Abstract
Diatom and chrysophyte assemblages from varved sediments of meromictic Crawford Lake, Ontario record major environmental changes resulting from spatially broadening anthropogenic environmental stressors related to the “Great Acceleration” in the mid-20th century. Biannual assessment of diatom and chrysophyte assemblages over the last ~200 years allowed for rate of change analysis between adjacent samples that increased substantially during the mid-20th century, concurrent with significant generalized additive model trends. Changes in diatom and chrysophyte assemblages were likely driven by multiple anthropogenic stressors including local forestry harvesting, agriculture, and milling activities, acidic deposition from regional industrial processes, and anthropogenic climate warming. Novel siliceous algal assemblages now exist in Crawford Lake, likely related to the complexities of the above mentioned local and regional stressors. The major assemblage changes at the proposed base of the Anthropocene Epoch detected in this study support the laminated sequence from Crawford Lake as a strong potential candidate for the Anthropocene GSSP.
Keywords
Introduction
The anthropogenic impacts on the environment and climate of our planet cannot be overstated. Human activities have been particularly detrimental to freshwater systems, many of which have experienced and/or continue to experience eutrophication (Carpenter and Lathrop, 2008), acidification (Stoddard et al., 1999), changes in nitrogen cycling (Elser et al., 2009), carbon cycling dynamics (Alin and Johnson, 2007), and biodiversity loss (Lake et al., 2000). These changes are encompassed within a rapidly warming climate (Winslow et al., 2017; Woolway and Merchant, 2019; Woolway et al., 2020) which has made the management of freshwater systems extremely difficult. This is particularly true as anthropogenic stressors have generally broadened from local to regional and global spatial scales through time. The resulting environmental changes are expected to increase as the climate continues to warm and a growing global population requires more freshwater resources to sustain itself (Arnell, 1999; Sahoo et al., 2020; Vörösmarty et al., 2000). Considering these growing pressures, highly resolved and nuanced understanding of past freshwater system responses to anthropogenic impacts are required to better prepare for future management and conservation challenges.
Global environmental changes resulting from anthropogenic stressors increased dramatically during the mid-20th century as human populations grew exponentially (Steffen et al., 2007, 2015). The environmental impacts resulting from increases in energy use, fertilizer use, food consumption, water use, land development, and non-renewable resource extraction have been described as the “Great Acceleration,” and mark an unprecedented interval of human impact on the planet that has resulted in a fundamental shift in the composition and function of global Earth systems (Steffen et al., 2007, 2015; Subramanian, 2019a). The Great Acceleration began around 1950 and its compounded global impacts, including environmental isotopic signatures resulting from post-World War II nuclear weapons testing, have been proposed as the markers representing the beginning of the Anthropocene Epoch (Lewis and Maslin, 2015; Steffen et al., 2015; Waters et al., 2018; Zalasiewicz et al., 2017). A mid-20th century starting date for the Anthropocene Epoch has been proposed by the Anthropocene Working Group (AWG) and candidate sites for the Global Stratotype Section and Point (GSSP) are presently under investigation (Subramanian, 2019b; Waters et al., 2018).
The Anthropocene GSSP must preserve excellent biological, chemical, and/or physical records of mid-20th century global environmental change related to the Great Acceleration in a geologic sequence and provide a high-precision estimate for the starting date of the epoch (Lewis and Maslin, 2015; Waters et al., 2018; Zalasiewicz et al., 2017). Lakes are ideal locations for the Anthropocene GSSP as environmental and climate conditions are recorded in their sediments as biological subfossils or biogeochemical/geochemical markers. These data can be collected from sediment cores and may be used in tandem with measured historical limnological and/or climate data, or be used to infer past environmental and climate conditions when historical data are lacking (Adrian et al., 2009). Dating resolution is a primary constraint on the power of paleolimnological studies. In most sediment cores, dating resolution is assessed by the activities of 210Pb and 137Cs radioisotopes (for decadal and centennial studies) in sediment layers (Appleby, 2001; Benoit and Rozan, 2001; Schelske et al., 1994) which in turn are used to estimate sedimentation rates and estimates of age-depth profiles (Appleby, 2001). Specific lakes may be dated without the use of radioisotopes if their sediments are composed of recognizable seasonal deposition patterns that are termed varves (Lamoureux, 2001). In ideal circumstances, varved sequences can be resolved annually and provide accurate and precise estimates of the time of deposition. Accuracy and precision in dating sedimentary sequences are required to determine the extent of changes in biological composition associated with the Great Acceleration, correlated with the impacts of spatially broadening anthropogenic stressors, and to provide a highly resolved estimate for the GSSP boundary of the Anthropocene.
Previous studies of the varved sediments of meromictic Crawford Lake (Milton, Ontario, Canada) provide evidence of past anthropogenic impacts (Ekdahl et al., 2007; McAndrews and Boyko-Diakonow, 1989), and the varved sequence is currently under consideration as a potential candidate for the Anthropocene GSSP. Crawford Lake has been used to reconstruct centennial-scale changes in climate and vegetation cover (McAndrews and Boyko-Diakonow, 1989; Yu, 1997), fire history (Clark and Royall, 1995), Indigenous peoples’ history and agricultural practices (Clark and Royall, 1995; Ekdahl et al., 2007; McAndrews and Boyko-Diakonow, 1989), and cultural eutrophication and other effects due to European colonization of the area (Ekdahl et al., 2004, 2007; McCarthy et al., 2018). These studies have relied on various biological and geochemical proxies stored in the varved sediments of Crawford Lake (Boyko-Diakonow, 1979; Dickman, 1985) that have allowed researchers to estimate the dates of environmental changes at a high temporal resolution. Crawford Lake is meromictic and consequently allows for exceptional preservation of biological subfossils which act as proxies for past environmental conditions, and makes sediments from this lake ideal for paleolimnological examination of changes over the last ~200 years. This period has received relatively little attention at Crawford Lake despite the well-documented increases in anthropogenic impacts that have occurred over the 20th century (Steffen et al., 2015). This gap, however, provides an opportunity to build upon the well-known millennial history of Crawford Lake (Boyko-Diakonow, 1979; Ekdahl et al., 2004, 2007) and to investigate rapid environmental changes connected with the Great Acceleration (Steffen et al., 2007, 2015).
This study examines siliceous diatom and scaled chrysophyte subfossils collected at a high temporal resolution to examine biological responses to spatially broadening anthropogenic stressors over the last ~200 years of Crawford Lake. The siliceous frustules of diatoms and scales of chrysophytes preserve readily in lake sediments and can be identified to precise (e.g. species, subspecies, strain) taxonomic levels (Battarbee et al., 2001; Siver, 2003; Smol and Stoermer, 2010; Zeeb and Smol, 2001). Because specific taxa have known optima and tolerances to various environmental variables, diatoms, and scaled chrysophytes have been used to reconstruct changes in pH (Cumming et al., 1992; Siver, 1991), nutrient levels and lake trophic status (Bennion and Simpson, 2011; Siver and Marsicano, 1996), carbon cycling (Kingston and Birks, 1990; Wolfe and Siver, 2013), and climate (Hadley, 2012; Rühland et al., 2008; Smol and Douglas, 2007) across multiple time scales. At Crawford Lake, diatoms have been used to examine centennial changes in nutrient levels (Ekdahl et al., 2007), and chrysophyte stomatocysts (i.e. resting stages) with identifiable morphotypes have shown changes associated with lake trophic status over the past 300 years (Rybak et al., 1987). We hypothesize that changes in the species composition of diatoms and scaled chrysophytes will respond to the rapidly changing conditions of the Great Acceleration in the mid-20th century (Steffen et al., 2007, 2015). A rapid change at this time could support the varved sediment sequence from Crawford Lake as suitable for defining the onset of the Anthropocene Epoch (Lewis and Maslin, 2015; Subramanian, 2019b).
Study site
The limnological, geological, and climate characteristics of Crawford Lake have been well-described in previous studies of this famous site (e.g. Dickman, 1985; Ekdahl et al., 2004, 2007; McAndrews and Boyko-Diakonow, 1989; Rybak and Dickman, 1988) and will only be briefly restated here. Crawford Lake (43°28′06″N, 79°56′55″W) is in the Crawford Lake Conservation Area, operated by Conservation Halton (Figure 1). The lake is small (~2.4 ha), but has a maximum depth of ~24 m. A small, spring-fed stream enters the lake’s north end and outflow is through a bog at the south end of the lake. Crawford Lake is part of the Bronte Creek watershed and lies within a karstic dolomitic limestone basin that results in high alkalinity in its surface waters (see below). Vegetation cover is composed of mixed-wood trees including elm (Ulmus), oak (Quercus), birch (Betula), cedar (Thuja), and pine (Pinus) but old-growth forests were cleared in the mid-19th century by European colonizers (McAndrews and Boyko-Diakonow, 1989). Crawford Lake is meromictic with a sharp chemocline at ~15 m isolating the dense monimolimnion from the overlying mixolimnion. Recent studies suggest that the monimolimnion of Crawford Lake is oxygenated due to groundwater inflows (Heyde et al., in revision) but seasonal varves accumulate in the deep basin (Boyko-Diakonow, 1979). The climate of the Halton region is humid continental with a mean annual temperature of ~7.9°C and ~930 mm of mean annual precipitation over the 1981–2010 period (Hamilton, Ontario; Environment Canada, 2020). Both temperature and precipitation records extend to 1866 (Adjusted and homogenized Canadian climate data; Environment Canada, 2018) and show increasing average temperatures and generally stable levels of annual precipitation over the past ~150 years (Figure 2).
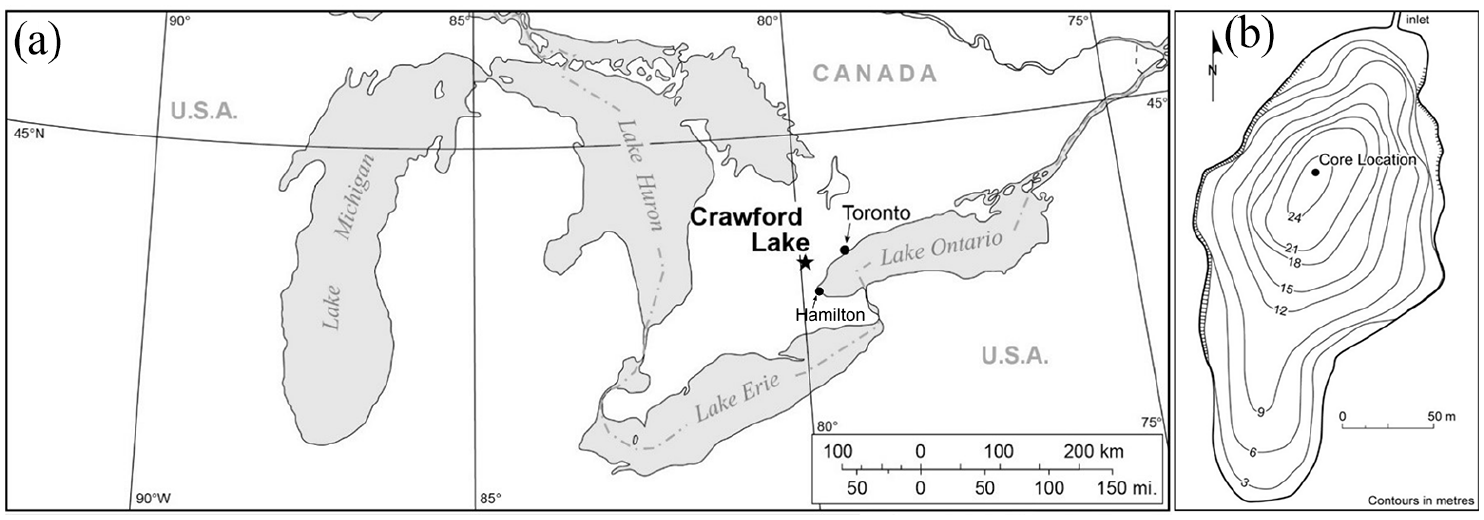
(a) Location of Crawford Lake in southern Ontario, Canada in relation to large population centers Toronto and Hamilton, Ontario, and the greater Laurentian Great Lakes region. (b) Bathymetry map of Crawford Lake with coring location denoted.
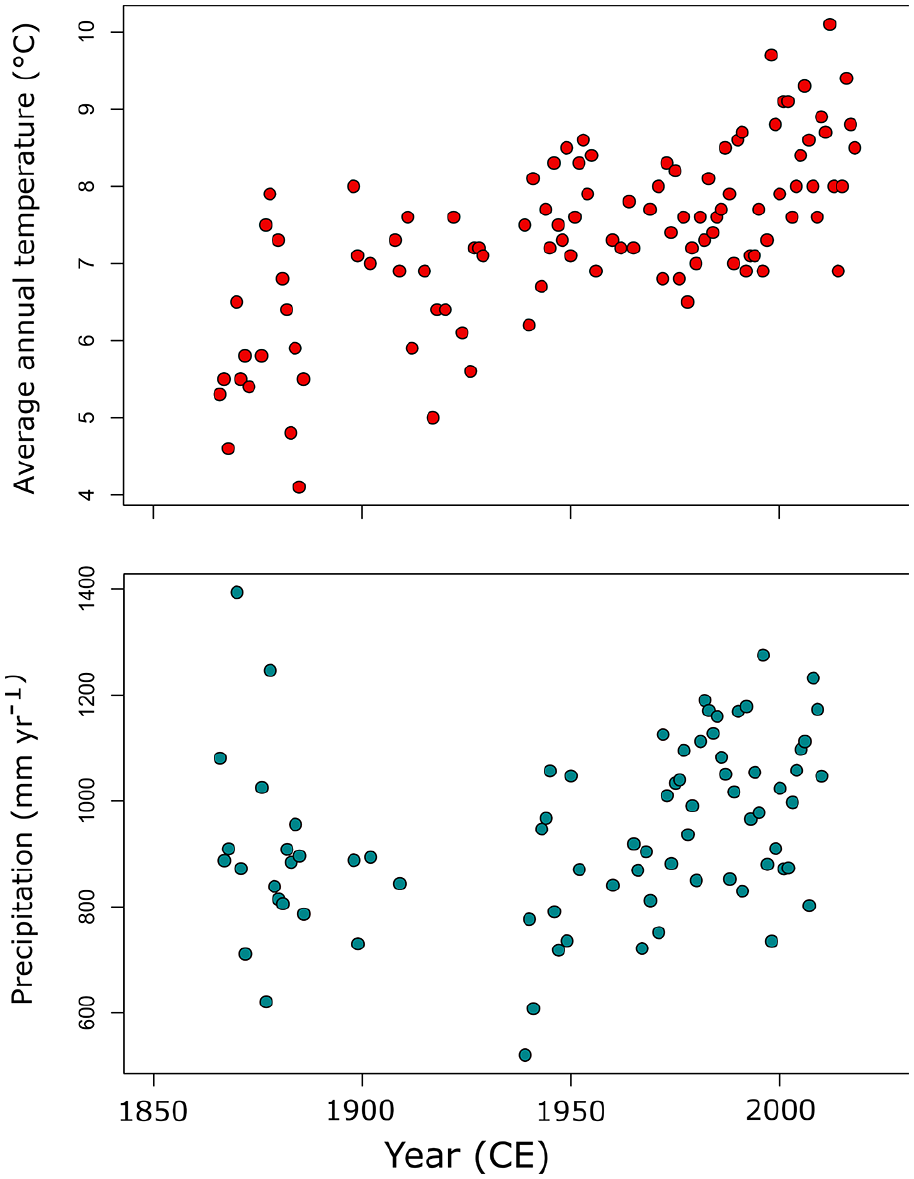
Homogenized historical average annual temperature (°C) and precipitation (mm year−1) data between 1866 and 2018 from Hamilton, Ontario, Canada.
Methods
Water chemistry
Physical and chemical variables of Crawford Lake were measured regularly between October 2019 and October 2020. Sampling was performed from a boat during the ice-free season, and from an augured ice hole in the winter months. Variables associated with water quality were measured using a Horiba U-5000 multiprobe sensor vertically in the water column of Crawford Lake at 1-m intervals. Of the variables measured in this manner, only pH data were incorporated into this study and the probe sensor was calibrated prior to every sampling date using a pH 4.00 buffer solution. For additional water quality data from Crawford Lake see Heyde et al. (in revision) and Llew-Williams et al. (in prep).
Discrete water samples were taken at selected depths in the water column using a Kemmerer water sampler and transferred into pre-rinsed 1-L high density polyethylene (HDPE) bottles. The bottles were labeled on site prior to sampling and transported in a cooler to E3 Labs (Niagara on the Lake, Ontario) where they were submitted for analysis of several geochemical variables (Llew-Williams et al., in prep), including alkalinity, total phosphorus (TP), and total nitrogen (TN), reported here. Samples underwent the APHA 2320B (mod) titration method to measure alkalinity and APHA 4500 methodology for nutrient analysis (APHA, 2005).
Sediment collection and siliceous algae analysis
An 89-cm length varved sediment core (CRA-19-2FT-B1) was collected a depth of 23 m from the ice (43°28′11.999″N; 79°57′0″W) over the deepest basin of Crawford Lake in February 2019 using freeze coring methods outlined in Glew et al. (2001); Figure 1. The freeze core was sub-sectioned in 2-year varve averages by hand at Carleton University (Macumber et al., 2011) with sub-samples being transferred to the PEARL laboratory at Queen’s University and stored at 4°C prior to the preparation of the samples for analysis of diatoms and scaled chrysophytes. A total of 34 samples between the varve-inferred dates of 1819 and 2011 were prepared for subfossil siliceous algae analysis. Varve analysis suggested that each interval contained 2-year increments and allowed for biannual resolution of siliceous algal data and statistical analyses (see below).
Sediment samples were prepared for both diatom and scaled chrysophyte analysis using the protocol outlined in Battarbee et al. (2001). Briefly, a small amount (~0.2 g) of wet sediment was digested in a 1:1 molar solution of concentrated nitric and sulphuric acid before being placed in a warm (~80°C) water bath for ~8 hours. Siliceous slurries were repeatedly aspirated each day to remove acidic supernatant which was replaced with deionized water. Eight rinses were necessary in order for the slurry to reach the pH of the deionized water. Slurries were then spiked with 1-mL of microsphere solution (copolymer 7508A series, Thermofisher, 7.9 µm; concentration = 2.0 × 107 spheres mL−1) and plated onto four coverslips with each coverslip being diluted from the original concentration by 50%. Coverslips were dried on a slide warmer, and were affixed to microscope slides using Naphrax®, with the aid of a hotplate. Diatom valves and chrysophyte scales were enumerated and identified using a Leica DMRB microscope with an oil immersible Fluotar objective (NA = 1.3) and oil immersible condenser (NA = 1.4) under 1000x magnification. Both diatom valves (Cumming et al., 1995; Krammer and Lange-Bertalot, 1986, 1988, 1991a, 1991b; Potapova et al., 2020; Spaulding et al., 2019) and chrysophyte scales (Siver, 1991) were identified to the finest possible resolution (i.e. species, strain) using taxonomic reference guides.
Numerical analyses
Diatom taxa which appeared at >1% relative abundance in at least two samples and all scaled chrysophyte taxa were square-root transformed (Legendre and Gallagher, 2001) and included in statistical analyses. Siliceous algae assemblages were separated into stratigraphically constrained zones using cluster analysis (CONISS) in Tilia (v. 2.0.2; Grimm, 2004) with a squared chord distance and broken-stick model validation using the rioja package (v. 0.9-26; Juggins, 2019) in R (R Core Team, 2019). Principal components analysis (PCA) was performed on siliceous algal data using the vegan package (v. 2.5-7; Oksanen et al., 2019) to determine directions and taxonomic drivers of diatom and chrysophyte assemblage changes through time. PCA axis-1 and -2 scores were extracted from the analyses and fitted with generalized additive models (GAMs) to determine periods of significant temporal changes in the siliceous algae assemblages. Thin plate regression splines, using restricted maximum likelihood (REML) and Gaussian error distributions were used in all models based on the recommendations of Simpson (2018). In all models, axis scores were modeled against varve-inferred date as the single covariate to determine temporal shifts in the data while temporal autocorrelation was assessed using continuous-time first-order autoregression (CAR(1)) correlation structure (Simpson, 2018). Model significance was determined by extracting the first derivative of the model trend based on the method described in Bennion et al. (2015). All GAMs were performed and analyzed using the mgcv package (v. 1.8-33; Wood, 2019) in R. Finally, Bray-Curtis dissimilarity (BCD) coefficients were calculated between adjacent diatom and scaled chrysophyte assemblages using vegan (Oksanen et al., 2019). These dissimilarity coefficients were standardized by the amount of time between samples as inferred by the varve date. Most samples (n = 31) were equidistant in time (biannual) based on varve year.
Results
Nutrients, pH, and alkalinity
Measurements of pH from Crawford Lake recorded slightly acidic conditions in the monimolimnion with an average of 6.1 in comparison to the slightly basic mixolimnion which had an average pH value of 7.25 (Figure 3). Density-derived stratification was evident in the alkalinity profile of Crawford Lake through the rapid and consistent increases in CaCO3 concentration across the chemocline (~15 m). The alkalinity of the mixolimnion was much lower (~278 CaCO3 mg/L) in comparison to the average value in the monimolimnion (~858 CaCO3 mg/L; Figure 3).
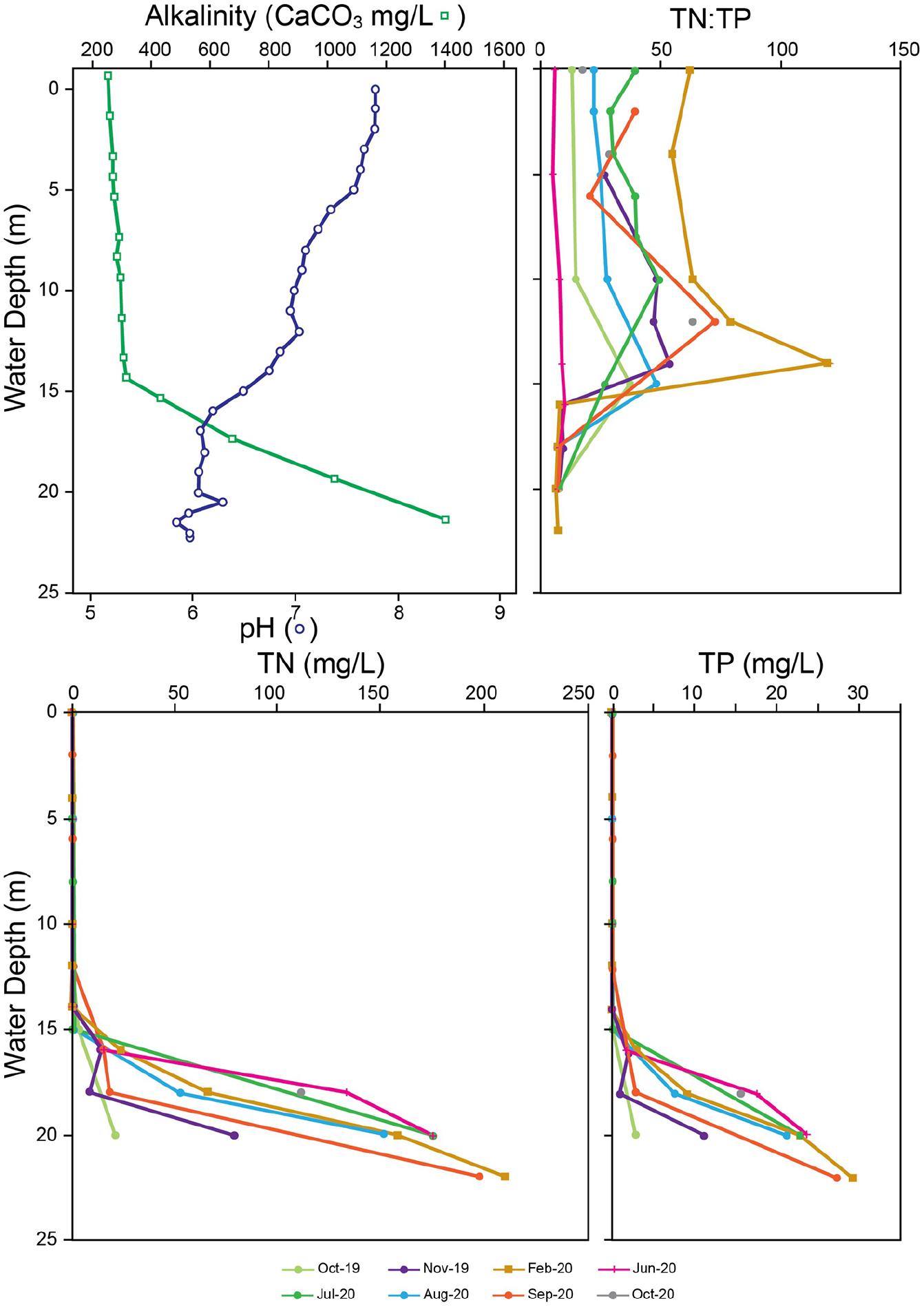
Measures of average pH and alkalinity (CaCO3 mg/L) between October 2019 and October 2020 of Crawford Lake plotted by water depth (m). Measures of total nitrogen (TN) and total phosphorus (TP) concentrations (mg/L), and TN:TP ratios from Crawford Lake between October 2019 and October 2020 plotted by water depth (m).
Similar to the trend in alkalinity, TN and TP concentrations increased sharply across the chemocline (Figure 3). The mixolimnion had average TN and TP values of ~0.6 and ~0.02 mg/L respectively. These nutrient values rapidly increased across the chemocline to averages of ~95.1 mg/L TN and ~13.1 mg/L TP within the monimolimnion (Figure 3). These patterns result in a sharp decrease in TN:TP molar ratios across the chemocline from variable values in the mixolimnion (Figure 3). TN:TP values were highest in the lower part of the mixolimnion in late summer and winter, and lowest during turnover in spring and fall (i.e. June and October; Figure 3).
Diatoms and scaled chrysophytes
CONISS analysis suggested the presence of three assemblage zones within the Crawford Lake diatom flora during the Canadian Zone (see Ekdahl et al., 2007) with high-level delineations occurring at ~1930 and ~1980 (Figure 4). Zone A1 occurred between ~1820 and ~1930 and was primarily composed of the planktic pennate diatoms Fragilaria crotonensis Kitton and Fragilaria tenera var. nanana Lange-Bertalot as well as the centric taxon Lindavia michiganiana Skvortzov with lesser occurrences of other planktic forms and minor abundances of benthic diatoms. Zone A2 occurred between ~1930 and ~1980 and was characterized by declines in planktic diatoms and increases in benthic taxa including Achnanthidium minutissimum Kütz, Navicula O. Müller, Cymbella Agardh, and Gomphonema Ehrenberg as well as several small benthic fragilarioid taxa (Figure 4). F. tenera var. nanana occurred at high relative abundances in this zone despite the decreases in other planktic taxa. Zone B occurred after ~1980 and contained large increases in Asterionella formosa Hassall from near negligible abundances. Abundances of Lindavia lemanensis (Chodat) T. Nakov et al. also increased in this zone while the previously common F. tenera var. nanana decreased. Benthic taxa also decreased in relative abundance in this zone.
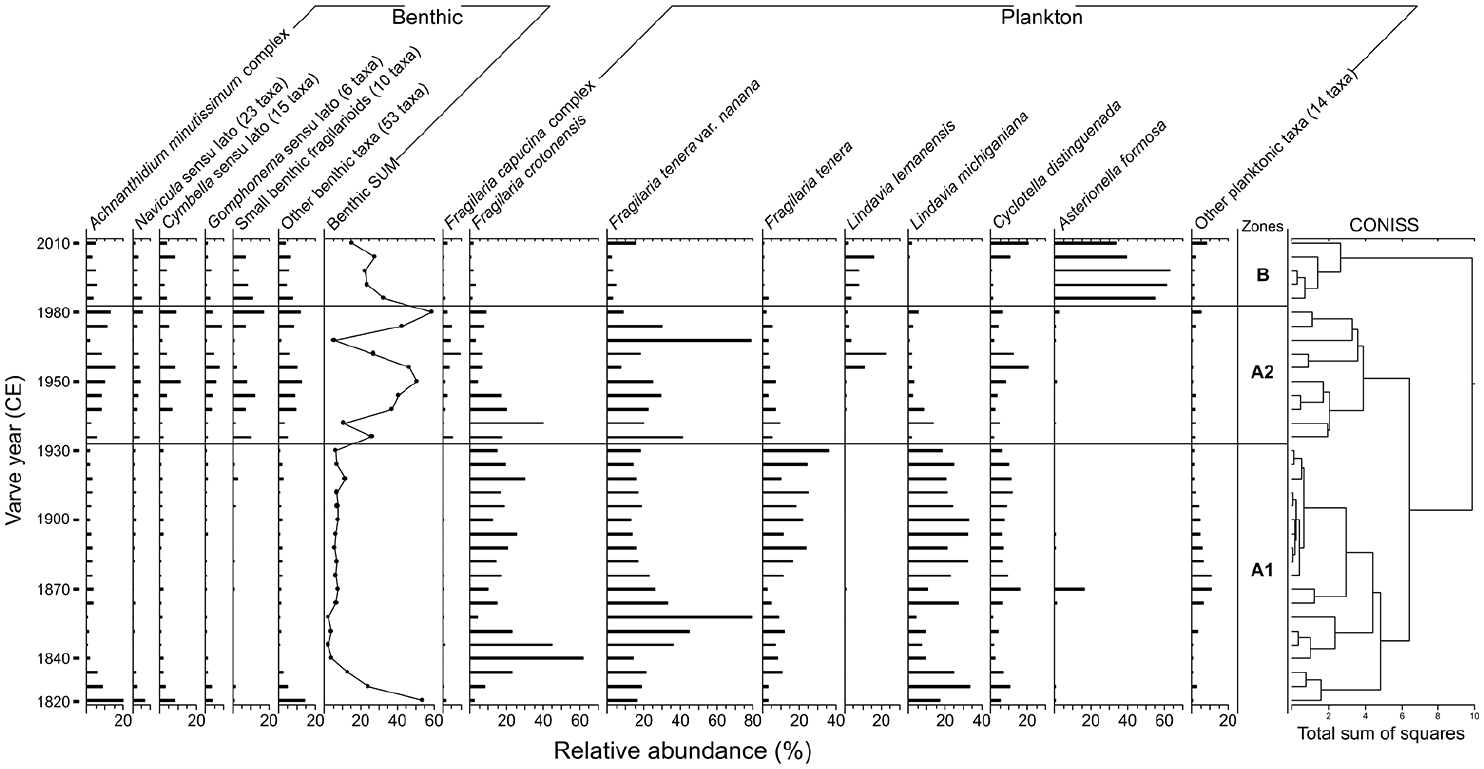
Relative abundances of common (>1%) and grouped diatom taxa from Crawford Lake plotted by varve year (CE). Taxa are split by functional ecology (i.e. benthic and planktic). Benthic taxa are summed. Diatom assemblage zones are separated by broken-stick model validated CONISS delineations.
CONISS analysis separated the scaled chrysophyte assemblage of Crawford Lake into two major zones with the delineation at ~1950 (Figure 5). Zone A occurred between ~1820 and ~1950 and contained high abundances of Mallomonas pseudocoronata Prescott as well as high abundances of Mallomonas tonsurata Teiling between ~1820 and ~1870. Zone B occurred after ~1950 and showed a decline in M. pseudocoronata and large increases in Mallomonas acaroides var. acaroides Asmund. In addition, abundances of Synura Korshikov taxa were high and variable throughout this zone, with the highest abundances occurring immediately after the assemblage transition at ~1950 (Figure 5).
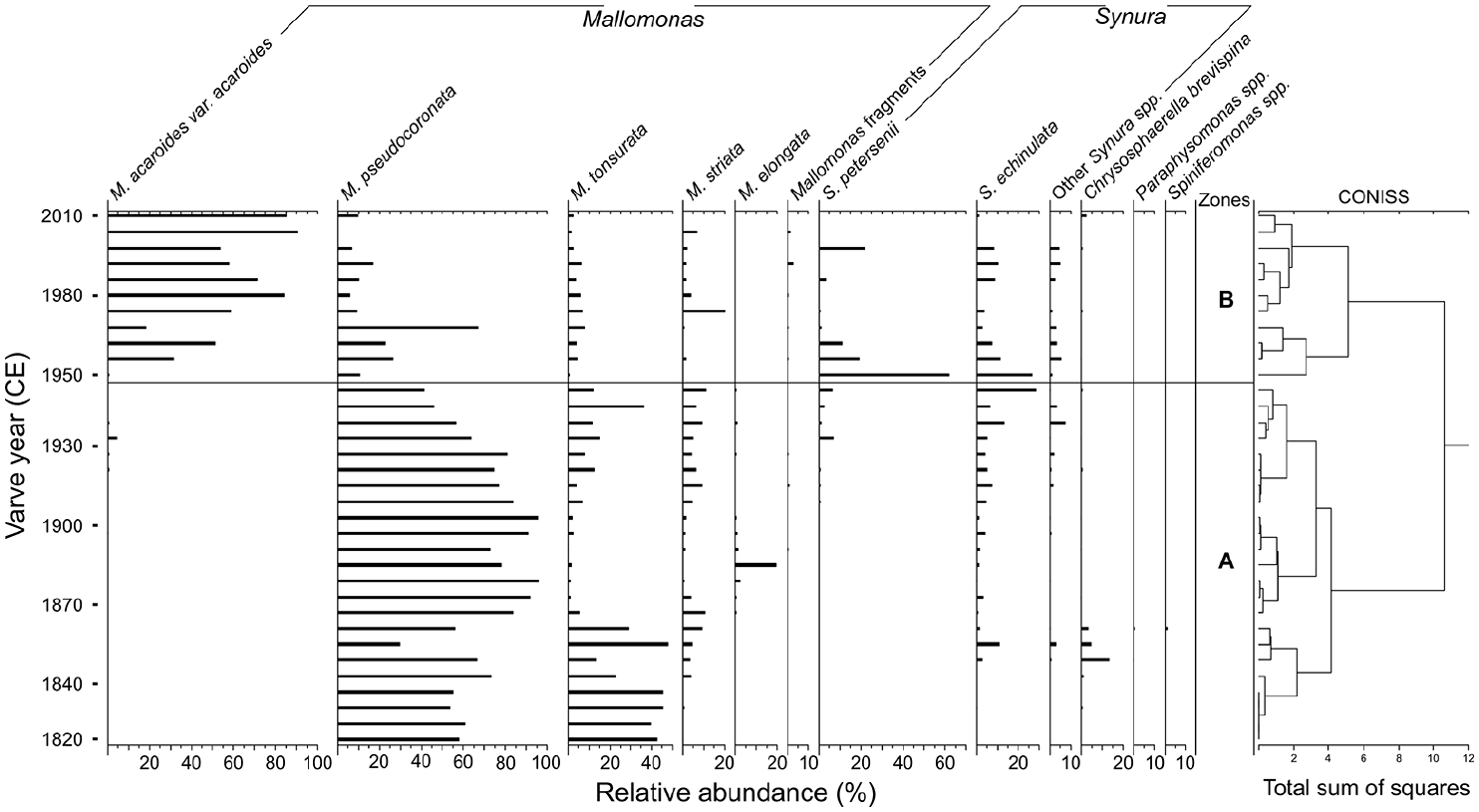
Relative abundances of chrysophyte taxa from Crawford Lake plotted by varve year (CE). Taxa are grouped by genera (i.e. Mallomonas and Synura) with additional taxa separated. Chrysophyte assemblage zones are separated by broken-stick model validated CONISS delineations.
Diatom PCA axis-1 explained ~45% of the variance in the diatom assemblage and was negatively correlated with high abundances of A. formosa and L. lemanensis and was positively correlated with high abundances of F. crotonensis, F. tenera W. Sm., and L. michiganiana (Figure 5a; Supplemental Appendix). Diatom PCA axis-2 explained an additional ~15% of assemblage variance and was negatively correlated with high abundances of F. tenera var. nanana, F. capucina var. gracilis Hust, and A. minutissimum (Figure 6a; Supplemental Appendix). Chrysophyte PCA axis-1 scores accounted for ~59% of the variance and was correlated positively with high abundances of M. pseudocoronata and low abundances of M. acaroides var. acaroides (Figure 6b; Supplemental Appendix). Chrysophyte PCA axis-2 explained ~14% of the variance of the assemblage and was positively correlated with high abundances of Synura taxa and was negatively correlated with high abundances of M. tonsurata (Figure 6a; Supplemental Appendix). In both ordinations, CONISS-derived zones were similarly separated from each other in ordination space (Figure 6).
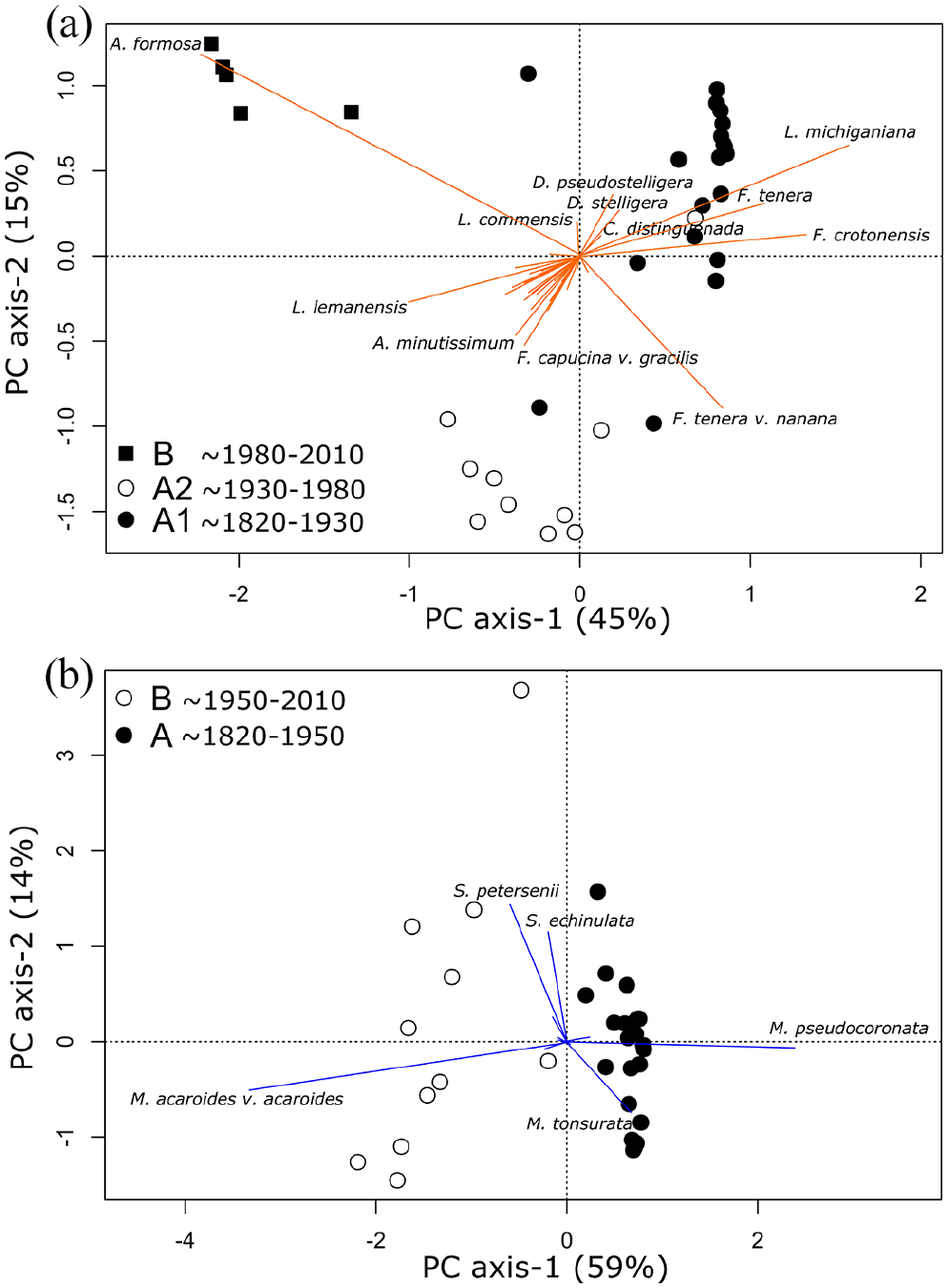
Biplots of PCA axis-1 v. PCA axis-2 of square-root transformed (a) diatom and (b) scaled chrysophyte abundances. In both biplots CONISS zones are marked by symbol shape and fill.
Diatom and chrysophyte PCA axis-1 and -2 scores served as the bases for generalized additive models to determine significant assemblage changes through time. The fitted GAM trend for diatom PCA axis-1 scores was stable from ~1820 to ~1920, after which significant changes occurred (Figure 7a). The fitted GAM trend for diatom PCA axis-2 scores showed similar, but more variable trends. Non-significant assemblage change occurred between ~1820 and ~1900 after which there were significant decreasing trends up to ~1950, followed by significant increasing GAM trends to ~2000 (Figure 7a). GAMs of chrysophyte PCA axis-1 scores were remarkably similar to the GAM of diatom PCA axis-1. Stable values began to change significantly at ~1930 and continued to do so up to ~2000 (Figure 7b). Chrysophyte PCA axis-2 scores, however, only showed a small period of significant trend change between ~1960 and ~1990 (Figure 7b).
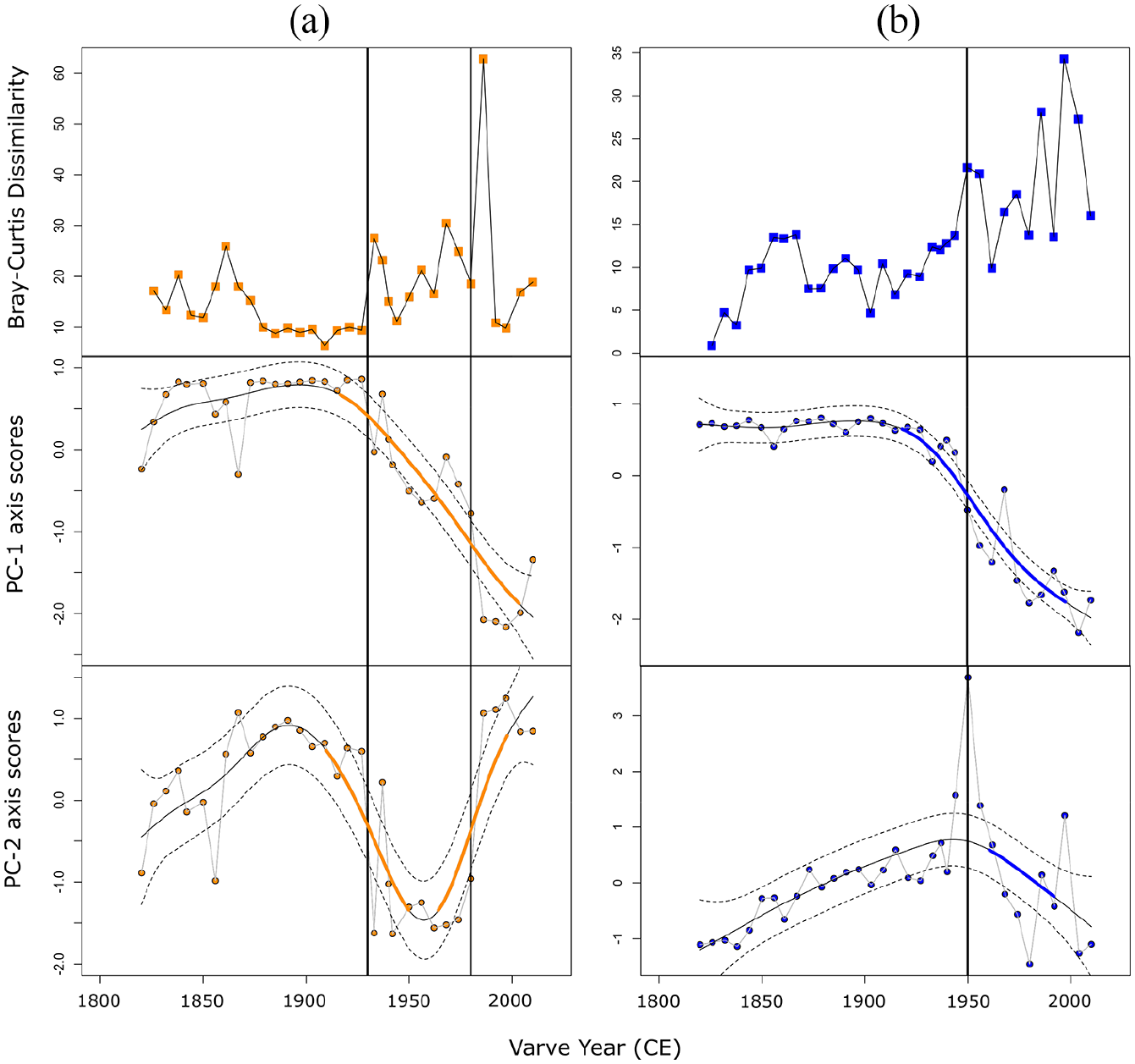
Bray-Curtis dissimilarity coefficients standardized for time (i.e. rate of change in assemblage structure between adjacent samples) and fitted generalized additive model (GAM) trends based on PCA axis-1 and -2 of Crawford Lake (a) diatom and (b) scaled chrysophyte assemblages plotted by varve year. For GAM plots, solid black lines are the fitted trends, dotted lines are 95% confidence intervals of the trend fit, and bolded lines represent periods of significant change. Vertical lines in all plots represent the times of CONISS-derived assemblage delineations (see Figures 4 and 5).
Diatom rate of change analysis (Bray-Curtis dissimilarity values between adjacent samples standardized by time) indicated highly similar assemblages up to ~1930, after which the rate of change between adjacent samples increased substantially. Values were dissimilar and variable between ~1930 and ~1980 (Figure 7a), with the greatest BCD coefficients at ~1980, after which A. formosa increased and maintained stable populations (Figure 3), resulting in a low rate of change between adjacent samples (Figure 7a). Chrysophyte rate of change increased slowly until ~1950 when large and variable changes in assemblage structure occurred (Figure 7b). Overall, chrysophyte BCD coefficients were lower than those from diatoms (Figure 7). Large changes in BCD coefficients, GAM-derived periods of significant assemblage changes, and CONISS-derived assemblage delineations generally occurred synchronously across both diatom and scaled chrysophyte assemblages over the past ~200 years of Crawford Lake (Figures 4–7), with increasing rates of change occurring in the mid-20th century and beyond.
Discussion
Analysis of subfossil diatom and scaled chrysophytes identified major environmental changes over the past ~200 years at Crawford Lake. Significant temporal trends in the GAM models of the main direction of variation of both the diatom and chrysophyte assemblages occurred in the mid-20th century (Figures 4, 5, and 7) and likely represent movement from local to regional stressors, including sulfur deposition and lake-water clearing due to increased industrial activity in nearby Hamilton, Ontario (~30 km upwind of Crawford Lake) during the Great Acceleration. Further changes in the diatom and chrysophyte assemblages after ~1980 may reflect a change to new ecological states potentially associated with climate warming and increases in dissolved organic carbon (Figure 2). These patterns are reflected in the greatest turnover in species composition of diatoms and scaled chrysophytes (i.e. highest BCD coefficients) in the late 20th century (Figure 7). Considering its accessible location, varved sediments which allow for accurate and high-precision dating, and the severity of mid-20th century anthropogenic impacts found in this study, Crawford Lake is a strong potential candidate for the Anthropocene GSSP.
Siliceous algal response to anthropogenic stressors at Crawford Lake
Previous studies of the diatom assemblages of Crawford Lake suggested that the lake experienced cultural eutrophication in the 14th century due to indigenous farming, and again by the mid-19th century due to deforestation and agriculture by European colonizers (Ekdahl et al., 2007). Our data agree with this assessment as high abundances of the meso-eutrophic diatom F. crotonensis prior to ~1860 would have required high nutrient levels (Cumming et al., 2015), consistent with land clearing and agriculture in the small (~126.5 ha) watershed during the mid-19th century. Relatively high levels of precipitation during the late 19th century (Figure 2) would have facilitated a high degree of terrestrial run-off and nutrient loading into Crawford Lake leading to increases in nutrients and dissolved organic carbon (DOC) from the cleared landscape (Ekdahl et al., 2004, 2007; McAndrews and Boyko-Diakonow, 1989; Rybak and Dickman, 1988). The resulting increase in biological production decreased light penetration in the lake (Morris et al., 1995; Williamson et al., 1996) and reduced the extent of the littoral area available for phytobenthic production in Crawford Lake and likely contributed to the lack of benthic diatoms between ~1840 and ~1930 (Gushulak and Cumming, 2020; Kingsbury et al., 2012), and the high abundance of meso-eutrophic pennate F. tenera and centric L. michiganiana planktic diatoms (Figure 4; Bracht et al., 2008; Cumming et al., 2015). Increased biological production would have also resulted in increased biochemical oxygen demand (BOD—the amount of dissolved oxygen required for the decomposition of organic matter by aerobic bacteria in the water column) and produced several decades-long intervals of hypoxic-to-anoxic conditions in the monimolimnion during this time (Heyde et al., in revision; Llew-Williams et al., in prep.). These are recorded by the exceptional preservation of cellulosic dinoflagellate thecae (Krueger and McCarthy, 2016) and the sharp decline in ostracods endemic to the monimolimnion (Chan, unpublished data; Finlayson et al., in prep.) in sediments dating to the intervals of Iroquoian and Canadian land use in the watershed. Groundwater recharge would have been relatively constant over the last millennium, with slight climatic variation, so estimates of phytoplankton influx are critical to modeling the balance between the inflow of DO through recharge and the BOD in the isolated monimolimnion (Llew-Williams, in prep.). Increases in terrestrial material and nutrient levels were exacerbated by the construction of a sawmill that expelled waste directly into Crawford Lake beginning at 1860 (Ekdahl et al., 2007), and may have caused increases in the scaled chrysophyte taxon M. pseudocoronata over M. tonsurata which declined substantially at this time (Figure 5; Siver, 2003; Siver and Marsicano, 1996). Crawford Lake retained high DOC and nutrient levels with associated depressed light penetration, as well as high BOD and bottom water anoxia until the sawmill closed in the early 20th century, and broad, regional-scale anthropogenic stressors, including sulfur deposition, were introduced to the site.
Significant changes in both diatom and chrysophyte assemblages, as determined by first-derivative extractions of fitted GAM trends, occurred during the mid-20th century (Figure 7). The abundances of benthic diatoms increased sharply at ~1930, resulting in high rates of assemblage change between samples (Figure 7a) and a relatively distinct separation of diatom assemblages (CONISS; Figure 4). The changes in the diatom assemblages were generally contemporaneous with a major shift in chrysophyte assemblages (Figure 5) where abundances of M. pseudocoronata decline substantially with increases in M. acaroides var. acaroides and Synura taxa. These changes suggest increases in light penetration and clearer water beginning at ~1930 (Gushulak and Cumming, 2020; Hadley, 2012; Kingsbury et al., 2012; Siver, 2003). Initial increases in benthic diatoms and Synura taxa may be attributed to decreases in DOC influx into the lake as local agriculture practices began to decline and forests were re-established (Ekdahl et al., 2007; McAndrews and Boyko-Diakonow, 1989), but further increases in these taxa may be due to increases in lake transparency as a result of disruption of DOC transport from the catchment from sulfur deposition in the mid-20th century (Hadley, 2012; Meyer-Jacob et al., 2020).
Deposition of sulfur oxides and other acidic compounds resulted in the acidification of numerous lakes in Ontario and the northeast United States (Meyer-Jacob et al., 2019; Stoddard et al., 1999). Crawford Lake was not acidified by this deposition, due to its limestone setting and high alkalinity in the surface waters (Figure 3; Ekdahl et al., 2007; McAndrews and Boyko-Diakonow, 1989) despite the likely dissolution and transport of carbonic acid from the limestones into the lake. Instead, the acidic compounds that were deposited to the soils surrounding Crawford Lake during the mid-20th century would have bonded to organic carbon sources and limited its transport to the lake thus reducing lake-water DOC concentrations (Ekström et al., 2011; Hall et al., 2021; Hruška et al., 2009; Monteith et al., 2007). This reduction of DOC concurrent with declines in local nutrient inputs due to reforestation (Keller and Fox, 2019) would have allowed for an extended littoral zone for phytobenthic production (Vadeboncoeur et al., 2008) that resulted in the increases in benthic diatoms in Crawford Lake centered on ~1950 (Ekdahl et al., 2007; Gushulak and Cumming, 2020; Kingsbury et al., 2012). In addition, nutrient and carbon declines would have lowered lake production and BOD resulting in higher oxygen content in monimolimnion as it was continually resupplied by oxygenated ground waters (Heyde et al., in revision; Llew-Williams et al., in prep). Furthermore, Hadley (2012) observed that increases in DOC in boreal lakes within Algonquin Provincial Park (Ontario) corresponded with declines in Synura taxa suggesting that the opposite relationship occurred during mid-20th century water clearing in Crawford Lake. Clear water in Crawford Lake occurred up to ~1980 when large declines in the deposition of sulfur and nitrogen oxides occurred due to the adoption of strict anti-pollution legislature (National Air Pollution Surveillance Program [NAPS], 2019).
The late 20th century was marked by significant diatom and scaled chrysophyte assemblage changes (Figure 7). Declines in benthic diatom taxa likely reflect decreases in light penetration as re-browning following acid deposition (Meyer-Jacob et al., 2019, 2020; Monteith et al., 2007) possibly due to an increase in DOC influx due to long-term reforestation and conservation efforts including the construction of an interpretive center and boardwalk around the lake as well as the archeological excavation of an Iroquoian settlement beginning in the mid-1970s. These projects would have also transported additional nutrients and solids into Crawford Lake, further increasing lake turbidity. Increases in DOC transport are likely responsible for the slightly acidic pH of the monimolimnion as associated humic acids would become constrained to below the chemocline following intrusion to the lake (Figure 3). Despite increases in nutrients, BOD in Crawford Lake remains low enough that the monimolimnion remains oxygenated throughout the year (Heyde et al., in revision). In both diatom and scaled chrysophyte assemblages, however, the post-1980 assemblages are novel (Figures 4 and 5). This is in part due to changes in land-use practices in the intervening ~150 years as well as lower surface water nutrient levels (TN = Figure 3) which do not support the more nutrient-rich assemblage of diatoms and chrysophytes from the agricultural period (Cumming et al., 2015; Ekdahl et al., 2004, 2007). Instead, diatom assemblages were primarily composed of planktic, colonial A. formosa, and large centric L. lemanensis, while M. acaroides var. acaroides has replaced the previously highly abundant M. pseudocoronata and Synura chrysophytes (Figures 4 and 5).
Sharp increases in A. formosa are not uncommon in relatively recent sediments across North America, although conflicting hypotheses make the interpretation of this phenomenon at Crawford Lake difficult. Abundances of A. formosa have been shown to increase under heightened total nitrogen to total phosphorus (TN:TP) ratios suggesting that regional increases in atmospheric nitrogen deposition may be related to the increase in abundance of this diatom (Saros et al., 2005, 2011; Sheibley et al., 2014; Wolfe et al., 2003). Water chemical analysis suggests that the surface waters of Crawford Lake have relatively elevated nutrient concentrations (TN = ~0.6 mg/L, TP = ~0.02 mg/L; Figure 3). The chemocline restricts additional high concentrations of nutrients to the monimolimnion where they cannot impact the phytoplankton communities of the mixolimnion (Figure 3; Ekdahl et al., 2007; Gulati et al., 2017). The high abundances of A. formosa in Crawford Lake post ~1980 may therefore be due to high TN:TP ratios that occur in the late summer and early fall of the site (Figure 3) as TP is used up throughout the growing season, and may allow this taxon to proliferate during the fall bloom prior to freezing (Saros et al., 2005, 2011).
Alternatively, increases in A. formosa have occurred in lakes which have experienced declines in nutrient levels, including nitrogen, in recent years. This pattern suggests that recent increases in A. formosa in Crawford Lake are tied to increasing temperatures or changes in internal lake thermal stratification patterns (Enache et al., 2011; Hadley et al., 2013; Sivarajah et al., 2016). Climate records from Hamilton, Ontario show that mean annual temperature has increased by ~1°C between 1970 and 2010 (Figure 2; Environmental Canada, 2021) which may partially explain the increase of A. formosa in the lake after ~1980. This would also allow for contemporaneous increases in L. lemanensis which proliferates in mid-depth waters when high temperatures result in deeper thermoclines and long periods of stratification (Saros et al., 2012). In Crawford Lake, the thermocline occurs between ~5 and 10 m (Ekdahl et al., 2007; Heyde et al., in revision), which may allow for high abundances of this taxon (Saros et al., 2012).
The replacement of M. pseudocoronata with M. acaroides var. acaroides is also most likely explained by climate warming. Mushet et al. (2017) found that increases in the concentration of M. acaroides var. acaroides occurred within northern Saskatchewan lakes that had experienced climate warming over the past century. This was despite varying amounts of nitrogen deposition from the nearby Athabasca oil sands into groups of lakes that were selected a priori to be either N or P limited. This pattern was also seen at the Experimental Lakes Area, which is relatively unimpacted by regional nitrogen deposition (Mushet et al., 2018), further suggesting the impacts of recent climate warming on chrysophyte assemblages (Laird et al., 2013; Mushet et al., 2018). Wolfe and Siver (2013) have also noted that chrysophyte concentrations increase dramatically during periods of enhanced pCO2 concentrations in lakes as these organisms rely on diffuse CO2 to enter their cells during photosynthesis. Concentrations of pCO2 have likely increased in Crawford Lake in recent decades as shown in elevated acidity in the monimolimnion (Figure 3) as a result of CO2 accumulation resulting from biological respiration and decomposition below the chemocline (Gulati et al., 2017). Although the mechanism of change is beyond the design of the current study, elevated nutrient levels, high DOC levels, and a warming climate, may be plausible explanations, alone or in combination, for the proliferation of M. acaroides var. acaroides during the late 20th and early 21st centuries at Crawford Lake (Lapierre and del Giorgio, 2012; Wolfe and Siver, 2013). When examined together, the increases in A. formosa and M. acaroides var. acaroides in recent decades suggests that increasing average temperatures greatly impacted the siliceous algal assemblages at Crawford Lake (Figure 2).
The Great Acceleration and the Anthropocene GSSP
Assessment of diatom and scaled chrysophyte assemblages from Crawford Lake record significant changes in the mid-20th century (Figure 7). These changes are tied to spatially broadening anthropogenic stressors beginning with early-19th century local deforestation and agriculture that resulted in non-significant assemblage changes, likely due to the lack of a pre-disturbance period in this core. Lake conditions were further changed by the impacts of regional urbanization and industry during the Great Acceleration (Steffen et al., 2015) as indicated by significant algal assemblage changes (Figure 7). Increases in both diatom and chrysophyte rates of change at ~1950 (Figure 7) highlights the broad-scale impacts of the Great Acceleration compared to previous local environmental stressors. High rates of change occurred in the diatom assemblage until ~1980 when major changes in diatoms occurred due to regional management of pollution and increasingly warmer temperatures due to global climate change. In contrast, rates of change in the scaled chrysophyte assemblages continue to increase during the late-20th and early 21st centuries. High rates of change in both diatom and scaled chrysophyte assemblages suggest that despite local and regional management, global stressors may continue to alter the ecosystem of Crawford Lake (Steffen et al., 2015).
The varved sediments of Crawford Lake allow for the highly resolved dating of the lake sediments in this study, and also provide opportunity for further research. Presently, the AWG will be voting on the establishment of an Anthropocene GSSP to mark permanent anthropogenic effects on the planet (Subramanian, 2019b). This study not only shows how freshwater biological proxies responded faster to spatially broadening anthropogenic stressors as a part of the Great Acceleration but notes that the most significant changes occurred in the mid-20th century, the date proposed for the Anthropocene GSSP (Subramanian, 2019b; Waters et al., 2018; Zalasiewicz et al., 2017). The varved sediments of Crawford Lake have been proposed as a potential GSSP and are currently under examination for high resolution 210Pb and 137Cs dating, changes in pollen and non-pollen palynomorph assemblages, as well as changes in biomarker pigments, elemental geochemistry, and plutonium as a marker of mid-20th century nuclear weapons testing (Waters et al., 2015). Should these additional proxies also record major anthropogenic environmental changes at the same horizon, then Crawford Lake may prove an ideal location for the Anthropocene GSSP. This study provides strong evidence for this position and suggests that the environmental changes recorded at Crawford Lake are indicative of increased global anthropogenic stressors during the mid-20th century and the beginning of the Great Acceleration.
Conclusions
High rates of significant changes in diatom and chrysophyte assemblages occurred during the mid-20th century of Crawford Lake and reflect spatially broadening anthropogenic impacts and the onset of the Great Acceleration (Steffen et al., 2015). Results from this study also show that global climate change can override freshwater biological responses to local and/or regional management of freshwater environmental stressors. At Crawford Lake, increasing temperatures have likely resulted in novel algal community states despite local reforestation, and regional reduction in atmospheric industrial pollution. These patterns highlight ongoing anthropogenic impacts on the freshwater environments.
The varved sediments of Crawford Lake have allowed for biannual temporal resolution in this study and resulted in confident estimates on the timing of major environmental and climate changes centered at the mid-20th century (Steffen et al., 2015). These characteristics are inline with the AWG’s conditions for an Anthropocene GSSP (Subramanian, 2019b). Additional environmental and climate proxies and key chemical markers of the nuclear arms race are under examination in the varved sediments of Crawford Lake to test the lake as the potential marker of the Anthropocene.
Supplemental Material
sj-docx-1-anr-10.1177_20530196211046036 – Supplemental material for Siliceous algae response to the “Great Acceleration” of the mid-20th century in Crawford Lake (Ontario, Canada): A potential candidate for the Anthropocene GSSP
Supplemental material, sj-docx-1-anr-10.1177_20530196211046036 for Siliceous algae response to the “Great Acceleration” of the mid-20th century in Crawford Lake (Ontario, Canada): A potential candidate for the Anthropocene GSSP by Cale AC Gushulak, Matthew Marshall, Brian F Cumming, Brendan Llew-Williams, R Timothy Patterson and Francine MG McCarthy in The Anthropocene Review
Footnotes
Acknowledgements
The authors thank the managers of Conservation Halton for access and permission to continue to work on Crawford Lake. Special thanks are given to Andrew L. Macumber, Riley Steele, and Carling Walsh (Carleton University) for field assistance, Krysten Lafond (Brock University) and Nawaf Nasser (Carleton University) for sediment preparation, and Mike Lozon (Brock University) for aid in figure drafting. Data can be made available from the authors at reasonable request. Samples and data that form the basis of this study were collected from land covered by Canada’s Upper Canada Treaties, the traditional territory of Anishnabek, Huron-Wendat, Haudenosaunee (Iroquois), and Ojibway/Chippewa First Nations people.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by an NRCan Clean Technology (#CGP-17-0704) grant to R.T. Patterson, and NSERC Discovery grants to R.T. Patterson, B.F. Cumming, and F.M.G. McCarthy with additional support from the Haus der Kulturen der Welt.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
