Abstract
Objective:
Antibiotic prophylaxis reduces infection risk following transrectal prostate biopsy. Studies in countries with high antibiotic resistance show that fosfomycin, given an hour or more before biopsy, has equal or better outcomes than ciprofloxacin. This study aimed to evaluate if fosfomycin, administered immediately before biopsy, is as effective as ciprofloxacin in Sweden, where antibiotic resistance is low.
Material and Methods:
A randomised, non-inferiority study of different antibiotic prophylactic regimes, including men undergoing transrectal prostate biopsy, was conducted. A total of 3448 patients were planned to be included. Primary outcome was hospitalisation due to urinary tract infection (UTI) within 14 days. Men without risk factors for infection received either fosfomycin or ciprofloxacin immediately before biopsy. Patients with risk factors received additional doses post-biopsy.
Results:
The study was stopped by the safety board after enrolment of 42 men. Four of 20 men (25%) in the fosfomycin group were hospitalised due to UTI, including two with positive blood cultures for Pseudomonas, whereas no hospitalisations (0%) occurred in the ciprofloxacin group. The main limitation was the small sample size.
Conclusion:
Fosfomycin administered immediately before biopsy was not supported by this study. The results may be skewed by the high incidence of Pseudomonas infections.
Level of evidence:
2
Introduction
Prostate biopsy is performed on approximately 1,000,000 men in Europe annually. 1 The most common complications are urinary tract infections (UTIs). 2 The risk of infections has increased over time.3–5 In Sweden, 1–4% of patients are hospitalised due to infections post-biopsy.3–6
Antibiotic prophylaxis reduces the risk of infections.7,8 The most common prophylaxis in Sweden is ciprofloxacin.9,10 Ciprofloxacin-resistant Escherichia coli is a strong risk factor for UTI after prostate biopsy. 11 The European guidelines do not recommend ciprofloxacin prophylaxis anymore, but in Sweden, the standard prophylaxis is ciprofloxacin. 12
The European Association of Urology (EAU) guidelines also recommend transperineal biopsies as the primary choice of prostate biopsy due to increasing antimicrobial resistance following transrectal procedures. Transrectal biopsies with targeted prophylaxis according to rectal culture are considered a secondary option. 13 A meta-analysis showed a lower risk of sepsis when multiple doses of antibiotics were used. 14
In Sweden, the primary route for prostate biopsy is still transrectal, and transperineal biopsies are the standard choice only at a few centres. 12 Swedish urology departments administer the prophylaxis just before the biopsy. 10 The rationale is a study where Lindstedt showed that late administration was as effective as prophylaxis administered 2 hours before. 15
A meta-analysis showed a reduction in infectious complications when fosfomycin was compared to ciprofloxacin as antibiotic prophylaxis before transrectal biopsy. 16 Fosfomycin inhibits the synthesis of the bacterial cell wall. 17 It is considered active against E. coli. 17 Administration of oral fosfomycin 1–4 hours prior to intervention results in concentrations in the prostate that are adequate to prevent infections, based on in vitro data. 18 There are no studies addressing administration of fosfomycin immediately before prostate biopsy.
The aim of this study was to assess if fosfomycin administered immediately prior to prostate biopsy was as effective as ciprofloxacin in a context with low rates of antibiotic resistance.
Material and methods
This was a randomised, controlled, open, multicentre, non-inferiority, phase II drug trial with parallel groups. The CONSORT guidelines for reporting and conducting randomised studies were followed. 19 An open design was used. Blinding of the study drugs was not expected to affect the primary outcome variable. Inclusion began on 5 June 2018 and ended prematurely on 23 May 2019, following a decision by the safety committee of the trial.
Population
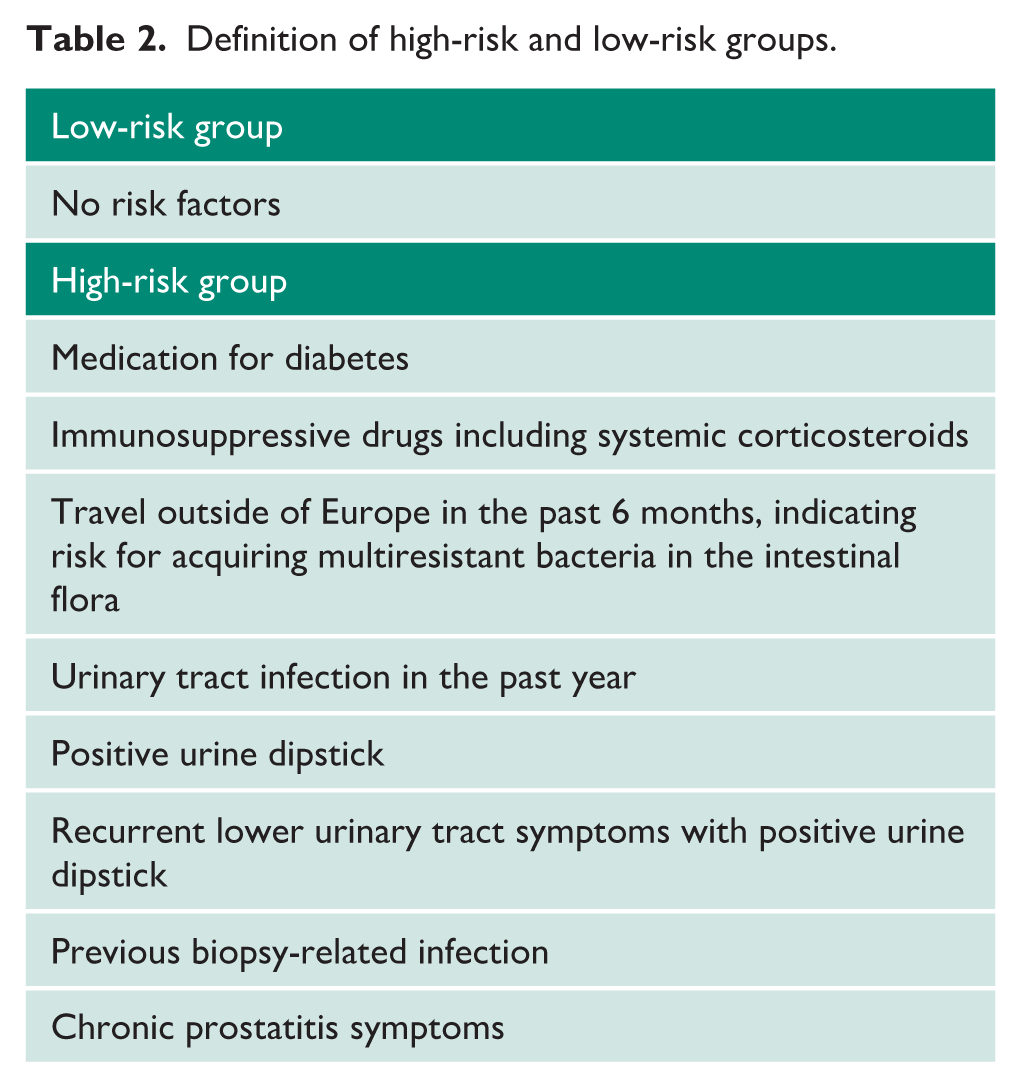
Men aged 18 years or older with an indication for prostate biopsy were included at three different Swedish hospitals (Örebro, Sundsvall and Linköping) after giving informed consent to participate in the trial. Table 1 shows the inclusion and exclusion criteria. The patients were divided into a low-risk group without risk factors for infection and a high-risk group (see Table 2).
Inclusion and exclusion criteria for randomised trial of fosfomycin versus ciprofloxacin as immediate antibiotic prophylaxis before prostate biopsy.

Definition of high-risk and low-risk groups.
Randomisation
The research nurse at each site entered the information on biopsy date, exclusion criteria and risk group classification into a web-based randomisation system, DynaReg. The system immediately reported drug, dose and treatment duration back to the clinic. Randomisation was done in blocks, stratified by study site.
Intervention and comparison
Study Group A: Low-risk group with no risk factors for infection. The patients were randomised to receive either oral granules of fosfomycin-trometamol (Zambon) 3 g × 1, immediately before the biopsy or an oral tablet of ciprofloxacin (Hexal) 750 mg × 1, immediately before the biopsy.
Study Group B: High-risk group with one or more risk factors for infection. The patients were randomised to receive either two doses of oral granules of fosfomycin-trometamol (Zambon) 3 g × 1, immediately before the biopsy and 24 hours after the biopsy or oral tablets of ciprofloxacin (Hexal) 500 mg × 2 for 3 days starting immediately before the biopsy.
Rectal povidone-iodine was not used in any of the groups.
Follow-up
A phone interview, conducted by a research nurse at the Research Unit at Sundsvall Hospital, was carried out 14 days after the biopsy to determine whether the patients had been hospitalised due to infection. The occurrence of fever, pain and bleeding was also recorded. An English version of the interview guide is provided in Supplemental Appendix 1.
Primary outcome variable
Hospitalisation due to febrile UTI within 14 days after prostate biopsy
Data were collected during the telephone interview. The choice of a 14-day limit for follow-up in the hospital registry was based on previous studies that used this limit.3,15,20
Secondary outcome measures
Hospitalisation for reasons other than infection within 14 days of the biopsy.
Treatment with antibiotics for symptomatic UTI or presence of fever within 14 days of the biopsy.
Pain, blood in the urine or stool, or urinary tract symptoms.
Blood in the semen.
Positive urine culture or positive blood culture with a urinary tract pathogen within 30 days after the biopsy. Data were collected from the local microbiological laboratories.
Screening cultures of rectal swabs
To screen for resistance to the antibiotics, a rectal swab (Eswab, Copan) was obtained from study participants before the first dose of antibiotics and at the follow-up visit 2 to 4 weeks after the biopsy. The samples were sent to a central laboratory (Clinical Microbiology, Örebro, Sweden). In total, 10 µL were cultured on selective agar (UriSelect, BioRad) supplemented with vancomycin (7.5 mg/L) and fosfomycin (32 mg/L) or nalidixic acid (16 mg/L). All cultures were incubated in ambient air (36°C), and growth was detected. Species identification was performed using MALDI-ToF (Bruker Daltonics).
Power calculation
Power was calculated for a non-inferiority trial with a binary outcome. The alpha value was set at 0.025 for a one-sided analysis. The beta value of 0.2 corresponded to a power of 80%. The non-inferiority limit of 0.5% was based on consensus among the authors on the highest acceptable difference in hospitalisation for febrile UTI between the study arms. The expected rate of hospitalisation in the combined low- and high-risk groups was 1.5% in the fosfomycin group and 3% in the ciprofloxacin group based on available studies at the time of the calculation3,15,21–23 and data from the National Prostate Cancer Register. There was a need for 862 patients in each study arm, 1724 patients in the low- and high-risk groups, respectively, totalling 3448 men.
Statistics
The primary endpoint was planned to be calculated with an intention-to-treat analysis using logistic regression providing a point estimate odds ratio (OR) with a 97.5% confidence interval (CI). However, because the trial was terminated with a very small population not including any cases of infections in one of the arms, the statistical plan was changed. The expected proportion of infections in the fosfomycin group, 1.5% according to the power calculation, was compared to the actual result using a binomial test. Secondary endpoints were presented using descriptive statistics.
Ethics
Ethical approval was obtained from the ethics committee in Umeå, no. 2017/144-31 and 2019-02239. The Swedish Medical Products Agency approved the study. The trial protocol was registered at the European Medicines Agency before study started: Eudra-CT nr 2017-000772-28. The principles of the Helsinki Declaration were adhered to.
Results
Details of the randomisation and follow-up of the 42 included patients are shown in the CONSORT diagram (see Figure 1). The median age of participants in the fosfomycin group was 66 years (interquartile range = 63–72) and 69 years (interquartile range = 63–74) in the ciprofloxacin group. The median prostate volume was 44.0 mL (interquartile range = 35.0–72.5 mL) in the fosfomycin group and 55.5 mL (interquartile range = 38.8–72.8 mL) in the ciprofloxacin group. The median number of biopsies performed was 8 (interquartile range = 4–10) in the fosfomycin group and 7 (interquartile range = 4–10) in the ciprofloxacin group. A minority of patients (9 out of 42) were classified as high risk for infection. Baseline data for the treatment arms are detailed in Table 3.
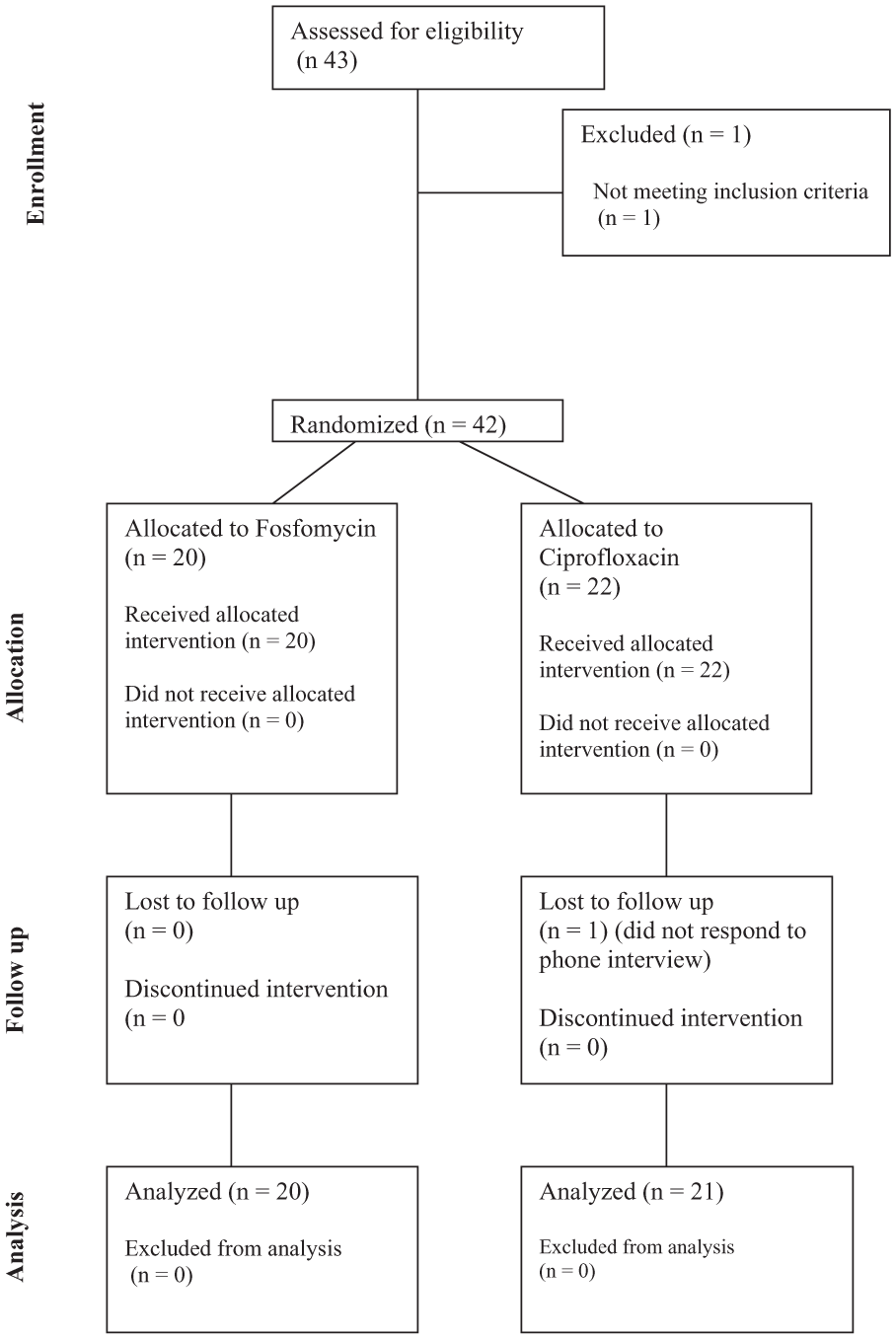
CONSORT diagram of the flow of the randomised trial.
Baseline characteristics of the study men before transrectal prostate biopsy procedure stratified by prophylactic agent.
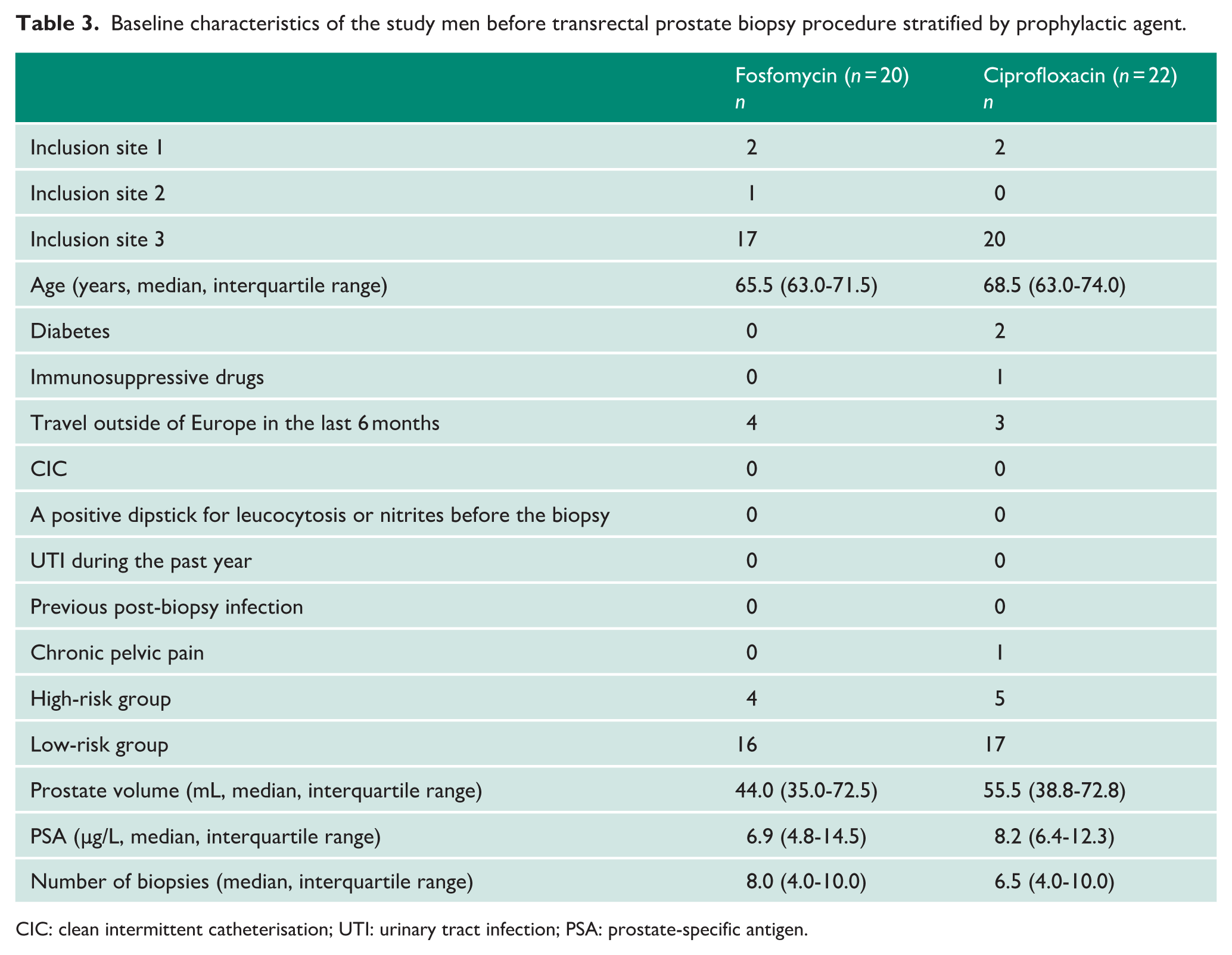
CIC: clean intermittent catheterisation; UTI: urinary tract infection; PSA: prostate-specific antigen.
The safety board prematurely halted the study after enrolment of 42 patients due to an unexpectedly high number of hospitalisations. In the fosfomycin group (n = 20), four patients (20%) were hospitalised due to infection – three from the low-risk group and one from the high-risk group. In contrast, none of the 21 patients completing the study in the ciprofloxacin group were hospitalised for infection. One patient in the ciprofloxacin group was lost to follow-up. Two patients, including one who received fosfomycin, reported fever but did not require hospitalisation. One of these patients received antibiotics but did not require hospitalisation. A one-sided binomial test indicated that the proportion of patients hospitalised with infection in the fosfomycin group was significantly higher than the predefined expected complication rate of 1.5% (p < 0.001).
Primary and secondary outcomes are detailed in Table 4. Blood in the semen was reported in a higher proportion of patients in the ciprofloxacin group (n = 15, 68%) compared to the fosfomycin group (n = 10, 50%). Conversely, blood in the urine was more common in the fosfomycin group (n = 3, 15%) compared to the ciprofloxacin group (n = 1, 5%). There were no major differences in screening culture results between the two groups (see Supplemental Table 1).
Adverse events reported by the patients during a telephone interview 2 weeks after prostate biopsy.
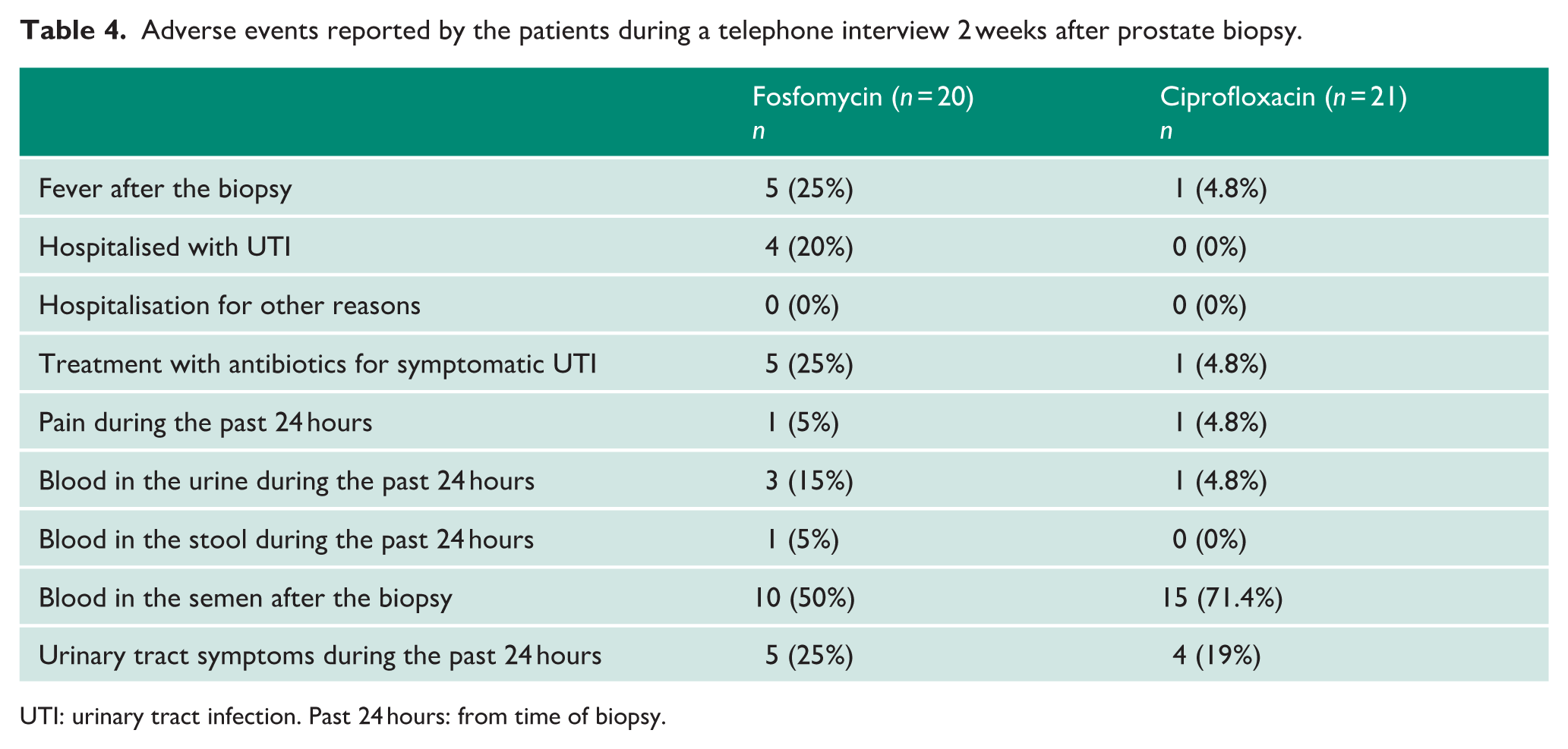
UTI: urinary tract infection. Past 24 hours: from time of biopsy.
Among the four hospitalised patients, two had positive blood and urine cultures for Pseudomonas aeruginosa, while the other two had negative clinical cultures. One of the former patients had positive rectal swab cultures for Pseudomonas species at both inclusion and follow-up. Another hospitalised patient, who received fosfomycin, had a positive rectal swab for quinolone-resistant E. coli. Of the two patients who were not hospitalised, one had E. coli growth in a blood culture. Additional data on the six patients who reported fever after biopsy are presented in Table 5.
Description of the outcome in terms of infections, hospitalisation and results from pre- and post-biopsy cultures for the six patients who reported fever within 2 weeks after prostate biopsy.
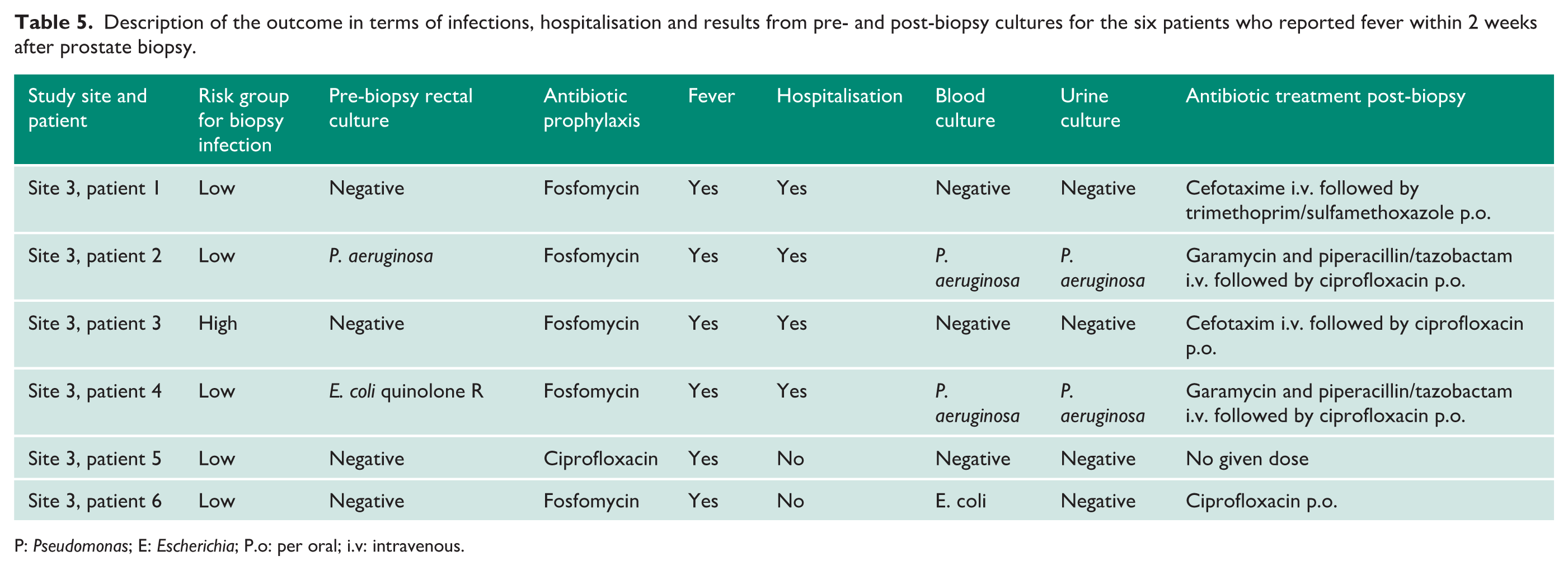
P: Pseudomonas; E: Escherichia; P.o: per oral; i.v: intravenous.
Discussion
This is the first randomised prospective study comparing ciprofloxacin and fosfomycin as antibiotic prophylaxis for transrectal prostate biopsies in a population with relatively low quinolone resistance. Antibiotic prophylaxis in both groups was administered immediately before the biopsy. The study was unfortunately prematurely halted after 20% of patients in the fosfomycin group were hospitalised with febrile UTI, compared to 0% in the ciprofloxacin group. The ratio of fever after biopsy was also 5:1 in favour of ciprofloxacin. Despite the early termination of the study, our results are statistically significant. These results contrast with previous studies, which have reported low infection rates with fosfomycin used as prophylaxis.
In a study by Sen, 300 men received fosfomycin or ciprofloxacin. The quinolone resistance in urinary cultures was 36%. Patients were administered fosfomycin the night before the biopsy or ciprofloxacin 60 minutes before the biopsy. The authors found no statistically difference between the groups. 21
Fahmy included 412 patients to receive either a single dose of fosfomycin 1–2 hours before biopsy or oral ciprofloxacin at least 1 hour before biopsy. Infectious complications were 4.5 times more frequent in the ciprofloxacin group. Urine cultures revealed that 73% of bacteria were quinolone-resistant. 22
Cimino showed that fosfomycin prophylaxis given in two doses was related to less UTI (34%) compared to beta-lactame or quinolones (43%). 24
A meta-analysis by Roberts showed that the relative odds of an infectious complication (OR = 0.22, 95% CI = 0.09–0.54) were significantly lower in patients receiving fosfomycin compared to those receiving fluoroquinolone. 16 All studies included were from countries with known high quinolone resistance. The studies were performed in Turkey, Egypt, Spain and Italy. 16
These studies generally support fosfomycin as an option for antibiotic prophylaxis in transrectal prostate biopsy when compared to ciprofloxacin, especially in settings with high resistance to ciprofloxacin. However, they all involved administering fosfomycin at least 1 hour before the biopsy.
Our study contributes data on the immediate administration of fosfomycin before the procedure. Although existing literature recommends the administration of fosfomycin 1–4 hours before biopsy, 18 it is important to emphasise that the timing of antibiotic prophylaxis for prostate biopsy remains unclear. The decision to administer fosfomycin immediately before the biopsy was based on current clinical practice in Sweden, where this approach is routinely employed due to logistical and procedural considerations. Immediate administration ensures timely prophylaxis, facilitating adherence to the recommended regimen clinically. Our results suggest that this practice may not provide the same level of efficacy as earlier administration of fosfomycin, highlighting the need for further research to clarify the optimal therapeutic window for fosfomycin in prostate biopsy prophylaxis.
In Sweden, it is common practice to administer antibiotic prophylaxis immediately before the prostate biopsy. 10 Our study suggests that fosfomycin should not be administered immediately before the biopsy. Fosfomycin has a slower pharmacokinetic distribution compared to ciprofloxacin, 18 indicating that it needs to be administered earlier.
Two of the four hospitalised patients in our study had positive cultures for Pseudomonas, a bacterium that is intrinsically resistant to fosfomycin 17 . Pseudomonas is an uncommon pathogen in non-hospitalised populations. 23 Nonetheless, if Pseudomonas is present, fosfomycin is likely to be a suboptimal prophylactic choice. Two of the four hospitalised patients in the fosfomycin group had Pseudomonas in cultures, while the other two had negative cultures.
While the use of ciprofloxacin for prophylaxis in prostate biopsy has been restricted by the EMA since 2018, our study reflects the ongoing clinical practice in Sweden and other Nordic countries where ciprofloxacin is still recommended in accordance with national guidelines. 12 These regional differences highlight the need for ongoing research to evaluate alternative prophylactic strategies in the context of local antibiotic resistance patterns.
It is possible that fosfomycin’s benefits against E. coli in countries with high levels of quinolone resistance might outweigh the drawbacks of requiring earlier administration. In a study from Spain by Lista et al, 25 quinolone resistance was 69%, whereas in Sweden, it is around 11%. 26 In the Swedish context, ciprofloxacin is still a valid option.
Due to the high infection rate in the experimental group, ethical considerations required the early termination of the study. While the small sample size is a limitation, we believe that publishing negative findings is crucial to contributing to the evidence base and informing future research directions.
The early termination of our study limits the ability to draw definitive conclusions. The primary limitation of the study was its design with the late administration of fosfomycin, which likely resulted in inadequate drug concentrations. The study’s strengths include its randomised prospective design, high internal validity and use of hospitalisation as the primary outcome. The safety committee’s prompt detection of the high risk associated with late fosfomycin administration also adds to the study’s strengths.
Future studies should explore the efficacy of fosfomycin with earlier administration in settings with low quinolone resistance.
Conclusion
Fosfomycin administered immediately prior to a prostate biopsy is inadequate. The high incidence of Pseudomonas infections, a bacteria against which fosfomycin is often ineffective, may have influenced the results. If fosfomycin is to be used, it should be administered with caution, particularly if Pseudomonas has been detected in prior cultures.
Supplemental Material
sj-docx-1-uro-10.1177_20514158251376398 – Supplemental material for Fosfomycin versus ciprofloxacin for transrectal prostate biopsy: An open randomised controlled multicentre drug trial
Supplemental material, sj-docx-1-uro-10.1177_20514158251376398 for Fosfomycin versus ciprofloxacin for transrectal prostate biopsy: An open randomised controlled multicentre drug trial by Andreasson Anders, Hällgren Anita, Georgeoulas Petros, Forsberg Jon, Fridriksson Jon, Granåsen Gabriel Sverige, Lundström Karl-johan Sverige, Resare Sven Sverige, Backman Lönn Beatrice, Grabe Magnus Sverige, Stattin Pär Sverige, Stranne Johan Sverige, Holmbom Martin Sverige, Sundqvist Martin Sverige and Styrke Johan Sverige in Journal of Clinical Urology
Supplemental Material
sj-docx-2-uro-10.1177_20514158251376398 – Supplemental material for Fosfomycin versus ciprofloxacin for transrectal prostate biopsy: An open randomised controlled multicentre drug trial
Supplemental material, sj-docx-2-uro-10.1177_20514158251376398 for Fosfomycin versus ciprofloxacin for transrectal prostate biopsy: An open randomised controlled multicentre drug trial by Andreasson Anders, Hällgren Anita, Georgeoulas Petros, Forsberg Jon, Fridriksson Jon, Granåsen Gabriel Sverige, Lundström Karl-johan Sverige, Resare Sven Sverige, Backman Lönn Beatrice, Grabe Magnus Sverige, Stattin Pär Sverige, Stranne Johan Sverige, Holmbom Martin Sverige, Sundqvist Martin Sverige and Styrke Johan Sverige in Journal of Clinical Urology
Footnotes
Acknowledgements
The authors would like to thank Maria Rolf for technical assistance with screening cultures.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded by the Swedish Research Council (2019-05913_VR), the Public Health Agency of Sweden, funds from the Swedish Nordic health care regions, Västernorrland County Council and grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (RV 939417).
Ethical approval
Ethical approval was obtained from the ethics committee in Umeå, no. 2017/144-31 and 2019-02239. The Swedish Medical Products Agency approved the study. The trial protocol was registered at the European Medicines Agency before study started: Eudra-CT nr 2017-000772-28. The principles of the Helsinki Declaration were adhered to.
Informed consent
Written informed consent was obtained from the patients to participate in the trial.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data availability statement
The data are available and sharable.
Guarantor
JS.
Contributorship
JS, GGS, KL, MG, PS and MS contributed to conception and design. PG, JF, JF, BBL and MH contributed to acquisition of data. AA, AH, GGS, JS, MS and JS contributed to analysis and interpretation of data. AA, AH, PG, JF, JF, GGS, KL, SR, BBL, MG, PS, JS, MH, MS and JS contributed to critical revision of the manuscript for important intellectual content. GGS and JS contributed to statistical analysis. JS contributed to obtaining funding. AA, BBL, MS and JS contributed to administrative, technical or material support. JS and JS contributed to supervision.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
