Abstract
Myocardial infarction is considered the most common cause of left ventricular pseudoaneurysm. Large missed pseudoaneurysm of the left ventricle incidentally diagnosed and treated conservatively.
Case description
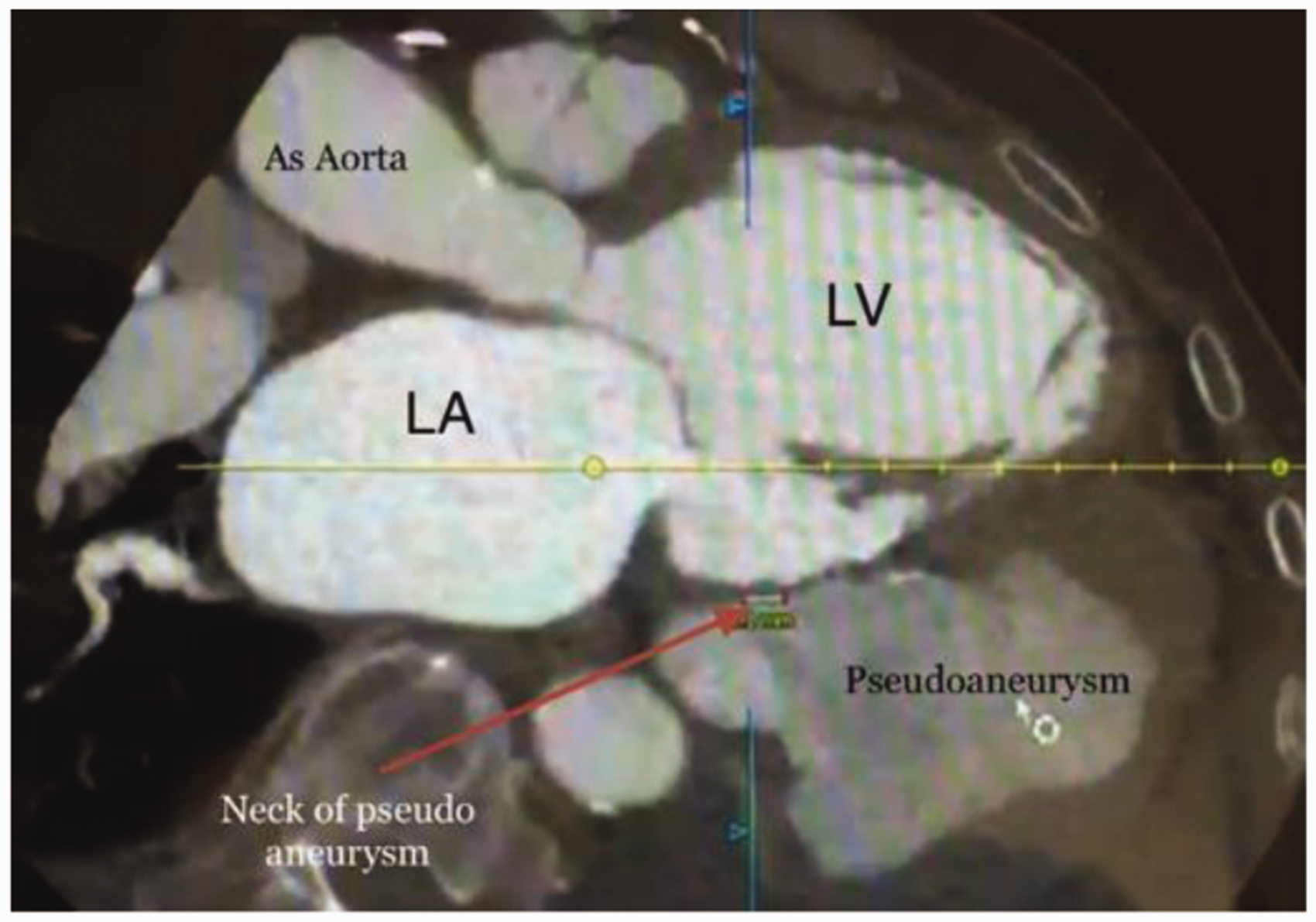
An 82-year-old male patient with a history of type 2 diabetes mellitus, hypertension, chronic obstructive lung disease, coronary artery disease, coronary artery bypass grafting in 2005, and ischaemic dilated cardiomyopathy with reduced systolic function presented with a picture of congestive heart failure. He had frequent admissions to different hospitals with similar picture and discharges after adjustment of his heart failure medications. The patient had no history of recent coronary events. His mobility had limited him to his home. After starting intravenous diuretics, he improved clinically. A routine transthoracic echocardiography showed dilated left ventricle with global hypokinesia, reduced systolic function of the left ventricle and an estimated ejection fraction of 30%. There was also a dilated left atrium and evidence of moderately severe mitral regurgitation. These parameters were similar to previous echocardiographic findings four months prior. Surprisingly a small endocardial defect in the inferolateral wall (posterior wall) of the left ventricle was observed, followed by a large oval anechoic area measuring 7.5 × 6.0 cm (Figure 1) containing a thrombotic mass surrounding its thin wall (Figure 2). This picture was suggestive of a large left ventricle pseudoaneurysm with a narrow neck of 5 mm. A colour-flow Doppler on the neck of the left ventricle pseudoaneurysm showed turbulence flow (Figure 3). The patient underwent a cardiac computed tomography and a computed tomography coronary angiography, which revealed an outpouching sac measuring 7.6 × 5.9 cm with a narrow neck and thrombotic mass adjacent to the wall of the sac and was strongly suggestive of a pseudoaneurysm (Figures 4 and 5). Patent grafts with severe native coronary artery disease were obtained on computed tomography coronary angiography. A surgical approach was considered the most effective way to avoid the dramatic scenario of the progression to spontaneous rupture; however, patient’s age and comorbidity limited the surgical option, and a conservative approach was taken.
Transthoracic echocardiography: Modified parasternal long axis view. Myocardial defect is located in the inferolateral wall (posterior wall) of left ventricle and Red arrow indicates neck of the cavity that connected to left ventricle. Green arrow indicates pseudoaneurysm. LA: left atrium; LV: left ventricle. Transthoracic echocardiography: Short axis view – pseudoanerysm seen behind the inferolateral wall of left ventricle (green arrow). A huge thrombus (T) lying out inside pseudoaneurysm. LV: left ventricle. Transthoracic echocardiography: Modified parasternal long axis view. Colour Doppler shows a systolic flow between left ventricle and pseudoaneurysm (red arrow) through the narrow neck (yellow arrow) of pseudoaneurysm. DA: descending aorta; LV: left ventricle; LA: left atrium. Cardiac computed tomography demonstrates the communication between left ventricle and pseudoaneurysm through the narrow neck (red arrow) measured 7 mm. LA: left atrium; AS aorta: ascending aorta; LV: left ventricle. Axial cardiac computed tomography shows pseudoaneurysmal cavity in relation to left ventricle. LA: left atrium; LV: left ventricle.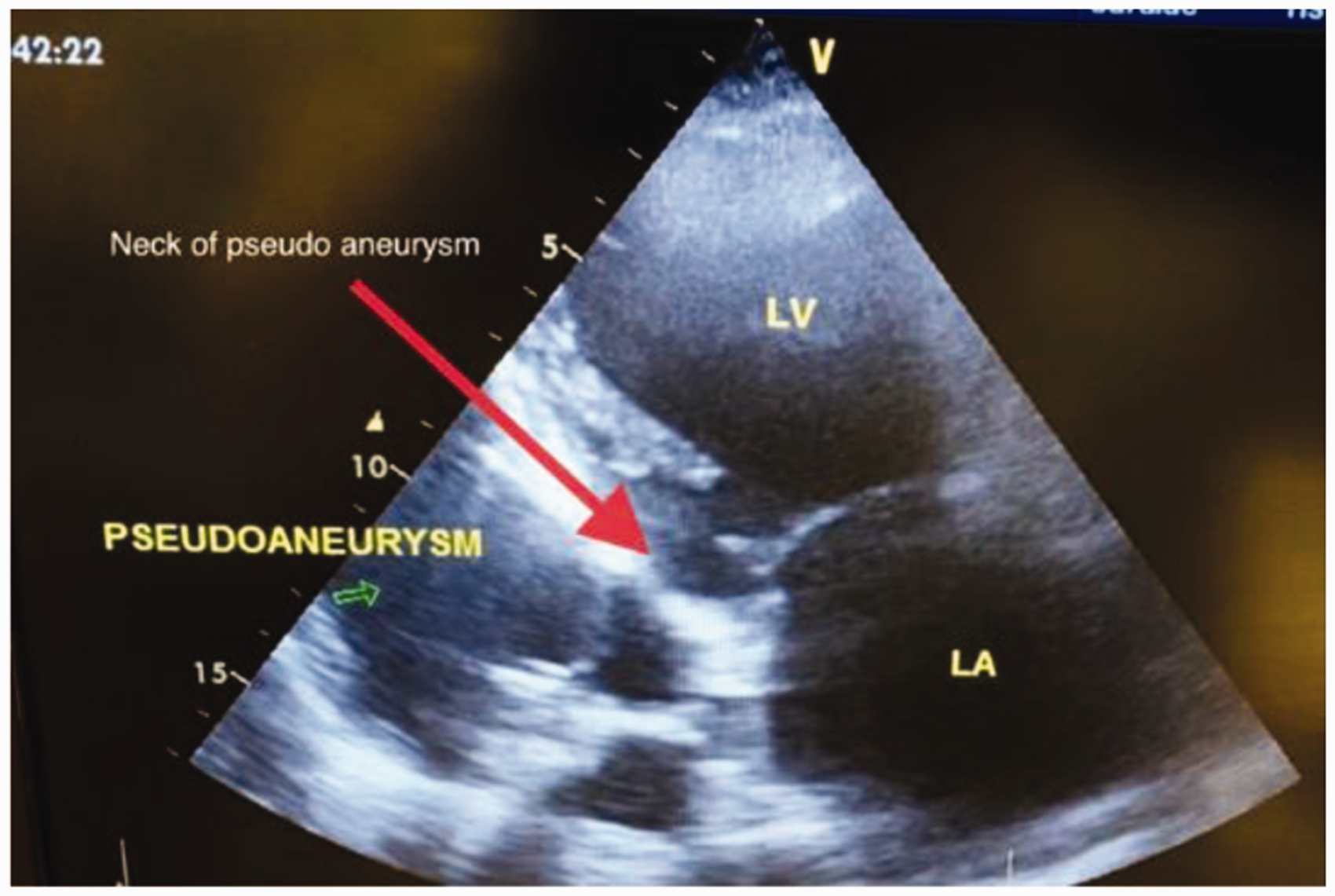



Discussion
Pseudoaneurysm of left ventricle is a rare and life-threatening complication of myocardial infarction. Pseudoaneurysm results from a contained myocardial rupture by overlying adjacent pericardium and scar tissue forming a wall containing only epicardium, pericardium and haematoma without any myocardium or endocardium. 1 Between 0.2 and 0.3% of myocardial infarction may become complicated with formation of left ventricle pseudoaneurysm, especially in elderly and male patients and myocardial infarction is considered the most common cause of left ventricle pseudoaneurysms. Cardiac surgery, infective endocarditis, previous thoracic trauma, tuberculosis, rheumatoid arthritis, Kawasaki’s disease and Behçet’s syndrome can also be an aetiology of left ventricle pseudoaneurysm. 2 Two case reports found in the literature demonstrated a left ventricle pseudoaneurysm with an unknown aetiology in young patients. 3
By definition, the wall of left ventricle pseudoaneurysms do not contain endocardium or myocardium, as opposed to true aneurysms where the wall consists of all the layers (endocardium, myocardium and epicardium). Due to such cardinal difference in wall structure between them, differentiation is essential to determine an appropriate method of management and can explain the high risk of rupture reaching 30–45% 4 if the pseudoaneurysm is left untreated surgically. However, it is not always easy to differentiate between these two entities. Left ventricle pseudoaneurysms typically have a neck narrower than the diameter of the aneurysm and are more often located on the posterior, lateral or inferior surface of the left ventricle and related more to inferior myocardial infarction. In contrast, a wide neck is characteristic of true left ventricle aneurysms and often seen in the anterior wall and apex. More than 10% of patients with left ventricle pseudoaneurysms are asymptomatic. Common reported symptoms in these patients are chest pain, dyspnoea, syncope or heart failure, and these are common symptoms in patients at risk, such as those with a history of myocardial infarction or previous cardiac surgery. Neither the symptoms nor signs are sensitive to left ventricle pseudoaneurysm. 5 In a series of 52 patients with pseudoaneurysms, 48% were diagnosed incidentally. 6 This obligates the use of imaging to diagnose or confirm suspected cases. A reasonable initial imaging modality is transthoracic echocardiography. In suspected cases, non-invasive modalities such as cardiac computed tomography and cardiac magnetic resonance imaging can be used since both can provide a high resolution and visualisation of each cardiac segment at any plane. Historically, the imaging modality of choice has been angiography of the left ventricle and coronary arteries. Failure to undergo surgical treatment of left ventricle pseudoaneurysm for any reason will keep the risk of pseudoaneurysmal rupture high, even after many years of diagnosis. 7 The treatment of choice in left ventricle pseudoaneurysm is a surgical option in most cases to prevent dramatic spontaneous rupture with subsequent tamponade of pericardium and cardiac arrest, whereas in true aneurysms of the left ventricle, conservative treatment is dominant. 7 In our case, although the patient had frequent admissions as a case of decompensated heart failure, it appears that a chronic large pseudoaneurysm of the left ventricle was missed before and incidentally diagnosed in the last admission. Although the surgical approach is the best option to avoid a dramatic scenario of pseudoaneurysmal rupture, in our case, the age, comorbidity and limited mobility of the patient limited the surgical option, and the conservative approach applied. Patient discharged home in compensated form after good diuresis and optimisation of heart failure medication. Follow-up in the outpatient clinic after four weeks revealed him in a good health with no increase of his weight since discharge.
Conclusion
Pseudoaneurysm of left ventricle is a rare and life-threatening condition. Myocardial infarction is considered the most common cause of left ventricle pseudoaneurysms. This case demonstrates a large chronic missed left ventricle pseudoaneurysm that was not suitable for surgical treatment and treated conservatively.
Footnotes
Declarations
Competing Interests
None declared.
Funding
None declared.
Ethics approval
Written informed consent for publication was obtained from the patient.
Guarantor
OM (Ossama Maadarani).
Contributorship
▪.
Acknowledgements
OM conceived the idea for the case report and wrote it. ZB provided the editorial input.
All authors were involved in managing the patient bedside.
Provenance
Submitted; peer reviewed by Arshiya Singhal.
