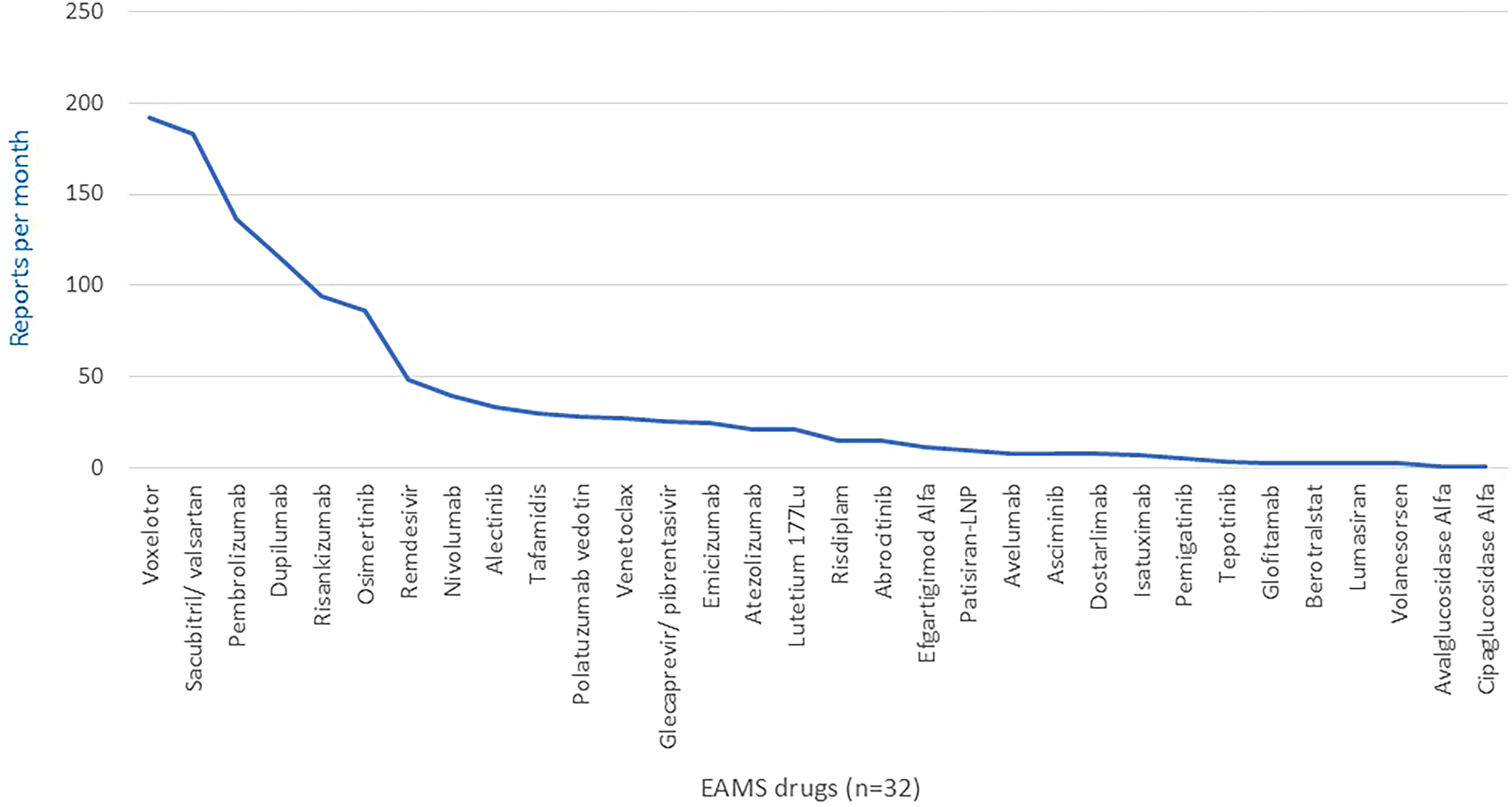
Monthly Eudravigilance suspected adverse reaction reports since EAMS commencement.

Monthly Yellow Card reports for serious adverse drug reactions and facilities since EAMS commencement.
Eudravigilance and yellow card reports.

Bold text indicates the drugs with elevated report rates.
Discussion
The pre-approval evidence upon which the MHRA based its decisions was limited; samples were generally small and most studies were non-randomised, unblinded comparisons. Most (76%) used surrogate outcomes. Eudravigilance and/or Yellow Card reports were elevated for almost half the drugs submitted to the EAMS. Most of the drugs had Black Triangle status and almost half had aRMMs.
To our knowledge, this is the first review of the EAMS to examine outcomes rather than process and to investigate the risks for patients rather than the benefits for industry. Nevertheless, limitations include the lack of a definitive ranking of risk and the lack of comparison with non-EAMS drugs. Another limitation is our use of spontaneous reporting systems as indicators of risk. Not only do these systems suffer from underreporting,30,31 they cannot establish a causal link between the drug and the reports. Nevertheless, the reports can signal real safety concerns: a month after our analysis, which found that voxelotor had the highest monthly rate of Eudravigilance reports, Pfizer withdrew voxelotor due to ‘an unfavourable imbalance in the number of vaso-occlusive crises and fatal events’. 32
Most of the EAMS submissions were for cancer, as has been found elsewhere, 4 and as with other fast-track schemes, 33 the majority of studies conducted for EAMS submissions used surrogate outcomes. While these shorten trial duration, enabling rapid approval, they correlate poorly with overall survival and lack relevance to patients who are more concerned with safety, survival and quality of life.6,9,11,13,34–38 Our finding that most of the studies conducted for the EAMS were not robustly designed has been found in the context of expedited schemes elsewhere8,33 as has the lack of consistency with which companies supplied methodological information and important details of study design. 33 The MHRA and other regulators do not appear to insist upon a specific format for supplying these data, although this would encourage companies to think more carefully about study design.
The high proportion of EAMS drugs with elevated pharmacovigilance reports, the ongoing Black Triangle status of most of the drugs and the high proportion with aRMMs all indicate a substantial risk to participating patients. The MHRA usually reviews Black Triangle status 2 years after marketing, so the fact that some EAMS drugs approved more than 2 years ago retain Black Triangle status indicates that their safety is not yet well established. Furthermore, the proportion of EAMS drugs with aRMMs (47%) is higher than for drugs in standard approval pathways (27% 29 ). Many EAMS drugs then, do not have an established safety profile, and several appear to have significant risks. Again, this is not uncommon for expedited drugs, as we noted in the introduction.13–15
The safety of expedited drugs might be improved with greater preclinical use of human biology-based technologies, the value of which is increasingly evident.39–43 The use of advanced
While the MHRA appears to rely on pharmacovigilance to monitor safety, it is less clear how it determines whether the EAMS drugs are effective, or more effective than existing treatments, since it does not demand post-approval trials. In response to our query about this, the regulator stated: ‘There is no requirement that EAMS products perform clinical trials at any part of their lifecycle,’ noting that companies perform Phase IV clinical trials at their discretion. However, it stated that if EAMS drugs receive a conditional marketing authorisation, conditions and expectations are stipulated on approval and the ongoing ability to market the product is subject to the provision of clinical or real-world data. 44
The FDA, however, not only requires post-approval trials to be conducted on expedited drugs to confirm or disprove earlier studies but also monitors
The approval rate for new drugs from Phase 1 is below 8%. 49 To expose the most vulnerable patients to expedited drugs that seem even less likely to be successful (given the lack of requirement for robust evidence) is far from compassionate. Cancer is the most common indication for accelerated approvals in the UK, yet cancer patients are a particularly vulnerable population, with evidence indicating that many feel pressured to take part in clinical trials (up to half of those in cancer drug trials appear to believe that participation is their only option). 35 Many cancer patients also seem unaware that for most, treatment prolongs life by around only 3 months.50,51 Would they take the drugs if truly aware of this, or of the potential severity of side effects, or the fact that they are more likely to die in hospital compared with patients who receive supportive care? 35 Pembrolizumab, an expedited immunotherapeutic for cancer, is the highest-grossing drug globally, with sales of $25 billion in 2023. 52 It is also one of the EAMS drugs with potential safety concerns according to our analysis. Are desperate patients, likely unaware of the uncertainty surrounding the risk-benefit profile of EAMs drugs, simply a money-making machine for pharmaceutical companies?
Conclusion
There needs to be honest communication with patients about the risks of participating in expedited drug trials. Fast-track schemes offer benefits to industry but the safety and well-being of patients cannot be assured while regulatory bodies fail to insist on robust evidence and when trial outcomes fail to reflect patients’ concerns. Given that one of the 15 drugs we highlighted as having potential safety concerns was recently withdrawn, it would seem prudent for the safety of the others to be reviewed as soon as possible, particularly since another, remdesivir for the treatment of COVID-19, continues to be administered to substantial numbers globally. We recommend that preclinical studies make greater use of human-relevant approaches, that clinical studies use patient-centred outcomes and are more robustly designed, and that the MHRA requires companies to provide consistent data in their submissions and demands post-approval trials. Future reviews of the EAMS need to centre patients’ concerns. The adoption of such measures will benefit us all since fast-tracked drugs show every sign of becoming the norm.
Footnotes
Competing interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no external financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable: this study does not involve human or animal participants. It is an analysis of publicly available data.
Guarantor
Pandora Pound is the guarantor for this paper. She accepted full responsibility for the conduct of the study, had access to the data, and controlled the decision to publish.
Contributorship
Kathy Archibald conceived the idea for the paper. Pandora Pound conducted the analyses and drafted the paper and revisions. Rebecca Ram and Kathy Archibald critically reviewed the paper for intellectual content and accuracy. All three authors approve the final version and agree to be accountable for all aspects of the work and to resolve any issues related to its accuracy or integrity.
Provenance
Not commissioned; peer-reviewed by Joel Lexchin.
Consent to participate
Not applicable: this study does not involve human participants.
Consent for publication
Not applicable.
