Abstract
Objective
To explore the feasibility for a set of hormone replacement therapy (HRT) eligibility guidelines that follow a similar structure and appearance to the UKMEC guidance for contraception. To enable non-specialists to feel confident in safely prescribing HRT and to aid selection of the most appropriate first line treatment.
Methods
A literature review was undertaken with evidence summarised on the topic of venous thromboembolism (VTE) which is an area frequently considered a barrier to prescribing. Medical eligibility tables which separated HRT by type were then produced for a set of VTE-related topics.
Results
The literature search confirmed the importance of distinguishing between different types and routes of administration when considering the suitability of HRT. Much of the evidence has been based on older synthetic types of HRT and whilst they still have a role in management, these medications carry different risks to the now more accepted use of body identical types. The search also highlighted the nuances involved, increasing the complexity of forming guidelines, with the need for consideration to be given to an individual’s own perception of risks and benefits.
Conclusion
The demand for HRT has risen in recent years and there is a need for this to be managed effectively, particularly for patients in primary care. The production of this type of guidance will enable the non-specialist to feel confident in safe and evidence-based prescribing. The guidelines are also designed to demonstrate to prescribers which complex patients should be referred onto menopause specialists.
Introduction
International medical organisations including the British Menopause Society, International Menopause Society and the Menopause Society have published best practice recommendations for menopause care.1–5 These provide evidence-based guidance for healthcare practitioners to refer to and are perhaps more accessible to those with a menopause interest. There are currently no guidelines available that provide specific recommendations on the prescription of HRT according to a medical co-morbidity. There are also no easy to access, risk-based stratification tools that can be used as a decision-making aid for prescribing HRT in primary care. A set of HRT eligibility criteria, with similarity to those established for contraceptive methods, 6 would enable the formation of this type of guidance. Such guidelines could be used from within an app or other digital interface and would help clinicians, particularly those with limited menopause experience, to feel confident in safely prescribing HRT, reducing the need for secondary care menopause related referrals. This review explores this concept using the topic of VTE. The examples produced are for illustration purposes only and a wider scope of topics with prior clinical advisory group review would be required before forming any final recommendations.
Methodology
The topic of VTE was chosen and a literature review undertaken with results from meta-analyses, systematic reviews and randomised trials evaluated. Observational studies were included with particular interest in population-based cohorts or large case-control studies. In addition, consensus papers, position statements and review articles were reviewed, with the reference list of relevant studies checked. Medical eligibility tables which separated HRT by type were then produced for a set of VTE related topics. The allocation of HRT to an eligibility category was judged by the strength of evidence available on that topic.
Eligibility categories
Definition of eligibility categories.

Key differences to contraception guidance
Although it is intended that this guidance will be aligned with that on contraception, there are key differences: (1) The indication for hormone treatment in this guidance will be for menopausal symptoms and not for contraception. When withholding treatment for contraception, this confers a potential higher VTE risk for the patient, should pregnancy occur. (2) When making decisions about risk stratification, the effect of relative risk on underlying absolute risk must be considered. The absolute risk of VTE is age dependent and in general will be higher for menopausal women, compared to younger women seeking contraception.
Discussion
Route of administration
There is good evidence from large observational studies,7–14 randomised controlled trials15–19 and meta-analyses20–22 that transdermal HRT, when given at licensed doses, does not increase the risk of VTE above that in non-users. This remains the case whether the transdermal preparation is estrogen only or a combined type.12,20 In contrast, for women that use oral HRT, including estrogen alone or combined preparations, risk of VTE is increased 2–4-fold and is greatest in the first year after initiation.1,23 Exceptions are for tibolone and preparations containing dydrogesterone or micronised progesterone which are discussed separately.
Progestogen choice
Classification of progestogens licensed for HRT.

The choice of progestogen can affect VTE risk. A large nested case-control study demonstrated a higher risk of VTE in women using preparations containing oral estradiol with MPA (OR 1.44 [1.09–1.89]) and oral estradiol with NET (OR 1.68 [1.57–1.80]) compared to oral estradiol with DYG (OR 1.18 [0.98–1.42]). 12 Similarly, a large meta-analysis of seven population-based observational studies found that in users of oral estrogen, there was higher VTE risk in women using MPA (RR 2.77 [RR 2.33–3.30]) than in those using other progestogens. 25 This study reported no change in VTE risk in users of transdermal estrogen with MP (RR 0.93 [0.65–1.33]) whereas the norpregnane derivatives, nomegestrol acetate and promegestone, were associated with an increased risk (RR 2.42 [1.84–3.18]) and risk related to other progestogens, which included pregnane derivatives, was intermediate with important heterogeneity. 25 In addition, two recent systematic reviews concluded that MP has a neutral effect on VTE.26,27
There is sparse data on NET or MPA prescribing and any associated VTE risk in post-menopausal women. Large studies have unanimously demonstrated no increased risk of VTE when low doses of progestogens are prescribed in pre-menopausal women for contraceptive purposes, and this includes norethisterone 0.35 mg taken daily.28–32 However, higher therapeutic doses of progestogens, when prescribed in pre-menopausal women for other reasons, have been shown to increase VTE risk which might suggest a dose dependent effect.33–38
The dose of NET is important to consider given it partly converts to ethinylestradiol in the liver. Current guidance recommends NET 5 mg is taken for 12 days in sequential HRT regimes and NET 0.5–1 mg is taken daily in continuous HRT regimes. 39 It is estimated that 1 mg of orally administered NET metabolises to 4–6 µg ethinylestradiol. 40 Considering the thromboembolic risk associated with ethinylestradiol, this conversion may be clinically significant in women at increased risk for VTE. Use of ultra-low doses, however, is unlikely to cause harm and safety data is available for doses ≤0.35 mg daily.
Unlike NET, the synthetic progestogen MPA does not convert to ethinylestradiol and MPA may therefore be safer from this point of view, although evidence is limited. There is some observational data that demonstrates no association of VTE with use of pregnane derivatives.8,41 However, as mentioned above, the large meta-analysis of seven population-based observational studies showed MPA was consistently associated with increased VTE risk in users of oral estrogen (RR 2.77 [2.33–3.30]). 25 In addition, in the Women’s Health Initiative (WHI) trial, the Women’s International Study of long Duration Oestrogen after Menopause (WISDOM) trial and the large nested case-control study by Vinogradova et al, the association of estrogen plus MPA was more thrombogenic than unopposed estrogen.12,18,19 Uncertainty therefore still remains regarding the impact of oral MPA on VTE risk and more research is required.
Evidence from large systematic reviews and meta-analyses has found that LNG intrauterine devices (LNG IUD) do not increase risk of VTE.29,30,42 Most women included in these studies were pre-menopausal in age, which limits generalisability. However, the relative safety of a 52 mg LNG IUD can be explained by its localised action. After placement, serum levels reach a peak and plateau after the first few weeks, thereafter the systemic concentration decreases. 43 It releases about 20 μg of LNG daily, most of which remains concentrated in the endometrial cavity. 29
Tibolone and combined preparations containing micronised progesterone or dydrogesterone
The safety of a combined estradiol 1 mg and MP 100 mg oral tablet was studied in the REPLENISH trial, a multicentre 12-month, phase 3, prospective, randomised, double-blind, placebo-controlled trial. 44 Results from this study found no significant changes in any coagulation parameters suggesting preliminary safety data for VTE risk is reassuring. However, long-term safety data are absent.
There is a body of evidence that suggests that when combined with oral estrogen, DYG delivered orally, like progesterone, has an overall better risk profile compared to other progestogens with regard to thrombotic risk.8,45,46 The large nested case-control study undertaken by Vinogradova et al reported that combined oral preparations containing estradiol with DYG were not associated with a statistically significantly increased VTE risk (OR 1.18 [0.98–1.42]). 12 However, further studies looking at various combinations of oral estradiol and DYG are needed.
Tibolone is a synthetic steroid with estrogenic, progestogenic and androgenic properties that has tissue-specific effects. A Cochrane review reported no increased risk of VTE with its use; however, it described the evidence included as being low or very low quality. 47 Further support has since come from a large UK nested case-control study which similarly found no association of tibolone with VTE (OR 1.02 [0.90–1.15]). 12
Vaginal estrogen
Vaginal preparations containing licensed doses of either estradiol or estriol do not elevate serum levels above normal post-menopausal ranges with long-term use. 48 Data from the large observational Women’s Health Initiative study showed no increase in VTE risk with use of any vaginal estrogen preparations at licensed doses. 49
Risk factors for VTE
A risk factor for VTE can be categorised as major, moderate or minor based on its odds ratio (OR) (Table 2). 50 In the presence of a minor risk factor, oral estrogen, including estrogen alone or combined preparations, can sometimes be considered, albeit with some caution. The UKMEC guidance for contraception, for example, denotes a BMI ≥30–34 kg/m2 as UKMEC 2 for combined hormonal contraceptive use. 6 Nonetheless, it is generally recommended that in the presence of any VTE risk factor, preparations with the lowest VTE risk should always be used first line where possible.
Where a moderate or major risk factor exists, or where there are multiple risks, oral preparations should be avoided. A similar approach should be taken when considering tibolone or oral preparations containing MP or DYG due to the limited number of studies available on these preparations.
Example eligibility tables
Obesity
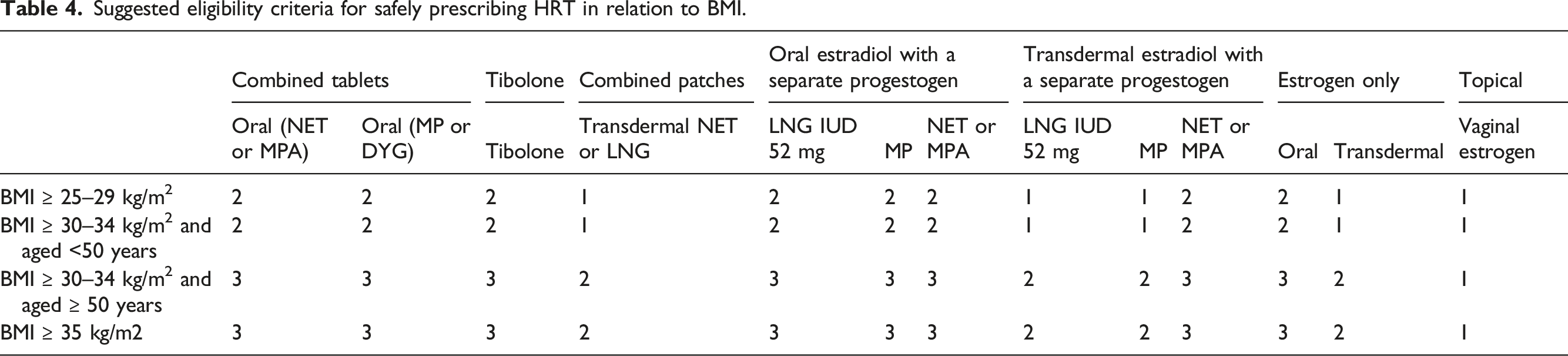
Increasing body mass index (BMI) has a linear association with the development of VTE.56–61 In one large prospective study, the OR for VTE in women for BMI 23.7–26.3 kg/m2 was 1.45 [CI 1.03–2.05], for BMI 26.4–29.9 kg/m2 was 1.81 [CI 1.27–2.56] and for BMI >29.9 was 2.82 [1.96–4.04]. 58 The risk of VTE is therefore more than doubled in obese subjects.
Suggested eligibility criteria for safely prescribing HRT in relation to BMI.
Recent surgery
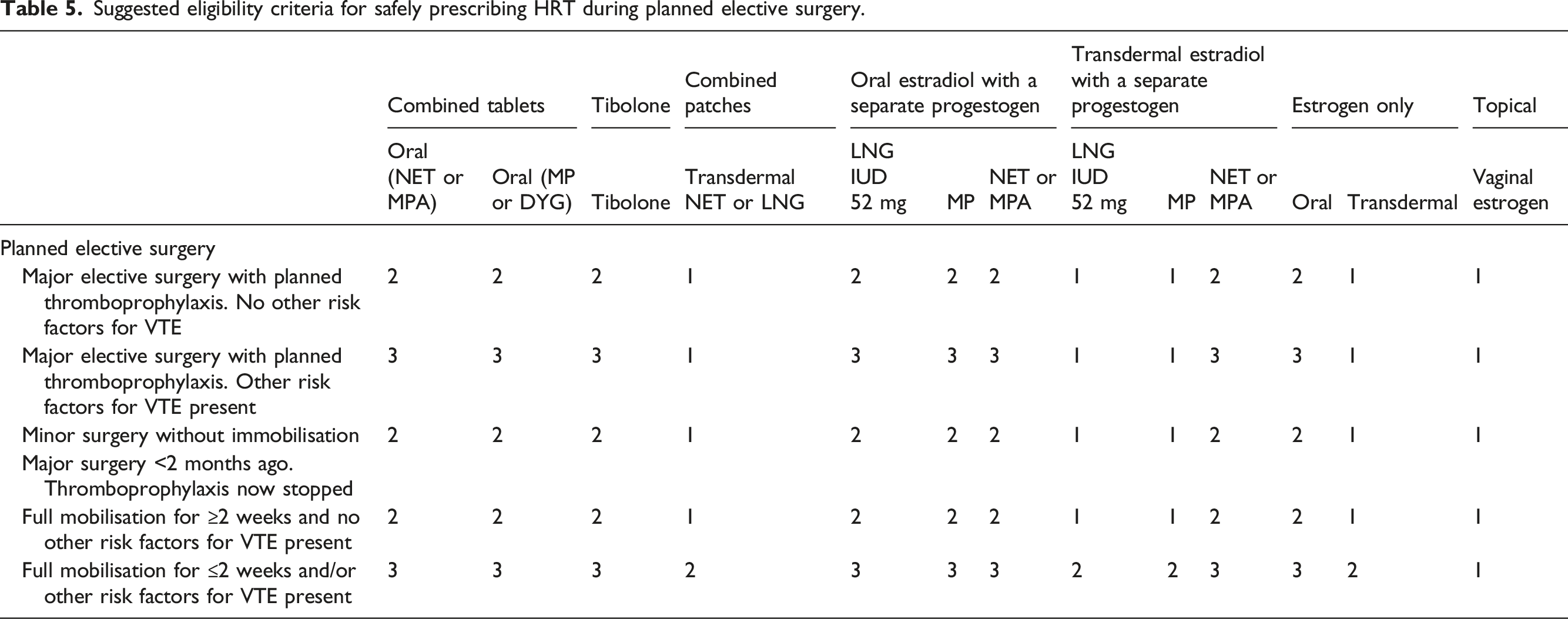
Although most surgical procedures increase VTE risk, this varies considerably across operation types. Elective procedures with a short duration of general anaesthesia (<30 min) and no associated immobilisation are considered low VTE risk.6,53,63 This can include some types of minor laparoscopic surgeries. 50 Elective procedures with a longer duration of general anaesthesia (>30 min) or associated with immobilisation are considered higher VTE risk.51,64 This will include surgery undertaken on the lower limbs such as hip and knee arthroplasties. 63
Where an individual is admitted electively to hospital, they should receive thromboprophylaxis as appropriate and if no additional risk factors for VTE are present, they do not need to discontinue their HRT whether transdermal or oral. 1 However, where additional risk factors for VTE are present, women using oral preparations of estrogen should switch to the transdermal route. 1
Suggested eligibility criteria for safely prescribing HRT during planned elective surgery.
Previous VTE episode
Whether an episode of prior VTE was provoked or unprovoked, it has implications for risk of recurrence. 53 If thrombosis was provoked by a transient risk factor, there is a low risk of recurrence. If thrombosis was provoked by a persistent or progressive risk factor, there is a high risk of recurrence. Patients with neither a transient or persistent risk factor are referred to as having an unprovoked VTE and have an intermediate risk of recurrence of VTE. 53
Suggested eligibility criteria for safely prescribing HRT in women with a past history of VTE.
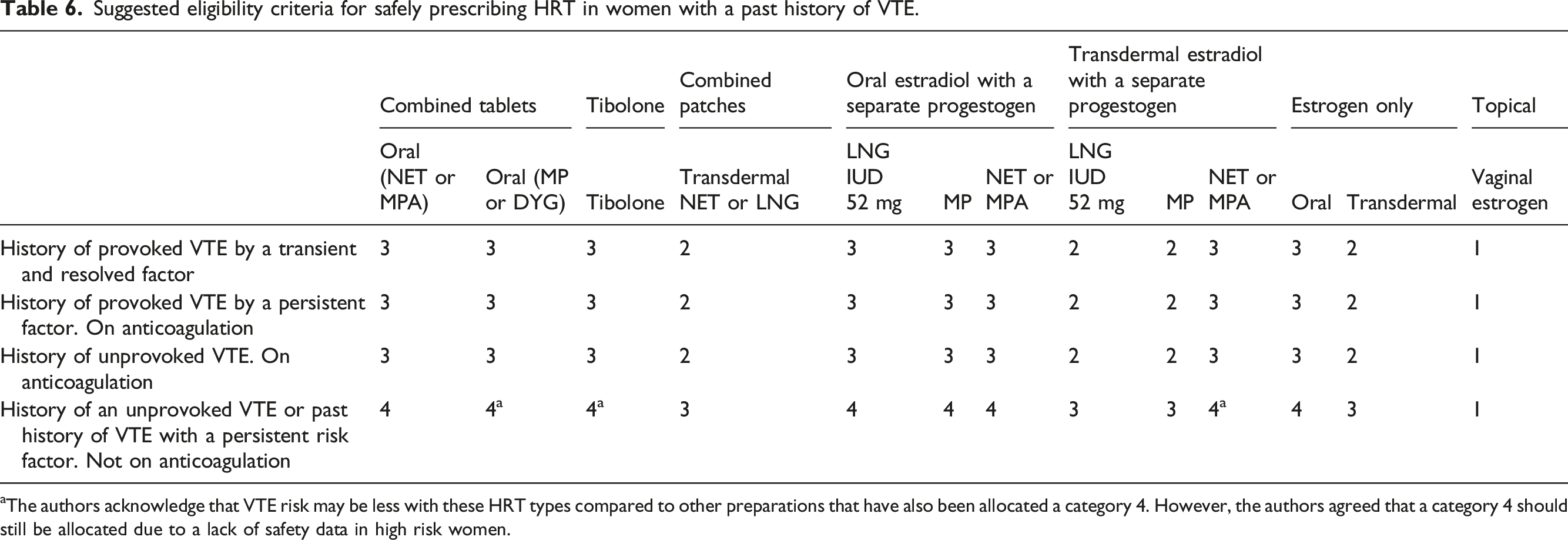
aThe authors acknowledge that VTE risk may be less with these HRT types compared to other preparations that have also been allocated a category 4. However, the authors agreed that a category 4 should still be allocated due to a lack of safety data in high risk women.
Family history of VTE
Suggested eligibility criteria for safely prescribing HRT in women with a family history of VTE.

Thrombophilia
The most common thrombophilias are heterozygous Factor V Leiden (FVL) and the prothrombin 20210A gene mutation. 23 Studies undertaken to date have mostly included women with these mutations. Data for more rare thrombophilias (including homozygous types, antiphospholipid (APL) syndrome, protein C deficiency, protein S deficiency and antithrombin deficiency) is limited and no trial data are available.
The risk of VTE is significantly increased in women with the FVL or prothrombin 20210A gene mutation using oral HRT preparations.15,67–71 However, a large systematic review and meta-analysis found that whilst the combination of oral estrogen with either of these gene mutations enhanced the risk of VTE, use of transdermal estrogen did not. 21 Similarly, a multicentre case-control study found that whilst use of oral estrogen in women with the FVL or G20210 A gene mutation increased risk of VTE 25-fold, transdermal estrogen did not confer any additional risk. 70
Suggested eligibility criteria for safely prescribing HRT in women a history of thrombophilia who are not using long-term anticoagulation for secondary VTE prevention. The included 'thrombophilias' are Factor V Leiden, prothrombin gene mutation (both homozygous and heterozygous), protein C deficiency, protein S deficiency, antithrombin deficiency, antiphospholipid syndrome and compound heterozygotes/combined thrombophilias.
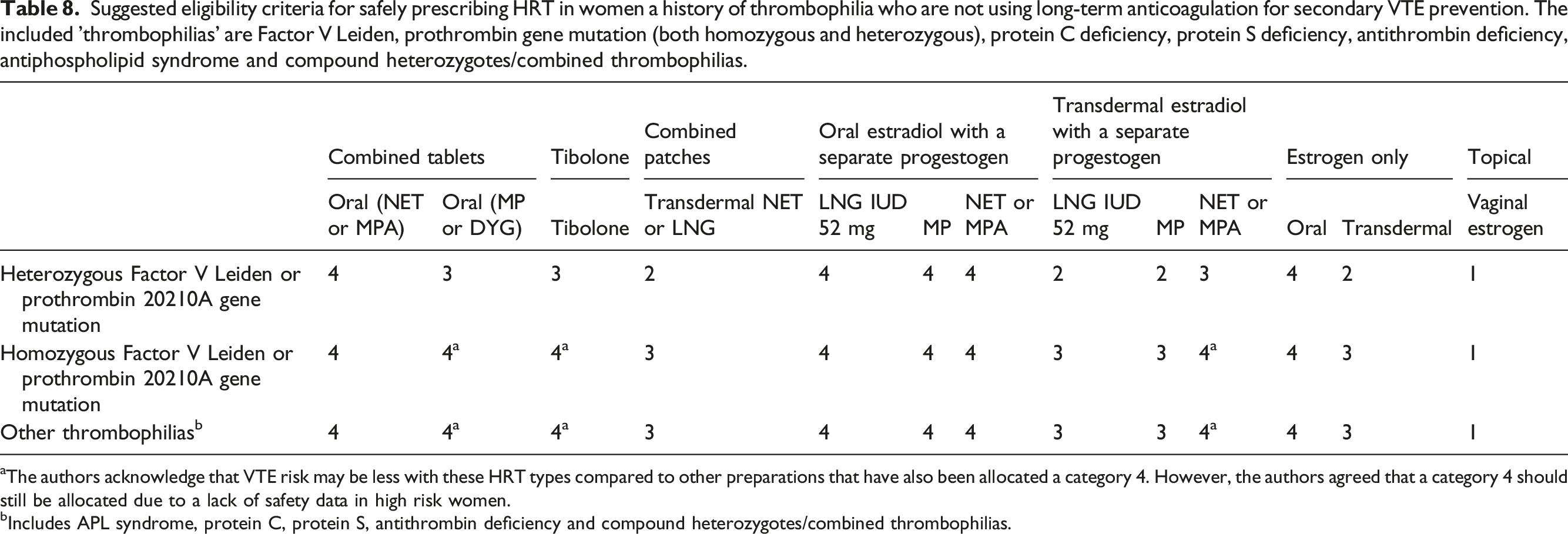
aThe authors acknowledge that VTE risk may be less with these HRT types compared to other preparations that have also been allocated a category 4. However, the authors agreed that a category 4 should still be allocated due to a lack of safety data in high risk women.
bIncludes APL syndrome, protein C, protein S, antithrombin deficiency and compound heterozygotes/combined thrombophilias.
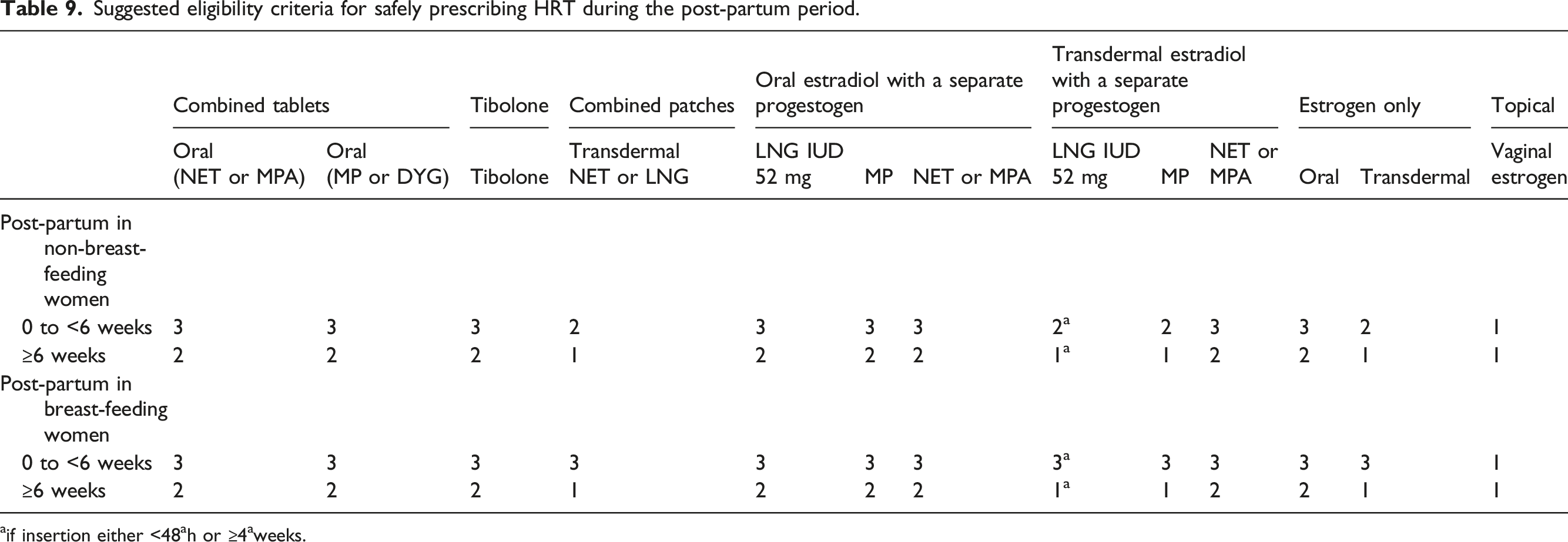
Post-partum
Pregnancy is uncommon in the menopause transition but not impossible and will be relevant to women with premature ovarian insufficiency who may have had fertility treatment. VTE risk is elevated during the post-partum period and this risk is most pronounced in the first 3 weeks after delivery declining to near baseline by 6 weeks.6,72–76 Other risk factors for VTE often co-exist and may pose additional risk, including immobility, transfusion at delivery, post-caesarean section, obesity, infection or pre-eclampsia. 6 Being post-partum includes any birth from 24 weeks. 6
Suggested eligibility criteria for safely prescribing HRT during the post-partum period.
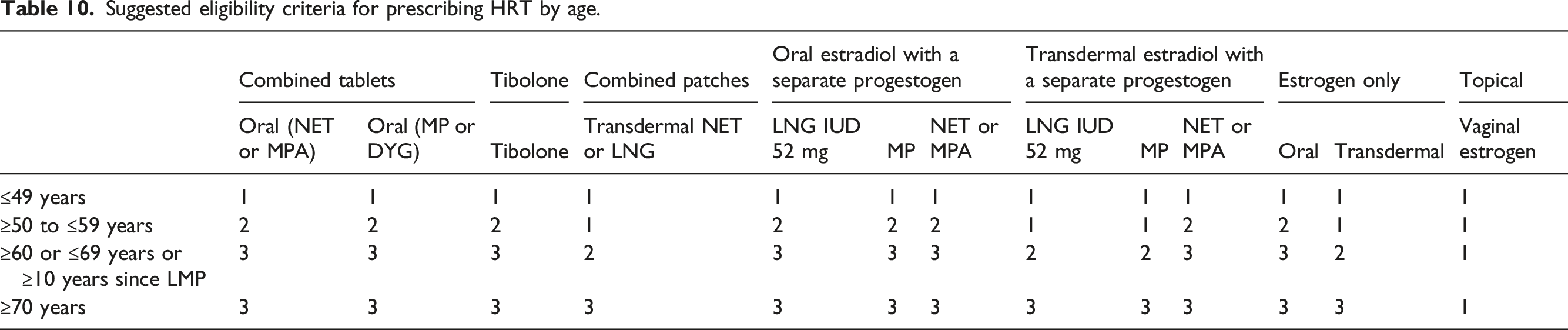
Age
Any decision to take HRT in older women should be made after an individualised discussion about the benefits and risks. 4 This should consider benefits to symptom control, quality of life, cardiovascular health and bone health, versus risks of developing a HRT-related health condition, such as VTE, MI, stroke or breast cancer. More careful considerations are required in older age groups, because the absolute risk of these health conditions, as well as other health conditions, increases with age.1,50,87,88
Suggested eligibility criteria for prescribing HRT by age.
Feasibility for development
This proposal demonstrates the potential for a set of eligibility criteria on the topic of VTE. Examples provided are for illustrative purposes only and do not represent any final guidance. The formation of final recommendations would require consensus from a panel of experts working within a clinical advisory group and would include a wider scope of topics such as cardiovascular health, stroke, migraine, previous cancers and drug interactions.
The feasibility of producing this type of guidance has already been demonstrated by the Spanish Menopause Society which used rigorous methodological tools to formulate eligibility criteria in nine clinical areas. 91 The authors described the most critical limitation being the lack of quality studies available for some subject areas and because of this, on occasions, a final recommendation was based on the best available evidence. 91 In some of the cases, this involved expert opinion based on data obtained in healthy women.
Conclusion
The creation of a set of HRT eligibility criteria would provide an easy to access, risk-based stratification tool that could be used as a decision-making aid for prescribing HRT in primary care. It has the potential to revolutionise prescribing by helping clinicians, particularly those with limited menopause experience, to feel confident in making decisions about HRT. It may benefit many women who have been denied HRT inappropriately, avoid prescribing in areas of risk, and help to reduce secondary care menopause related referrals. It could be developed into an app or other user-friendly digital interface and be aligned with current guidance on contraception. Any next steps would involve the formation of a clinical advisory group and formation of an evidence to decision framework. Even a small set of eligibility criteria would be likely to be beneficial to many healthcare professionals.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mr Tim Hillard BM DM FFSRH FRCOG. Declarations: Speaker and consultancy fees from Astellas and Besins; Prof. Nick Panay BSc MB BS FRCOG MFSRH. Declarations: Speaker and consultancy fees from Abbott, Astellas, Bayer, Besins, Gedeon Richter, Mithra, Mylan, Novo Nordisk, Roche, SeCur, Theramex and Viatris.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

