Abstract
Ureteroscopy has undergone many advances in recent decades. As a result, it is able to treat an increasing range of patient groups including special populations such as pregnancy, anomalous kidneys and extremes of age. Such advances include Holmium laser, high-power systems and pulse modulation. Thulium fibre laser is a more recent introduction to clinical practice. Ureteroscopes have also been improved alongside vision and optics. This article provides an up-to-date guide to these topics as well as disposable scopes, pressure control and developments in operating planning and patient aftercare. These advances allow for a custom strategy to be applied to the individual patient in what we describe using a new term: Tailored endourological stone treatment (TEST).
Introduction
As the incidence of kidney stone disease (KSD) rises worldwide, this is mirrored by the volume of minimally invasive endourological procedures performed. 1 Based on findings shared from national databases of hospital records, this trajectory is most notable for ureteroscopy (URS). 2 For example, Perera et al. 3 reported that over the past 15 years in Australia, the volume of URS procedures has increased by 9.3% per year while shockwave lithotripsy (SWL) has decreased by 3.5% per year. Indeed, the technology now available is hugely different to the early descriptions recorded by Pérez-Castro Ellendt and Martinez-Piñero 4 in 1982 at a time when the authors stated that URS was considered ‘a forbidden field’. In the contemporary era, URS is routinely performed in the day case setting and is a treatment option for many non-indexed patients.5,6 This includes special populations such as pregnancy, the extremes of age and individuals with anomalous anatomy (e.g. transplant kidney).7–9 Advances that have fuelled this growth include development of next-generation ureteroscopes, the introduction of laser and a range of ancillary devices (Figure 1). 10
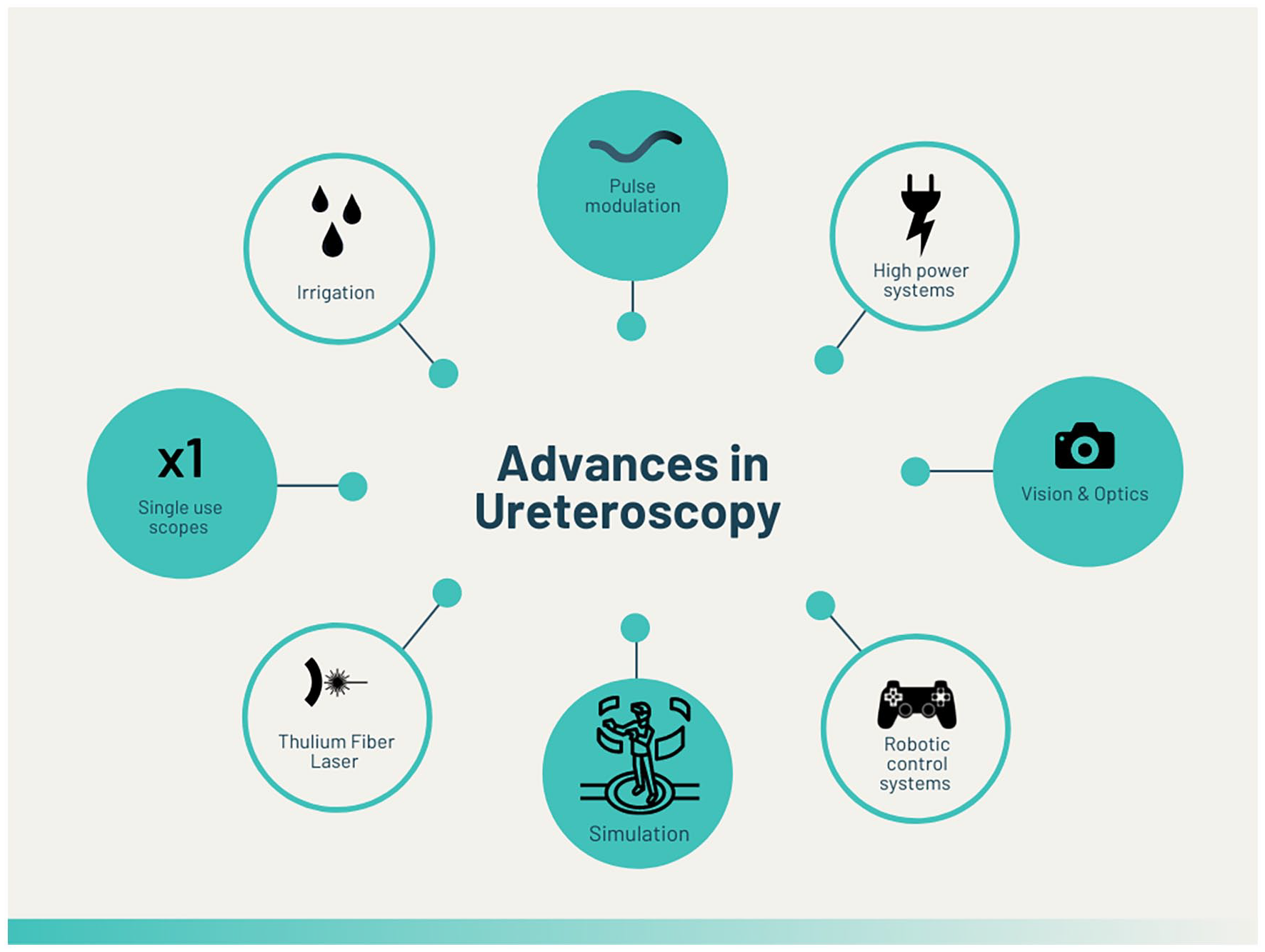
Summary of advances in ureteroscopy.
This review article serves to provide an overview of recent technological advances in URS for treatment of urolithiasis including ancillary devices and the principles they are founded upon.
Lasers
Holmium lasers
Holmium:yttrium–aluminium–garnet (Ho:YAG) also referred to as simply ‘Holmium’ laser is the current laser recommended by the European Association of Urology (EAU) guidelines for intracorporeal lithotripsy during URS and is able to ablate all stone types. 11 The most commonly accepted principle of function is that it relies principally on a photothermal effect with energy absorption mainly into water molecules within pores and cracks of stones, resulting in stone breakage and chemical decomposition. 12 A key contributing factor for its success in the therapeutic setting of stone lithotripsy is the high peak power (ca. 1000 to 2000 W) emerging from pulsed energy delivery to stones, rather than a continuous mode laser. The benefits of a pulsed laser can be compared to using a hammer drill versus rotary drill to bore a hole in a hard surface such as concrete. 13 The former is more efficient, faster and generates less heat. Two key parameters, which the surgeon can adjust are pulse energy (PE – total energy emitted in a single pulse) measured in Joules (J) and frequency (Fr – pulse repetition rate i.e. number of pulses emitted per second) measured in Hertz (Hz). Power (Watts (W)) is the product of PE multiplied by the Frequency. 14 Increasing PE is directly proportional to the stone ablation volume. 15 Increasing PE results in improved fragmentation but at the cost of increased retropulsion. 16 The latter is most probably a result of the vapour bubble collapsing, as well as stone particles being released from stone surface. Using high PE also results in increased degradation of fibre tip (so-called ‘burnback’ effect), which in turn may reduce laser ablation efficiency, but most importantly causes regular interruption of the operation cycle due to the need for laser fibre manipulation to compensate the length loss.15,17
Next-generation holmium systems
Earlier low-power Ho:YAG laser generators were only capable of a limited set of ‘traditional’ operating ranges (e.g. > 0.5 J, < 15–20 Hz and < 15–20W).5,18 For this reason, stone fragmentation and active stone retrieval (e.g. basketing) was the default technique until next-generation, high-power Ho:YAG systems (e.g. 0.2–6.0 J, 5–100 Hz and 2–120 W) became available. 18 Applying high-power settings to traditional systems was not possible as the flash lamp system would quickly overheat. 19 Collimation of multiple synchronised laser cavities enabled the construction of high-powered Ho:YAG systems. By maintaining low PE but increasing Fr, the stone can be ablated into fine particles, which can pass spontaneously and do not require active removal. 16 A recent study aimed to determine what size particles should be defined as dust and proposed an upper limit of 250 µm to be the cut-off for the so-called ‘stone dust’. 20 After performing this technique known as ‘dusting’, it is possible to aspirate the fluid and submit the dust for composition analysis – although some stone composition changes may have occurred from stone lithotripsy. 21 These techniques are referred to as contact laser lithotripsy, where the operator tries to maintain a minimal distance between the laser fibre tip and stone surface. An alternative strategy, which has gained popularity, is ‘popdusting’. 22 This form of non-contact lithotripsy is usually selected as a completion method when the stone fragment has been reduced in size. When the stone mass becomes less, fragments will bounce in front of the laser fibre when it is activated, which makes it difficult for the surgeon to ablate it efficiently. The surgeon can then change the settings (e.g. 0.3–0.6 J x 20–40J) and adopt this ‘end-game strategy’ (a termed coined by Aldoukhi et al. 16 ). The laser fibre is held in a fixed position close to the fragment collection. 23 The resultant interaction between laser fibre and stone fragment(s) delivers a ‘popcorn’ effect.24,25 This delivers greatest effect if positioned in a small calyx, based on in vitro studies. It is now possible to customise these strategies even further through adjustment of the pulse width (PW), also referred to as pulse duration (PD). This offers the ability to alter the emission period of the pulse over a microsecond (μs) although the energy output itself does not differ.12,26 In practice, PW settings are typically categorised as short pulse (SP, 180–330 μs) or long pulse (LP, 650–1215 μs). 15 Early lithotripter models were restricted to SP only. In the in vitro setting, LP mode offers advantages of reduced retropulsion, fibre burnback and lower risk of fibre fracture.17,27 Whether LP is superior to SP for pop-corning is under debate. Overall, it is PW combined with PE and Fr which determines the volume of fragments, which can be generated in a fixed time period (referred to as ablation efficacy). 19
Pulse shape modulation
A more recent innovation for Ho:YAG systems and pulse modulation is Moses™ technology (MT; Lumenis®), which takes advantage of the known ‘Moses effect’ and refers to how the laser creates a vapour channel, which separates the fluid. 28 Moses™ technology generates a ‘split-pulse’ by modulating the pulse shape to more efficiently deliver the energy transmitted to the stone in this channel created. If retropulsion can be minimised, this offers potential for improved operative efficiency as less time is lost in re-locating the displaced fragment(s). While dissemination of this novel technology is limited by higher costs, Elhilali et al. 29 demonstrated that its application reduced stone movement by 50 times (0.8 J x 10 Hz). In a double-blind randomised trial of 72 patients reported by Ibrahim et al., 30 this newer technology was associated with significantly reduced procedural time compared to regular mode (50.9 minutes versus 41.1 minutes, p = 0.03), but there was no significant difference in SFR at 3 months follow-up (83.3% versus 88.4%, p > 0.05). EAU guidelines do not currently make any recommendations regarding this technology yet and further studies are warranted to determine its true clinical benefits. 11 Nomenclature can be confusing, and it is, therefore, worth noting that several other systems are available that rely on similar pulse modulating principles, e.g. Virtual Basket® & Vapor Tunnel® technologies (Quanta™).
Thulium fibre laser
Thulium fibre laser (TFL) is a newer laser platform to the field of urology and has now been introduced to clinical practice although benchside studies were investigating its potential role more than 15 years ago.5,13,15,31 It has attracted considerable attention due to several favourable properties. 32 These offer the potential to overcome key limitations of Ho:YAG, namely energy transmission through water and retropulsion. 18 In vitro studies have demonstrated that its higher water absorption coefficient (WAC) results in a fourfold lower stone ablation threshold compared to Ho:YAG. 5 Similar to Ho:YAG, TFL can fragment all stone types. 33 Fr levels exceeding 2000 Hz are possible as well as lower PE settings than what can be achieved with Ho:YAG. These characteristics set it up to be efficient at dusting. Interestingly, while extremely high-frequency values are possible in theory, early clinical experiences reported with the technology reveal that even settings below 100 Hz generate a blizzard effect, which cause the surgeon to be forced to pause and wait for adequate vision is regained.32,34 Use of lower settings and patience can, therefore, pay dividends in such situations. Ho:YAG is restricted to a fibre size lower limit of 200 µm, whereas TFL theoretically can use sizes down to 50 µm. General advantages of smaller fibres are improved fibre and endoscope deflection, improved irrigation, lower risk of fibre fracture and lower risk of stone retropulsion.15,17,35 The latter can serve to not only improve vision but reduce intra-renal temperatures. 36 In a recent prospective randomised trial comparing the clinical outcomes of URS lithotripsy using TFL and Ho:YAG, superior SFR (92% versus 67%, p = 0.001), less intraoperative adverse events (8% versus 27%, p = 0.011) and shorter operative time (49 minutes versus 57 minutes, p = 0.008) were achieved using TFL. 37 TFL may, therefore, evolve to take over as the preferred and recommended laser of choice for URS lithotripsy.
Modern ureteroscopes
In addition to lasers, the anatomy of modern ureteroscopes has also undergone many changes. This includes the switch from fibre-optic to digital systems with improved vision accordingly, albeit this has taken time to achieve. 10 The spaces between the fibre-optic strands result in a honeycomb (also termed ‘moiré’) appearance. These quartz bundles are susceptible to damage particularly in circumstances where extreme deflection is applied. 38 Vision can steadily be reduced with repeat use of the scope until repair is mandated. Digital systems aim to overcome these limitations. Since the first ACMI (later purchased by Olympus) model was released in 2006, they have continued to improve and address issues such as image flickering caused by acoustic shockwaves of the laser interacting with the image sensor. 39 The image sensor is located at the tip (hence referred to as a ‘chip on tip’ set up). 38 Miniaturisation of endoscopes offers potential advantages of lower insertion-failure rates (and, therefore, lower pre-stenting rates), better irrigation outflow (and, therefore, irrigation turnover, hence visibility), better manipulation and possibly lower risk of ureteral damage. New models, such as the Uscope 3022 (Pusen™), are the slimmest (7.5Fr tip and shaft) single-use ureteroscopes available and early reports indicate they may serve to offer such advantages without compromising factors such as deflection, and vision quality.40,41 However, miniaturisation of digital ureteroscopes is still hindered by the current sizes possible for these chips. It is for this reason that the URS models with the slimmest tips are still fibre-optic systems such as the Olympus URF-P7 (4.9Fr tip and 7.95 proximal insertion shaft), which allows for easier access into the ureteral orifice and negotiating narrow segments of the ureter. The smaller fibre sizes possible with TFL could allow for the working channel to be made smaller from the conventional 3.6Fr. It is worth noting that improved image resolution has supported advances in kidney-sparing management of upper tract urothelial cancer. 42 Further studies are required to assess how smaller calibre ureteroscopes could affect rates of stenting post-URS.
Disposable technology
Ureteroscopes require careful handling and are at risk of damage particularly when associated with high-volume use and especially in cases of complex anatomy such as urinary diversion. Decontamination, sterilisation and the repair process are costly and time intensive. Careful handling is required during reprocessing as damage can occur during this period too. Single-use (‘disposable’) scopes such as LithoVue™ (Boston Scientific) are one solution to this problem, and there are now over 20 models available. 43 The performance outcomes (e.g. manoeuvrability, vision and deflection) associated with these single-use devices appear comparable to digital reusable ureteroscopes. 44 The critical question that remains unanswered is the true cost-benefit ratio. Consensus in the literature is lacking and this is largely because the rates of number of URS uses/repair vary. Talso et al. 45 found that the number of procedures varied from eight to 29 before repair was required. However, higher number of procedures before repair is probably possible. It is, therefore, difficult to accurately estimate cost burden for repairs, which has been reported between $120 and $957. 44 Contributing factors to these differences include hospital setting (e.g. academic, teaching and district), different handling protocols for URS repairs and the individual financial contract each centre has with suppliers. We recommend that clinicians perform a cost analysis at their own institutions to determine an accurate and individualised cost-benefit ratio. The key disadvantage of single-use scopes is the high cost (range $700–$1500), and so their implementation is unlikely to be feasible in many health systems. 44 Most studies reported have not been funded independently. Rather than using disposable ureteroscopes routinely for all cases, a hybrid model for use in select cases appears to be the current modus operandum in high-volume centres that have introduced disposable scope technology. This includes use for complex anatomy such as lower pole stones with a difficult infundibulopelvicureteric angle, e.g. < 50°, patients with a history of recurrent urosepsis post-intervention and/or multi-resistant urine strains. 46 Another important point discussing the role of disposable ureteroscopes, which is sometimes forgotten in this debate, is the impact on the environment and carbon footprint.
Ureteral access sheaths
Attention surrounding use of UAS has increased in recent years and has become more popular both in adult and paediatric populations. 47 Initial designs were introduced in the 1970s but really it was not until 30 years later when modern versions were introduced, which had hydrophilic coatings and a hub-locking mechanism that outcomes improved considerably. 48 In a global survey of endourologists, 76% responded that they routinely use UAS for renal stones. 49 UAS offers the benefits of improving visibility, reduced intra-renal pressure (IRP) and ease of access for multiple passages to the upper urinary tract during active stone retrieval. 50 It can also offer some protection against high intra-renal temperatures. It should be noted, however, that the protective features, such as reducing IRP and temperatures, using UAS apply for the larger size sheaths only. This should be considered carefully when recommending routine use of smaller UAS for URS. The positive characteristics using large-size UASs lend themselves to the treatment of complex and large renal stone burdens. However, long-term follow-up data are lacking, and there remain concerns regarding trauma incurred to the urothelium with subsequent stricture formation and risk of early postoperative complications such as infection and pain.51,52 In 2021, Meier et al. 53 shared findings from a state-wide study, which included data from over 5000 URS procedures and revealed that UAS use significantly increased risk of hospital visit and did not increase likelihood of becoming stone free. Recent meta-analysis by Huang et al., 54 which included data from eight trials and over 3000 patients, concluded that use of UAS does not lead to any significant difference in SFR but was associated with higher complication rates.
Pressure control
Physiological IRP is normally only a few cmH2O; however, this is increased when URS is performed owing to use of irrigation. 55 The latter offers benefits to the surgeon including improved vision. Intra-operative IRP is multifactorial and is influenced by scope size, UAS, UAS/scope calibre ratio and size of the occupied working channel. 56 Elevated IRP can lead to complications such as infection and forniceal rupture. Pyelovenous backflow can occur as low as 13.6–27.2 cmH2O. 57 It is generally recommended to aim to maintain pressures below 30 cmH2O. Multiple systems now exist to improve irrigation delivery such as continuous flow single action pumping system (SAPS™), foot-operated systems (Peditrol™) as well as using manual control such as Pathfinder Plus™ or a simple syringe. However, a recent in vitro study, which performed a comparison of six such models, concluded that all of them can result in excessively high pressures even if the working channel is occupied. 58 Despite the availability of many novel systems of this kind, routine use of passive gravitational pressure is the safest approach. To improve surgeon control of IRP, monitoring devices have been trialled. This includes continuous monitoring using a wire with an embedded sensor (PressureWire, St. Jude Medical, USA). 59 The next step is for an automated system, which can not only measure but also control pressure. One example is a patented intelligent system reported by Deng et al., 60 which maintains stable IRP using pressure feedback technology via a vacuuming platform and UAS with a pressure-sensitive tip. Early experiences in humans are promising but further research remains warranted.
Operative planning and aftercare
In addition to new technologies at the surgeon’s disposal, other innovations have been introduced to optimise patient care. One example is the use of nomograms, which can aid operative planning and patient counselling. 61 An example is the Resorlu-Unsal Stone Score (RUSS) which has a scoring system based on the following factors, which they found to predict SFR: stone size, stone number, stone composition, infundibulopelvicureteric angle and renal malformations. 62 Understanding has also improved regarding patients most at risk of infectious complications. Southern et al. 63 performed a study of 2746 patients undergoing URS and found female gender, longer surgical time, medical complexity and positive preoperative urine culture to be risk factors for postoperative fever and systemic inflammatory response syndrome. Pietropaolo et al. 64 recently devised a predictive model for predicting infectious complications post-URS. As well as objective outcome measures such as SFR, subjective parameters are being increasingly used both in the clinical and research setting. 65 This includes implementation of patient-reported outcome measures (PROMs), which have been specifically designed for KSD such as Wisconsin Stone Quality of Life Questionnaire (WISQOL). 66
Future directions for URS
Further advances are anticipated in all areas discussed in this review (Figure 2). Several surveys have been published, which highlight the diversity of practice patterns such as regarding use of UAS, high-power laser systems and post-endoscopic ureteral stenting.49,67 Further studies will help direct the most evidenced-based choices.
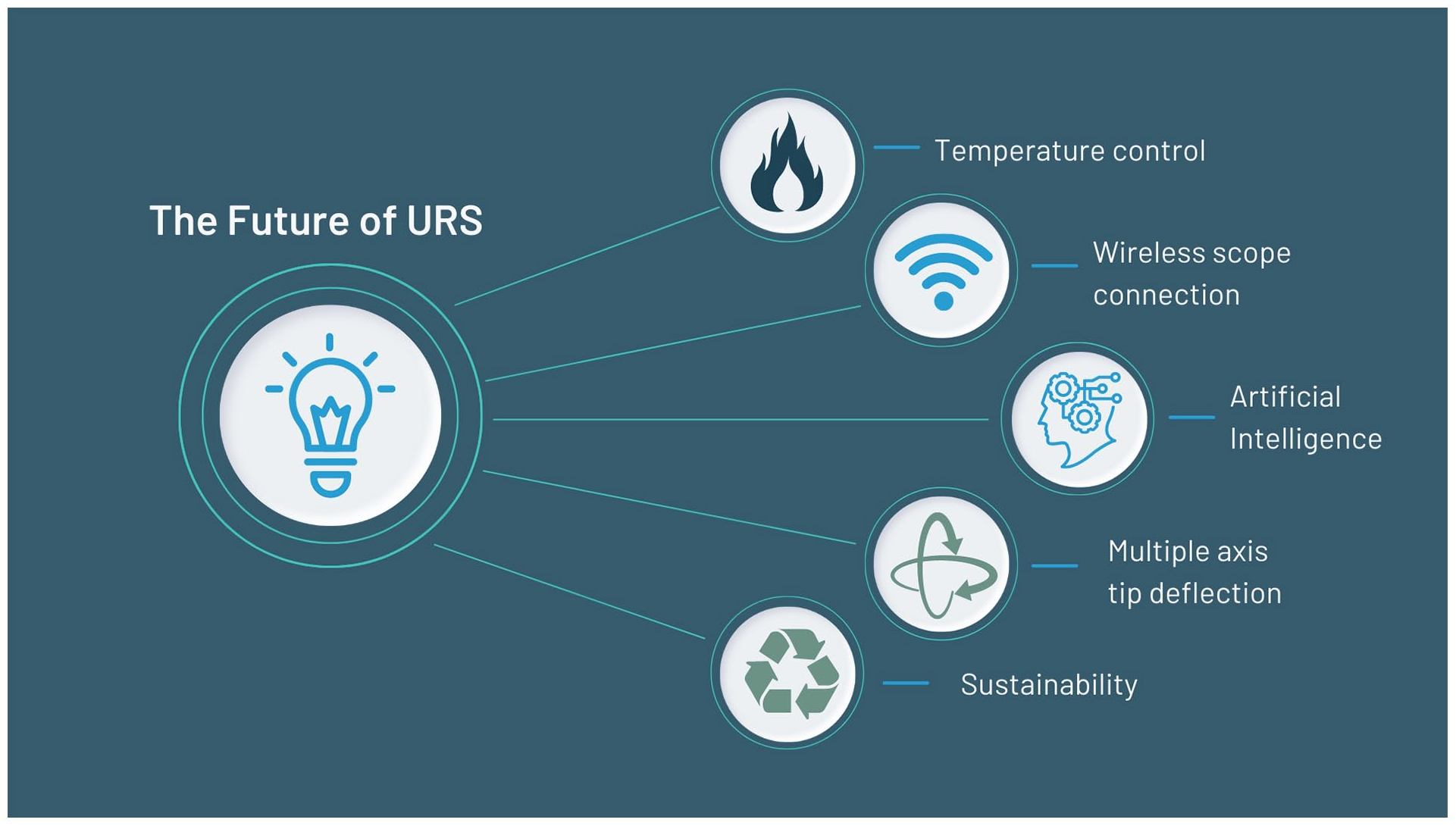
Future of URS.
Regarding new technology, robotic platforms for URS have been introduced and early results have been reported but the high upfront costs, space requirement and successful outcomes associated with conventional URS render it unlikely to achieve dissemination in the near future at least. 68 While such technology does reduce the ergonomic burden for the surgeon, the tactile feedback that is enabled through handheld manipulation of the scope is also lost. 10 Introducing such haptics and multiple axis tip deflection would be valuable additions to robotic systems. Regarding improving peri-operative care, enhanced recovery after surgery (ERAS) protocols have recently been introduced for stone surgery and this is likely to become increasingly popular, particularly in an effort to reduce opioid use in the recovery period.69,70 This issue has attracted increased attention in North America where over 50% of patients receive opioid prescription post-URS. 71
TEST
These advances across all areas of endourological treatment allow for a tailored strategy to be planned and implemented for a particular patient. To this end, we propose a new term to capture this, TEST (Figure 3).
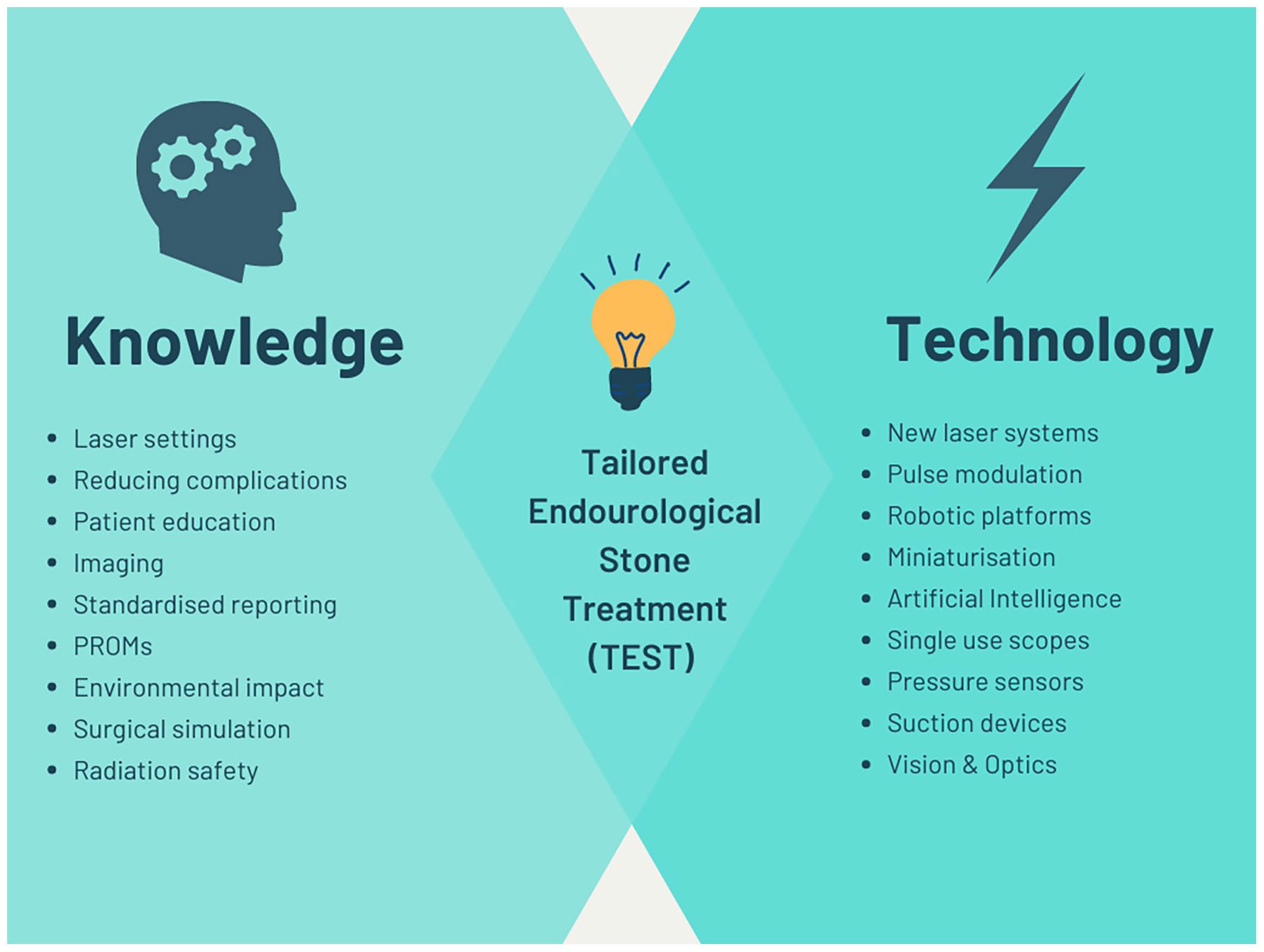
Tailored endourological stone treatment.
Conclusion
There have been many advances in URS over recent decades. While new technology stands at the centre of this, innovations have also introduced such as those to improve operative planning and aftercare, which bring health professionals another step closer to stone surgery, which is truly customised. As such, the clinician is now able to offer a TEST.
Footnotes
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: E.X.K. is a speaker/consultant for Coloplast, Olympus, Boston Scientific, Recordati, Debio.pharm, Alnylam but has no specific conflict of interests relevant to this work. Ø.U. is a consultant for Olympus but has no specific conflict of interests relevant to this work. All other authors have no conflicts of interest to declare. No external company has been involved in writing or content of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Figures created using Canvas for educational purposes.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
P.J.
Contributorship
P.J. and Ø.U. conceived the idea for the article. All authors reviewed the literature to find relevant material. All authors evaluated and interpreted this material. P.J. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript including the figures. Ø.U. supervised the project.
