Abstract
Purpose:
The natural history and clinical manifestations of dural metastases (DM) in castrate-resistant prostate cancer (CRPC) will change with advances in new hormonal therapy. Here, we characterised the incidence, clinical presentation, and outcomes of patients with DM in a contemporary patient cohort with CRPC.
Methods:
We retrospectively reviewed our CRPC database from 2012 to 2020. The primary outcome was the diagnosis of DM, defined as metastasis to the dura mater in the brain or spine. We describe the presenting symptoms, biochemistry, radiologic findings, and therapy sequence for all DM patients. Multivariable logistic regression was performed to identify predictors of DM.
Results:
Six of the 275 patients (2.2%) with CRPC developed DM. The average age of CRPC diagnosis for patients with DM was 65.6 years. Mean patient survival was 4.5 months after the diagnosis of DM. At the time of CRPC diagnosis, patients who developed DM were significantly younger, had lower baseline haemoglobin, higher lactate dehydrogenase (LDH), and elevated alkaline phosphatase (ALP) compared to those without DM. On multivariable analysis, younger age of CRPC diagnosis was found to be a predictor for DM.
Conclusion:
The presence of neurological symptoms in the context of younger age, anaemia, and elevated baseline LDH and ALP are associated with DM in CRPC.
Level of Evidence:
4
Introduction
Dural metastases (DM) in advanced prostate cancer are rare, with a historical incidence between 0.3% and 8.1%, approximately six times less common than breast or lung cancer.1–4 Routine brain scanning is thus not routinely recommended as part of the metastatic workup for patients with prostate cancer,5,6 aside from those with advanced neuroendocrine features. 7 DM in association with prostate cancer has been reported in small observational studies, case reports, and autopsy series,8,9 and can be classified as intracranial, intraspinal, epidural, or a combination thereof. 10 The mechanism of spread to the dura is thought to be via Batson’s plexus to the bone and subsequent extension into the central nervous system or via a direct haematogenous route. 11
The management of castrate-resistant prostate cancer (CRPC) has evolved with the introduction of novel hormonal therapies (NHTs) such as abiraterone, enzalutamide, apalutamide, and most recently, darolutamide.12,13 Improved survival characteristics support a central role of the androgen receptor pathway in disease pathogenesis, and as a successful target for current and future therapy. These agents may impact the pathogenesis of metastatic disease, including the development of DM. 14
To direct patient therapy in CRPC, clinical and biochemical predictors are often used.15,16 Elevated serum alkaline phosphatase (ALP) and lactate dehydrogenase (LDH) levels have been associated with worse overall survival and increased metastatic burden.17,18 However, the impact of clinical and biochemical factors in specifically predicting DM is less certain. The aim of this study is to characterise the incidence of DM in a contemporaneously treated CRPC cohort with a focus on clinical and radiological features and identify predictors that may prompt earlier investigation and treatment of DM in advanced patients.
Methods
Study design and population
Following institutional ethics approval, a retrospective chart review was conducted of CRPC patients seen at the Carlo Fidani Regional Cancer Centre, Trillium Health Partners (THP) from 2012 to 2020. Patients were included regardless of prior primary tumour treatment or CRPC treatment sequence.
Variables and outcomes
Clinical variables collected include age, prior prostate therapy, sequence of therapy including chemotherapy and NHT, presenting symptoms at DM, prostate-specific antigen (PSA) kinetics, and full biochemistry including LDH and ALP. PSA doubling time was determined at the time of diagnosis of CRPC and DM, respectively. At each diagnosis, the PSA at the three most recent timepoints were fit with a line and the slope was used to determine the doubling time. Prostate pathology was reviewed by a uropathologist and determined from prostate biopsy in three patients, transurethral resection of prostate (TURP) specimen in one patient, and radical prostatectomy specimens in two patients.
The primary outcome assessed was radiographic progression to DM, defined as the presence of primary intracranial or intraspinal tumour localisation. All DM-positive cases were reviewed at multi-disciplinary tumour boards. The radiographic findings were reviewed by a dedicated neuroradiologist (YAC).
Statistical analysis
Descriptive statistics were tabulated on patients with or without DM. Univariable analysis was performed utilising Student’s T test for continuous variables assuming equal variance. Odds ratios and 95% confidence intervals were calculated. Multivariable logistic regression was performed using chosen covariates to determine potential predictors of developing DM. All statistical analyses were performed using SPSS Version 26 (IBM) with a p value of <0.05 used for statistical significance.
Results
Of the 275 CRPC patients reviewed, 46 (16.7%) patients developed visceral metastases and of these, six (2.2%) patients had confirmed DM, of which one of six had pathological confirmation following surgical resection. The mean follow-up of our CRPC cohort was 21.8 ± 17.8 months.
Management of prostate cancer and CRPC
The timeline of prostate cancer progression for all DM patients from initial diagnosis of prostate cancer to progression to CRPC to death is outlined in Figure 1(a). At the time of initial prostate cancer diagnosis, two of six patients were treated with primary radiation, two of six underwent radical prostatectomy, and two of six received docetaxel as initial therapy for de novo metastatic disease.19,20 All patients had confirmed castrate levels of testosterone at diagnosis of CRPC with continued luteinising hormone-releasing hormone (LHRH) agonist therapy throughout treatment. Following time of CRPC, three of six received enzalutamide, one of six received abiraterone, and two of six received both therapies. In addition, three of six patients received radium-223 and one of six patients received cisplatin and etoposide for confirmed neuroendocrine differentiation confirmed by pathology following palliative TURP for bladder outlet obstruction.
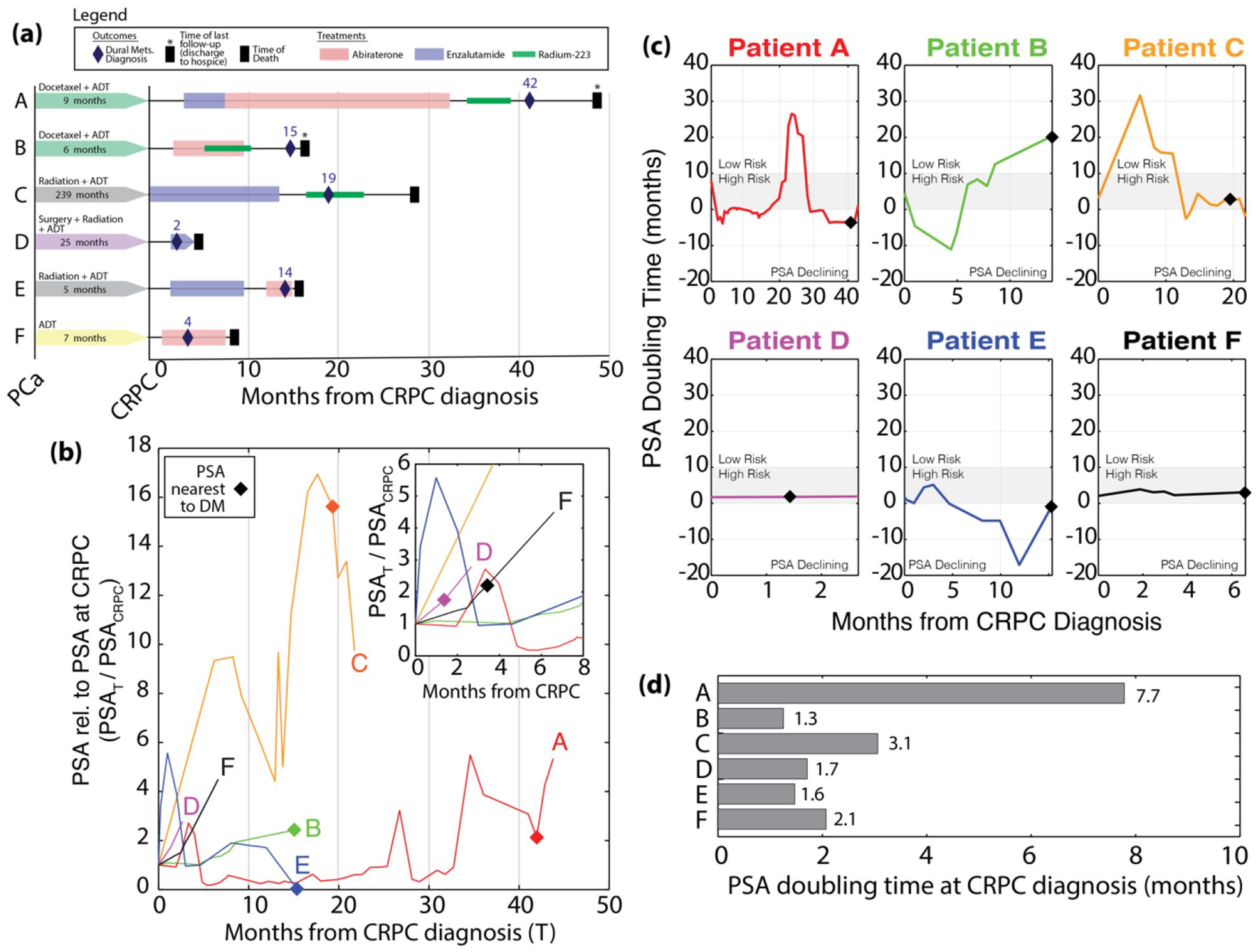
(a) Clinical history and treatment of each patient with the onset of DM and time to death, (b) PSA normalised to PSA at CRPC until time of death from CRPC, (c) PSA doubling time for each patient over course of follow-up, and (d) PSA doubling time at CRPC diagnosis for patients who developed DM.
All six patients had documented osseous metastases of which four of six experienced a symptomatic skeletal event (SSE), defined as pathologic fracture, spinal cord compression, or bony pain requiring radiation. All four SSEs were treated with palliative external beam radiation and supportive care. With regards to visceral metastases, one of six had previous choroidal metastases treated with radiation, and one of six had lung metastases which were treated conservatively. One patient had concurrent parenchymal metastases which were treated with radiation.
Clinical, pathological, and biochemical features at initial CRPC diagnosis
Prostate cancer pathology was available for all patients. Of these, five patients had adenocarcinoma on pathology, and one was found to have neuroendocrine disease following a palliative TURP. Total Gleason score was available for five of six patients, of which three had a Gleason score 9 or above. The average time from initial prostate cancer diagnosis to the development of CRPC was 54.6 ± 80.5 months. Mean PSA at CRPC diagnosis was 420.8 ± 597.5 ng/mL and PSA over the course of CRPC is provided in Figure 1(b). The mean PSA doubling time was 2.9 ± 2.4 months with all six patients meeting high-risk criteria with a doubling time <10 months 21 as shown in Figure 1(c) and (d).
The time to progression to DM and biochemical profile of the patients with and without DM at CRPC diagnosis are summarised in Supplementary Table 1. Patients with DM had significantly lower haemoglobin of 108.8 ± 22.9 g/L, elevated ALP of 478.7 ± 621.8 U/L, elevated LDH of 860.2 ± 618.9 U/L, and were diagnosed of CRPC at a younger age of 65.2 ± 9.0 years compared to those who did not progress to DM. On multivariable regression, younger age was found to be a statistically significant predictor of DM (Table 1).
Multivariable logistic regression analysis of features associated with the development of dural metastases.
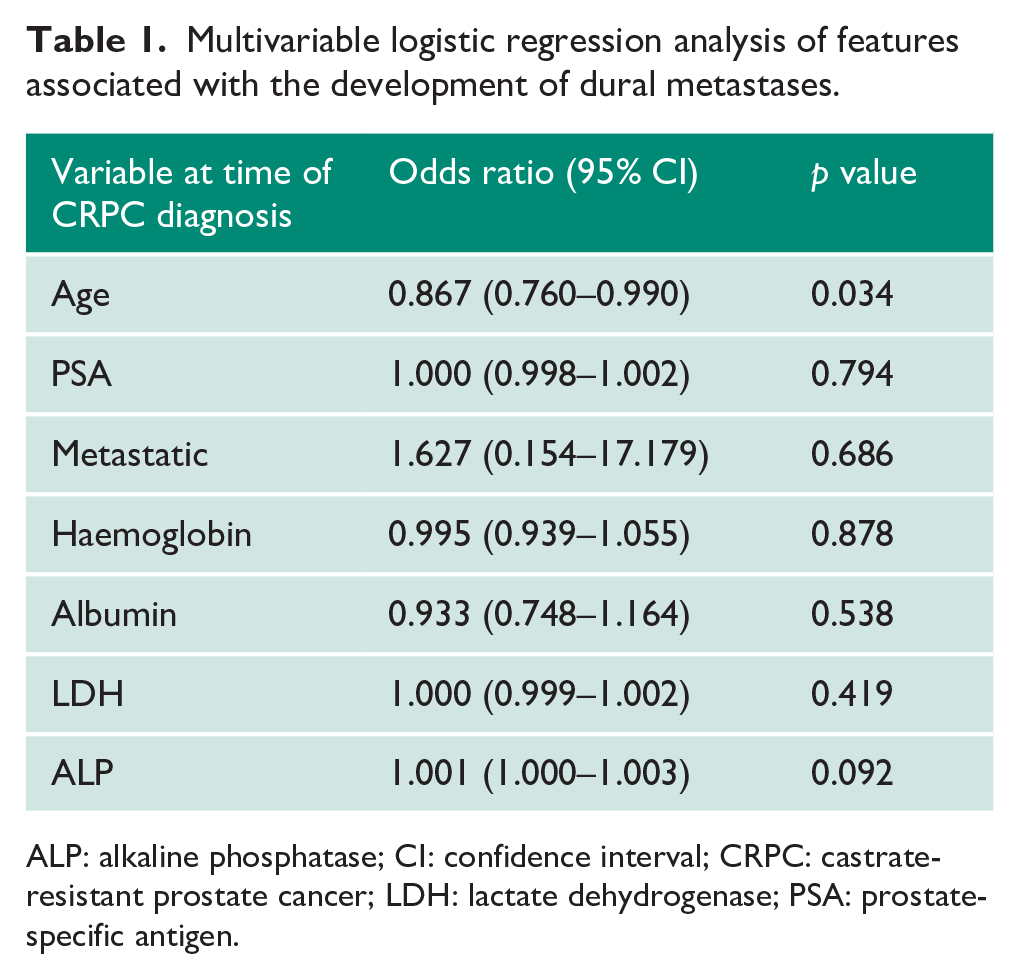
ALP: alkaline phosphatase; CI: confidence interval; CRPC: castrate-resistant prostate cancer; LDH: lactate dehydrogenase; PSA: prostate-specific antigen.
Clinical and biochemical features at progression to DM during CRPC
The average time from the diagnosis of prostate cancer to DM was 64.5 ± 95.8 months, and the average time from CRPC to DM was 16.0 ± 14.4 months. The mean age of DM presentation was 66.6 ± 8.4 years. The time between diagnoses of DM and the last follow-up or death was 4.3 ± 3.6 months. All six patients were confirmed to have died from DM. The medical record of death was available for four of six patients, and the date of discharge to hospice was used in two of six cases for time-to-event analysis.
As shown in Table 2, presenting symptoms included motor deficits (three patients), neuropathic pain (two patients), seizures (one patient), and headaches with cranial nerve deficits (one patient). At DM, patients had reduced mean haemoglobin to 91.7 ± 21.8 g/dL, elevated mean LDH of 1247.8 ± 1033.9 U/L, and an elevated ALP of 390.2 ± 480.5 IU/L compared to the time of initial CRPC. Compared to the time of initial CRPC diagnosis, the mean PSA was significantly elevated to 976.8 ± 1061.7 ng/mL at DM onset.
Clinical, pathological, and timepoints for patient outcomes included in this case-series.
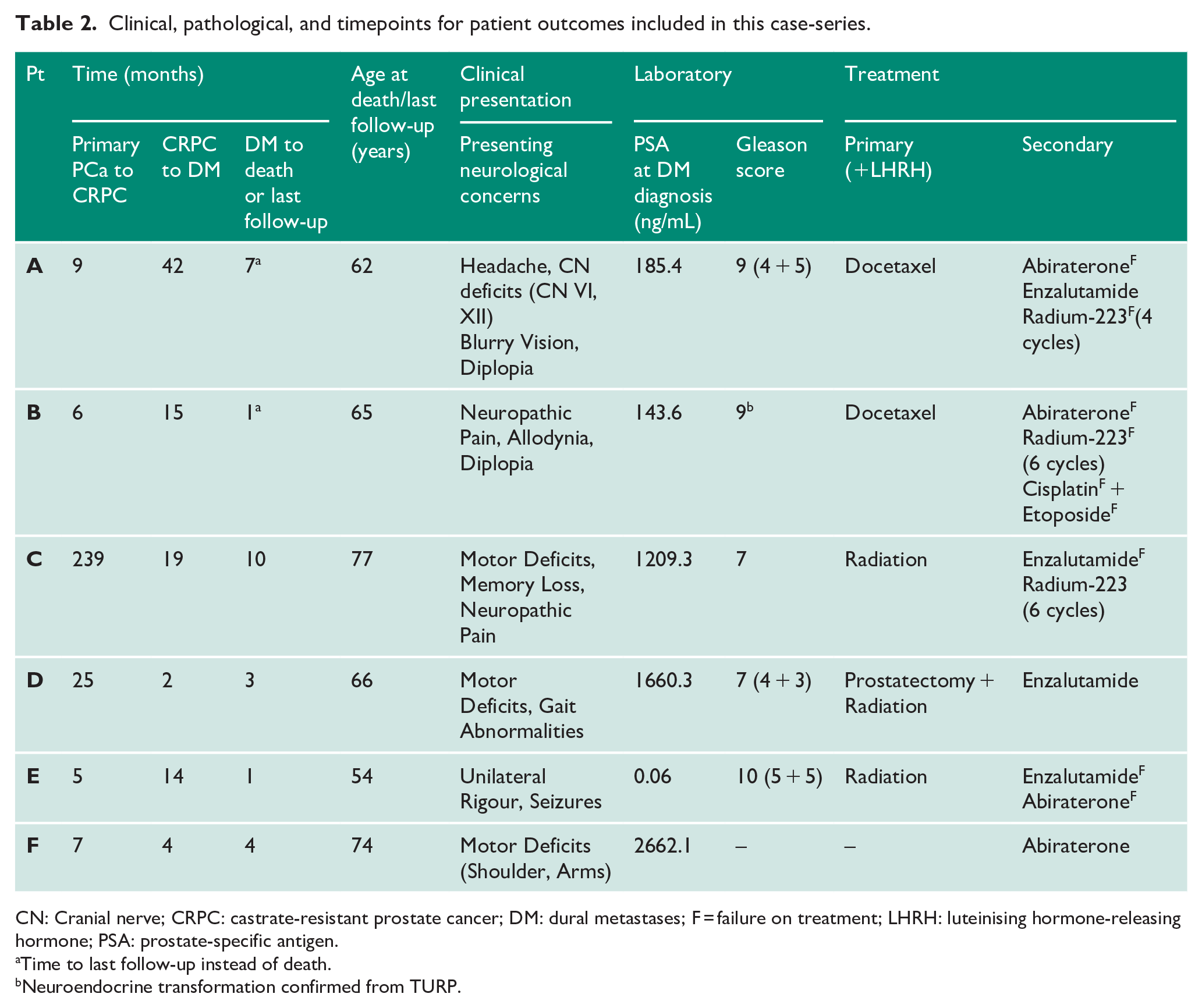
CN: Cranial nerve; CRPC: castrate-resistant prostate cancer; DM: dural metastases; F = failure on treatment; LHRH: luteinising hormone-releasing hormone; PSA: prostate-specific antigen.
Time to last follow-up instead of death.
Neuroendocrine transformation confirmed from TURP.
Radiographic findings of DM
Radiological findings for each patient are summarised in Table 3. Five patients presented with multifocal dural lesions. One patient had epidural metastasis at C4T1 with central canal compression and was treated with palliative radiation and dexamethasone. Of the remaining five patients with intracranial DM, two were treated with whole-brain radiation, one was treated with stereotactic radiosurgery, one was surgically resected, and one patient could only be provided best supportive measures.
Radiological findings and treatment for each patient included in this case-series.
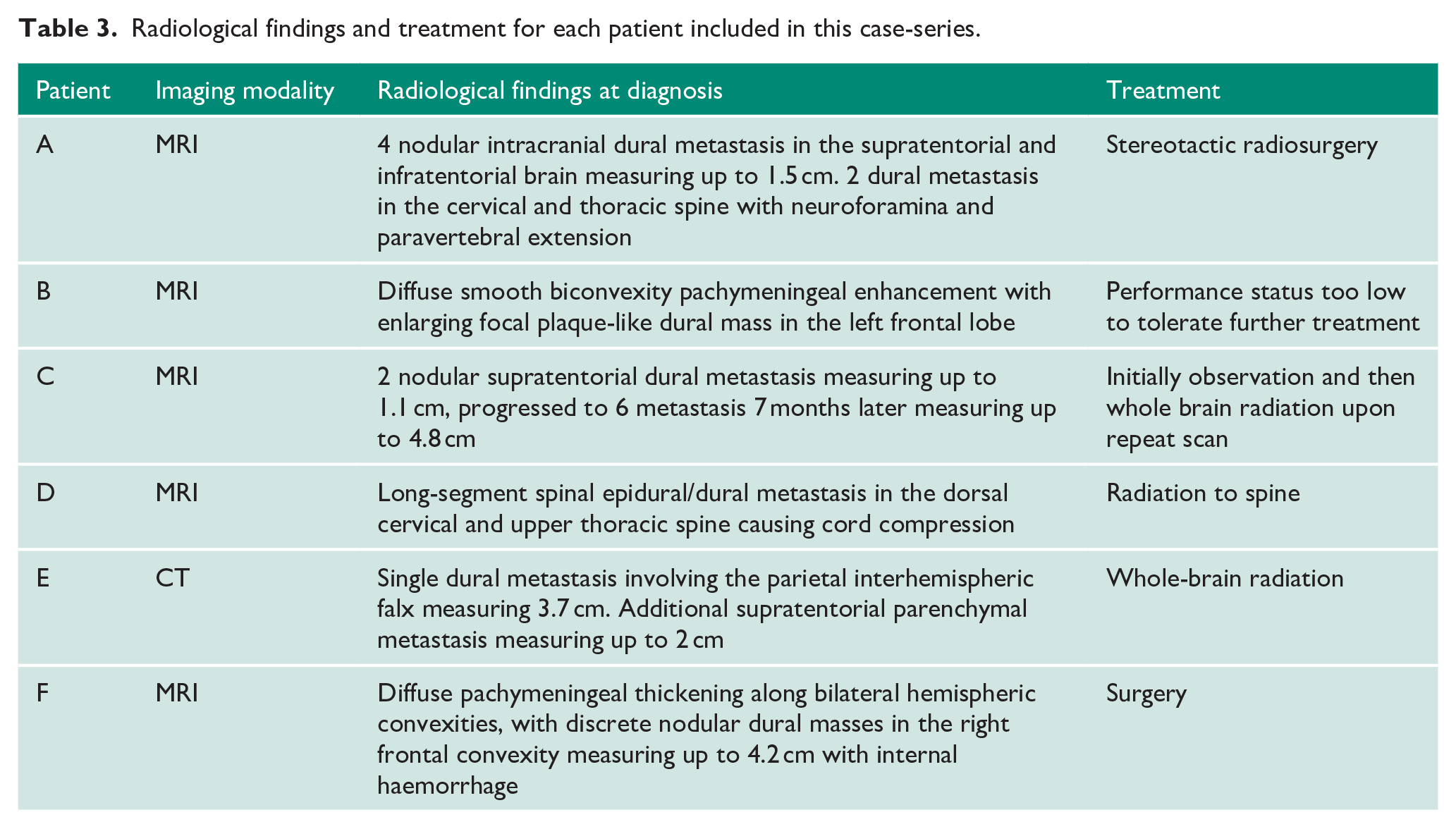
Notably, patient C had two small (<1 cm) dural lesions at diagnosis of DM, but then had radiographic progression of disease to six dural lesions at 7 months. Two of six patients were initially thought to have primary meningiomas on radiological interpretation before further review and consensus at multidisciplinary tumour boards. Of the five patients with intracranial DM, four presented with multiple dural lesions and secondary vasogenic oedema. All patients were provided supportive and palliative care as necessary.
Discussion
While overall survival has improved in the era of NHTs, 16 the incidence of rapidly disseminated disease upon failure of NHT is increasing. 22 This may be due to the earlier selection of highly resistant and proliferative clones. 23 The most common sites for visceral metastases in CRPC are the lungs and liver. 24 In our series, we found that DM is a rare but clinically significant finding in the CRPC population. The presence of CNS symptoms, younger age, anaemia, and an elevated baseline LDH and ALP were associated with the development of DM. To our knowledge, this is the first study that assesses clinical and biochemical factors to predict the development of DM in a contemporary CRPC cohort. Whether one NHT is more associated with the development of DM cannot be assessed in this cohort due to the low event rate.
The literature on DM in CRPC is scarce, with most experience emanating from single-centre case series (Table 4). In other series, the average age at DM was between 62 and 66 years, the average time from prostate cancer diagnosis to DM was between 43 and 56 months, and the average survival time for patients after DM was between 2.6 and 8.1 months.2,3,25,26 In our experience, the mean age at DM was 67 years, the mean time from prostate cancer diagnosis to DM was 65 months, and the mean survival time was 4.5 months, in keeping with previous series (Table 4). Our cohort had a longer time to DM from prostate cancer diagnosis compared to other series. This delayed presentation of DM until later in the course of disease may be attributed to earlier administration of second-line therapy which may alter the natural history of DM. 3
Presentation and time to clinical outcomes for patients with DM in the context of CRPC recorded in the literature.
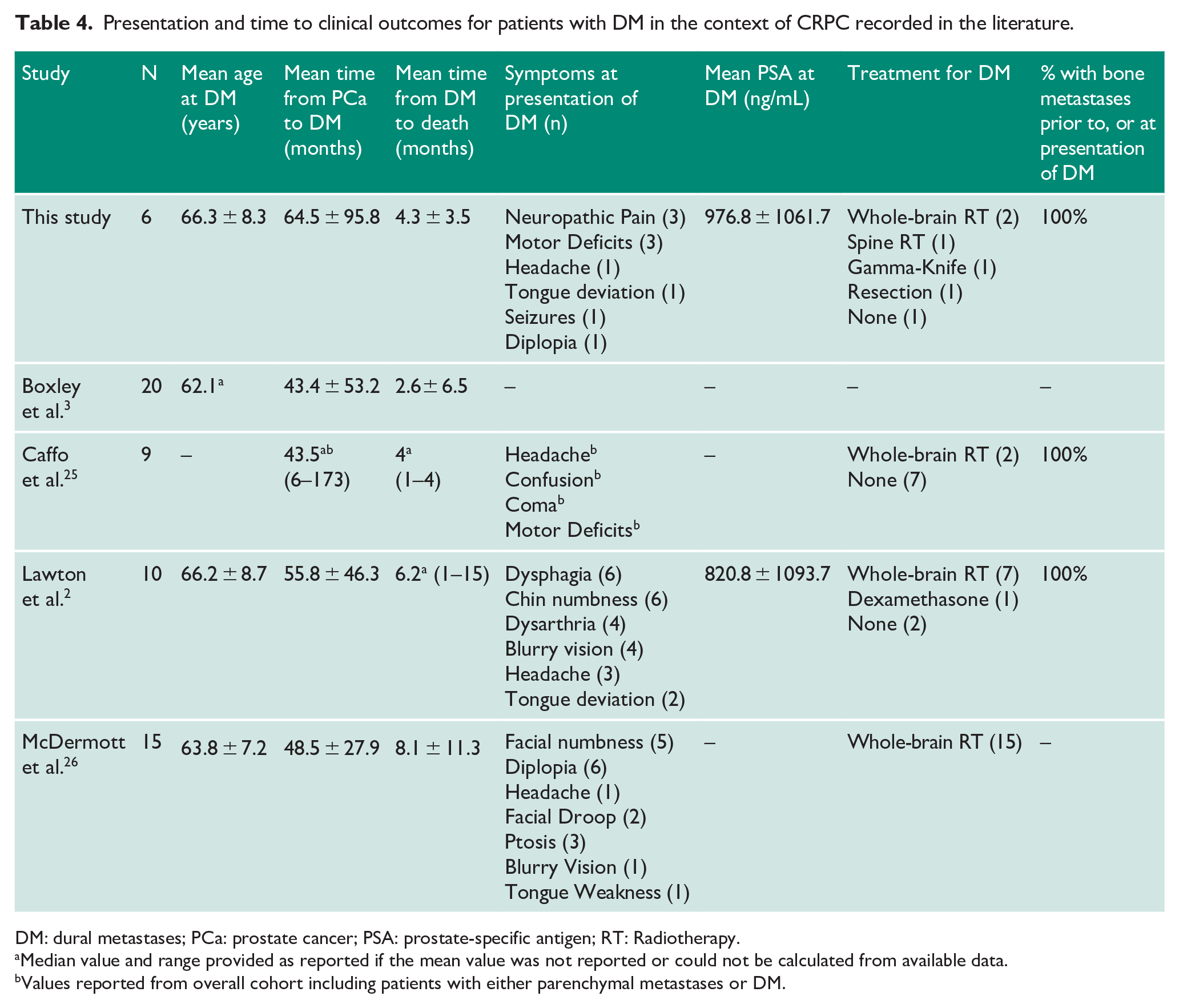
DM: dural metastases; PCa: prostate cancer; PSA: prostate-specific antigen; RT: Radiotherapy.
Median value and range provided as reported if the mean value was not reported or could not be calculated from available data.
Values reported from overall cohort including patients with either parenchymal metastases or DM.
We found that the most common presenting symptoms for DM included focal cranial nerve deficits, motor deficits, headaches, and neuropathic pain.2,25,26 Early symptom recognition and treatment may play a central role in extending survival. 1 Despite it being highly associated with a terminal event, timely investigation of DM may provide an opportunity for earlier palliation via whole-brain radiation, surgical resection, or stereotactic radiosurgery. 1 The incidence of DM in prostate cancer cohorts ranges from 0.3% to 8.1%, while the incidence of DM in our series was 2.2%, which is in agreement with previous literature.1–3
In this analysis, we evaluated the predictive capacity of numerous clinicopathological variables for DM. Patients who develop DM tended to be younger, have lower baseline haemoglobin values, and have higher comparative ALP and LDH at the time of CRPC diagnosis. Interestingly, PSA was not an independent predictor of DM in our series. Although many studies support PSA as a major driver of disease course, 27 PSA performs less optimally in advanced patients compared to LDH and ALP. 28 Importantly, PSA doubling time of less than 10 months at the time of CRPC diagnosis was highly indicative of DM, 21 supporting its role as a key predictive factor. These predictors are consistent with known adverse prognostic risk factors.29,30 The consistency of these results in our analysis could support more ardent clinical screening for neurological symptoms during follow-up, with a low threshold for earlier CNS surveillance for these patients. Future analysis combining genetic and immunohistochemical features of the primary tumour with the present findings may further stratify the at-risk patient cohort.
Limitations of the current study include its retrospective methodology and a relatively small event rate. Treatment heterogeneity among the six patients who developed DM is a potential confounder in the determination of predictors. Asymptomatic patients with CRPC may have undiagnosed or latent DM in this study population. Pathologic confirmation of DM was only available in one of six patients, with five of six based on strict radiographic criteria only.
Conclusion
The rate of DM is rare in the CRPC population and is associated with a poor prognosis. Clinical manifestations of DM include a younger age, anaemia, and an elevated baseline LDH and ALP at CRPC diagnosis. Further study to stratify these poor-risk patients and to describe hormonal therapy impact is warranted.
Supplemental Material
sj-docx-1-uro-10.1177_20514158221090040 – Supplemental material for Incidence of dural metastases in castrate-resistant prostate cancer
Supplemental material, sj-docx-1-uro-10.1177_20514158221090040 for Incidence of dural metastases in castrate-resistant prostate cancer by Adree Khondker, Jethro CC Kwong, Christopher Tran, Emily Evans, Ishan Aditya, Lucshman Raveendran, Yingming A Chen, Amna Ali and Andrew Feifer in Journal of Clinical Urology
Footnotes
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Trillium Health Partners Research Ethics Board.
Informed consent
Informed consent was not sought for the present study because due to retrospective nature of the study, a waiver of consent was approved by the research ethics board.
Trial registration
Not applicable because this study uses retrospective chart review methodology.
Guarantor
AF
Contributorship
AK, JK, AA, and AF conceived the study. All authors collected data and performed analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
