Abstract
Objective:
Currently the National Institute for Health and Care Excellence (NICE) recommends an abnormal digital rectal examination (DRE) as a standalone referral criterion for suspected prostate cancer. Unlike referrals for a raised prostate-specific antigen (PSA) which are triaged directly to magnetic resonance imaging (MRI), an abnormal DRE requires re-examination in a secondary clinic first. Here, we investigated the ongoing value of the abnormal DRE as a referral criterion.
Methods:
This study is a retrospective review of patients referred to secondary care for suspected prostate cancer based on an abnormal DRE over a 15-month period at a single UK hospital (n = 158). Age, PSA, primary and repeat DRE findings and eventual diagnosis were collated.
Results:
A concurrent raised PSA was present in 65/158 (41%). Concordance between primary and secondary care DRE was only 72/158 (46%). The overall and significant cancer detection rate was 26/158 (16%) and 22/158 (14%), respectively. Among men with a concurrent raised PSA, 19/65 (29%) had significant cancer found, whereas with an abnormal primary care DRE and normal PSA (n = 93), only 3/93 (3%) had a significant cancer. Mandating a PSA before referral for an abnormal DRE would have redirected 65/158 (41%) of men to MRI first, negating the need for a repeat DRE (p < 0.0001). This finding was recapitulated in a second prospective validation cohort (n = 30) with 9/30 (30%) redirected to MRI first.
Conclusions:
This is one of the first studies to investigate the value of the DRE in contemporary practice. We propose that PSA is used to triage men with an abnormal DRE to MRI without needing a repeat DRE. If the PSA is normal, the diagnostic yield is low but may still warrant a repeat DRE to assess the need for further investigations. Additional multi-centre studies are required to further validate our findings.
Level of evidence:
4
Introduction
The current UK recommendations from the National Institute for Health and Care Excellence (NICE) 1 advise primary care doctors to refer patients to prostate cancer diagnostic clinics mainly on the basis of (1) an abnormal digital rectal examination (DRE) or (2) an age-referenced elevated prostate-specific antigen (PSA). These guidelines have been in place for over 20 years, and primary care DRE remains an important part of the routine assessment for patients with suspected prostate cancer. However, the evidence underpinning these recommendations comes from historic population-based case–control studies. 2 In these, the positive predictive value for a ‘malignant-feeling’ DRE was as low as 12% but is known to improve as PSA increases. 3 Screening studies have also shown that DRE, in turn, can improve PSA sensitivity.4–6
The continued utility of DRE remains controversial.4,7,8 Much of the data are derived from studies performed in the pre-magnetic resonance imaging (MRI) era of prostate cancer diagnostics and before the current imperative to diagnose only clinically significant disease.7,9 With the use of MRI much earlier in the referral pathway, the role of an isolated abnormal DRE is becoming increasingly uncertain. 8 In particular, whether a primary care-based DRE (in the absence of a raised PSA) could be triaged straight to MRI (as currently happens with an elevated age-referenced PSA) or mandates a repeat re-examination by a specialist first. At present, most units would arrange a repeat secondary care examination which incurs clinic and staffing resources. A patient may then still go onto an MRI and further investigations based on this. Reducing unnecessary clinic visits has become particularly important, given the increased risk of Covid-19 to the typical prostate cancer patient demographic. 10
There is a lack of up-to-date evidence supporting the role of DRE in the modern prostate cancer referral pathway. To address this, we undertook a retrospective study to assess the outcomes of investigations in men referred from primary care to our centre with a suspicious DRE.
Methods
Cohort
Between January 2019 and March 2020, using our hospital’s electronic record system, we undertook a retrospective case note review. We included all men referred for suspected prostate cancer on the 2-week wait pathway where the primary referral criterion was an abnormal DRE (institutional review board approval: Service Evaluation PRN8595). Figure 1 illustrates the two current referral pathways based on whether referral is triggered through a raised PSA or an abnormal DRE. For every patient, we collated the following: age (years), PSA (ng/ml) at referral, primary care DRE findings, secondary care DRE findings, if an MRI and/or biopsy occurred and if a diagnosis of prostate cancer was made. Repeat DREs at our hospital were all performed by senior staff grades or consultants. We recorded if any cancer was found and if this was clinically significant (defined as histological ⩾ grade group 2 on a biopsy or a clinical diagnosis of cancer based on a high PSA + T3 on clinic DRE). All information was anonymised at the point of data collection and entered into a database. An abnormal PSA was defined as a threshold PSA ⩾2.5 ng/ml if younger than 50 years, ⩾3.0 ng/ml for men aged 50–69 years and ⩾4.0 ng/ml for patients aged 70 years and above. This allowed stratification of patients with an abnormal DRE into two groups: those with an abnormal PSA and those without an abnormal PSA. For each of these groups, we assessed the diagnostic path taken and investigation outcome. To validate our findings, we applied our models (see analysis) to a second prospective cohort (n = 30) of men referred between April 2020 and December 2020. Data collection and analysis were similar to the primary cohort above.
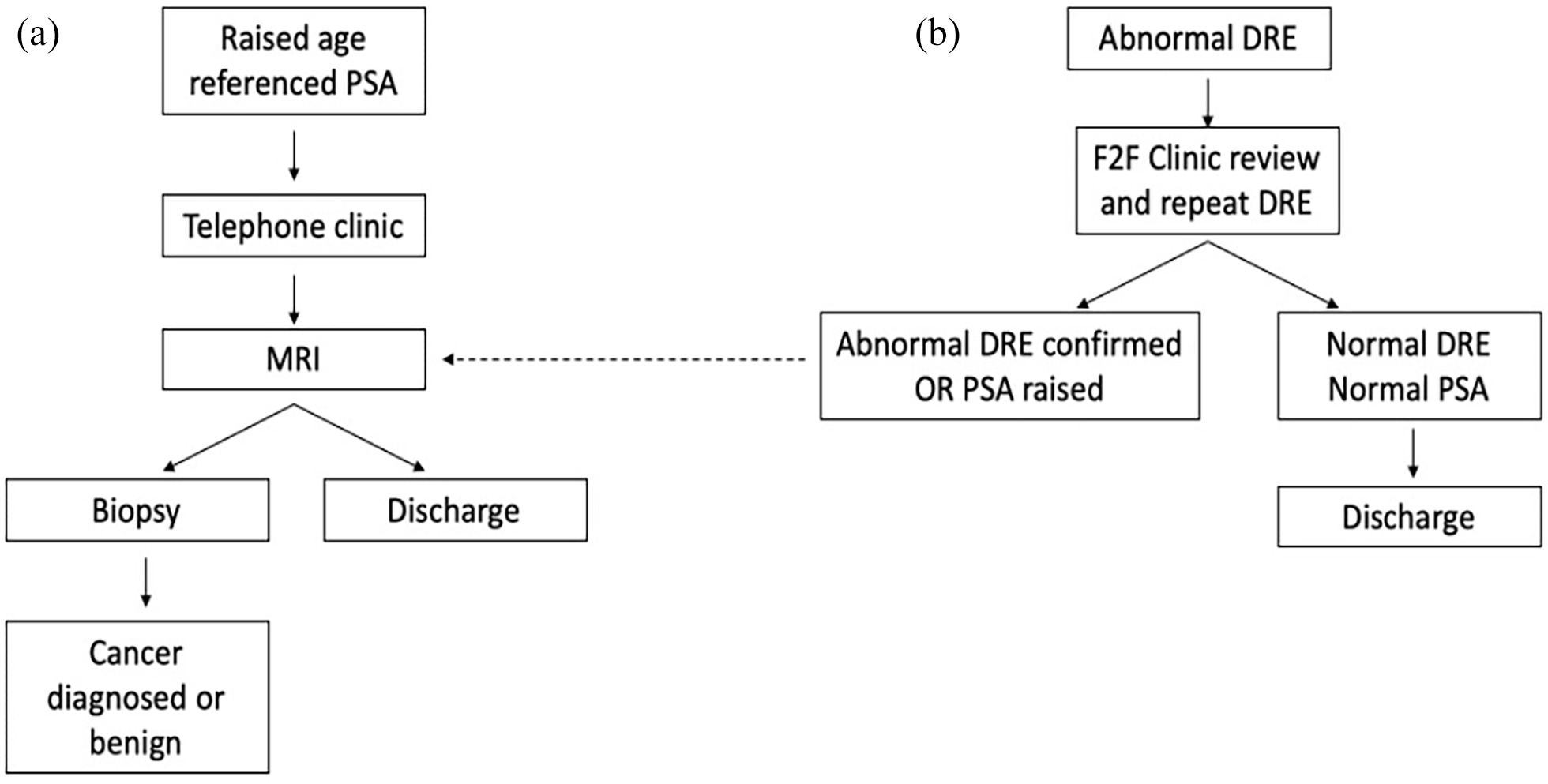
Current pathway for men referred for suspected prostate cancer with (a) raised PSA and (b) an abnormal DRE. The need for a repeat assessment in the current pathway necessitates an extra clinic visit and resources. In contrast, treating an abnormal DRE from primary care the same as a raised PSA would result in a large number of unnecessary MRI scans.
Analysis
The concordance between primary care DRE and secondary care DRE was assessed, as was the number of prostate cancer diagnoses in patients with an abnormal DRE alone or an abnormal DRE and abnormal PSA. Based on this, we proceeded to test models capable of reducing unnecessary clinic visits. Two alternative diagnostic pathways were tested. Under model A, no referrals would be accepted to secondary care clinics unless the PSA was tested and raised above the age-referenced range (Figure 2). Under model B, following an abnormal DRE, a PSA measurement is mandated before a referral is made. If the PSA is elevated, then the patient proceeds directly to prostate MRI (similar to standard of care). If the PSA is normal, then a DRE is repeated in secondary care. We applied these alternate pathways to compare the net effect on the number of secondary care visits needed and impact on cancer diagnoses. Simple descriptive analysis was performed using proportions and percentages within and between the groups. Fisher’s exact test was used to compare the cancer diagnosis rates between the current and proposed new pathways and to assess the significance in the observed difference in secondary clinic visits between pathways. Our sample size had a power of 0.94 (alpha 0.02) to detect significant differences. The McNemar test was used to look for any concordance between a raised PSA and a confirmed second abnormal DRE. The standardised difference in mean number of visits was compared using Student’s t test.
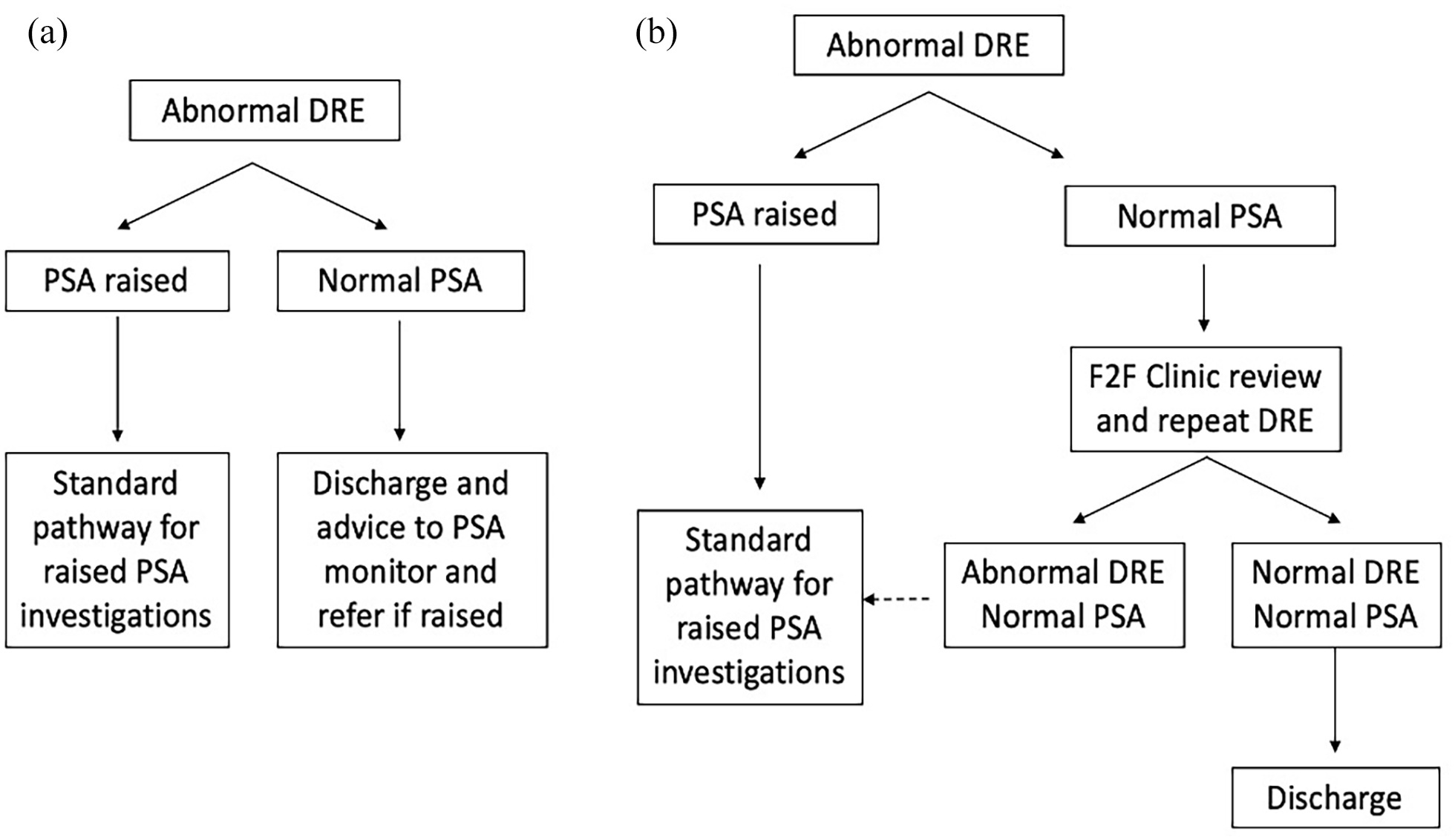
Alternative model pathways for men referred for suspected prostate cancer with model (a) only accept referrals for an abnormal DRE if there is a concomitant raised PSA and model (b) triage referrals to MRI pathway if there is a raised PSA, regardless of an abnormal DRE. A normal PSA results in repeat clinical examination in secondary care.
Results
Primary cohort analysis
A total of 158 patients were included in the primary study group (Table 1). The median age was 69 years. An elevated age-referenced PSA was observed in 65/158 (41%) of men. The concordance rate between primary care and repeat DRE in the secondary care clinic was 72/158 (46%). Of the remaining patients, seven did not have a repeat DRE (deemed unnecessary as the PSA was raised) and the remainder were considered normal examinations. Of these 158 patients, 82 went onto have an MRI and 38 subsequently had prostate biopsies. The overall cancer detection rate was 26/158 (16%) and for clinically significant cancers, 22/158 (14.0%). One case of non-Hodgkin’s lymphoma involving the prostate was identified, and this case was excluded from the analysis.
Baseline characteristics of patients from the primary cohort and validation cohort.
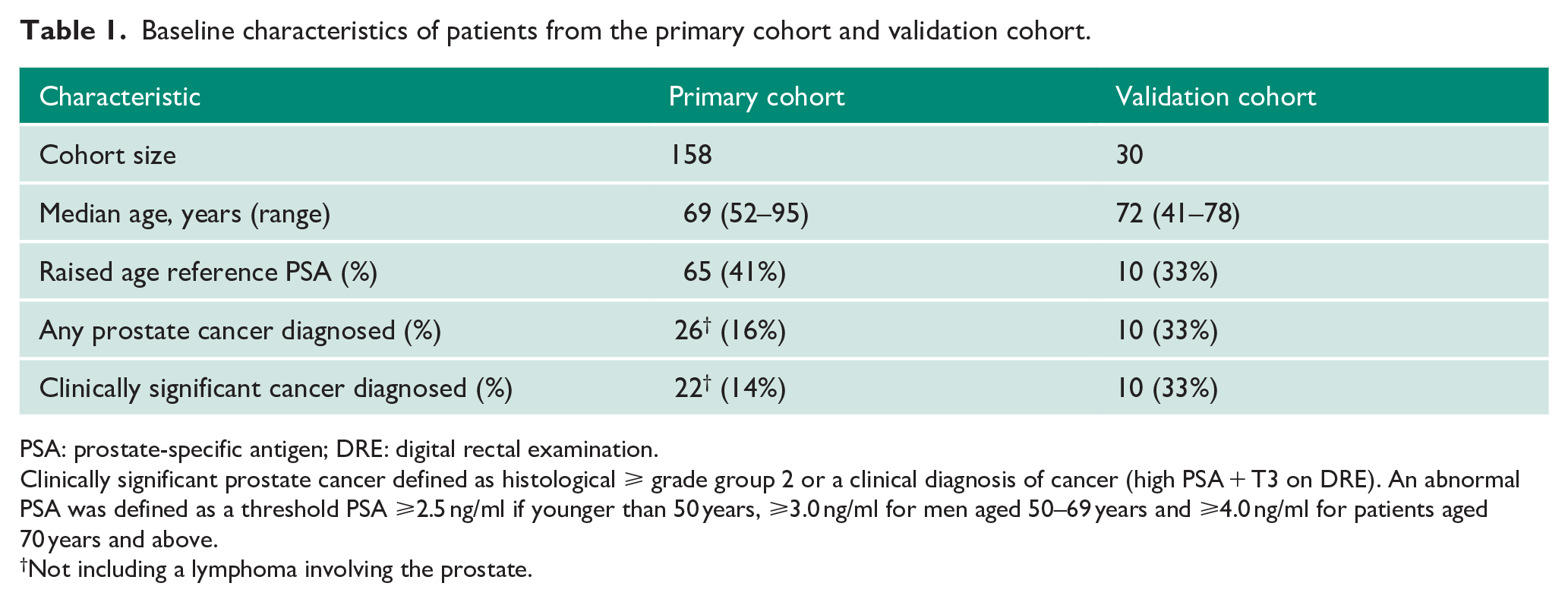
PSA: prostate-specific antigen; DRE: digital rectal examination.
Clinically significant prostate cancer defined as histological ⩾ grade group 2 or a clinical diagnosis of cancer (high PSA + T3 on DRE). An abnormal PSA was defined as a threshold PSA ⩾2.5 ng/ml if younger than 50 years, ⩾3.0 ng/ml for men aged 50–69 years and ⩾4.0 ng/ml for patients aged 70 years and above.
Not including a lymphoma involving the prostate.
Subgroup analysis
The cohort was next sub-divided by whether or not there was a concurrent age-elevated PSA.
Concurrently raised PSA
Of those with an abnormal primary care DRE and a concurrent raised PSA (n = 65), 45/65 (69%) went on to be referred for MRI after being seen in clinic and 23/65 (35%) went on to undergo prostate biopsy. A total of 21/65 men (32%) were subsequently diagnosed with prostate cancer of which the majority (n = 19) were clinically significant cancers (Table 2). Thus, the overall significant cancer detection rate was 29%. The primary care and repeat DRE concordance among these men was 38/65 (58%).
Distribution of cancer diagnosis by PSA at presentation in the primary cohort.
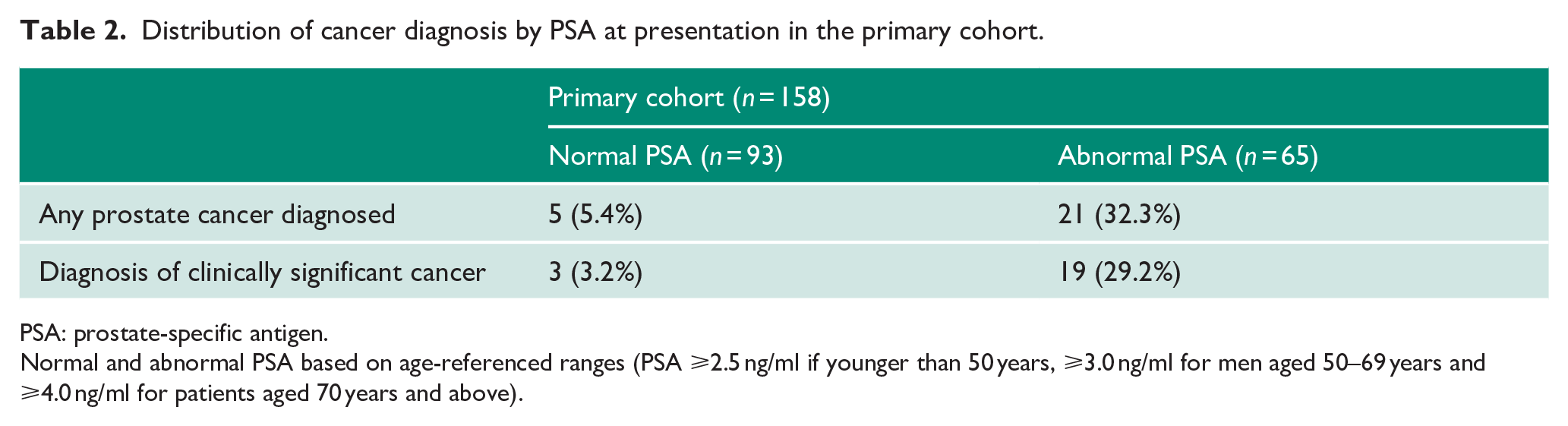
PSA: prostate-specific antigen.
Normal and abnormal PSA based on age-referenced ranges (PSA ⩾2.5 ng/ml if younger than 50 years, ⩾3.0 ng/ml for men aged 50–69 years and ⩾4.0 ng/ml for patients aged 70 years and above).
Normal PSA
Among men with an abnormal primary care DRE and a normal age-referenced PSA (n = 93), 37/93 (40%) were referred for MRI and 15/93 (16%) subsequently had a prostate biopsy. A total of 5/93 men (5%) were found to have prostate cancer of which 3/93 men (3%) had clinically significant prostate cancer (Table 2). The primary care and repeat DRE concordance among these men was 34/93 (37%).
We further looked for any concordance between a normal or raised PSA and a confirmed secondary care DRE and found no significant association (p = 0.44).
Modelling alternative pathways
The previous analysis had shown differences in cancer detection rate depending on the presenting information. To test how we could refine our pathway, we modelled two decision algorithms (Figure 2). If model A were implemented, then 93 referrals would be rejected (59% reduction). However, this would miss 3/22 (14%) patients with clinically significant cancers (excluding the prostate lymphoma presentation). If model B were implemented, then no referrals would be rejected. Secondary care repeat DRE visits would however be reduced by 65 (41% reduction) with no cancers missed (p < 0.0001 compared to the current prevalent diagnostic method). Overall, the mean number of clinic visits for a repeat DRE was 0.58 in the new pathway compared to 1.0 in the prevalent method (p < 0.0001). Table 3 also shows the comparative performance of the current and proposed pathways in cancer detection efficiency. The current pathway (based on repeat DRE for all) had a p value of 0.0006 for detection of significant cancer (odds ratio 5.9 (1.82–25)) and a pathway based on triaging with a raised PSA had a p value of <0.00001 (odds ratio 11.8 (3.19–64.7)). To further validate this alternative pathway, we applied this to a second prospective cohort derived from the time period subsequent to the primary dataset (Table 1). In this cohort, there were proportionally more cancers identified; 33% (10/30) and all were considered clinically significant (Supplemental Table 1). Nearly all also occurred in men with an abnormal PSA (9/10) (Table 2). Applying model A, 21 referrals would have been rejected (70% reduction) but one patient with clinically significant cancer missed. Applying model B, nine secondary care DRE visits would be saved (30% reduction) but with no significant cancers missed.
Comparative performance of different pathways for cancer detection based on either (a) traditional pathway with repeat secondary care DRE for all or (b) new pathway using PSA to triage to MRI.
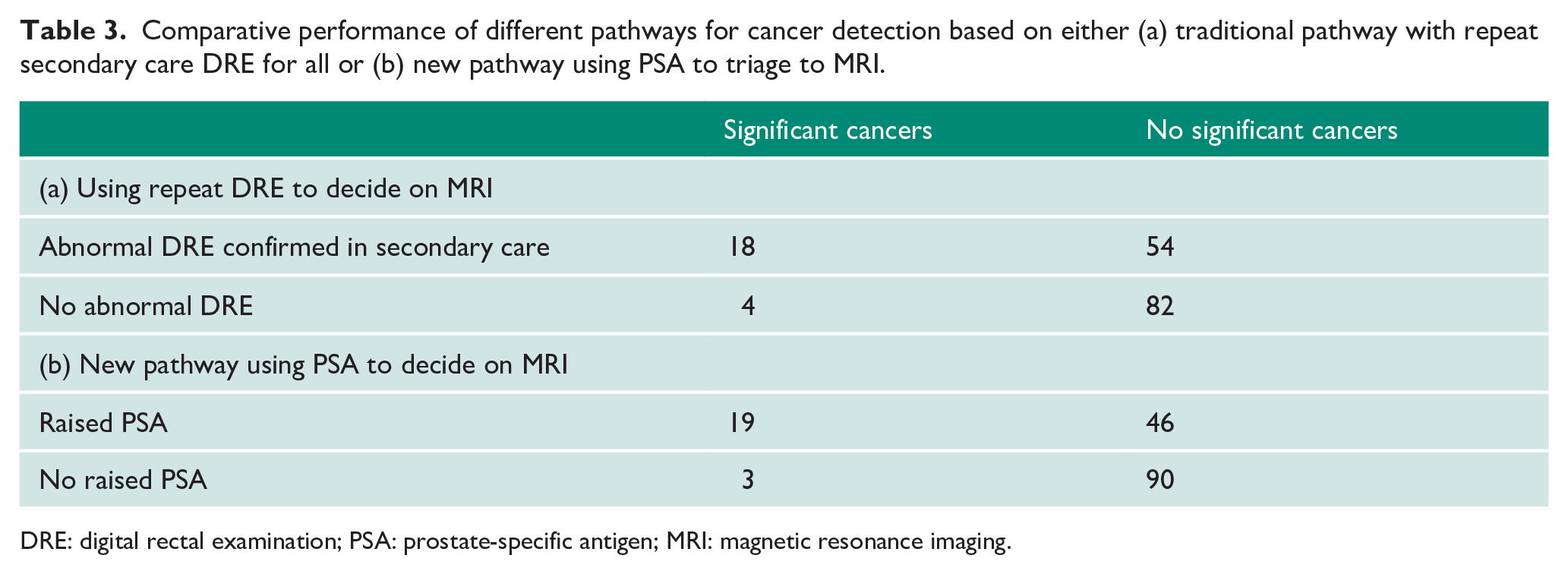
DRE: digital rectal examination; PSA: prostate-specific antigen; MRI: magnetic resonance imaging.
Discussion
In this paper, we analysed the cancer diagnosis yield from referrals based on an abnormal DRE in primary care. We demonstrate that concordance between primary and secondary care DRE is poor. We further show that using PSA levels can reduce unnecessary repeat examinations and instead re-direct many patients to MRI instead. Using this simple decision tree, at least 30% of repeat DRE clinic visits can be saved.
The rationale for an abnormal DRE as a trigger for referral is based on the notion that PSA alone may sometimes miss cancers. Furthermore, previous studies have shown the added value of DRE if a PSA is also raised, particularly in detection of higher volume and more advanced cancers. 11 However, these studies and others pre-date the use of MRI which is intended to both exclude the need for a biopsy (if negative) and earlier detect significant cancer while still organ confined. The widespread availability of MRI and its use as an upfront tool therefore questions the value of DRE in modern cancer diagnosis. In this study, for example, the significant cancer rate among men with an abnormal primary care DRE was only 3% if a PSA was normal but nearly 10 times higher if the PSA was also abnormal. This is in line with previous work which has shown the inefficiencies of DRE, with regard to sensitivity and specificity, at lower PSA levels.6,8,12,13
Mandating a PSA to accompany a DRE should be easy to implement and is probably already a common practice (though not required by current guidelines). In our series, we found that PSA results were often available after the referral had been made. Reasons for referrals that are not accompanied by a PSA result included the following: not a mandate to decide on referral, administrative errors or differing PSA age-referenced thresholds. On this latter point, we have previously reported that at least 10 different PSA range thresholds are used across England at the present time. 14 Another finding was the poor concordance between primary and secondary care DRE. In our study, this was less than 50%. Walsh et al. 7 however reported better concordance of 74%. There is however surprisingly little in the way of other literature on this issue. No doubt, there are a number of confounding factors to consider including the experience of the primary care doctors, the number of DREs they have done and/or if they have any experience in urology. Of more value would be an assessment of prostate volume, as the PSA corrected for volume (PSA density) is known to be a much better indicator for prostate cancer presence, though this may be challenging to do in primary care at the current time. 15
Strengths of our study included the relative completeness of our data and the use of a test and validation cohort. However, our study has the expected limitations of a retrospective single-centre design and is reliant on the reporting and documentation of DRE results by different primary care doctors. Our sample size is also relatively small and there were no fixed criteria for the secondary care decision to investigate, and hence, this relied on clinician discretion in the clinic. We also did not do any a priori sample size and power estimates before this study. We do believe however that this report is one of the only UK studies to assess the value of DRE in this setting and also reflects real-world practice.
To conclude, we have presented here an analysis of the value of the primary care abnormal DRE as a single or combined test with a raised PSA. With modern MRI-based pathways, many referrals for primary care abnormal DRE can follow the standard route if a PSA is raised. For those with normal PSA, the diagnostic yield is very low but men probably still warrant repeat DRE in clinic from which onwards investigation can be made. Although further validation in other centres is warranted to test the veracity of our findings, we believe that there are sufficient reasons to now consider changing practice and guidance to better streamline referrals. We therefore call for a simple but potentially significantly impactful resources saving change in the current prostate cancer referral guidelines on abnormal DRE.
Supplemental Material
sj-docx-1-uro-10.1177_20514158221091402 – Supplemental material for Diagnostic value of the abnormal digital rectal examination in the modern MRI-based prostate cancer diagnostic pathway
Supplemental material, sj-docx-1-uro-10.1177_20514158221091402 for Diagnostic value of the abnormal digital rectal examination in the modern MRI-based prostate cancer diagnostic pathway by Wasiq Sajjad, Vineetha Thankappannair, Syed Shah, Adham Ahmed, Kasra Saeb-Parsy, Christof Kastner, Benjamin Lamb and Vincent J Gnanapragasam in Journal of Clinical Urology
Footnotes
Acknowledgements
We are grateful to Mrs Christina Stephens for administrative help for data collection. V.J.G. acknowledges infrastructure support from the National Institute for Health Research (NIHR) Cambridge Biomedical Research Centre (BRC-1215-20014). The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care. We are very grateful to Dr Artitaya Lophatananon, University of Manchester, for expert additional statistical advice.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Addenbrooke’s Hospital (institutional review board approval: Service Evaluation PRN8595).
Informed consent
Informed consent was not sought for this study because all data were fully anonymised.
Guarantor
V.J.G.
Contributorship
V.J.G., W.S. and V.T. were involved in the collection and analysis of data. W.S. analysed data and wrote the first draft of the manuscript. V.J.G. reviewed data and analysis and edited the manuscript, and conceived and proposed the project. All authors were involved in reviewing and drafting the manuscript and approving the final content.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
