Abstract
Summary
Objectives
To systematically review the effects of radiofrequency catheter ablation (RFCA) on left atrial (LA) size, volumes and function in patients with atrial fibrillation (AF).
Methods
We searched MEDLINE, EMBASE, ScienceDirect, Highwire, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews and the reference lists of retrieved reports in July 2012.
Setting
China
Participants
Twenty-six studies (enrolling 1821 patients) were included in the final analysis.
Main outcome measures
Changes of LA size or volumes and/or function in patients with AF after RFCA.
Results
Compared to pre-ablation values, there were significant decreases in LA diameter and LA volumes at post-ablation follow-up. However, compared to pre-ablation values, there were no significant differences in LA ejection fraction/LA active emptying fraction and LA strain at post-ablation follow-up. Decreases in LA diameter and LA volumes remained significant in those without AF recurrence but not in those with AF recurrence. LA ejection fraction/LA active emptying fraction did not decrease in patients without AF recurrence, whereas they decreased in patients with AF recurrence. As for LA strain, it seems that LA strain increases in patients without AF recurrence, with less fibrosis and with more LA volumes decrease, but the differences were not significant.
Conclusions
Successful RFCA in patients with AF significantly decreases LA size and volumes and does not seem to adversely affect LA function.
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia. It impairs cardiac function and increases the risk of stroke. Treatments of AF include restoring normal sinus rhythm or controlling rate only and preventing thromboembolism. 1 Rate control is the preferred management option in most patients. Rhythm control is an option for patients in whom rate control cannot be achieved or who have persistent symptoms even with good rate control. Pharmacological therapy to restore and maintain sinus rhythm in some patients is often unsuccessful. For this group, catheter ablation is an important treatment option. Catheter ablation is indicated to prevent the recurrence of symptomatic AF. 2 Recently, clinical trials have clarified the role of catheter ablation in the treatment of AF. 3 But most randomized studies only included patients with paroxysmal AF and the FDA has approved catheters for use only in such patients. 4 However, ablation of persistent or permanent AF in symptomatic patients in whom medical therapy has failed is reasonable, since such patients have been shown to have considerable symptom relief with a successful ablation. Jeevanantham et al. had performed a meta-analysis in 2010 to assess the effects of radiofrequency catheter ablation (RFCA) on left atrial (LA) size, volumes and function and found that there were significant decreases in LA diameter and LA volumes at post-ablation follow-up. However, compared to pre-ablation values, there were no significant differences in LA ejection fraction (LAEF) and LA active emptying fraction (LAAEF) at post-ablation follow-up. 5 But the exact effects of RFCA on LA size, volumes and function in patients who undergo RFCA are not well understood.
Review aims
We reviewed the studies about the effects of RFCA on LA size, volumes and function in patients with AF, aiming to update the effects of RFCA of previous studies and to find whether the effects were different between the patients with AF recurrence or without.
Methods
MEDLINE, EMBASE, ScienceDirect, Highwire, Cochrane Central Register of Controlled Trials, and Cochrane Database of Systematic Reviews were searched before conducting the review on May 2012 to ensure that there was no recent review. The review was reported according to the PRISMA guidelines for systematic reviews. 6
PICOS identifiers from research questions (key terms) and database- and thesaurus-derived alternatives (additional terms) used to generate database searches.

Terms within each column were distinguished using the OR function, and the terms in different columns combined using the AND function.
Search
We searched MEDLINE, EMBASE, ScienceDirect, Highwire, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews and the reference lists of retrieved reports in July 2012 for studies of RFCA in patients with AF using the terms identified by PICOS (Table 1).
Selection
Inclusion/exclusion criteria.

RFCA: radiofrequency catheter ablation; LA: left atrial; LAEF/LAAEF: LA ejection fraction/LA active emptying fraction; AF: atrial fibrillation.
Data extraction
Two reviewers (YZ and YHY) extracted relevant data from the included studies using a standardized data extraction form. Randomized and non-randomized studies with follow-up imaging done at least one month after RFCA were considered for inclusion. Primary outcome measures were changes in LA diameter (LAD), changes in LA maximum volume (LAVmax; defined as the maximal LA volume before the opening of the mitral valve), changes in LA minimum volume (LAVmin; defined as the minimal LA volume at the closure of mitral valve), changes in LA ejection fraction (LAEF; defined as [LAVmax − LAVmin]/LAVmax), changes in LA active emptying fraction (LAAEF; defined as [LAmid-diastolic volume at onset of p wave on surface electrocardiogram − LAVmin]/LA mid-diastolic volume at onset of p wave on surface electrocardiogram) and changes in LA strain. Studies reporting changes in LAD, LA volumes and function on the basis of AF recurrence were analysed separately to assess effects of RFCA in patients with AF recurrence compared to those without AF recurrence. Studies that did not separate the results on the basis of the recurrence of AF were analysed together as combined studies. The total changes in LA size, volumes and function for all studies (recurrent, no recurrence and combined) were also analysed.
Quality assessment
The quality of included studies was assessed by exploring (1) study design, (2) the representativeness of the study participants with regard to patients who undergo RFCA for AF, (3) reporting of loss to follow-up and (4) limitations and biases.
Data synthesis
Statistical analyses were performed using RevMan version 4.2 (The Cochrane Collaboration, Oxford, England), and the results are expressed as weighted mean differences (WMDs) for continuous outcomes, with 95% confidence intervals (CIs). We calculated the
Results
We identified 26 studies (enrolling 1821 patients) that reported changes in LA size, volumes or function in patients with AF who underwent RFCA (Figure 1). Baseline characteristics of the studies included in the meta-analysis are listed in Table 3. The primary outcomes of the studies included are listed in Table 4.
Reports evaluated for inclusion in meta-analysis. Characteristics of studies included in the meta-analysis. R: responder (LAVmax after ablation decrease ≥ 15%); NR: non-responder; RE: recurrence of AF; LF: left atrial wall fibrosis <10%; ZF: paroxysmal AF; CX: nonparoxysmal AF. Primary outcome variables before & after ablation of studies included in the meta-analysis. R: responder (LAVmax after ablation decrease ≥ 15%); NR: non-responder; RE: recurrence of AF; LF: left atrial wall fibrosis <10%; ZF: paroxysmal AF; CX: nonparoxysmal AF.


Management I – interventions
All patients had underwent RFCA, There were two studies had repeated RFCA. 7,10 Liu et al. had compared different treatment strategies on left atrial size in patients with lone paroxysmal atrial fibrillation. 17 Nineteen studies had included patients with paroxysmal or nonparoxysmal AF, 7 –9,13,16 –30,32 four studies only included patients with paroxysmal AF, 12,14,15,31 one study had found that ablation in no paroxysmal AF patients with heart failure and low ejection fraction could reversing atrial and ventricular remodelling. 23 Nine studies had reported changes in LAD, LA volumes, or function on the basis of AF recurrence. 9,11,15,17,19,23,26,27,31
Management II – outcomes
Compared to pre-ablation values, there were significant decreases in LAD (WMD − 1.52 mm, 95% CI − 2.57 to − 0.47; Figure 2), LAVmax (WMD − 6.12 mL, 95% CI − 9.46 to −2.78; Figure 3) and LAVmin (WMD − 2.59 mL, 95% CI − 4.88 to −0.29; Figure 4) during follow-up after ablation therapy. However, when analysed on the basis of AF recurrence, decreases in LAD, LAVmax and LAVmin remained significant among studies which reported no AF recurrence but not in those with AF recurrence (Figures 2 to 4).
Forest plot of comparison of change in LAD before ablation and after ablation at follow-up. Forest plot of comparison of change in LAVmax before ablation and after ablation at follow-up. Forest plot of comparison of change in LAVmin before ablation and after ablation at follow-up.


Compared to pre-ablation values, there were no significant differences in the LAEF/LAAEF (WMD − 0.58%, 95% CI − 3.64% to 2.47%; Figure 5) after RFCA during follow-up. However, when analysed on the basis of AF recurrence, there were significant decreases in the LAEF/LAAEF in patients with AF recurrence, whereas there were no significant decreases in the LAEF/LAAEF in those with no AF recurrence (Figure 5).
Forest plot of comparison of change in LAEF/LAAEF before ablation and after ablation.
As for LA strain, it seems that LA strain increases in patients without AF recurrence, with less fibrosis and with more LA volumes decrease, but the differences were not significant (Figure 6).
Forest plot of comparison of change in LA strain before ablation and after ablation.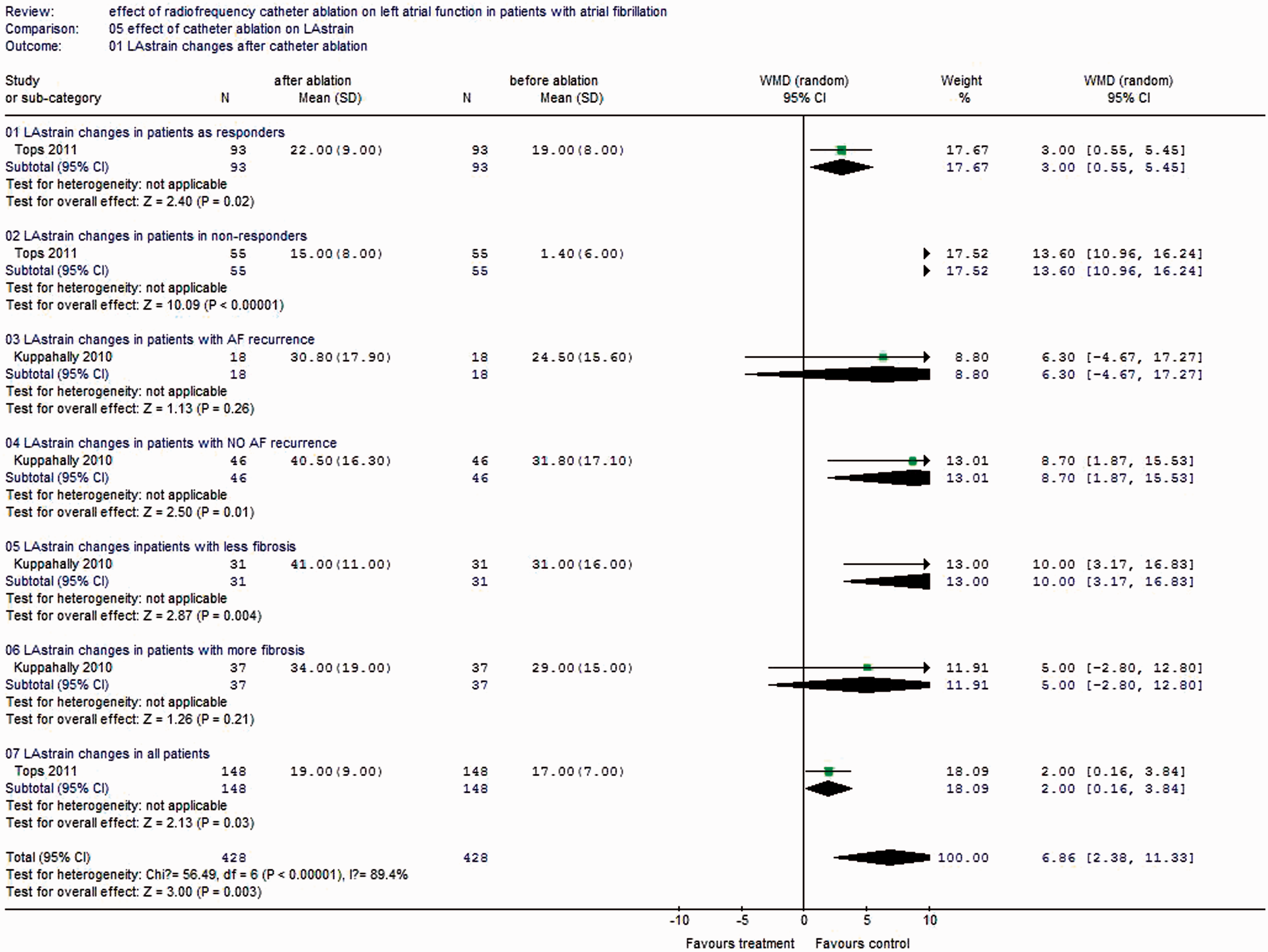
Quality assessment of the studies
The quality of the studies analysed, using predefined criteria, showed that most of these studies were retrospective in nature and used consecutive sampling designs. Sampling bias and smaller sample sizes limited the external validity of these results.
Heterogeneity and sensitivity analyses
Heterogeneity was explored using sensitivity analyses. We reported the results using a random-effects model because significant heterogeneity was noted in all total outcomes. The results did not differ when analysed using a fixed-effects model.
Sensitivity analyses based on studies with ≥3 months of follow-up showed that LAD (WMD − 1.49 mm, 95% CI − 2.58 to − 0.4), LAVmax (WMD − 5.47 mL, 95% CI − 8.83 to −2.11) and LAVmin (WMD − 6.23 mL, 95% CI − 9.54 to − 2.91) decreased significantly after RFCA, whereas the LAEF/LAAEF (WMD − 0.12% 95% CI − 3.45 to 3.22%) did not significantly change at follow-up after RFCA.
Sensitivity analyses on the basis of studies including ≥30 patients showed that LAD (WMD − 1.41 mm, 95% CI − 2.48 to −0.33) and LAVmax (WMD − 5.60 mL, 95% CI − 9.00 to −2.20) decreased significantly after ablation, whereas LAVmin (WMD − 2.19 mL, 95% CI − 4.54 to 0.16) and the LAEF/LAAEF (WMD 0%, 95% CI − 3.32% to 3.31%) did not significantly change at follow-up after ablation.
Publication bias
On the basis of funnel plot analysis, publication bias was noted for the primary outcomes (Figure 7).
Funnel plot of change in LAD before ablation and after ablation at follow-up.
Discussion
Principal findings
Our study shows that LAD and LA volumes decrease after RFCA during follow-up. However, LA function (as measured by the LAEF/LAAEF and LA strain) does not change after RFCA. When studies were separately analysed on the basis of AF recurrence, we found that, in patients with AF recurrence, there were no significant decreases in LAD and LA volumes after RFCA. More importantly, there were significant decreases in the LAEF/LAAEF in patients with AF recurrence after ablation therapy. In contrast, in patients without AF recurrence, RFCA therapy clearly decreased LA size and volumes, and there was no change in LAEF/LAAEF. It seems that LA strain increases in patients without AF recurrence, with less fibrosis and with more LA volumes decrease, but the differences were not significant.
Interpretation of findings in relation to previously published work
There are many factors that could influence the structural and functional remodeling of left atrium after RFCA. Those factors included the amount of LA scar produced by RFCA, or LA fibrosis before ablation and restoration of sinus rhythm and/or reduction of AF burden (defined as the frequency times the duration of AF). Currently, the amount of RFCA in different areas of the left atrium required for creating an ideal scar volume that would prevent AF recurrence and not decrease LA function are unknown. Hall et al. 33 showed that wall thickness of LA is variable and the roof and posterior wall are thinnest. Recently, Peters et al. 34 showed that patients with more RFCA scars in the right inferior pulmonary vein area had less AF recurrence than those with fewer scars in the same area. But Wylie et al. 20 showed that excessive scar volume decreased the LA function. Also, there are studies that showed that the extent of LA wall injury correlates with arrhythmia recurrence at three months. 35 So, the recovery of LA function after RFCA is a comprehensive result of amount of LA scar tissue and the restoration of sinus rhythm and/or a reduction in AF burden. It is difficult to distinguish whether the decrease in LA function in those with AF recurrence after RFCA is related to the continued presence of AF (any AF or some magnitude of AF burden) or excessive scar tissue produced by RFCA therapy.
The time of research of LA function or LA dimensions is another important factor that needs more attention. Studies with longer durations of follow-up 15,16,22,30 have found no significant increases in LA function, whereas studies with shorter durations of follow-up 20 have shown decreases in LA function. Our sensitivity analysis on the basis of the duration of follow-up did not get a conclusion, which may be caused by different sample size and lack of individual patient data. Studies have found that the detection and quantification of LA scar is feasible three months after RFCA. 35,36 Therefore, LA function should be evaluated over a longer duration of follow-up. Future studies should consider the timing of LA functional assessments as an important factor in their study designs and allow adequate time for the recovery of LA function.
Another challenge about studying LA function is in patients with permanent AF. Because LA function could not be accurately assessed in AF rhythm, one possible way to assess baseline LA function of patients with chronic AF is to measure it within days after the RFCA procedure and then repeat measurements three to six months after RFCA. 30
What is more, the method to determine LA function is of upmost importance. Until now, there is no standardization in the measurement of LA function. During the initial part of left ventricular diastole, the left atrium has a passive conduit function, whereas it has an active pump or booster function during the later part of ventricular diastole, as the atrium contracts. More importantly, the imaging techniques used in these studies were different and included two-dimensional trans-thoracic echocardiography, three-dimensional trans-thoracic echocardiography, computed tomography with three-dimensional reconstruction and cardiac magnetic resonance imaging. Although echocardiography is an established tool in cardiac imaging, it has significant limitations in patients with poor acoustic windows, such as obese patients and those with severe obstructive pulmonary disease. Two-dimensional echocardiography may underestimate true LA size compared to computed tomography or magnetic resonance imaging. Multi-slice computed tomography has excellent spatial and temporal resolution in quantifying LA volumes, but radiation exposure and contrast dye exposure limit its use. Magnetic resonance imaging has unique advantages over other modalities because it can simultaneously measure the pulmonary vein anatomy and detect pre-ablation fibrosis and post-ablation scar in patients who undergo RFCA. 37
Our results show that RFCA therapy favours structural remodeling of the left atrium by decreasing size and volumes during follow-up. This is important because LA size and volumes are powerful predictors of cardiovascular outcomes. Increased LA size and volumes are associated with increased risk for developing AF and congestive heart failure. Therefore, a consistent decrease of LA volumes seen across all the studies may be an important outcome of an RFCA procedure. Another important observation, although inconclusive, is that successful RFCA procedures did not adversely affect LA function. It is important to understand the effect of RFCA therapy on LA function because the left atrium modulates left ventricular filling and performance. Moreover, elderly patients are more dependent on LA contraction for left ventricular filling, and a loss of LA function could lead to decreased exercise capacity and an increased incidence of heart failure.
Weaknesses of this study
Our meta-analysis had limitations that deserve further consideration. Variations in study imaging techniques, differences in ablation strategies and different follow-up durations among these studies make it difficult to draw definitive conclusions about LA functional change produced by RFCA. Also, a lack of individual patient data on the post-RFCA duration of sinus rhythm or AF burden and the amount of LA scar makes it difficult to differentiate the effects of RFCA on LA function. Another important limitation is that LA function could be measured only during sinus rhythm. A lack of standardization of measurement of LA function among the included studies is another limitation. Additionally, our systematic review could not distinguish cause from effect. We were unable to distinguish whether the differences in the effects of RFCA on LA size and function cause AF recurrence or whether AF recurrence after RFCA cause the differences in LA size and function. Perhaps studies that methodically analyse scar volume and quantity AF burden will shed light on the cause-and-effect relations.
