Abstract
Objectives
To evaluate the short-term 24 h urinary excretion of platinum, arsenic, selenium, magnesium and zinc in patients with lung cancer and with cancer other than lungs treated with cisplatin or/and carboplatin from Antofagasta, Chile.
Design
Urine measurements of Pt and Se were made by inductively coupled plasma optical emission spectrometry, As by hydride-generation atomic absorption spectrometry and Mg and Zn by means of flame furnace atomic absorption spectrometry.
Setting
All samples were provided by the Oncological Centre of Antofagasta Regional Hospital (Region of Antofagasta, Chile).
Participants
Ninety 24-h urine samples from cancer patients after the infusion of Pt-base drugs and 10 24-h urine samples from cancer patients not treated with metal-base drugs.
Main outcome measures
Concentrations of Pt, Se, As, Zn and Mg coming from 24-h urine samples.
Results
Pt excreted was not significantly different between patients with lung and other cancers treated with cisplatin. The excretion of Mg, Zn and Se was greater than As. Then, Pt favours the excretion of essential elements. For lung and other types of cancers treated with drugs without Pt, excretion of Mg, Zn and Se was also greater than that of As, suggesting antagonism Mg-Zn-Se–anti-cancer drug relationship.
Conclusions
The amounts of Mg, Zn and Se excreted were greater than for As either with or without Pt-containing drugs, suggesting antagonist Mg-Zn-Se–anti-cancer drug relationships. The excretion of As, Mg, Zn and Se is induced by Pt. Knowledge obtained can contribute to understanding the arsenic cancer mechanism and the As-Mg-Zn-Se-Pt inter-element association for lung cancer and other types of cancer.
Keywords
Introduction
The deterioration in the environmental health of people living in Antofagasta, Chile could be associated with medical geology characteristics of this part of the Atacama Desert ecosystem and the extensive mining related activities carried out in this geographical zone that exposes the inhabitants to an increasing risk of cancer, cardiovascular diseases and neurological diseases. Geological structures in certain places on Earth influence human health1,2 and the underlying cause could be the multi-metallic synergy of heavy metals. 3 Arsenic (As) is one of the main environmental stressors that affects the quality-of-life of the population4–7 in the Antofagasta region in Chile; Antofagasta is a highland–coastal desert ecosystem in which large-scale mining activities have been conducted for a long time. 8 These activities have affected the ecosystems and the quality-of-life of the people living there. 9
Cancerous and non-cancerous diseases associated with As contamination are known in several parts of the world. 10 However, this association reaches alarming levels in the Antofagasta region, as evidenced by the incidence of cardiovascular diseases 5 and cancer.4,7,11 The mortality rates due to cancer in the Antofagasta region are higher than those in any other population exposed to As in the world. 12 Most therapeutic treatments are based on the action of platinum group metals under the form of metal-based drugs.13–15
There is a large body of evidence about the trace element ecosystem impact and the cancer development. 3 The toxic effect of metals are involved in a variety of diseases, some related to heavy metal stress and others to the imbalance of essential metals due to human multi-elemental exposure.3 So, both approaches need multi-element hypothesis.
The precious heavy metal Pt is of considerable interest because of its emission from automotive catalysts, its use in prostheses and in Pt-based anti-tumour drugs.16,17 Administration of Pt-based drugs has an impact at the cellular level, affecting metabolism, homeostasis and physiology in humans.18,19 The main side effects of the application of Pt drugs are loss of hearing (ototoxicity), kidney dysfunction (nephrotoxicity) 20 and resistance to anti-cancer drugs. 21 As in other anti-cancer drugs, the principal disadvantage of these treatments is the metastasis. 22
Despite of the advances in cancer treatments, the cytotoxicity of chemotherapies continues to be an important problem. Problems arising from erroneous calculation of the Pt-based dose drugs when the body surface area (BSA) method is applied,23 which can influence the Pt inter-element interactions with elements such as Mg, Zn, As and Se and the corresponding metal-creatinine ratios values. Nevertheless, failure in correctly applying the BSA method does not account for the marked inter-patient variation in drug handling. Therefore, drug effects such as toxicity or consequences due to unexpected under-dosing are unpredictable. 24
Most toxicology studies focus on a single agent, but this approach does not reflect real-world scenarios in which humans are exposed here and now to multiple chemicals. The toxicity of an element may be reinforced or weakened through its inter-element interactions. Such interactions are, in general, classified as being ‘adversely additive’, ‘synergistic (greater than additive)’ or ‘antagonistic’ (less than additive or even acting as antidotes to one another). Antagonistic behaviour between As-Se and Pt-Se pairs has been observed in humans.25,26,27–29 Hence, the level of Se in urine30,31 could be particularly of interest when assessing exposure to heavy metals. Inasmuch cisplatin and carboplatin caused suppression of anti-oxidant enzyme protection via mitochondrial dysfunction.32,33 Selenium would strengthen the capacity of antioxidant-protecting components of cells (e.g., nucleic acids) from damage of reactive oxygen species22,30 through antioxidant Se-based and Zn-based enzymes such as glutathione peroxidase 31 and thioredoxin reductase, which require Se(IV) for their expression28,34 and Zn-superoxide dismutases. Several hypotheses have been stated to account for experimental data, which indicates that Se compounds 35 have anti-cancer properties. However, although evidence has been accumulating in support of the ‘Se chemo protective hypothesis’, definitive proof is lacking.36–40 Otherwise, the vulnerability of the antioxidant-protecting components of cells can also result in side effects, such as metastasis37,38 and resistance to Pt-based drugs. 21
Evidence suggests that As inhibits the potential anti-cancer effects of Se, and also its interaction with essential trace elements and other heavy metals such as copper, zinc, cadmium, mercury, tin, lead, nickel, cobalt, antimony, silver, gold and molybdenum.39,41 A study on the urinary excretion of Se and As in pregnant women from northern Chile 42 found a significant association between the urine concentrations of these elements. Drugs that contain Pt (e.g. cisplatin and carboplatin) have been shown to increase the biliary excretion and distribution of Se in tissues, but Pt has not been shown to significantly influence the urinary or respiratory excretion of Se. 43 The distribution of Se in tissues is altered by cisplatin, which results in high concentrations of Se in the liver, kidney and plasma, and low levels of Se in the testicles and brain. The underlying mechanism of Se homeostasis is incompletely understood. 20
Studies looking at urinary excretion of Pt among patients treated with cisplatin have been conducted on average between 6 months and 16.8 years after treatment. 44 In the present work, we investigated the excretion of Mg, Zn, As, Se and Pt in 24-h urine samples after the infusion of Pt based-drugs to cancer patients. Short-term monitoring of urine after the first cycles of treatment with Pt-based drugs could be a good approach for studying the inter-element interaction among Mg, Zn, As, Se and Pt in patients whose cancer was probably triggered by As exposure. Such knowledge could contribute to better understanding of the mechanisms of side effects 21 and resistance to Pt-based drugs, 45 improving the calculation of optimal doses of anti-cancer drugs 23 and understanding the resistance (or permissiveness) to metastasis. 46
In this work, we study the amounts of magnesium, zinc, arsenic, selenium and platinum in five groups of urine samples of patients with cancer coming from the Region of Antofagasta – Chile. Information was obtained about the inter-element relationships Mg–Zn 47 , Zn–Se 48 , Zn–As 49 , As–Se, Zn–Pt and As–Pt pairs in short-term 24-h urine excretion of persons with cancer from Antofagasta – Chile; platinum has been supplied as cisplatin and/or carboplatin.
Materials and methods
The work was carried out in accordance with the Health Service of Antofagasta (Antofagasta, Chile) according to ORD 4391; the study protocol was also authorised by the Ethics and Biosafety Committee of Antofagasta University.
Study population and collection of samples
Ten 24-h urine samples from cancer patients of the Antofagasta region were collected before Pt-based drugs chemotherapy was initiated. Ninety 24-h urine samples from cancer patients were collected immediately after the infusion of the Pt-based drugs; and 10 24-h urine samples were collected from patients subjected to chemotherapy not involving metal-based drugs. All samples were provided by the Oncological Centre of Antofagasta Regional Hospital (Region II, Chile). The 90 cancer patients are distributed in the following cases: 32 lung cancer, 16 ovarian cancer, 12 cervical-uterine cancer, 8 testicular cancer, 5 thymus cancer, 3 oesophageal cancer, 3 gastric cancer, 3 Hodgkin lymphomas, 2 vagina cancer, 2 bladder cancer and one each of gallbladder, tonsil, salivary gland and osteosarcoma cancers. The dose calculation for chemotherapy had been made by the BSA method. 24 Three of the four patients with ovarian cancer initially treated with cisplatin were again treated with cisplatin after 28 days, whereas the fourth patient was treated with carboplatin. Cancer affecting other parts of the human body than lungs was categorised as ‘other cancers’; only patients in this group were treated with carboplatin. Samples were collected between May and August 2005.
Chemicals, solutions, materials and instruments
The chemicals employed were Instra (J.T. Baker, Phillipsburg, NJ, USA) and Suprapur (Merck, Whitehouse Station, NJ, USA), both of which were of trace-element grade. Solutions were prepared in deionised water that was subsequently distilled in quartz (conductivity, <0.5 µMHO/cm at 25℃). The deionisation was undertaken in a deioniser with two ion-exchange cartridges – a Metex 01506-45 and a Research 01506–35 (Cole-Parmer, Waltham, MA, USA) – connected in tandem.
The glass materials used were sequentially treated for 24 h with 2-M solutions of hydrochloric acid and nitric acid prepared from Merck pro analysis quality grade chemicals. After being washed with water, the glass materials were treated for another 24 h with a 0.025 M disodium salt of ethylenediamine tetraacetic acid solution prepared from Titriplex III (Merck), after which they were rinsed with water.
Pt(II) standard dilutions were prepared from a primary solution of Certipur (Merck) and those of Mg, Zn and Se(IV) were prepared from Titrisol (Merck) primary solutions; the concentration of primary solutions was 1000 mg L–1. An intermediate 50 mgL–1 standard solution was made, which was used to prepare the working dilutions. Standard As solutions were prepared from a 1000 mgL–1 primary solution of disodium arsenate Titrisol (Merck), from which a 500 mgL–1 intermediate standard dissolution was prepared for the working dilutions. To generate arsenic hydride, 10 M chlorhydric acid solution was used, which was prepared from Instra (J.T. Baker). A 3% p/v sodium borohydride solution was prepared in a 1% p/v sodium hydroxide solution, both of which were Merck pro analysis grade quality chemicals.
Urine samples were transported in a cooler at 4° C–5° C to an acclimatised and pressurised Pre-treatment Sample Room equipped with a laminar-flow hood (Labconco Purifier, class II; Labconco, Kansas City, MO, USA). Samples were microfiltered in a Nalgene system through a Millipore cellulose ester membrane (pore size, 0.45 µm; diameter, 47 mm; Millipore, Bedford, MA, USA). Samples were then stored at −20° C. Immediately before analyses of metals were carried out, the samples were thawed and again microfiltered, this time through a Millipore cellulose ester membrane of pore size 0.22 µm protected by a borosilicate glass pre-filter (Advantec-MFS, Japan) (pore size 0.7 µm).
Matrix-matched ionic strength surrogate urine was prepared by diluting subtidal seawater twice with deionised glass-distilled water. As a reference, the measured average salinity of urine samples was 13 parts per thousand. This was measured in a previously calibrated Orion Ion Meter 1260 (Thermo Scientific, Waltham, MA, USA).
Urine samples digestion
Urine samples and controls were mineralised using the wet method in Teflon bombs with 10 ml of nitric acid (Instra; J.T. Baker, USA) for 2 h at 150℃ in a homemade ceramic oven with an internal temperature sensor and external temperature control. After the samples were cooled, the oven was opened and an additional 0.25 mL of perchloric acid and 0.50 mL of sulphuric acid were added to the Teflon bombs with the digested samples; both acids were of Suprapur grade (Merck). Samples were then reheated under the same conditions of time and temperature as described above. Finally, samples were transferred to Erlenmeyer flasks and heated at 300℃ under a gas extraction hood to eliminate excess acids. Sample volumes were made up to 50 mL with deionised water, and then micro-filtered through a 0.22-µm Millipore membrane with a 0.7-µm fibreglass pre-filter (Advantec-MFS, Japan).
To determine total As concentrations, 5-mL aliquots of samples and controls were digested to semi-reflux under a temperature programme between 85℃ and 300℃. 44 After cooling, samples and controls were diluted volumetrically with 0.5 M HCl prepared from Instra quality grade chemical (J.T. Baker, USA).
Determination of total Pt and Se in urine through inductively coupled plasma optical emission spectrometry
With the aim of meeting the matrix-matching conditions, aliquots of three samples of digested urine were titrated with NaOH secondary standard solution, yielding an average acidity equivalent to a pH of 2. To make compatible the matrix-matching conditions of the analytical blanks, seawater was diluted 8-fold, 10 mL of sample was diluted to a final volume of 50 mL and, when necessary, the solution was adjusted to pH 2 with Instra nitric acid (J.T. Baker, USA). The bimetallic Pt–Se standard was also subsequently prepared in this medium.
Instrumental optimised conditions for urine analysis of Mg, Zn, As, Se and Pt by means HGAAS, FFAAS and ICP-OES.

HGAAS: hydride-generation atomic absorption spectrometry; ICP-OES: inductively coupled plasma optical emission spectrometry; FAAS: furnace atomic absorption spectrometry
Determination of total As, Mg and Zn concentrations in urine by hydride-generation atomic absorption spectrometry and flame furnace atomic absorption spectrometry
Determination of As was undertaken using an atomic absorption spectrophotometer (GBC 909 AA; GBC Victoria, Australia) equipped with a continuous-flow hydride generator (GBC HG 3000) and a electro-thermal atomization system (GBC EHG 3000). GBC quartz cells and an As-boosted current lamp from Photron (Narre Warren, Victoria, Australia) were also used. The optimised conditions for measuring As through hydride-generation atomic absorption spectrometry (HGAAS) are presented in Table 1. Multiple-standard-addition methodology was also applied and the additions were made from an intermediate 500 mg/L standard solution prepared in 0.5 M HCl from a primary standard arsenic solution (Titrisol, Merck). Additions from this solution were made to surrogate-sample urine, which had been made up to 2.0 µg/L in As. The addition volumes were 50, 100, 150 and 200 µL; the concentrations added were therefore 2, 4, 6 and 8 ng/mL, respectively.
For Mg and Zn, urine analysis FFAAS was applied 50 in the GBC 909 AA atomic absorption spectrometer employing a GBC quartz atom trap cell (Australia) and Photron hollow cathode lamps (Australia). The optimised conditions for Mg and Zn analysis are presented in Table 1. For Mg, a standard dilution of 100 mg/L in 0.3 M HCl was prepared from the Titrisol standard solution (Merck). From this solution, the standard measurement dilutions were prepared using seawater as ‘surrogate urine’. The analytical concentrations for the calibrating curve standard dilutions were 5.0, 10.0, 15.0, 20.0 and 25.0 mg/L. For Zn, the multiple standard additions methodology was also applied. In this case, the additions were also made over the ‘surrogate urine’ from one zinc standard dilution of 100 mg/L prepared in 0.3 M HCl from the corresponding Zn Titrisol standard solution (Merck). Surrogate urine was made 400 µg/L in zinc and the additions were of 100, 200, 300 and 400 µL. So, the added concentrations were of 400, 800, 1200 and 1600 ng/mL, respectively.
The baseline was adjusted with the analytical blank solution, i.e. diluted surrogate urine. QC tests were conducted to evaluate the recovery of Mg, Zn and As from Mg-, Zn- and As-spiked real samples, measurement of secondary standards were also made. Urine samples of patients underwent the abovementioned procedures and analytical methods.
Urine and serum creatinine levels and calculation of creatinine clearance
Levels of urine and serum creatinine were measured using the Jaffé method. 51 Creatinine clearance rate (CrCl) was calculated using the Cockcroft–Gault equation. Serum creatinine is the blood clearance or endogenous blood creatinine depuration, 52 and their values were provided by the Oncological Center of Antofagasta Regional Hospital. Urine and serum CrCl values are used to evaluate renal dysfunction. The estimated CrCl for normal renal function should be >80 mL/min. 53
Statistical analyses
Statistical analyses were conducted using the Statistica 10.0 MR1 software (StatSoft, Tulsa, OK, USA). p < 0.05 was considered significant.
Results and discussions
Determination of Mg, Zn, Pt, As and Se
The ICP-OES calibration curves for total Pt and Se concentrations in urine were linear. In the case of Pt, the parameters of the calibration equation were intercept = 93.45 counts s–1, slope = 1.079 counts × s–1/concentration and r = 0.9998. For Se, the parameters were intercept = 55.47 counts s–1, slope = 1.720 counts × s–1/concentration and r = 0.9880.
The resulting standard-addition methodology parameters for As urinary analysis were also analytically suitable: intercept = 0.0375 µA, slope = 0.01615 µA/concentration and r = 0.9977.
Traceability and quality control of urinary analyses of Mg, Zn, As, Se and Pt in cancer patients.
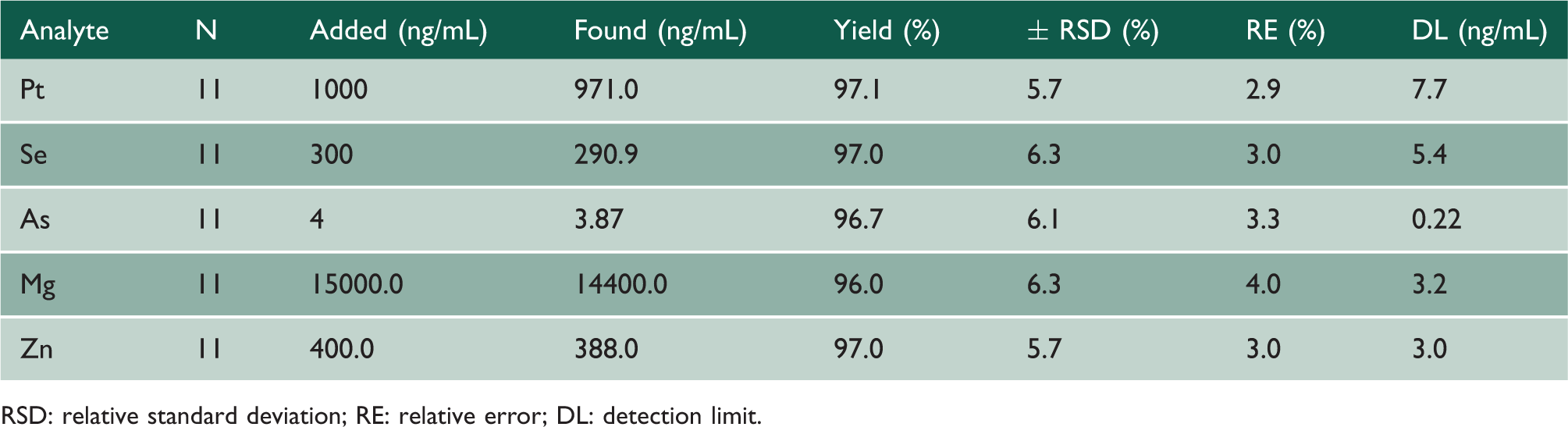
RSD: relative standard deviation; RE: relative error; DL: detection limit.
The analytical validation and QC results for Pt and Se by ICP-OES, As by HGAAS, Mg and Zn by FFAAS analyses in 24-h urine samples of cancer patients indicated that the techniques and methodologies applied in this work were analytically suitable. Surrogate urine prepared with clean seawater was important for optimisation of the analytical parameters and appropriate representativeness of the results.
Inter-element relationship of Mg, Zn, Pt, As and Se in urine of cancer patients from the Antofagasta Region of Chile treated with Pt-based drugs
Knowledge in this area is mainly based upon single elements; a better-known case is that of As, which has been associated with several diseases, including cancer.54–57 However, recent developments in multi-elemental analytical techniques and methodologies have enabled exploration of inter-elemental relationships and toxicities of essential elements and heavy metals, which coexist in environmental and biological matrices.58–63 However, it has become increasingly clear that simple, single-element models are often inadequate to explain the relationship between disease and nutrition, element toxicity or deficiency of elements in food. Improved understanding of the synergistic and/or antagonistic interactions between trace elements is needed. 64 A multi-elemental hypothesis may be a better approach to understanding these relationships.
Renal function before and after cancer treatment with Pt-based drugs is an important parameter. Before starting anti-cancer treatment, it is advisable that the CrCl is >60 mL/min.52,65 Patients with lower values have an increased risk of severe suppression of the bone marrow. The CrCl for normal renal function is > 80 mL/min.52,66 The CrCl values before Pt-based drug chemotherapy for the control group 2 (baseline patients; see below) were 93.3–114.6 mL/min; serum and urine creatinine values were 0.6 – 0.9 and 0.8–1.1 mg/dL, respectively. Evidently, Pt-based drug chemotherapy impacts the CrCl and the metal/creatinine quotient values; both parameters are considered controversial.67,68 According to this approach, after chemotherapy, 82 of 90 cancer patients had kidney damage.
Statistical parametric treatment
Basic statistical values for the group patients treated with Pt-based drugs and control group treated without Pt-based drug and the baseline group. Amounts of supplied Pt and Pt, Mg, Zn, As and Se in 24-h urine samples.

Supp: supplied; X: mean; Med: median; SD: standard deviation; n: number of cases; Min: minimum value; Max: maximum value. (a) Due to the small number of cases, the median values were not reported; (b) non-platinum drugs treatment; (c) patients without chemotherapy (two gastric cancer, two bladder cancer and five lung cancer).
Basic statistical values for the group patients treated with Pt-based drugs and control group treated without Pt-based drug and the baseline group. Pt, Mg, Zn, As and Se amounts in 24-h urine samples expressed as metal (g creatinine)–1.

Supp: supplied; X: mean; Med: median; SD: standard deviation; n: number of cases; Min: minimum value; Max: maximum value. (a) Due to the small number of cases, the median values were not reported; (b) non-platinum drugs treatment; (c) patients without chemotherapy (two gastric cancer, two bladder cancer and five lung cancer).
Although the number of patient with cancer of the control groups was low, we can observe that in both cases the Se levels tend to be higher than those of As. However, in control group 2, the range of As and Se levels tends to be comparable or slightly smaller than those of control group 1. In Antofagasta Region, the Se concentrations have been shown to be higher than those As in the umbilical cords and placentas of normal and malformed newborn born babies, as well as in the tissues of patients and tissues of patients subjected to cardiovascular surgeries.69,70 This characteristic was also observed in the present study, inasmuch that the Se and As concentrations in group 3 were comparable with those of control 1 and control 2 groups.
Multivariate statistical treatment
The cluster technique was applied to the involved variables (i.e. calculated amounts of Pt supplied through Pt-based drugs and the urinary amounts of Mg, Zn, Pt, As and Se excreted in 24-h urine after the cancer patients' chemotherapy with cisplatin). Cluster analysis is a multivariate statistical methodology applied to datasets if there is a lack of information about the classes comprising the data. The basic objective is to group the variables by similarities. 71 The technique is based on two aspects: the way the distance between the variables is measured (metric) and the groupings or clusters (linkage or amalgamation rule). 72 The technique was applied to the most representative sets of values in accordance with the Shapiro–Wilk normality test. Ward’s method was applied with Pearson’s 1-r approximation to measure the distance between variables and the groupings or clusters.
Figure 1 shows the dendrogram of metal amounts in 24-h urine after the cisplatin first cycle treatment to lung-cancer patients, and Figure 2 shows the two main variables grouping, that is to say, (Pt exc, Se exc) and (Mg exc, Zn exc, As exc) when the principal component multivariate technique was applied, that is to say (Pt exc and Se exc) and (Mg exc, Zn exc and As exc). Then, Pt sum coming from cisplatin is not a principal component for lung cancer patients under orthogonal conditions. Therefore, this result would confirm that platinum from cisplatin is predominantly distributed and bio-accumulated in the cancerous and healthy tissues under less active anti-cancer platinum forms.
Tree diagram for six variables: Pt amounts supplies from Cisplatin drug and 24 h urine excreted amounts of Pt, Se, As, Zn y Mg from lung cancer patients of the Antofagasta Region in Chile. Factor loadings, Factor 1 versus Factor 2, for extraction of principal components and varimax normalized rotation for six variables: Pt amounts supplies from Cisplatin drug and 24 h urine excreted amounts of Pt, Se, As, Zn y Mg from lung cancer patients of the Antofagasta Region in Chile.

The effect of Pt sum is critical over As exc, Se exc, Zn exc and Mg exc. So, besides Pt and As, two heavy metals, Se was considered as a critical essential and anti-cancer 34 element, and Mg and Zn 47 two acknowledged essential elements were also considered in this work. Consequently, the information obtained in this work about urinary levels of this set of elements with less active d orbital electrons immediately after cancer patients were subjected to chemotherapy with Pt-based drugs could contribute to a better understanding of the adverse effects of Pt and help in the more accurate estimation of drug doses.24,73 Consideration of urinary levels of carcinogens such as As and cancer-fighting nutrients such as Mg, Zn and Se could allow more individualised or dedicated strategies for predictive calculations of drug dosages to be applied to cancer patients.
A well-established correlation has been found between As exposure in the Antofagasta region of Chile and the risk of cancer of the bladder, lung and skin. 74 Consequently, urinary levels of As and Se after each treatment cycle with Pt-based chemotherapeutic drugs could be important for a better understanding of the carcinogenic mechanism of As exposure 75 and about the hypothetic cancer-protective effects of Se. Genetic and epigenetic processes appear to be involved in cancer mechanisms.76,77 Epigenetic studies focus on the stress that As provokes in cells without affecting DNA, but directly influences the genome expression. In this complementary mechanism, As participates as an ‘opportunistic carcinogen’ that enhances the genotoxicity and mutagenicity of other environmental stressors, 74 for instance, other trace elements. During Pt-based drug treatments, patients are exposed to an acute impact of Pt. This subjects cellular homeostasis to deliberate stress by Pt, inducing toxic and, perhaps, hormesic responses. Among cancer patients exposed chronically to arsenic and treats with Pt-drugs based Mg, Zn, Se, As and Pt can participate synergistically in the regulated and unregulated activation of apoptosis, phagocytosis and cellular homeostasis involved in toxicological and hormesis mechanisms.18,20,76,78–80
Conclusions
Overall, despite significant variation in Pt amounts supplied from Pt-based drugs, the amounts of Pt in 24-h urine samples were not significantly different between patients with lung cancer and those with other cancers treated with cisplatin. With respect to the baseline status of patients, the analytical amounts of excreted Mg, Zn and Se were greater than those for As, particularly, for cisplatin chemotherapy. This finding could imply that, with regard to As displacement, Pt favours also the excretion of Mg, Zn and Se. For other types of cancers treated with drugs without Pt, excretion of Mg, Zn and Se was also greater than that of As, suggesting an antagonist Mg, Zn, Se–anti-cancer drug relationship.
The knowledge obtained in this work and in the related work 81 can contribute to understanding the cancer mechanism of As in a multi and inter-element scenario among Mg-Zn-As-Se-Pt for lung cancer as well as other types of cancer. Elements with similar electron arrangements are often antagonists in biological systems, 82 which can be rationalised by the Hard/Soft/Acid/Base principle. 83 The antagonism and/or synergism among these elements can be intrinsic to the cancerous metalloid-dependent mechanism of the As-Se pair, the exogenic Pt participation in healing, cancer propagation or metastasis and the essentiality of the Mg-Zn pair. Further research including more elements is in progress.
Footnotes
Declarations
Acknowledgements
Part of this research was supported by resources generated by the Bioinorganic and Environmental Analytical Chemistry Laboratory, Chemistry Department, Basic Science Faculty, University of Antofagasta, through the University of Antofagasta Technical Assistance Office. The authors are grateful for having been authorised to access urine samples at the Oncological Center of Antofagasta Regional Hospital.
Provenance
Not commissioned; peer-reviewed by Antonia Maria Palacios.
