Abstract
Lesson
We report a rare case of Myasthenia gravis and overactive bladder successfully treated with pretibial nerve stimulation.
Case report
A 24-year-old nulliparous Lithuanian lady with Myasthenia gravis was referred by her General Practitioner to the urogynaecology clinic with a complaint of urinary incontinence for the last three years. This had become worse in the last few months.
She had symptoms of urgency, urge and stress incontinence with urge being reported as the predominant symptom. This was severely affecting her quality of life. There were no other complaints. She had previously undergone a thymectomy and interestingly reported that her leakage started following this surgery. She was taking pyridostigmine bromide (60 mg qds po) as treatment for her Myasthenia. Examination findings were normal, and she had a normal body mass index.Investigations included midstream urine (MSU), frequency – volume chart, urodynamics and an ultrasound scan of the pelvis. She was provided with a copy of the overactive bladder questionnaire (OAB-q). She was advised to cut out caffeine from her diet, limit daily fluid intake to 1.5 litres, perform bladder drill regularly for six weeks, and given instruction to complete a course of pelvic floor exercises for 12 weeks.
Investigations revealed a normal MSU and pelvic scan. Urodynamic studies revealed a normal maximum flow rate of 49 ml/s on a total voided volume of 592 ml with a residual volume of 20 ml. The filling cystometry revealed unprovoked detrusor contractions with urgency. Pdet maximum during filling was 29 cmH2O and voiding cystometry parameters were normal. There was no demonstrable stress incontinence. The patient reported no improvement with conservative management.
She was treated with a course of 30 min sessions of pretibial nerve stimulation (PTNS) given weekly over a three-month period (Uroplasty®). This is delivered percutaneously through the tibial nerve via temporary insertion of a 34 gauge needle electrode, inserted approximately 5 cm cephalad to the medial malleolus and slightly posterior to the tibia and connected to a hand held electrical stimulator. 1 The patient completed the OAB-q following completion of treatment, as well as having repeat bladder diary and urodynamic studies.
At follow-up post-PTNS treatment, the patient reported being very happy, as all of her symptoms were relieved. There was a reduction in the frequency of micturition as well as absence of the previously reported leakage as measured by the frequency-volume charts. The repeat urodynamic study was normal, with a normal maximum flow rate of 46 ml/s on a total voided volume of 540 ml with a residual volume of 22 ml. Pdet maximum during filling was 20 cmH2O and voiding cystometry parameters were normal. There was absence of unprovoked detrusor contractions.
OAB-q subscale and total scores pre- and post-treatment with PTNS.
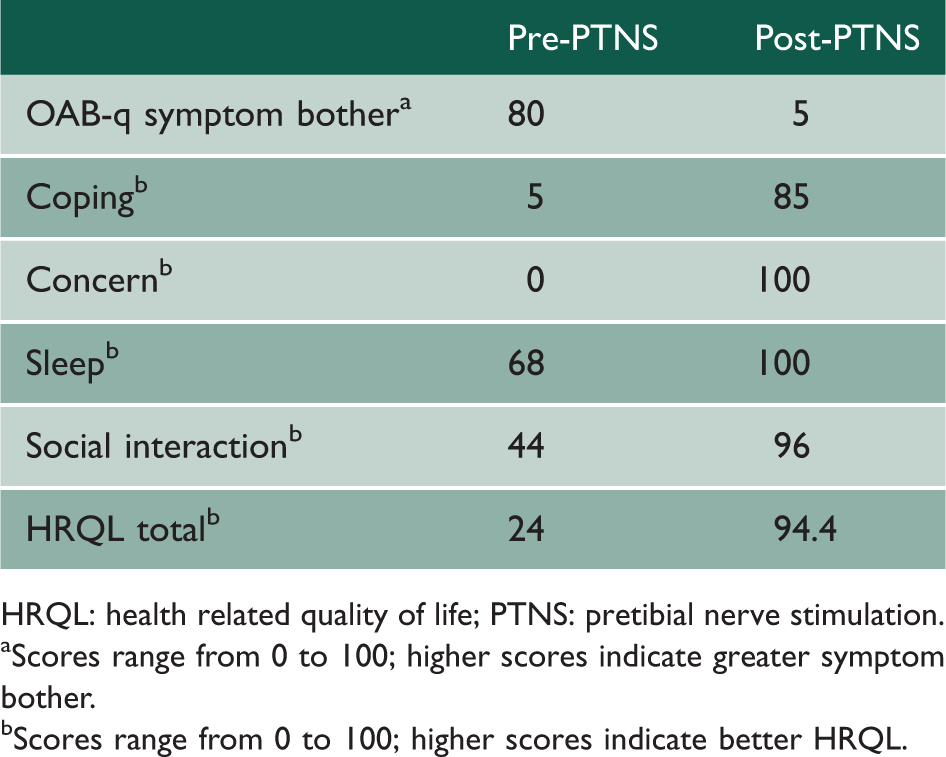
HRQL: health related quality of life; PTNS: pretibial nerve stimulation.
Scores range from 0 to 100; higher scores indicate greater symptom bother.
Scores range from 0 to 100; higher scores indicate better HRQL.
Follow-up of the patient up to two years reveals her to be well with no return of the urinary symptoms.
Discussion
The mainstay treatment of detrusor over-activity and urge urinary incontinence is drug treatment in the form of antimuscarinic anticholinergic drugs. 2 These drugs are absolutely contraindicated in patients with Myasthenia gravis, an autoimmune neuromuscular disorder characterised by circulating antibodies that block acetylcholine receptors at the postsynaptic neuromuscular junction. Other treatment options available include β3-adrenergic receptor agonist drugs such as Mirabegron (unavailable at the time of managing this patient) and neuromodulation in the form of PTNS and sacral neuromodulation. Neuromodulation is currently reserved for patients who are unresponsive to drug treatment as per NICE guidelines. 2 Botulinum toxin is relatively contraindicated in patients with Myasthenia gravis.
Overactive bladder (OAB) is defined by symptoms rather than objective measures, and many authors have tried to assess treatment outcomes from a patient perspective, with the development of questionnaires as patient reported outcome measures. 3 These have been used to more reliably assess treatment effectiveness. The overactive bladder questionnaire (OAB-q) is a questionnaire designed to assess symptom bother and health related quality of life in patients with both continent and incontinent OAB. The OAB-q has demonstrated reliability, validity and responsiveness, and test–retest reliability. 4 The OAB-q consists of an 8-item symptom bother scale and 25-item health related quality of life (HRQL) scale with four domains: coping, concern/worry, sleep and social interaction.
The patients’ maximum flow rate of 49 ml/s may appear to be high however it is within the 75th centile of the reported Liverpool nomograms for maximum flow rates and voided volume. 5
There is a dearth of data in the literature regarding the management of overactive bladder in patients with Myasthenia gravis with only one case report, in which a six-year-old boy was treated with electrical stimulation using suprapubic surface electrodes with improvement after six weeks. However, there was a return of symptoms of incontinence immediately upon discontinuation of the stimulation. 6 The difference in this report is use of a percutaneous technique with nerve stimulation over a 12-week period as compared to the surface electrodes used in the case of the six-year-old boy, with a six-week period, and use of the pretibial rather than suprapubic approach. Detrusor underactivity though rare in Myasthenia is more common than is overactive bladder in this disease.
Mirabegron offers a new alternative for treating overactive bladder in patients where antimuscarinics are either not tolerated or contraindicated. Botulinum toxin (Botox) is relatively contraindicated in patients with Myasthenia gravis and there are very few reports of the use of Botox in Myasthenia patients, none of which involve overactive bladder.
There is now evidence for the efficacy of PTNS in OAB comparable to the effect of antimuscarinic drugs but with a better side effect profile.1,2 The mechanism of action of PTNS, however, remains poorly understood. 7
We may speculate that the effective use of PTNS in a patient with an autoimmune condition may involve a possible immunological mechanism of action, particularly since only one course of PTNS was given, and the patient was followed up for two years with no recurrence of the condition. It is more usual for patients to require repeat courses of PTNS. Additionally, there is evidence of neural-immune interactions with a bidirectional relationship;8–10 therefore, the possibility of an immune mechanism exists, although there is currently no evidence to support this. This possibility will be explored further in a randomised controlled trial.
Footnotes
Declarations
Acknowledgements
None
Provenance
Not commissioned; peer-reviewed by Marios Hadjipavlou
