Abstract
Objective:
Stress urinary incontinence (SUI) is a common problem affecting up to 46% of women. Urethral bulking agents (UBAs) are a popular minimally invasive procedure, being used increasingly commonly to avoid complications of more invasive procedures. The purpose of this scoping review is to evaluate systematic reviews on UBAs for adult women.
Methods:
MEDLINE, Embase and CINAHL databases were electronically searched for relevant studies. We included systematic reviews reporting outcomes of currently available UBA for adult women with SUI published from 2017 to October 2022.
Results:
Overall, 243 studies were identified, and eight studies were eligible for inclusion. Objective cure rate for UBAs ranged from 25.4% to 93.3%. Subjective cure rates of UBAs were significantly lower compared to mid-urethral slings (MUSs) (28% vs. 77.6%, p < 0.01). UBAs showed quality of life benefit compared to pelvic physiotherapy at 3 months (mean difference (MD) = 0.54, 95% confidence interval (CI) = 0.16–0.92), but were inferior to MUS (59.2% vs. 91.0%, p = 0.01). Post-intervention complications were similar between UBAs and other surgical procedures (Relative Risk/RR = 1.30, 95% CI = 0.30–5.66). Urinary tract infection rate was similar among the different types of UBAs, with a pooled rate of 13.0%. Pooled rates of urinary retention for all bulking agents were 10.0% (95% CI = 6.0–14.0%). Outcome reporting was heterogeneous between studies, and some outcomes were quite poorly reported.
Conclusion:
Data from systematic reviews support the use of UBAs. However, success rate ranges were wide. MUS have higher success rates than UBAs. Long-term data using a standardized set of outcomes with direct comparisons between currently available bulking agents are needed.
Level of evidence:
Not applicable
Keywords
Introduction
Stress urinary incontinence (SUI) affects 46% of women. 1 The gold-standard surgical treatment for SUI is the mid-urethral sling (MUS); however, MUS have been under media scrutiny due to a 3–5% risk of potentially debilitating mesh complications.2,3 As a result, many countries have banned or suspended use of MUS. 2 Urethral bulking agents (UBAs) have gained recent popularity as an alternative, minimally invasive surgical procedure for SUI. UBAs provide a cushion in the urethral submucosa to improve urinary continence.4,5 Injection of UBAs is a relatively simple procedure and can be done under local anaesthesia. The ideal UBA would be biocompatible, durable, non-absorbable and lead to minimal inflammatory response.5,6 Currently available bulking agents include polyacrylamide hydrogel (PAHG–Bulkamid™), calcium hydroxylapatite (CaHA–Coaptite™), carbon beads (Durasphere™), silicon particles (Macroplastique™), and dextranomer hydrospheres (Deflux™).
We aimed to summarize evidence from systematic reviews regarding success rates, adverse effects, and impact on quality of life (QOL) for UBAs compared to other interventions for SUI.
Methods
Eligibility criteria
Search and selection of eligible studies for this scoping review was based on predetermined inclusion and exclusion criteria, based on the PICO (patient, intervention, comparisons, outcomes) format (Appendix 1). We included adult women with primary or recurrent SUI or stress-predominant mixed urinary incontinence (MUI). Studies in males, pregnant and postpartum women, women with overactive bladder (OAB), urgency urinary incontinence (UUI), unspecified urinary incontinence or neurogenic SUI or women with SUI/MUI belonging to a subgroup with a specific comorbidity were excluded. Interventions included all currently available bulking agents (excluding no longer recommended options such as glutaraldehyde cross-linked bovine collagen). Comparisons were made against any currently recommended interventions for SUI, placebo, sham or no treatment. Search was limited to systematic reviews, and English or French language. We included reviews published after 1 January 2017, as older reviews would likely duplicate studies already included in more recent reviews. In addition, recent studies/reviews would include UBAs applicable to current practice. Only studies with accessible full-text were included. Outcomes of interest are listed below.
Information sources
With the assistance of a medical librarian, MEDLINE, Embase, CINAHL and Cochrane Database of Systematic Reviews were searched on 23 October 2022. Grey literature was not searched.
Selection of sources of evidence
After excluding duplicates, title and abstract screening was performed (CW), followed by full-text screening (CW). A study log including main interventions in each study, and reasons for inclusion or exclusion was kept in Microsoft Excel.
Data items
Data extraction was performed on Microsoft Excel using a template document designed specifically for this study. Data extraction included study reference, study design, population age, intervention group, comparison groups, specific outcomes, study number per outcome and number of participants per outcome. Outcomes were grouped into the following categories: cure and improvement rates (objective and subjective), adverse effects and complications, de novo urgency, infection, pain, QOL, reoperation/reinjection rates, sexual dysfunction, urinary retention/voiding dysfunction and urodynamic parameters. Data were extracted from a review when an attempt was made to synthesize, pool or meta-analyse data when two or more studies reported on the same outcome. We did not extract data from reviews that only reported outcomes of several studies separately without any attempt to pool results.
Results are presented as means, percentages, pooled rates or ranges, as reported in included studies. Follow-up time was extracted when reported. However, this information was missing in many cases. When possible, 95% confidence intervals (95% CI) and heterogeneity (I2) were presented.
Review quality appraisal
Two reviewers (CW, ML) assessed methodological quality of included systematic reviews using A MeaSurement Tool to Assess Systematic Reviews 2 (AMSTAR-2) checklist. The AMSTAR-2 gives users a rating of the confidence in results of systematic reviews that include randomized, non-randomized or both study types. Overall scores are classified as high, moderate, low or critically low – highlighting potential impacts of inadequate rating within each category. 7 A ‘high’ rating indicates zero or one non-critical weakness, whereas ‘critically low’ indicates there is more than one critical flaw, with or without non-critical weaknesses. 7
Results
Selection of sources of evidence
A total of 245 articles were found, and 187 were screened after removing duplicates. Eighteen studies were assessed for eligibility through full-text review, and eight were included in the review (Figure 1).
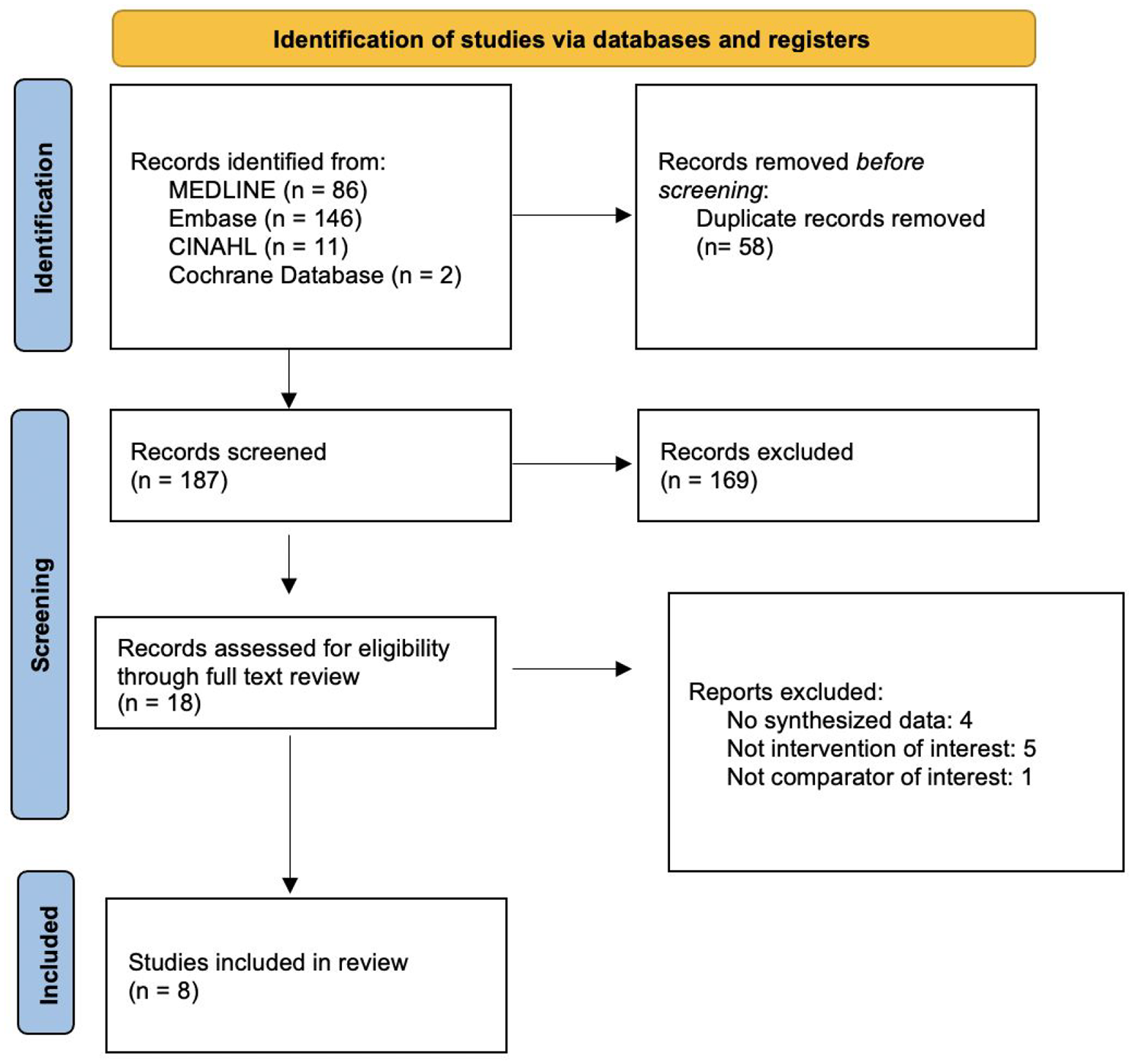
PRISMA flow diagram.
Characteristics of sources of evidence
Included studies’ characteristics are summarized in Table 1.
Characteristics of systematic reviews included.
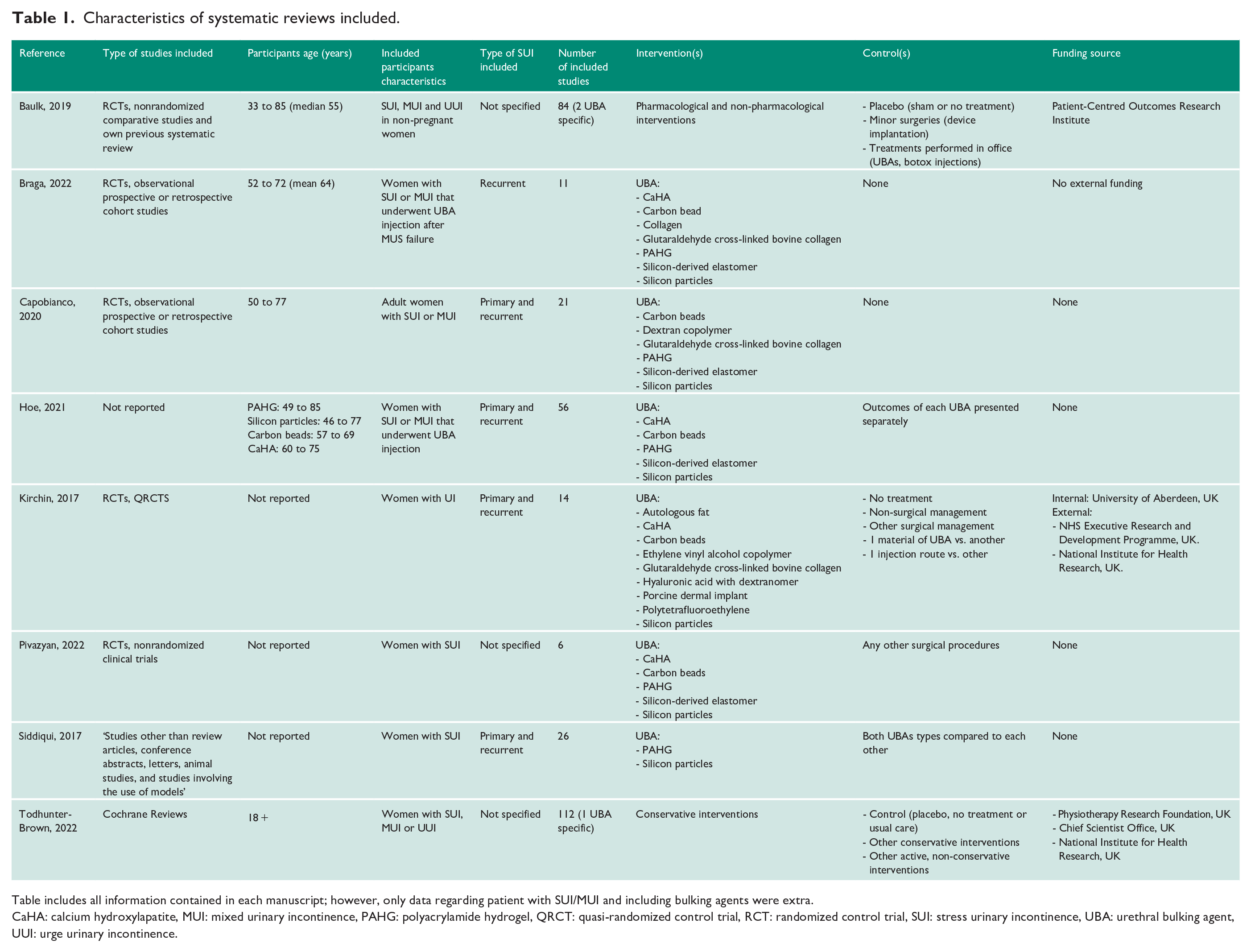
Table includes all information contained in each manuscript; however, only data regarding patient with SUI/MUI and including bulking agents were extra.
CaHA: calcium hydroxylapatite, MUI: mixed urinary incontinence, PAHG: polyacrylamide hydrogel, QRCT: quasi-randomized control trial, RCT: randomized control trial, SUI: stress urinary incontinence, UBA: urethral bulking agent, UUI: urge urinary incontinence.
Confidence assessments
Of the eight systematic reviews included, one was rated ‘high’ by the AMSTAR-2 guidelines, 5 one was rated ‘moderate’ 8 , one was rated ‘low’ 9 , and five were rated ‘critically low’4,6,10 –12 (Appendix 2). The most common issues were a lack of explanation on selection of study designs included in the review (Item 3), not including details on excluded studies (Item 7) and not reporting sources of funding for studies included in the review (Item 10).
Synthesis of results
Overall, commonly reported outcomes and reported rates of these outcomes are presented in Table 2.
Summary of outcomes of all included urethral bulking agents.
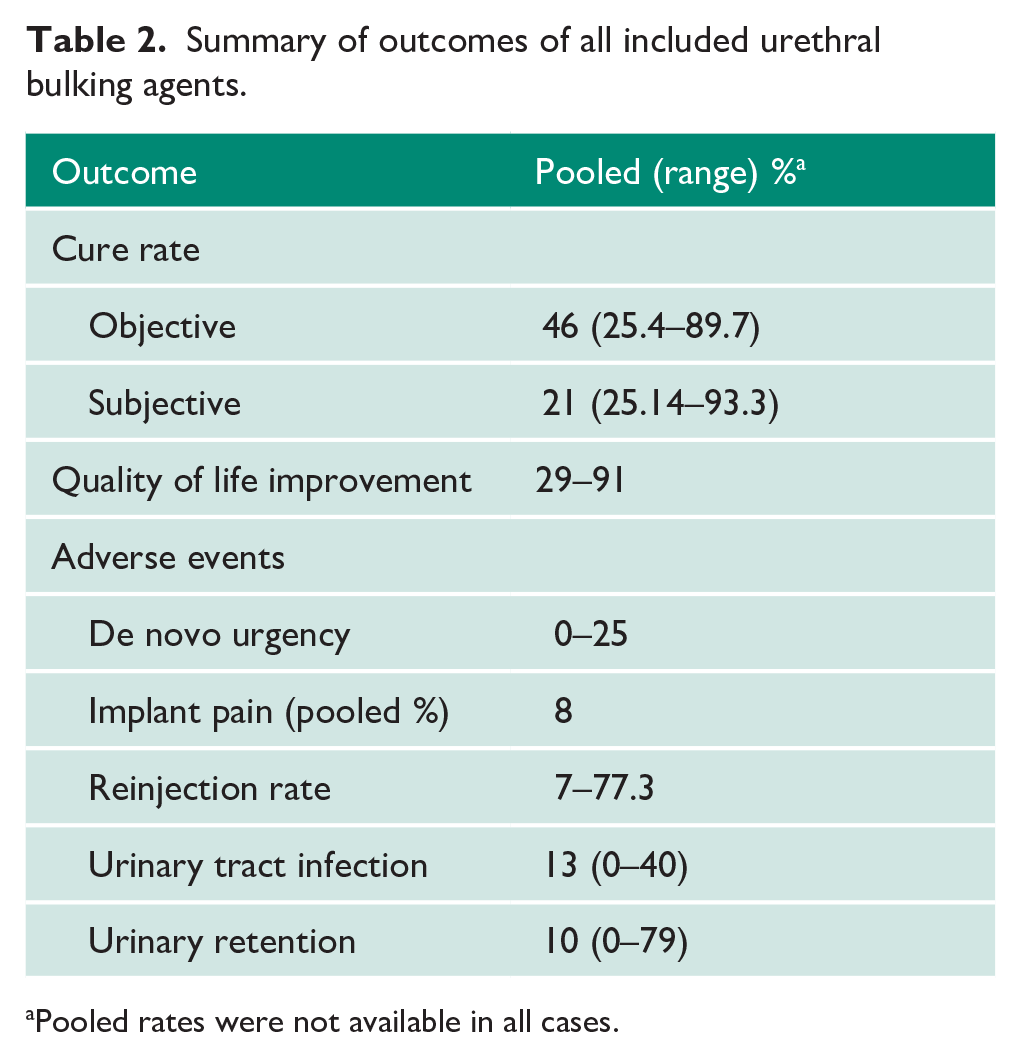
Pooled rates were not available in all cases.
Cure and improvement rate
Objective
Five reviews 4 –6,11,12 reported on objective cure rates of UBAs with various definitions. Overall, objective treatment success ranged from 32.7% to 93.3%, with a pooled rate of 67% (95% CI = 59–75%) at ⩽ 12 months and 46% (95% CI = 37–55%, I2 = 55.3%) over 12 months. 11 PAHG objective cure rates ranged from 19.6% to 79.3% (five studies, 12 months).4,6 Silicon particles showed an objective cure rate of 44% to 73.3%. 4 Pad weight was significantly reduced by all UBAs,4 –6 daily pad count decreased with PAHG,6,13 CaHA, 6 and silicon particles,4,14 and incontinence episodes were significantly less after PAHG4,6 (see Appendix 3).
PAHG was inferior to retropubic MUS at 12 months for objective cure (66.4% vs. 95%, one trial, p < 0.001).6,15 Similarly, silicon particles were inferior to traditional slings (6-month objective improvement 9% vs. 81%, one trial, n = 45, RR = 4.77, 95% CI = 1.96–11.64, p < 0.001).5,12,16 Daily number of pads used decreased significantly with silicon particles (from 3.4 to 1.9), compared to pelvic floor muscle training (PFMT) (from 2.7 to 2.5) (one trial, n = 45, p = 0.015, 3 months).5,17
Subjective
PAHG subjective cure rates at 12 months ranged from 66% to 89.7%. 4 Reported cure or improvement for carbon beads at 1 year was 66% (one trial).5,18 In one trial, 12-month subjective cure of silicon particles was 37%.5,19 In total, 32% of patients reported slight or no improvements in their symptoms after silicon particle bulking agents (one trial).5,19 Using CaHA, 37% of patients had a clinically significant improvement (one trial).5,20
Subjective improvement after bulking agents was consistently lower than after other surgical procedures (RR = 0.70, 95% CI = 0.53–0.92, p = 0.01). 12 PAHG and silicon particles had significantly lower subjective cure rates than retropubic MUS (28% and 77.6% respectively; one study, n = 1386 bulking agent, n = 18,763 retropubic MUS, p < 0.01).12,21 However, subjective success rates of silicon particles were similar to that of traditional slings (77% vs. 90%, one trial, n = 45, p = 0.41, 6 months).6,16 Compared to a home PFMT programme, silicon particles showed a significant difference in subjective improvement defined as ‘cured’ or ‘markedly improved’ (19.0% vs. 62.5%, one study, n = 45, p = 0.002, 3 months).6,17
Success
When treatment success was not specifically defined as objective or subjective, 15 studies showed a pooled cure rate of 26% at ⩽ 12 months with UBAs (n = 1131, 95% CI = 21.0–32.0%, I2 = 89.9%). 11 Meanwhile, five studies found a 21% pooled cure rate at > 12 months (n = 217, 95% CI = 16.0–27.0%, I2 = 34.2%). 11 Another review found a success rate range of 30–90% at ⩽ 24 months, and 21–80% at > 24 months. 6 Baulk et al. 10 found no significant difference in achieving cure or improvement of SUI between bulking agents and no treatment (odds ratio (OR) = 1.32 (95% CI = 0.42–4.16) and 1.97 (95% CI = 0.74–5.20), respectively; low quality, indirect evidence only).
Individual UBAs’ success rates varied: PAHG’s was 29.8–89.7% at ⩽ 24 months and 42–70% after > 24 months. 6 CaHA success rate was 39% at 12 months 5 , 54.8–90% at ⩽ 24 months, and 60–74.7% at > 24 months. 6 Carbon beads had a cure and improvement rate of 40% and 80%, respectively, at 1-year follow-up (one trial)5,22; cure or improvement of 33.3–65.5% at ⩽ 24 months, and 21–80% at > 24 months (two studies). 6 Silicon particles’ success rate ranged from 48% to 84% at < 24 months and 21% to 80% at > 24 months (nine studies). 6 Compared to 69% for traditional slings, success rate for silicon particles was 21% at > 62 months (one trial, n = 45).12,16
A review 9 looking specifically at use of UBAs for recurrent SUI after MUS found an overall cure or improvement rate between 64% and 85% (pooled rate = 75%, I2 = 88.9%, 11 studies). Specific agents’ rates of cure or improvement for recurrent SUI were: PAHG 80% (95% CI = 74.0–85.0%, I2 = 5%) and silicon particles 84% (95% CI = 77.0–90.0%, I2 = 0%), respectively. 9 When comparing UBAs to repeat MUS surgery following MUS failure in a managed care population, UBAs were associated with a higher risk of subjective or objective failure (38.8% vs. 11.2%, one study, n = 165, OR = 3.49, 95% CI = 1.34–9.09, p = 0.01).12,23
QOL
Five reviews 4 –6,8,12 addressed patient QOL after UBAs. Using various validated questionnaires, QOL was improved after PAHG,6,12,24 CaHA,6,25 and silicon particles.4,5,26 Silicon particles showed improved QOL compared to conservative treatments such as PFMT at 3 months (n = 45, I-QOL questionnaire score mean difference (MD) = 0.54, 95% CI = 0.16–0.92, moderate certainty, one study).5,8,17 PAHG and silicon particles were inferior to retropubic MUS for QOL outcomes, although improvements were noted in both groups12,15,21,24 (see Appendix 3).
De novo urgency
De novo urgency after UBAs was reported in 0–25% of patients.5,6 Specifically, different rates were found for each UBA: 0–10% after PAHG (nine studies), 6 0–5.7% after CaHA (two studies), 6 21% after silicon particles, and 25% after carbon beads. 5
Capobianco et al. 11 investigated urgency and UUI among all UBAs, finding a pooled urgency percentage of 7.0% (95% CI = 3.0–13.0%, n = 952, I2 = 89.9%, eight studies) and pooled UUI percentage of 5.0% (95% CI = 2.0–9.0%, n = 897, I2 = 77.5%, eight studies).
Urodynamic parameters
Maximal urethral closing pressure (MUCP) increased following both UBAs and MUS; however, the increase was greater following MUS (one trial, n = 50, p < 0.001, 6 weeks).12,27 Silicon particle UBA did not significantly change urodynamic parameters including peak flow, flow time and post void residual volume (one trial, n = 27, 12 months).4,26 One trial found a 73.3% improvement in the valsalva leak point pressure with UBAs.4,14
Adverse effects and complications
Siddiqui et al. 4 found that PAHG caused more adverse effects (urinary tract infection (UTI), implantation site pain, de novo urgency, persistent UUI and haematuria) than silicon particles, except for dysuria and acute urinary retention. However, Hoe et al. 6 concluded that PAHG had the lowest number of complications compared to other agents. Silicon adverse effects included haematuria (7%) and transient dysuria (47%). 5
A review 12 found no significant difference in post-intervention complications when comparing UBAs with surgical procedures (RR = 1.30, 95% CI = 0.30–5.66, three studies), although one trial reported a higher proportion of complications with retropubic MUS compared to PAHG (44.6% vs. 19.6%, difference = 24.9%, 95% CI = 12.3–36.6%, n = 108).6,15
Urinary retention/voiding dysfunction rates
Postoperative transient urinary retention ranged from 0 to 79% (pooled rate = 10.0%, 95% CI = 0.06–0.14; 10 studies, n = 1092, I2 = 73.0%) after UBAs. 11 Again, reported rates varied between different agents: 0–20% after PAHG (15 studies), 6 11.7–50% after CaHA (three studies),5,6 and 95.8% after silicon particles (one study, n = 24).5,17 Urinary retention was higher with silicon particles, compared to PFMT (one trial, n = 45.8% vs. 0%, RR = 34.32, 95% CI = 2.2–535.8).5,17 Compared to traditional slings, silicon particles had a significantly shorter catheterization time (one trial, n = 23, 1 day vs. 5 days, p < 0.0001).6,16 Carbon beads had a urinary retention rate of 17%, which resolved in all cases within 7 days.5,18
Reoperation/reinjection rates
There is a wide-reported range of reinjection rates associated with UBAs (7–70%).5,6,28 While the total number of reinjection per patient was rarely reported, a maximum of two to three injection sessions per patient appeared to be the norm.29,30 Reinjection rate was reported as 15.6–70% after CaHA (four studies), 7.4–52.5% after silicon particles (18 studies), 15.4% after carbon beads (one study), and 7–70% after PAHG (12 studies).5,6,28 Compared to traditional slings, silicon particles required more repeat incontinence procedures for failure (one trial, n = 45, 5% vs. 32%).5,16 PAHG had a lower rate of reoperation due to complications such as haematomas or retention compared to retropubic MUS (0% vs. 6.0%, respectively). 6
When using UBAs for recurrent SUI after failed MUS, pooled reoperation rate was 25% (95% CI = 17–34%, I2 = 76.7%, nine studies). 9 Pooled reoperation rates of specific UBA agents used for recurrent SUI were 24% (95% CI = 13.0–37.0%, I2 = 46%) after PAHG, and 31% (95% CI = 20.0–43.0%, I2 = 33%) after silicon particles, respectively. 9
Pain
Type and characteristics of pain after UBA, including its timeline, were poorly described. Pooled implantation site pain after UBA averaged 8.0% (95% CI = 3.0–13.0%, six studies, n = 812, I2 = 83.5%). 11 One study specified that implantation site pain was transient. 29 Similarly, pain rate after silicon particles was 8%. 5 Tension Free Vaginal Tape (TVT) and PAHG appeared to similarly increase pain from baseline (one trial, p = 0.78).12,24
Infection
Pooled rate of UTI following any UBA was estimated at 13.0% (10 studies, 95% CI = 6.0–21.0%, n = 1051, I2 = 91.3%, range = 0–40% between studies).6,11 UTI rates appeared similar across bulking agent types. 6 PAHG UTI rates ranged from 1.6% to 40% (13 studies). 6 Silicon particles had similar incidence of UTI as traditional slings (one trial, n = 45, 9% vs. 13%).5,16 No studies reported on UTI rates after carbon bead UBAs. 6
Sexual dysfunction
There was limited information regarding sexual dysfunction and UBAs. Sexual function was improved with both PAHG (p = 0.01) and retropubic MUS (p < 0.001), with significantly higher scores for MUS (one trial, n = 224).12,24 Another study reported that 23 of 29 women (79.3%) continued to be sexually active following treatment with PAHG.4,31
Discussion
This paper aimed to summarize evidence from recent systematic reviews regarding UBAs. Cure rate for UBAs ranged from 25.5% to 93.3%, showing potential for high efficacy although less efficacious than MUS. QOL improvements were shown; however, PFMT showed lower risks for urinary retention than UBAs. Repeat injections of UBAs can be required and UBAs after failed MUS appear effective; however, more literature is needed into factors that could affect success rates in those cases. We found a disproportionate representation of outcomes, with most papers and reviews discussing cure rate (subjective and objective), and much fewer papers discussing other outcomes. There was a lack of information surrounding complications and sexual function despite their importance to patients’ QOL (two reviews only). Current literature lacks systematic definitions for each outcome, and more importantly a core outcome set for SUI management research. Encouragingly, some research groups have begun investigating patient-preferred outcomes, 32 while others are creating a core outcome set 33 and a minimum standard data set. 34 Outcomes such as complications and pain should be better defined and include duration, location, management and impact. Long-term outcomes are also urgently needed to evaluate effectiveness of UBAs.
Very few trials and reviews compared efficacy of different UBAs to each other or to other interventions or a control (e.g. saline solution or sham). The most recent Cochrane Review conducted about UBAs only included 14 eligible trials, with a single trial comparing UBAs to PFMT, two trials comparing them to surgery, and eight comparing agents to each other, but including some agents that are no longer recommended or off the market. 5 The review concluded that current evidence on UBAs was insufficient to guide practice and that no clear-cut conclusions could be made from the trials that were comparing UBAs due to heterogeneity and wide CIs. 5 They also found that patients were as likely to report cure or improvement of SUI symptoms while using saline injections as they were when using autologous fat injection (an agent no longer recommended); the improvements were analysed through both subjective cure rate via patient symptom reporting and objective cure rate via one-hour pad tests. 5 This further supports the need for comparisons to controls and placebos in UBA research. High rates of heterogeneity and wide CIs have also led to further conflicting information; an example of which included reviews evaluating safety profiles, with Hoe et al. 6 finding that PAHG had the best safety profile while Siddiqui et al. 4 found that silicon particles were safer than PAHG.
Limitations
Limitations of this review include the use of systematic reviews to extract data. While this is an overview of the current systematic reviews, systematic reviews synthesize evidence from several studies, as such, extracting data from different reviews increases the chance that specific trials may be overrepresented within the data if they are present across several reviews. Some outcomes were poorly reported, and follow-up times for each outcome were frequently omitted in included reviews, affecting interpretation of findings. Although authors made an effort to review currently available UBAs, it is possible that some newer underreported agents or agents used in countries outside of North America may not have been represented, limiting generalizability.
Conclusion
Data from current systematic reviews support the use of UBAs, although cure and improvement rate ranges were wide. MUS had higher success rates than UBAs. Risks and adverse effects following UBAs appeared relatively low. Outcome reporting was heterogeneous between studies, and some outcomes, such as pain and sexual function, were poorly reported due to lack of standardized definitions and lack of core outcome set for SUI research.
There is a need for more long-term studies, with a standardized set of outcomes and direct comparisons between currently available UBAs. Further reviews with more conclusive data are needed to ensure both clinicians and patients can make better informed decisions about SUI management, specifically with the use of UBAs.
Footnotes
Appendix 1: Sample search strategy
Appendix 2
AMSTAR-2 methodological quality assessment of included reviews.
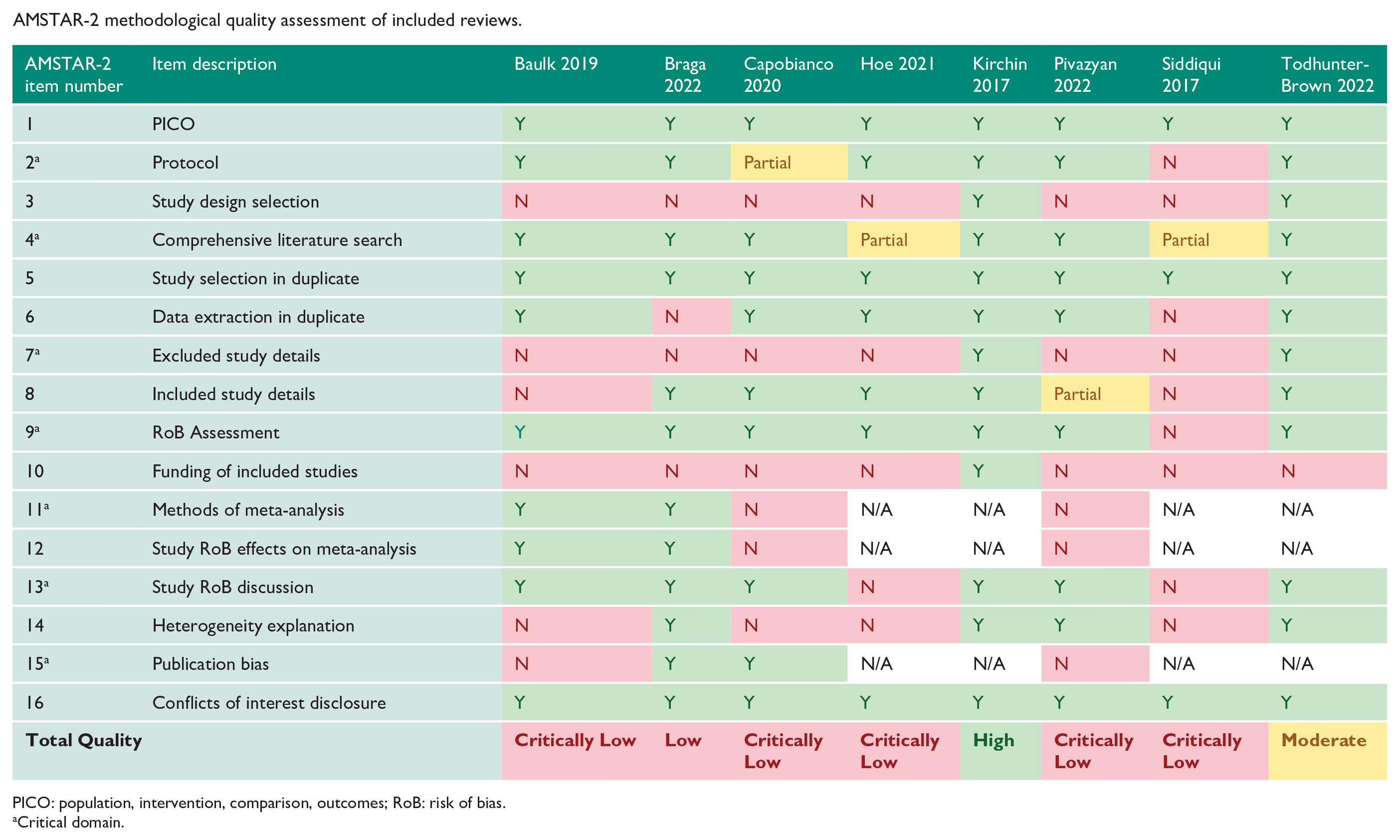
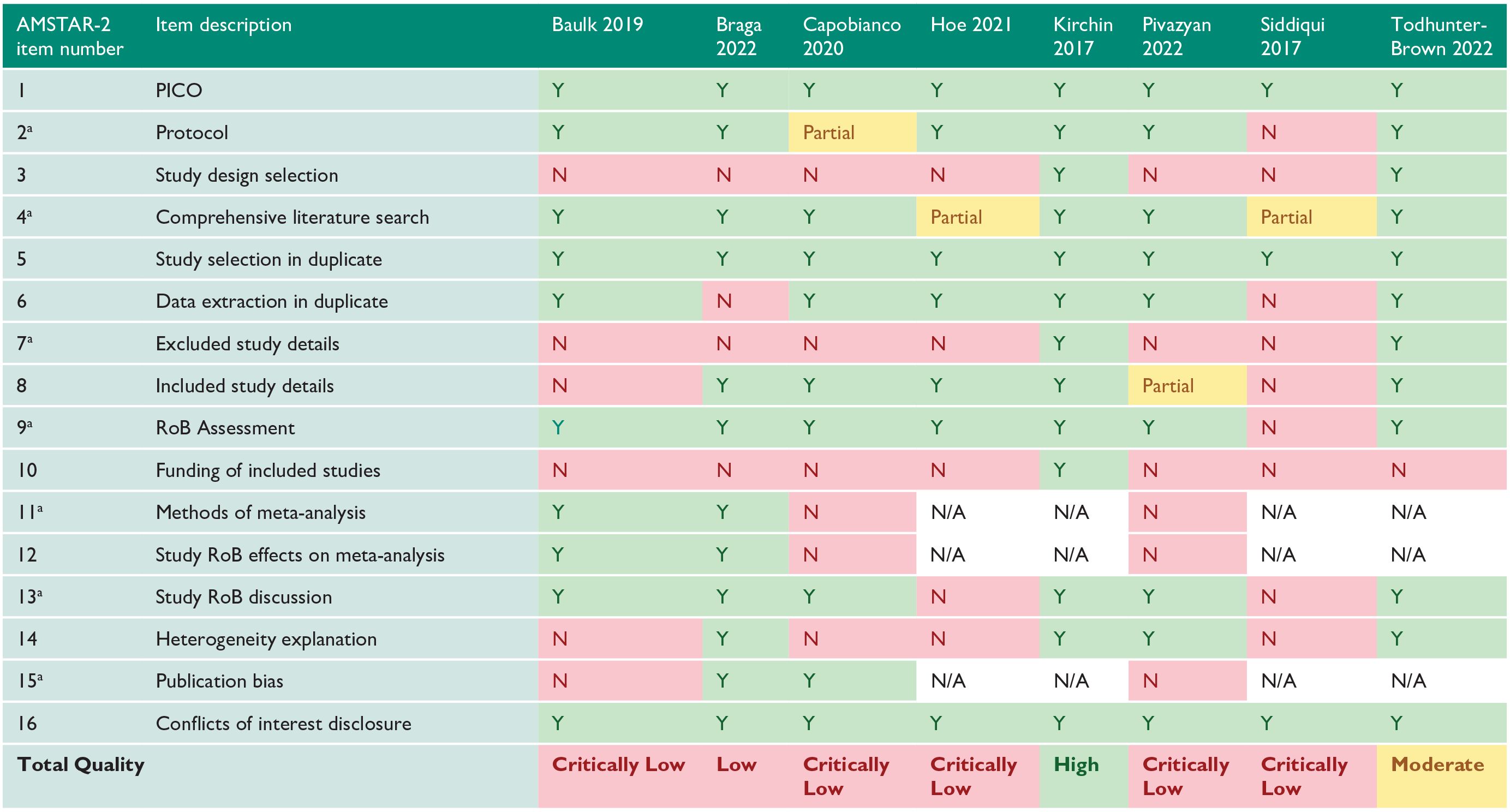
|
PICO: population, intervention, comparison, outcomes; RoB: risk of bias.
Critical domain.
Appendix 3: Detailed supplementary data
Author contributions
CW and ML conceived the study and wrote the first draft of this manuscript. Material preparation, data collection and data synthesis were conducted by CW. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Ethical approval
Ethics approval was not required, as only publicly available anonymized data from published systematic reviews was used.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declare the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: (details omitted for anonymized peer review). M.L. is a consultant for AbbVie, on the advisory board for FemTherapeutics, and received research funds from the St. Mary’s Research Centre. G.N. has been an advisory board member for AbbVie and Astellas; a consultant for AbbVie, Boston Scientific, and Searchlight Pharma; and a speaker for AbbVie and Laborie.
