Abstract
Introduction
Integrated Care Pathways (ICPs) are a method for the mutual decision-making and organization of care for a well-defined group of patients during a well-defined period. The aim of a care pathway is to enhance the quality of care by improving patient outcomes, promoting patient safety, increasing patient satisfaction, and optimizing the use of resources. To describe this concept, different names are used, e.g. care pathways and integrated care pathways. Modern information technologies (IT) can support ICPs by enabling patient empowerment, better management, and the monitoring of care provided by multidisciplinary teams. This study analyses ICPs across Europe, identifying commonalities and success factors to establish good practices for IT-supported ICPs in diabetes care.
Methods
A mixed-method approach was applied, combining desk research on 24 projects from the European Innovation Partnership on Active and Healthy Ageing (EIP on AHA) with follow-up interviews of project participants, and a non-systematic literature review. We applied a Delphi technique to select process and outcome indicators, derived from different literature sources which were compiled and applied for the identification of successful good practices.
Results
Desk research identified sixteen projects featuring IT-supported ICPs, mostly derived from the EIP on AHA, as good practices based on our criteria. Follow-up interviews were then conducted with representatives from 9 of the 16 projects to gather information not publicly available and understand how these projects were meeting the identified criteria. In parallel, the non-systematic literature review of 434 PubMed search results revealed a total of eight relevant projects. On the basis of the selected EIP on AHA project data and non-systematic literature review, no commonalities with regard to defined process or outcome indicators could be identified through our approach. Conversely, the research produced a heterogeneous picture in all aspects of the projects’ indicators. Data from desk research and follow-up interviews partly lacked information on outcome and performance, which limited the comparison between practices.
Conclusion
Applying a comprehensive set of indicators in a multi-method approach to assess the projects included in this research study did not reveal any obvious commonalities which might serve as a blueprint for future IT-supported ICP projects. Instead, an unexpected high degree of heterogeneity was observed, that may reflect diverse local implementation requirements e.g. specificities of the local healthcare system, local regulations, or preexisting structures used for the project setup. Improving the definition of and reporting on project outcomes could help advance research on and implementation of effective integrated care solutions for chronic disease management across Europe.
Keywords
Introduction
Diabetes represents a growing challenge for health care systems everywhere. Its prevalence has been growing continuously over the last decades, 1 with the financial and human burden on healthcare systems increasing as a result. In addition, diabetes is linked to a high risk of comorbidities including depression and cardiovascular diseases, resulting in complications such as retinopathy, nephropathy, neuropathy, stroke, dementia, or lower extremity amputations. 2 The need to treat such long-term chronic conditions, which often require multiple complex interventions, only adds to the pressure on health systems already dealing with the impact of an aging population. 3
Finding sustainable solutions to curb the morbidity, mortality, and disability associated with major chronic diseases has become an urgent priority for health practitioners and policymakers. 4 Moreover, there is a need for a paradigm shift in how health services are funded, managed, and delivered. 3 In Europe, such services are being restructured to meet changing care needs, via the development and implementation of new, more innovative approaches. 5
Of these, integrated care (IC) has emerged as one of the most promising strategies, with many countries prioritizing it as a means to support the improvement of health outcomes for people at risk of or living with chronic illnesses.4,6 More specifically, integrated care pathways (ICPs) are personalized, structured multidisciplinary care plans, which outline essential steps for the care of patients with specific clinical and/or social problems. 7 They also support the transposition of national guidelines into local protocols and daily care practice. ICPs differ from practice guidelines in that they are used by a multidisciplinary team, and focus on improving the quality and coordination of care. 8
From a patient’s perspective, integrated care aims to meet both their health and social needs, taking these as the starting point for redesigning their care. Better coordination and continuity of care not only make for an easier patient journey through treatment; they also contribute to improved outcomes.9,10 To this end, integrated care can drive greater efficiency and value within health care delivery systems, benefiting patients, healthcare providers, and care systems alike.9,10
Multidisciplinary care delivery is to a large extent an information-driven process. Technology for collecting and exchanging information is considered essential to make integrated care work for all stakeholders involved. 11 Current developments in information technologies (ITs) can be the ultimate facilitators for integrated care models. 11 Modern diabetes management is an example of this, making use of internet-enabled devices such as glucose meters, insulin pumps, activity trackers, and calorie counters (through wearables and smartphones), or allowing patient–carer interaction. 11 Furthermore, the data generated by such devices and systems can provide insights into how to further improve operations and decision-making within healthcare services. 12 ICPs also enable the systematic collection and analysis of relevant data for the purposes of audit, or to promote changes in practice. 7 However, it is worth noting that important and necessary functionalities for health technology remain limited, as do sufficient interoperability and an effective roadmap for the implementation and deployment of such technologies.11,13
Integrated care hinges on a tailored combination of the right structures, processes, and methodologies to address a patient’s needs, alongside specific institutional and community circumstances. 10 Thus, local settings are the most appropriate context for the tailored development, implementation, and deployment of ICPs. 14 Since there is no “one size fits all” or silver-bullet solution for ICPs,1,10 it’s important to study the features of successful IT-supported ICPs for diabetes patients.
The aim of this study was therefore to identify commonalities in good practices of IT-supported ICPs for patients with diabetes mellitus, and subsequently better understand when these ICPs are successful, and for what kind of patients.
Definitions applied in the research
The European Commission (EC) defines “good or best practices” in the area of active and healthy aging as real-life examples of successful applied innovations. 5 Meanwhile, the European Innovation Partnership on Active and Healthy Ageing (EIP on AHA) initiative, which is supported by the EC, identifies a “good practice” by its feasibility and potential for replicability under various conditions, 5 considering “good” and “best” practices to be interchangeable.
To date, there is no standardized definition of integrated care pathway that has been developed for the management of chronic diseases or, more specifically, on diabetes. Several organizational models have been created to implement integrated care pathway approaches that are known under the name of care management programmes, disease management, population management, and chronic care models, to mention but a few. 15
According to Perleth et al., 16 an activity, discipline, or method available for identifying, implementing, and monitoring available healthcare evidence is dubbed a “best practice”. These can be further identified according to three criteria: input, implementation or dissemination, and outcome. To measure the improvement of a health care system’s performance, Perleth et al. 16 suggest further criteria related to safety, efficacy, effectiveness, cost-effectiveness, the appropriateness of health care interventions, the quality of healthcare, and public perception. In addition, the authors recommend taking a collective approach to information-gathering and management since no single one of these approaches forms an entirely successful and all-embracing strategy for ascertaining what constitutes best practice.
Several examples of IC “good practice” models are described in the scientific literature.5,6,16–26 Integrated care is associated with a number of positive outcomes, including an improvement in system performance, clinical results, service delivery, and patient satisfaction. 17 However, due to the complex, disruptive and context-specific nature of health care service transformation, it is often difficult to directly measure its effects in terms of efficacy and efficiency. 5 Failure to recognize the widely varying definitions and components of ICPs can lead to inappropriate conclusions being drawn in regards to the effectiveness of such programmes. 19
According to Busetto et al., 6 the differences in definitions and operationalization of IC interventions, as well as the different classifications of interventions and/or of their components, can make it nearly impossible to compare reviews investigating the relative effectiveness of IC, or to draw overarching conclusions. With this in mind, the chronic care model (CCM) has been proposed in order to narrow down an operational definition of IC that allows for the systematic, reliable identification, and categorization of different kinds of IC interventions.6,20 This model suggests that improving integrated chronic care requires changes in six areas: community, the health system, self-management support, delivery system design, decision support, and clinical information systems. 20 Of these six, the latter four are considered to be the priority when it comes to improving matters. 6
A systematic review of integrated healthcare models delivered at the primary–secondary care stage identified the characteristics of models that lead to favorable clinical outcomes. 18 The six common features are interdisciplinary teamwork, communication/information exchange, shared care guidelines or pathways, training and education, patient access and acceptance, and a viable funding model. 18
These overlap with the World Health Organization’s concept of “building blocks” for care; an expansion of the earlier mentioned CCM 21 entitled the Innovative Care for Chronic Conditions (ICCC) Framework. This is based on a set of guiding principles fundamental to the micro- meso-, and macro-level organization of the healthcare system. 22
Based on the above, we developed a set of process and outcome indicators by using Delphi-technique described in the methods section, which is listed in Table 1, to select “good practices” and to identify the criteria for successful IT-supported integrated care pathways.5,6,16–26
Methods
A methodological triangulation strategy was used to identify, describe and compare IT-supported ICPs for diabetes, which were recognized as “good practices” in their original documentation. In doing so, three related methods were applied: desk research, a non-systematic, narrative literature review, and a survey, which was followed by in-depth interviews with project participants to gather additional data and expand the insights obtained via the other data collection methods.
The desk research took as its primary source a publication of the EIP on AHA, 26 outlining a new approach to research and innovation bringing together all relevant actors at all levels. The EIP on AHA’s goal is to share knowledge on good practices for active and healthy aging, including integrated care, and to deploy these across a network of European regions across Europe known as reference sites.
In November 2015, the EIP on AHA’s Action Group on integrated care for chronic diseases presented a report compiling such 86 good practices. 26 Of these, we selected several projects fulfilling the following inclusion criteria: they related to people with chronic diseases, in particular diabetes, with comorbidities in European countries, and they incorporated any kind of IT support where digital information between people and/or devices was exchanged.
Given the European Commission’s current work in the area of chronic disease management, we complemented the research with an examination of other good practices identified within the Chrodis Joint Action. 27 JA-CHRODIS is a European collaboration that brings together over 70 associated and collaborating partners from national and regional departments of health and research institutions in 25 European countries. These partners work together to identify, validate, exchange, and disseminate good practice on chronic diseases across EU Member States.
To capture other good practices of IT-supported ICPs beyond those featured within the EIP on AHA, 5 a non-systematic, narrative review was performed using PubMed, screening all articles published between 1984 and May 2016 using the terms “diabetes”, “IT”, and “integrated care” in any field (research date: 30 May 2016). Non-European projects, studies focusing on technical developments, and studies focused on diseases other than diabetes mellitus were excluded.
To describe and compare selected ICPs, the research team used the process and outcome indicators listed in Table 1. We applied a Delphi technique as a structured process of “rounds” to collect information among our investigation group. Rounds were held until group consensus has been reached. An initial face-to-face meeting aligned experts with the purpose of the consensus building process; this was followed by two anonymous survey rounds using an internet platform on the different dimensions on process and outcome indicators to characterize ICP.
The following steps were completed in this modified Delphi process: (1) the investigators agreed upon a set of principles to guide the development of the indicators’ list; (2) a small working group of investigators scanned the available literature to identify key resources on ICP; (3) a draft document was created based on these reviews and inputs; (4) an expert stakeholder meeting was held to discuss and confirm the issues; (5) two rounds of an on-line survey were completed to revise and reach consensus on the pathway; (6) all the investigators participated in focus groups to validate the list, and (7) a final meeting confirmed the final version of the indicators.
As far as possible, interviews were conducted with representatives of the projects selected from the EIP on AHA catalogue26,27 to gather additional data. Interviewees were invited to take part in a 30-min phone interview and to provide additional information on the metrics and data used to assess the impact of the practice. The survey included the process and outcome indicators mentioned in Table 1. A complete questionnaire can be found in the Appendix of this article.
Results
Desk research
A total of 16 IT-supported ICPs recognized as “good practice” were identified as meeting our inclusion criteria (Table 2, see Appendix).26,28–30
The selected projects came from several European countries and from different regions, including Spain, 6 Italy, 4 and one each from Denmark, France, Germany, the Netherlands, the United Kingdom, and Ireland. The ICPs differ in their stated goals, the composition of the multidisciplinary teams, patient characteristics, number of integrated patients, and local environment (clinic and ambulant settings). According to information available in the EIP AHA catalogue and in the literature, the 16 projects featured outcome indicators such as hospitalization, 11 cost savings, 9 patient satisfaction, 4 and the satisfaction of health care professionals (HCP). 2
Literature review
The non-systematic literature review yielded 434 results. After exclusion of reviews and other comparative analysis (N = 61), non-European projects (N = 56), articles on technical development or other common care subjects (N = 128), and articles focused on diseases other than diabetes mellitus (N = 175), 13 articles remained. Of these, five articles included redundant information which was covered in other references and were therefore removed from this analysis, resulting in a final total of eight original projects (Figure 1).31–40
Flow chart of research results from the PubMed research (search criteria: “diabetes”, “IT”, and “integrated care”).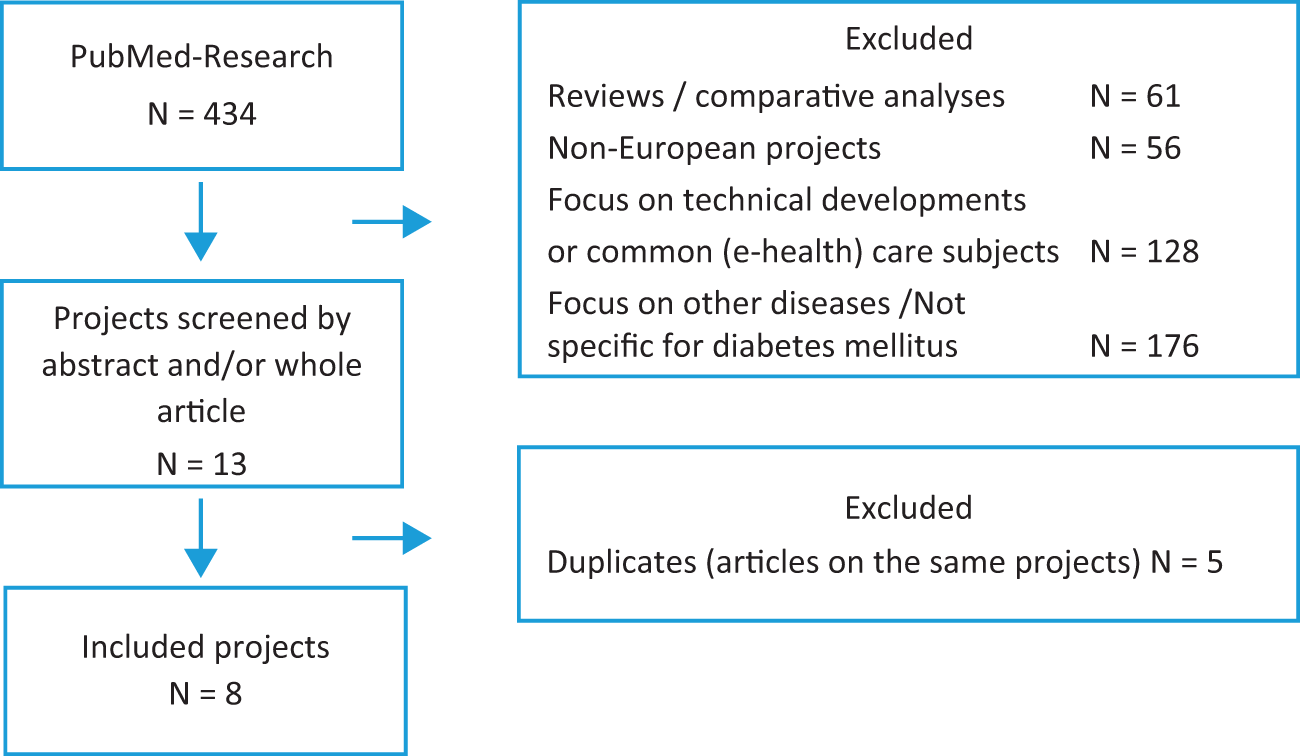
The majority of the projects were located in Spain and Norway (see Table 2, Appendix). Similar to the desk research, the projects identified in the non-systematic review research differed with respect to their objectives, patient and staff characteristics, organizational structure, and local environment. Projects from the nonsystematic review integrated a measurement of different outcome indicators such as hospitalization, cost-savings, and physiological parameters. Four projects implemented a comparator group to control the effectiveness of the projects.31,35,36,38
Survey results
Participants from 9 out of the 16 selected projects (56%) agreed to take part in the follow-up interviews. These were conducted with representatives of the projects (Table 3, Appendix).
All projects investigated received public funding and relied on multidisciplinary teams for implementation. Seven projects involved public authorities at various stages, seven used eHealth tools, and provided patient education and capacity-building. 41 The few practices relying on m-Health reported using apps or Skype to connect patients and services, and vice-versa. Seven projects reported involving patients at different stages of the practice. “eHealth” generally refers to the use of information technology, including the Internet, digital gaming, virtual reality, and robotics, in the promotion, prevention, treatment, and maintenance of health. “mHealth” refers to mobile and wireless applications, including text messaging, apps, wearable devices, remote sensing, and the use of social media such as Facebook and Twitter, in the delivery of health-related services.42,43
Usually patients were involved to solicit feedback on the practice worked and how it could be improved. Further patient empowerment was recognized as an area for improvement—respondents believed that there were not enough existing tools and activities available to empower patients.
The respondent identified an improvement in communication between patients and HCPs, as well as an increase in patient satisfaction (evaluated by surveys, semi-structured interviews or workshops within the projects), as advantages of IT-supported ICPs in diabetes. However, it was found that patient access to health records remains unequal and that social and psychological support is not sufficiently embedded in the practices. Most practices did not measure outcomes such as impact on physiologic parameters, hospitalization, or cost-savings.
Discussion
Our research reviewed 24 examples of IT-supported ICPs in diabetes and chronic diseases which have been applied in different regional settings and were identified as “good practices”. A comparative analysis nevertheless demonstrated a surprising level of heterogeneity.
While their implementation is—as a whole—likely to have positive impacts on process, outcome and economic dimensions of care, 44 the ICPs showed differences in objectives, the composition of the multidisciplinary teams, patient characteristics, organization, and local environment. In addition, the assessment of potential success factors did not reveal commonalities which could serve as a starting point to develop ICPs in diabetes throughout Europe.
However, the survey results did provide insights into the possible motivations for local healthcare systems to implement integrated care pathways for diabetes. Representatives surveyed reported improved communication between HCPs and patients, a greater satisfaction for both groups, and/or increased patient empowerment. However, these process indicators were not systematically analysed, and the understanding of what was meant by “improved communication” varied according to respondents; some took it to mean an increase in the level of communication, while other understood it as an improved perception of the doctor–patient relationship, and/or better adherence to treatment.
When focusing on ITs we found that within the majority of the ICPs, ITs were used to facilitate remote management between patients and HCPs. The use of ITs, in particular tele-health, for the support of integrated care for long-term chronic conditions has been described as useful and effective. 36 However, the existing status of IT-supported ICPs suggests that they are only currently being used to a fraction of their full potential.
Our analysis shows that, while good practice projects should have predefined objectives and outcome indicators, only a few of the selected projects actually measured hard outcomes, such as impact on physiologic parameters, hospitalizations, or cost-savings. The number of ICP projects, which applied a comparator group to assess the effectiveness of ICPs31,35,36,38 was even lower. It is therefore difficult to derive conclusions about the comparative effectiveness of ICPs.
We applied the Delphi technique as described by Kröger et al. 45 to develop process and outcomes indicators, since such an approach has been applied on chronic illness, 46 performance indicators, 47 and indicators for cardiovascular disease. 48
We are aware that a certain degree of uncertainty exists concerning the parameters of the Delphi technique such as the definition of group consensus, Delphi technique variants, expert selection, number of rounds, and reporting of the method and results. 49 In addition, the set of indicators identified throughout the process might well be incomplete, thus preventing the discovery of potential commonalities. Taken together, these limitations of our methodological approach may have lead to gaps in the set of indicators and the subsequent identification of commonalities.
By using a methodological triangulation strategy, not only could we include projects which have been extensively evaluated, but we were also able to include ongoing projects which have not yet been published in a scientific journal or other formal report. In addition, the projects from the EIP on AHA were followed-up with a structured survey to garner further insights. However, since only 9 out of 16 project representatives took part in an interview, it was difficult to draw a complete picture of all identified projects. This illustrated the difficulty of collecting data around ICP projects even when making use of multiple methods.
The current use of the labels “good practice” or “best practice” should concern policymakers and healthcare providers as much as researchers. It remains unclear what the labels “good practice” and “best practice” mean exactly, since no homogeneous definition has emerged from the collected data, making it difficult to distinguish and evaluate the two. To monitor the ongoing development and success of a project, it is essential to identify, document, and systematically evaluate goals and outcomes, rather than relying on arbitrary labels of “good practice” or “best practice”.
When comparing the dataset obtained through desk research with the literature review results, we found that no articles on the EIP on AHA projects had been published in the timeframe covered in the PubMed research (1984 until May 2016). The EIP on AHA report on good practices was published in November 2013, and most of the projects had been running for longer than one year at that time. The follow-up interviews could only partly fill this gap; interviewees were available for only 9 of the 16 projects and the information obtained did not always clarify why the respective projects had been labelled as “good practice”.
The lack of published data raises the question of how to draw substantial conclusions about the success of the projects, as well as limiting the ability to design a blueprint for future projects based on these practices, as there are not enough learnings on how to avoid missteps made in the existing good practices. Hence, the lack of published data risks hampering further research.
The conclusion must be drawn that any support or funding for projects in the field of ICP should require robust documentation, and the expectation that as far as possible, the outcomes of these projects be published according to acknowledged standards. This may not be feasible for all projects, such as those with limited resources and the need to put patient care above research efforts, but raising these issues before embarking on a project would be a first step towards greater transparency and consistency.
Currently available data display too much heterogeneity in all aspects of the ICP projects identified as good practices. The reasons behind this are diverse, but likely include the variance between different national European healthcare systems. In many countries, the organization of healthcare delivery is largely a regional responsibility, but funding mechanisms and healthcare provider responsibilities may also be organized differently. It is also arguable that variance may be linked to a lack of standardized definition of integrated care pathways. Efforts to seek consensus on a definition of integrated care pathway for chronic diseases may help address some of the weaknesses identified in this paper.
Conclusions
European health systems are still “disease oriented” and managed around single medical specialties, thus fragmenting and poorly coordinating care. IT-supported integrated care pathways in diabetes show a great variability in their practical implementation. As a multi-dimensional tool, ICPs differ in their objectives, design, process, and outcome indicators. This makes them adaptable to local needs and different health care systems, but complicates the identification of general criteria for ICP in the field of diabetes.
That being said, and on the basis of our findings, we recommend that an ICP on diabetes ideally should include:
A multidisciplinary and multistakeholder project group involving representatives of all HCPs, public stakeholders and patients for the definition of the Diabetes IC Protocol and for the monitoring of indicators; A detailed protocol which translates and standardizes the best evidence into the local context, and defines the roles, tasks, responsibilities, indicators, and targets (to be updated regularly); A disease registry and computerized system for process monitoring to assess the epidemiology dynamics, the application of ICP protocol, management costs, and the achievement of objectives; An evaluation framework collecting quantitative and qualitative information including patient-reported outcomes; A continuous education programme for all HCPs, public stakeholders, and patients to ensure the upscaling of the IC model among European countries, regardless of the healthcare systems governance, and the collection of organizational, financial, and technological needs for the improvement of the care model, including primary, and secondary care.
Authors’ contributions
Study conception, design of the work, collection, analysis and interpretation of data: HV, NG, AGdB. Discussions on data interpretation: HV, NG, AGdB, BH. Manuscript drafting and scientific writing: HV, NG, AM. Critical revision of the manuscript: ALL. Final approval of the manuscript version to be published: ALL.
Footnotes
Acknowledgements
This article is based on the discussions and recommendations of the Diabetes Sherpa Group, consisting of healthcare professionals, working on the field of diabetes. Meetings of the Diabetes Sherpa group were supported by Roche Diabetes Care and some members of the working group received an attendance honorarium.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HV receives compensation from Roche for scientific input to European Working Group Integrated Chronic Disease Pathway. AGdB has been compensated for developing and delivering educational presentations for Abbvie, Celgene, Novonordisk, and Menarini in the past three years. BH has been working as a consultant for Roche. AM is an employee of Roche. MS received Honoraria for lectures from Abbott Diabetes Care, Eli Lilly, Novo Nordisk, and Sanofi a member of an advisory board of Medtronic Scandinavia and part-time employed by Diabetespolyclinic of Neliapila, which is a subsidiary of Quattrofolia Inc. NG, MdlC, DP, and SdS declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
