Abstract
Introduction
Small cell carcinoma of the prostate (SmCCP) is a rare and aggressive prostate malignancy. It is commonly observed in men who have previously undergone hormone treatment with androgen deprivation for prostatic adenocarcinoma. 1 SmCCP carries a poor prognosis with a mean survival rate of less than 1 year after diagnosis. 1 Our current literature lacks both pathological and imaging description of this rare malignancy, especially when it occurs de novo. In this report, we present both histological and magnetic resonance imaging (MRI) findings of SmCCP in a gentleman without a previous history of prostatic adenocarcinoma.
Case report
A 50-year-old male presented with visible haematuria and clot retention. The patient reported a 3-month history of rectal pain and worsening lower urinary tract symptoms. He denied melena or haematochezia. Past medical history included ischemic heart disease and atrial myxoma resection 10 years ago. He had a 30-pack-year history of smoking and took regular aspirin.
On examination, the abdomen was soft and non-tender. He had a firm and enlarged prostate on digital rectal examination. His Hb was 147 g/L with a white cell count (WCC) of 11.2 × 109/L. Serum prostate-specific antigen (PSA) was 1.2 ug/L. Renal function was preserved with a serum creatinine of 88 umol/L and estimated glomerular filtration rate (eGFR) of 83 mL/min/1.73 m2. International normalised ratio (INR) was 1.1. He remained haemodynamically stable and afebrile. Urine culture was sterile.
Our patient underwent continuous bladder irrigations with a 22-Fr three-way indwelling catheter. An initial computed tomography (CT) intravenous pyelogram (IVP) demonstrated a large heterogenous fluid and soft tissue attenuating mass lying within the rectovesical pouch. There was associated presacral, bilateral common, external iliac and left para-aortic lymphadenopathy. A subsequent gadolinium-enhanced 3-Tesla MRI of the pelvis confirmed a primary prostate malignancy with peri-prostatic, pelvic sidewall and retroperitoneal nodal metastases and meso-rectal vascular invasion. Furthermore, MRI appearance was suggestive of direct tumoral invasion into the bladder base, left pelvic side wall musculature and into the anterior rectum (see Figures 1 and 2). There were no thoracic or intracranial metastases on staging scans.
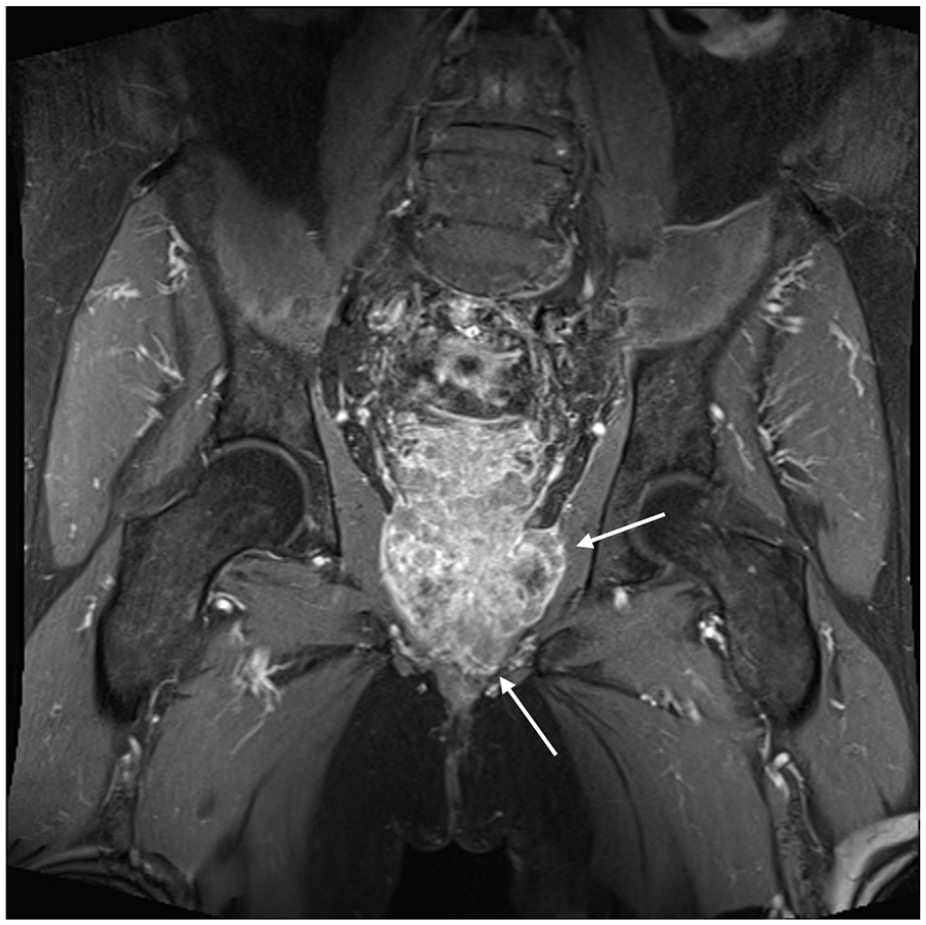
Coronal T1 fat-saturated post-gadolinium MRI sequence showing invasion of tumour into pelvic sidewall musculature (left > right) and into the pelvic floor. T1 hypo-intense non-enhancing areas in the centre likely represent necrosis or haemorrhage.
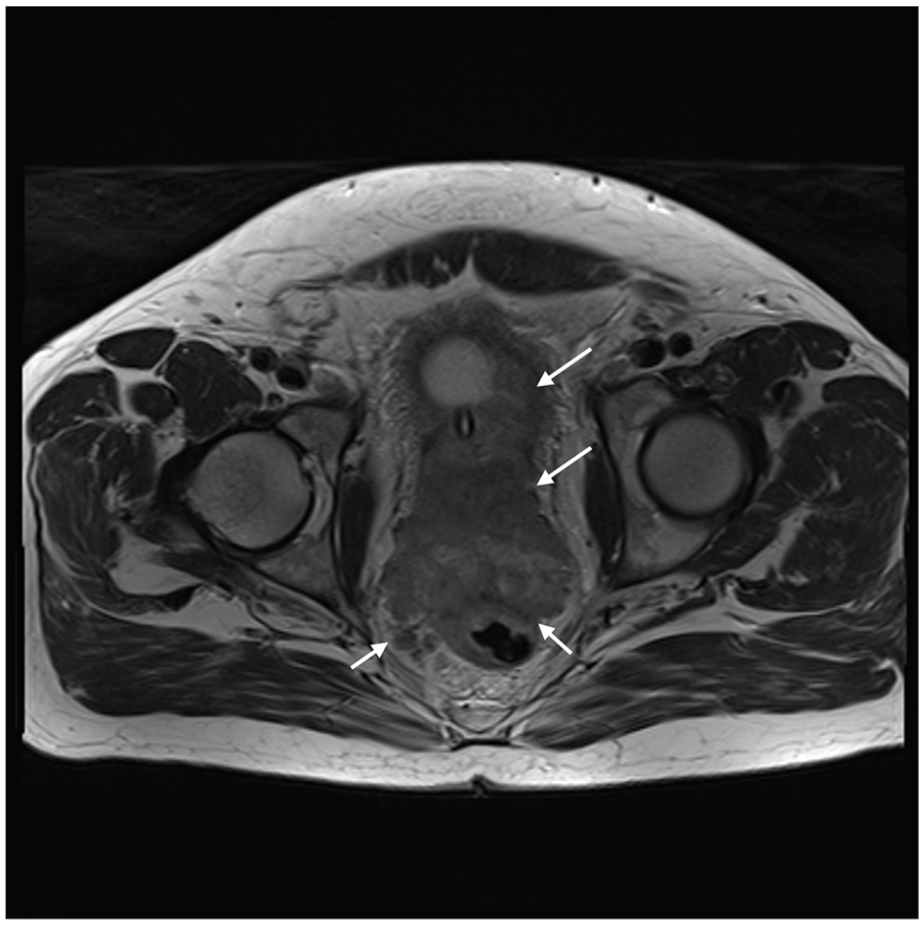
Axial T2 non-fat-saturated MRI sequence demonstrating direct tumoral invasion into the bladder, seminal vesicles, anterior rectal wall and meso-rectal vascular invasion. There is loss of peri-rectal fat planes and Denonvillier’s fascia.
Trans-rectal prostate biopsy was performed. Final histopathology confirmed smooth muscle extensively infiltrated by small cell neuroendocrine carcinoma in one out of six cores (see Figure 3). Immunohistochemistry stains showed positivity for AE1/AE3, MNF 116 and synaptophysin and were negative for desmin, chromogranin, WT 1, S100, PSA and prostate specific acid phosphatase (PSAP). Referral was made to oncologists, given extensive nodal disease.
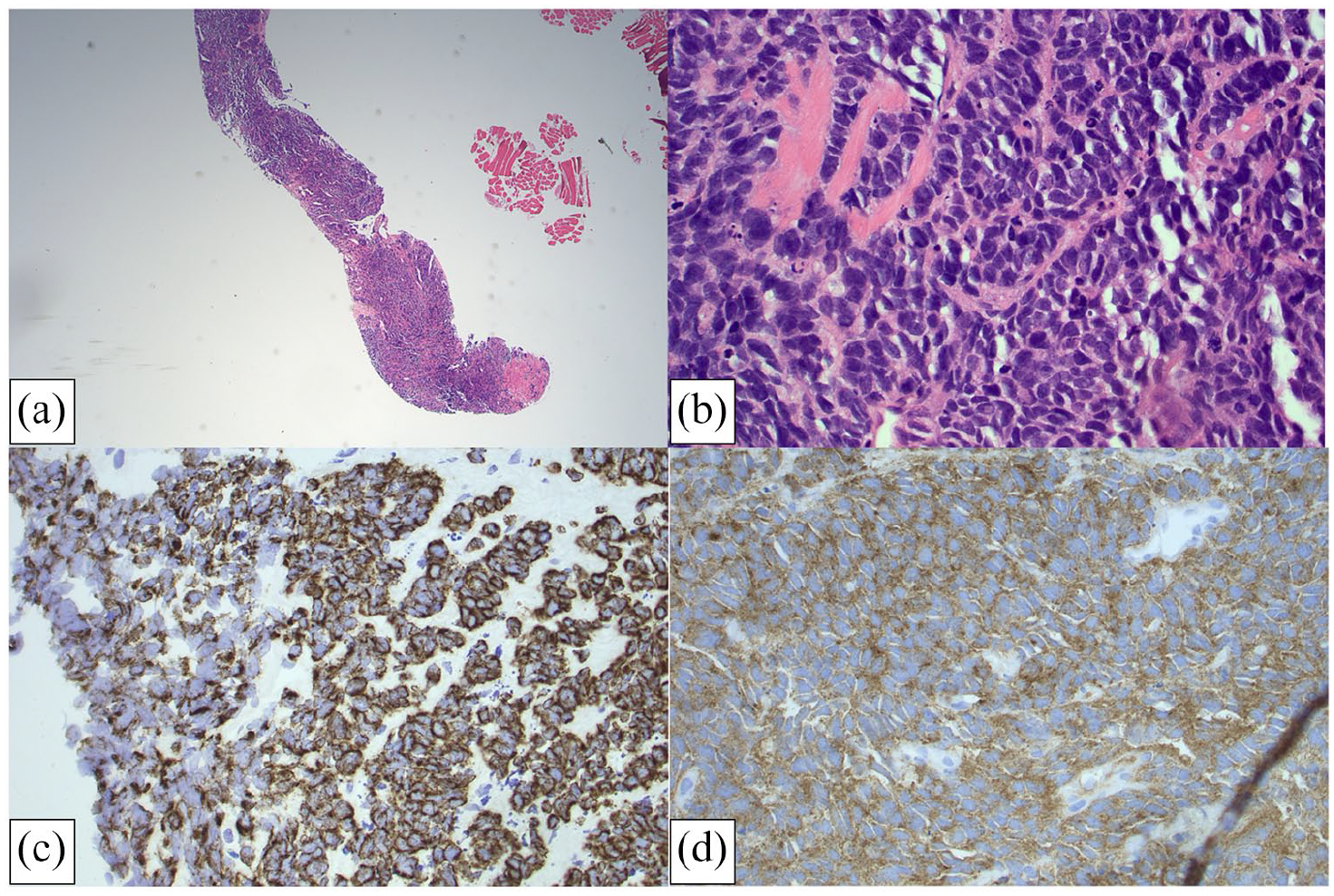
Photomicrographs demonstrating sections of biopsied prostate tissue. Haematoxylin- and eosin-stained sections at lower power (×40) (a) and higher power (×200) (b) show diffuse infiltration with small round blue cells displaying features of small cell carcinoma. The chromatin pattern has a characteristic ‘salt and pepper’ appearance. Cytokeratin MNF 116 (c) and synaptophysin (d) positive immunohistochemistry was observed.
The patient proceeded with four cycles of carboplatin and etoposide chemotherapy followed by pelvic external beam radiotherapy. A dose of 60 Gy was delivered in 30 fractions to the prostate with 50.4 Gy delivered in 30 fractions to the pelvic nodal area. This gentleman remained clinically well 6 months post-treatment with no radiological progression of disease.
Discussion
SmCCP accounts for <1% of all prostate cancers. 1 It is a rare malignant neoplasm exhibiting neuroendocrine differentiation and may occur pure or mixed with acinar adenocarcinoma. According to the American Urological Association, about 50% of patients have a history of acinar adenocarcinoma or prior hormonal treatment with androgen deprivation therapy. 1 Interestingly, PSA levels remain normal, despite extensive disease or metastatic spread.1,2
Patients generally present with rapid onset of urinary tract obstruction and a large tumour burden, extra-prostatic extension and/or distant metastases, making them poor candidates for surgery. The optimal treatment for men with SmCCP is not well established but platinum-based combination chemotherapy is often first line.2,3 Regimens including cisplatin or carboplatin with either etoposide or docetaxel have been described. 2 Consolidation radiotherapy may help with palliative symptom control by shrinking tumour size. Hormone therapy has no role in treatment as neuroendocrine cells are devoid of androgen receptors. 1 Despite treatment, SmCCP remains highly aggressive and carries a poor prognosis with mean survival of <1 year after development. 3
Histological features of SmCCP are indistinguishable from SmCCP of other organs. There are sheets of small round blue cells with ‘salt and pepper’ chromatin that infiltrate prostatic tissue in a diffuse pattern.1,2 Crush artefact, nuclear moulding, high mitotic activity, apoptosis and necrosis are commonly observed features.1,4 Immunohistochemistry is usually positive for cytokeratins and neuroendocrine markers such as synaptophysin and chromogranin. 4
Due to its rarity, MRI findings of SmCCP are not well documented in literature. Tumours are generally large and heterogenous and have low to intermediate signal intensity on T1-weighted imaging (T1W1), high intensity on T2-weighted imaging (T2WI) and are hyperintense on high b-value with moderate low signal density on apparent diffusion coefficient (ADC) imaging.5,6 These tumours enhance avidly with gadolinium. Findings from our report are concordant with those of He et al. 7 who describe a similar case of SmCCP in a 78-year-old man with MRI demonstrating low signal on T1WI, higher signal on T2WI and diffusion weighted imaging (DWI) with an ADC value of 0.860 ± 0.130 mm2/s. Other imaging features include bladder or rectal invasion, enlarged pelvic lymph nodes, metastases to other organs other than bone (e.g. brain, lung or liver) and osteolytic bone metastases. 7
The usual imaging gold-standard for workup of haematuria is a CT-IVP with limited role of MRI. In this scenario, the use of MRI in conjunction with CT was particularly useful in the diagnosis of SmCCP. This case demonstrates the utility of MRI in defining pelvic pathology and suggests that the increased use of MRI, especially in patients with suspected prostate malignancy, could improve outcomes.
Footnotes
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethics approval for reporting individual cases or case series.
Informed consent
Verbal and written informed consent was obtained from the patient for the anonymised information to be published.
Guarantor
P.S.
Contributorship
P.S. and A.A.A. were involved in the patient’s care and performing the diagnostic biopsy. P.S. and A.A.A. researched the literature and contributed to writing the case report and discussion. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
