Abstract
Background:
Intracranial metastases from prostate adenocarcinoma are very unusual and typically occur late in the course of the disease, and initial presentation with symptomatic brain involvement, especially vision loss is very rare (with this being only the sixth such reported case to the best of our knowledge). The present case elucidates how a diagnosis was reached in such a scenario and the management provided.
Case presentation:
A 66-year-old gentleman presented with loss of vision and headache with no other ocular or neurological complaint. Computed tomography (CT) of his head revealed a destructive lesion involving the clivus and a space-occupying lesion (SOL) in occipito-parietal region. Detailed inquiry regarding the possible primary source of suspected the metastatic lesion revealed an increased frequency of urination, nocturia, and significant weight loss. His serum prostate-specific antigen (PSA) levels were raised. He was treated by surgical hormonal therapy and his visual symptoms improved.
Conclusion:
Awareness of such a presentation can lead to an accurate diagnosis. Initiation of appropriate therapy can successfully alleviate the neurologic deficits.
Background
Prostate carcinoma metastasis to the brain is extremely rare with its incidence in patients with systemic metastasis elsewhere being around 0.38%. However, only 0.86% of all patients having brain metastasis are reported to have prostate adenocarcinoma as its primary tumour.1,2 Most commonly, the neurological presentations seen are non-focal resulting from raised intracranial tension and frontal lobe syndrome. Seizures are also very common. 3
Here, we present a case of an elderly gentleman with loss of vision as the initial presentation of metastatic prostate carcinoma who underwent orchidectomy and received docetaxel. His vision has slightly improved since the treatment.
Case presentation
A 66-year-old Indian gentleman, belonging to the upper-lower socioeconomic class, presented to the ophthalmology clinic of a tertiary care centre in Mumbai with chief complaints of headache and sudden onset of loss of vision. He denied any other ocular or neurologic symptoms. He had no history of trauma or any significant past medical history. The patient was evaluated further and computed tomography (CT) of his head revealed a destructive lesion involving the clivus and a space-occupying lesion (SOL) in occipito-parietal region of the brain with midline shift and cerebral oedema (Figures 1 and 2). The patient was admitted to the medical ward where, upon careful history, the patient revealed a slightly increased frequency of urination with nocturia about twice every night in the previous 6 months. He had also lost 10-pound weight over the past few weeks. He denied fever, haematuria, dysuria, straining or urinary incontinence. In view of urinary complaints, a urology reference was sought. A digital rectal examination was performed which revealed an enlarged hard nodular prostate. Laboratory testing demonstrated a serum prostate-specific antigen (PSA) level of 1321 ng/mL. The other laboratory parameters were – alkaline phosphatase of 1317 IU/L, serum creatinine of 1.23 mg/dL and haemoglobin of 6.7 g/dL. Prostate volume was 51 mL on transrectal ultrasonography. Random-guided biopsies of prostate were obtained. The microscopic examination of the core biopsies revealed a high-grade prostatic adenocarcinoma with Gleason score 10 (5 + 5). There was bilateral extensive and diffuse involvement of all cores from the apex, middle and base of the prostate (Figures 3 and 4).
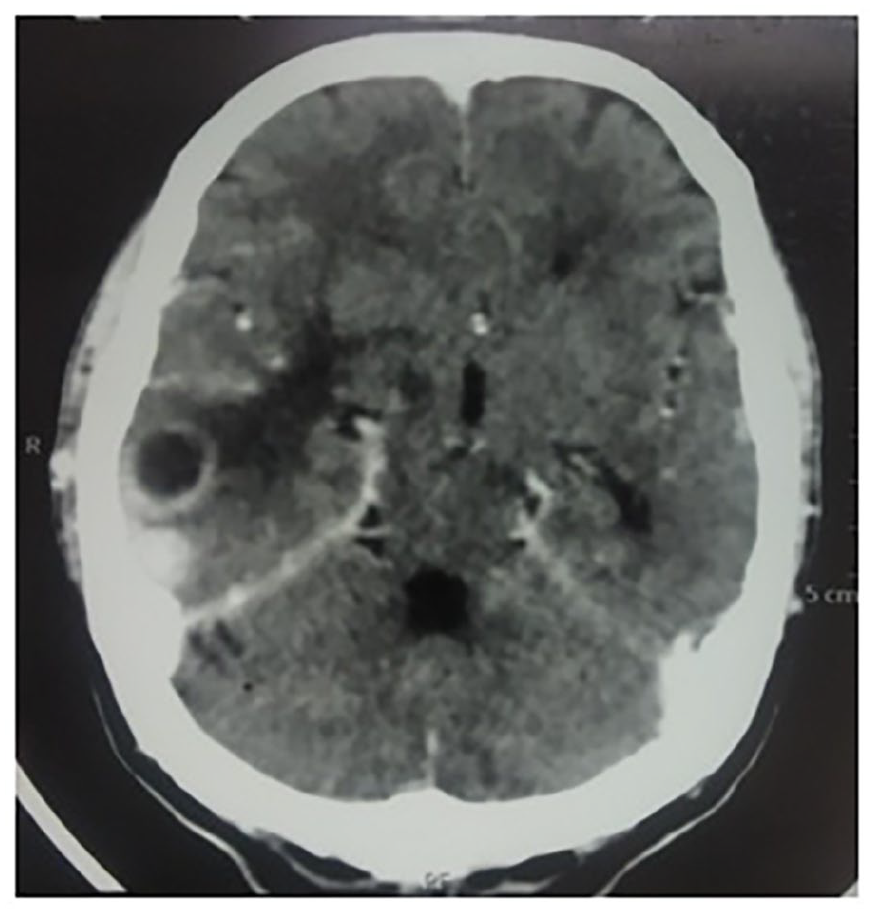
Axial section shows a peripheral enhancing cystic lesion in the right occipito-parietal region. An intensely enhancing smaller lesion is seen posterior to it with moderate perilesional oedema causing mild effacement of the third ventricle.
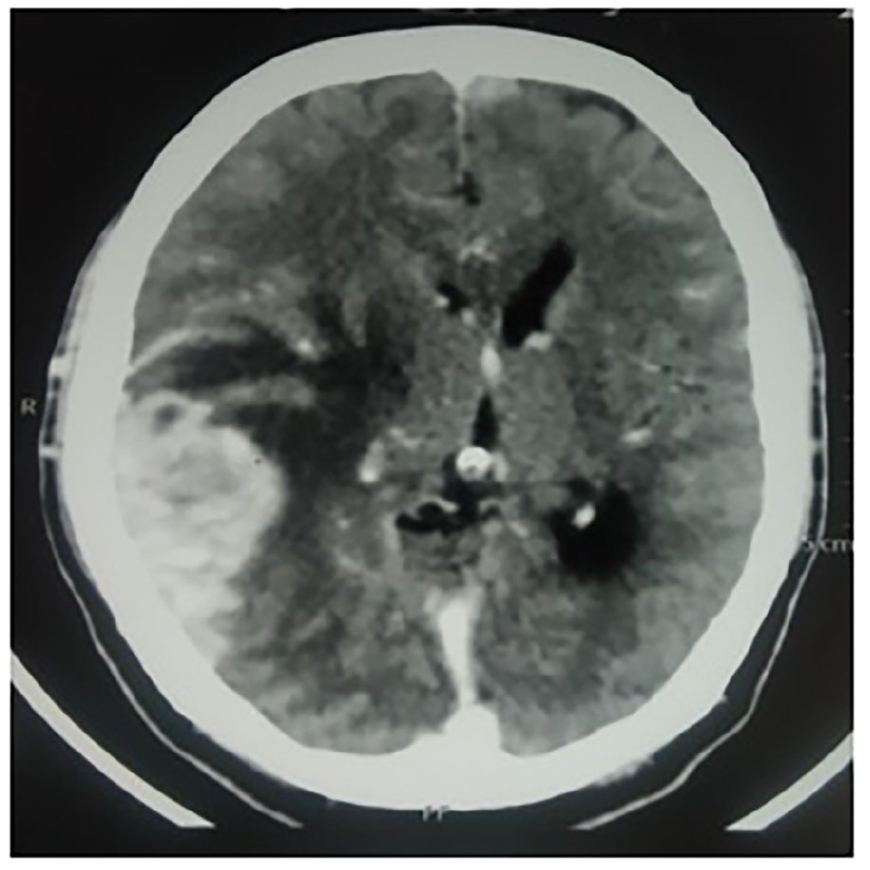
Axial section of the brain shows enhancing mass lesion in the right occipito-parietal region with moderate perilesional oedema causing effacement of the basal ganglia and right lateral ventricle, right thalamus and midline shift to the left.
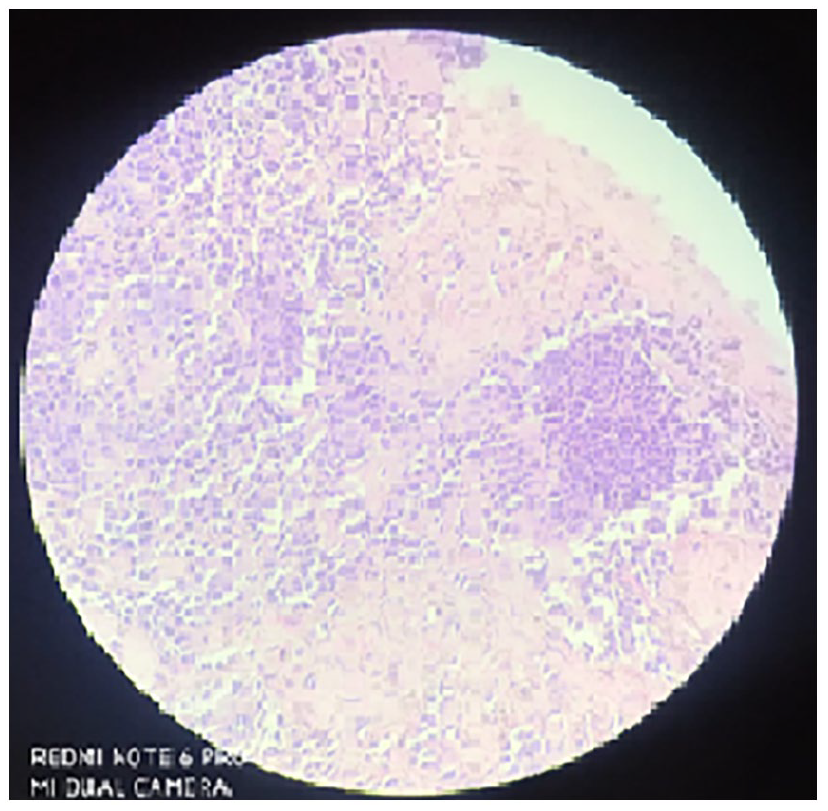
Transrectal ultrasonography (TRUS)-guided biopsy of left lobe of prostate apex showing adenocarcinoma with Gleason score of 8 (5 + 3) – Gleason group 4 with 30% of biopsy volume shows tumour.
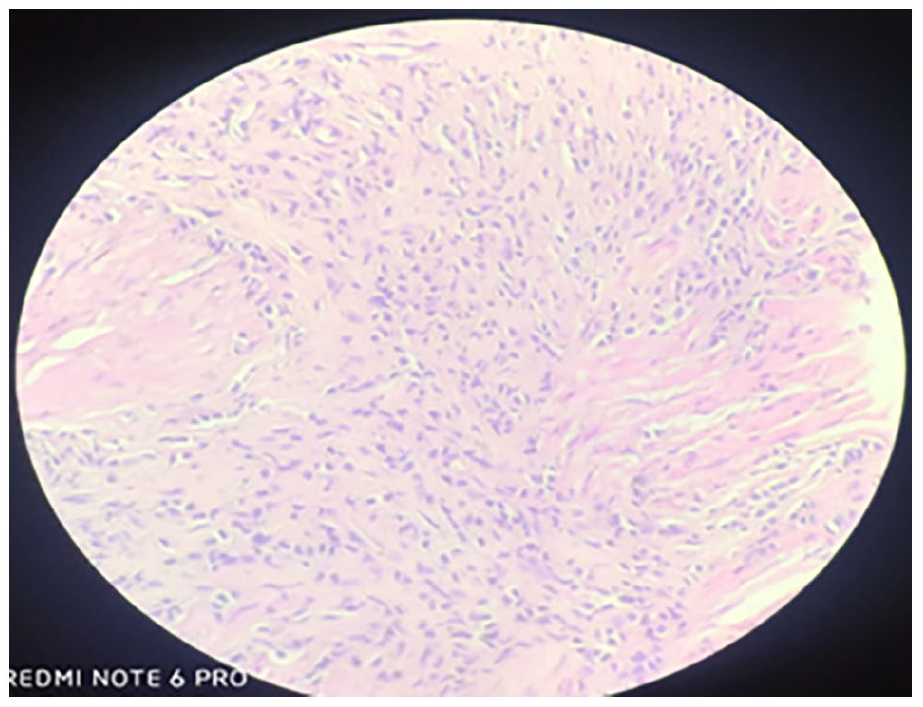
TRUS biopsy of right lobe of prostate base showing adenocarcinoma with Gleason score of 10 (5 + 5) – Gleason group 5 with 5–10% of biopsy volume shows tumour.
Metastatic workup was done for prostate cancer. Bone scan and CT chest and abdomen and pelvis which revealed heterogeneous enhancing lesion of the prostate extending into bilateral vesicoureteric junction, posterior and inferior wall of urinary bladder with a heterogeneous enhancement of bilateral para-aortic lymph nodes, adrenal and segment 4 of liver (Figures 5 and 6). Multiple peritoneal deposits were seen throughout the axial skeleton with right nodular pleural thickening.
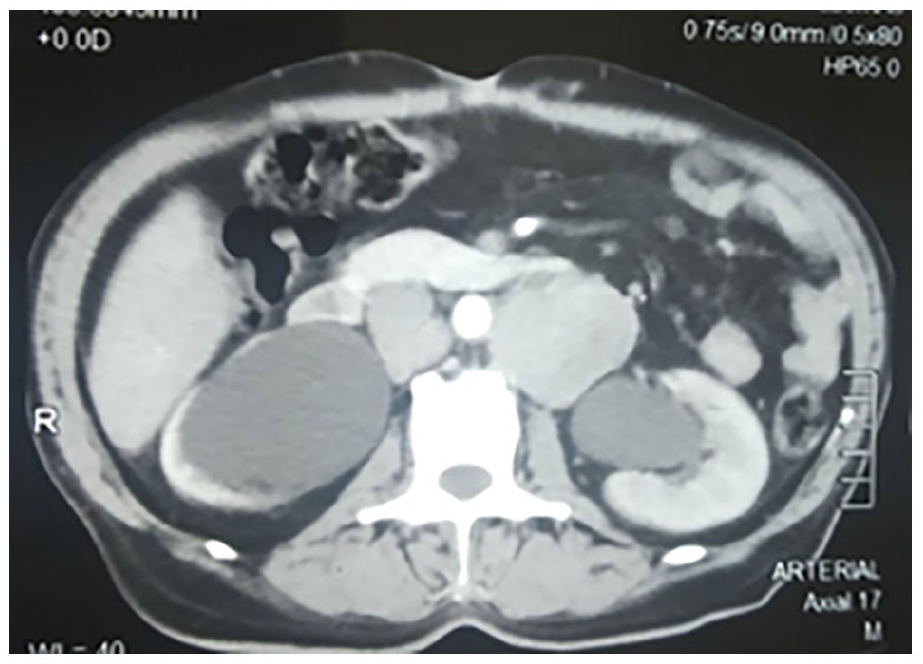
CECT shows severe dilation of bilateral renal pelvis with enhancing deposits in bilateral para-aortic regions.
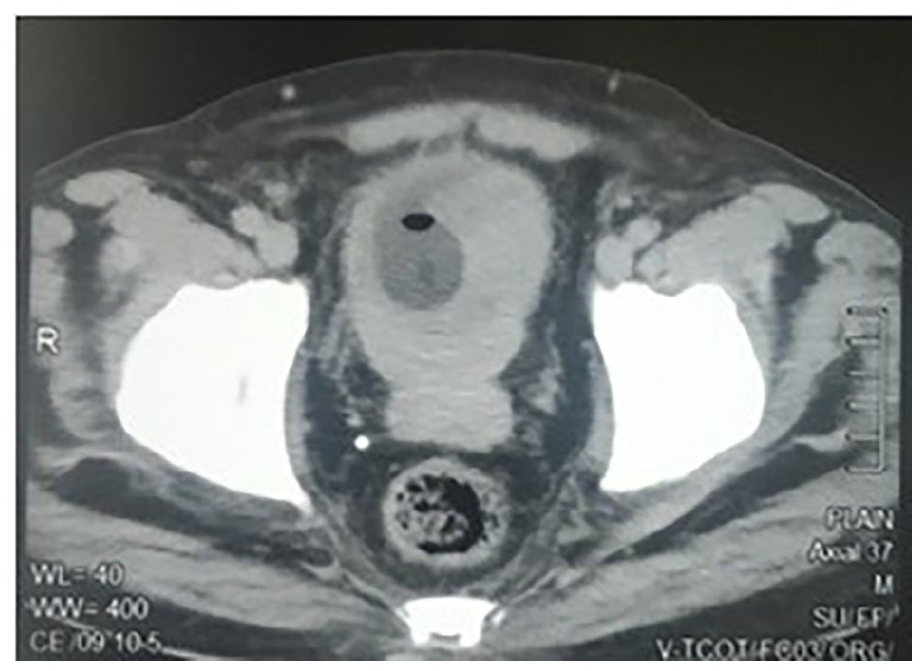
Axial section of prostate shows heterogeneous enhancing lesion extending into bilateral VUJ, posterior and inferior wall of urinary bladder.
The patient was counselled on medical versus surgical options of hormonal treatment and the patient opted for surgery. He subsequently underwent bilateral orchidectomy and bilateral percutaneous nephrostomy around 2 months after the first medical contact, the delay being due to the COVID-19 pandemic-related restrictions.
Initial ophthalmological examination showing bilateral papilloedema with flame-shaped haemorrhages and blurring of optic showed an improvement within 7 days of orchidectomy. At 4-month postoperative period, the patient gained projection of rays and perception of light.
The patient responded remarkably well and had normalisation of PSA after 6 months of therapy. The patient continues to be in remission at a follow-up of 12 months.
Discussion
Usually, after a median of 81 months from initial prostate cancer diagnosis, brain metastasis is diagnosed. Moreover, although some cases of intracranial metastasis as the first presentation have been reported, they generally present with non-focal signs and symptoms.1,3,4 Authors of a retrospective study of 7994 cases with intracranial metastasis found 38 cases from prostate cancer (0.7%). 5 The reported incidences of intracranial metastases from prostatic carcinoma at autopsy range from 0.6% to 4.4% and predominantly involve the leptomeninges (67%), followed by the cerebrum (25%) and cerebellum (8%).6,7 Clivus metastasis from prostate cancer presenting with nerve compression is a very rare entity, but it has been described previously. 8
A decrease in vision as the presenting symptom of prostate carcinoma has been reported in only five cases to date. Among them, Rao described intracranial metastasis causing mass effect at the cavernous sinus, leading to compression of cranial nerves II, III, IV, V and VI with minimal symptoms of bladder neck obstruction. Surgery and radiation therapy were the used modalities to manage the patient. 9 Kohri et al. reported a case involving frontal lobe mass with a left visual field defect. 10 Saito et al. reported a case in which diplopia was the chief complaint with hardly any complaints suggestive of bladder outlet obstruction. 11 The lesions in these two cases responded to hormonal therapy alone, with the resolution of the lesion demonstrated at a repeat CT and magnetic resonance imaging (MRI), respectively.10,11 An interesting cause of the diminution of vision was reported by Duncan et al. as retinopathy as a paraneoplastic manifestation of prostatic carcinoma. 12
The present case was similar to that described by Malloy, with prostate cancer metastasis to the clivus and presentation of deficit in eye abduction and sixth cranial nerve involvement. Neuroimaging in the Malloy case revealed metastasis to the clivus, involvement of the cavernous sinus and smaller metastasis to the left temporal lobe with extensive bony involvement in the vertebral column. 8 In our case, the CT scan of the skull showed destruction of the clivus and involvement of the Dorello canal, with multiple metastatic calvarium lesions without pituitary or cavernous sinus involvement. Furthermore, parieto-occipital SOL was also present. However, there was no compression of the cavernous sinus. Detailed scans revealed extensive bone metastases to the spine, ribs and pelvic bones, as well as extensive retroperitoneal and thoracic nodal disease, with involvement of the pleura and liver.
It has been postulated that metastasis to the brain either occurs by: (1) direct access through the paravertebral venous plexus, avoiding the bone and viscera; or (2) a multistep chronic process whereby the initial metastatic foci are the bone or lung, with secondary seeding of tumour cells to the brain.13,14 Our case can be explained by the latter postulation. Rapid progression of these steps with initial clivus destruction was thought to be the cause of the isolated sixth cranial nerve palsy.
The patient described by Malloy was treated with intracranial radiotherapy for visual symptoms. The patient’s visual symptoms responded, but within a year, he presented with widespread metastatic lesions in the entire vertebral column. 8 Interestingly, no recurrence of the intracranial lesion was noted. Our case presented with the neurologic deficit as the chief complaint despite multiple areas of widespread metastasis. The patient responded remarkably well, with complete recovery of his visual symptoms and PSA normalisation after 6 months of therapy. The patient continues in remission at a follow-up of 12 months.
Conclusion
Prostate cancer can occasionally present with neurologic manifestations, predominantly due to metastatic involvement of the skull bone. Awareness of this possibility could lead to accurate diagnosis, and the initiation of appropriate therapy can successfully reverse the neurologic deficits. With treatment, the response rates and prognosis are similar to other types of metastatic prostate cancer.
Footnotes
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent for publication has been obtained.
Guarantor
A.J. the guarantor of this study.
Contributorship
A.J. and S.P. were involved in active management of the case. A.N. did the literature search. A.N. and A.J. wrote the original draft. S.P. modified the case report. All authors have read and approved the manuscript submitted.
