Abstract
Rare case of concurrent subarachnoid haemorrhage and ST elevation myocardial infarction, highlighting the importance of detailed history in an emergency.
Case history
A previously fit 66-year-old Caucasian man was admitted to hospital following an out of hospital cardiac arrest. He complained of chest pain followed by dizziness and lost consciousness thereafter. He received bystander cardiopulmonary resuscitation for 10 min. Spontaneous circulation returned following a single defibrillation and was found to have ST elevation in leads V1, V2 and V3 with reciprocal changes in leads II and III on the 12 lead electrocardiogram. Primary percutaneous coronary intervention pathway was activated, and he was transferred to the angioplasty centre. Left heart catheter (Figure 1(a)) revealed a thrombotic subtotal occlusion of the mid-left anterior descending artery which was treated with a single 3.0 mm× 32 mm synergy drug eluting stent. This was optimised and post-dilated with a 3.5 mm non-compliant balloon, with excellent angiographic result. Thrombolysis in myocardial infarction grade 3 normal flow was restored. The rest of the coronary arteries were unobstructed.
(a) Thrombotic subtotal occlusion of the mid left anterior descending (LAD) artery on coronary arteriogram (right anterior oblique caudal view), (b) Computer tomography head post-percutaneous coronary intervention confirming the presence of a subarachnoid haemorrhage mainly in the interhemispheric fissure (white arrow) and (c) digital subtraction angiography of the brain revealing a 6 mm aneurysm of the anterior communicating artery (white arrow).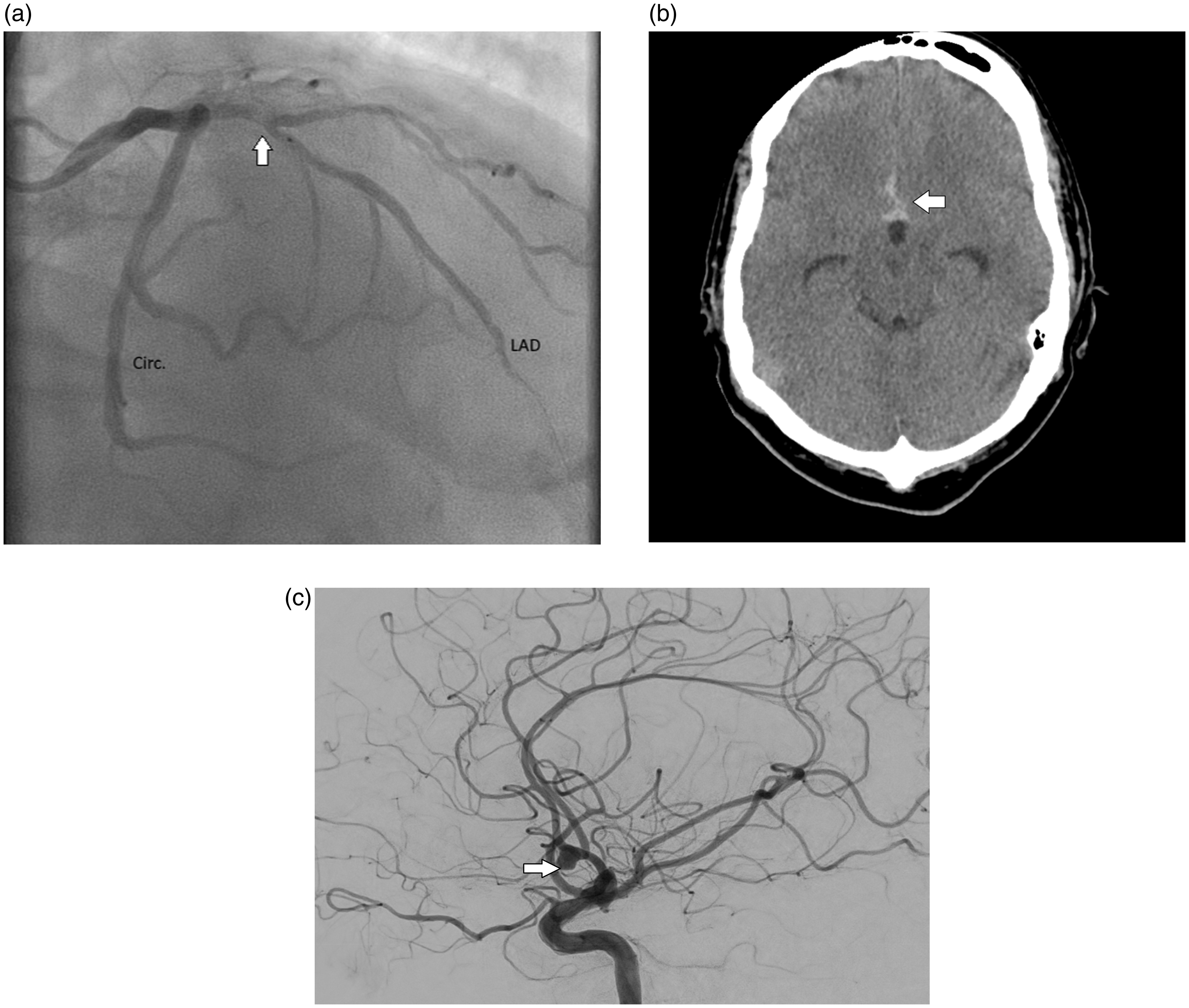
Aspirin (75 mg a day) and ticagrelor (90 mg twice daily) were administered post-primary percutaneous coronary intervention along with 18 hours of Eptifibatide (Glycoprotein IIb/IIIa receptor blocker). Transthoracic echocardiogram post-primary percutaneous coronary intervention showed moderate impairment of left ventricular systolic function (ejection fraction 35%) and mild-to-moderate aortic regurgitation.
The next day, the patient described severe generalised frontal headache with nausea and photophobia. On further questioning, it transpired that the headaches actually preceded the chest pain and cardiac arrest. The full blood count, coagulation, renal and liver function were normal (haemoglobin 132 g/L, white blood cells 9.3 × 109/L, platelets 229 × 109/L, neutrophils 7 × 109/L, international normalised ratio 1.0, sodium 136 mmol/L, potassium 3.6 mmol/L, urea 6.5 mmol/L, creatinine 72 µmol/L, estimated glomerular filtration rate (eGFR) >90, bilirubin 17 µmol/L, alanine transaminase 26 U/L, alkaline phosphatase 72 U/L, albumin 30 g/L). Urgent computer tomography head and computer tomography cerebral angiogram were performed (Figure 1(b) and (c)). Computer tomography brain confirmed the presence of a subarachnoid haemorrhage mainly in the interhemispheric fissure, while computer tomography angiogram revealed a 6 mm aneurysm of the anterior communicating artery. No cerebrospinal fluid sample was obtained for xanthochromia as radiological imaging was significantly diagnostic and any lumbar puncture attempt would pose a significant bleeding risk due to the dual anti-platelet therapy and eptifibatide. Dual antiplatelet therapy was not stopped due to the high risk of stent thrombosis. Coil embolisation of the aneurysm was performed with platelet cover and excellent occlusion of aneurysm was achieved.
Patient made a good gradual recovery and four weeks after initial cardiac arrest was discharged from hospital.
Discussion
Subarachnoid haemorrhage is commonly associated with electrocardiograph and echocardiographic changes. A patient with SAH may have ST segment changes, T-wave inversion, or prolongation of the corrected QT interval on their electrocardiograph, an elevated troponin level and transient global or segmental hypokinesis of the left ventricle on echocardiogram despite having unobstructed coronaries. The precise mechanism of these electrocardiograph changes is not clear; however, a possible explanation is that catecholamine excess during an subarachnoid haemorrhage causes a neurogenic stunned myocardium, mimicking myocardial infarction. 1
We present a case which highlights the rare correlation between true ST elevation myocardial infarction and subarachnoid haemorrhage. Only two other cases of subarachnoid haemorrhage with obstructive coronary arteries have been reported and in both cases, the predominant symptom was headaches with meningism. 2 This gentleman had no neurological signs and the predominant symptom was chest pain.
We postulate that the subarachnoid haemorrhage was the initial event, causing secondary plaque rupture in a patient with pre-existing coronary artery disease. Our case supports cardio–cerebral interaction beyond the hypothalamic–pituitary–adrenocortical and sympatho-adrenomedullary axes. Noradrenaline surge from hypothalamus ischaemia and vasospasm of epicardial vessels are probable hypotheses. Noradrenaline stress response causing real cardiac injury is supported by cases where takotsubo cardiomyopathy is seen following a subarachnoid haemorrhage.3–5 However, the pathophysiology behind true ST elevation myocardial infarction following subarachnoid haemorrhage has not been established.
The learning point from this case is to continue to obtain a detailed history even in the setting of a primary percutaneous coronary intervention pathway where time critical interventions need to be made. There are other causes of ST elevation such as acute pericarditis, left ventricular aneurysm and takotsubo that a physician needs to consider in the context of cardiac arrest. 6 This case also highlights that bystander cardiopulmonary resuscitation helps to reduce cognitive impairment and increases the chances of survival.
