Abstract
Background
Few data exist regarding the long-term effectiveness of golimumab in ulcerative colitis. No data have been reported on real-world continuous clinical response.
Objective
This study aimed to describe the long-term outcomes in a large cohort of patients on golimumab who had ulcerative colitis.
Methods
Consecutive patients with active ulcerative colitis, started on golimumab, were enrolled and prospectively followed up. The primary end point was to evaluate the long-term persistence on golimumab therapy.
Results
A total of 173 patients with ulcerative colitis were studied. Of these, 79.2% were steroid dependent, and 46.3% were naïve to anti-tumour necrosis factor alpha agents. The median duration of golimumab therapy was 52 weeks (range 4–142 weeks). The cumulative probability of maintaining golimumab treatment was 47.3% and 22.5% at 54 and 108 weeks, respectively. Biological-naïve status (odds ratio (OR) = 3.02, 95% confidence interval (CI) 1.44–6.29,
Conclusions
Biological-naïve status and not requiring steroids at weeks 8 and 14 seem to be associated with a longer persistence on golimumab therapy in ulcerative colitis.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory disease involving the colon, characterised by a relapsing/remitting course and requiring lifelong medical therapies. Biological drugs and, more recently, JAK inhibitors such as tofacitinib are the best medical option for patients with moderate-to-severe disease with an inadequate response or intolerance to conventional therapies (5-amynosalicilates, steroids and/or thiopurines). 1 Golimumab, a fully human IgG1 kappa monoclonal antibody, subcutaneously administered, has now been used in clinical practice for more than five years for the treatment of adult subjects with UC. 2 , 3 The efficacy of golimumab for the induction and maintenance of clinical remission in biological-naïve UC patients has been studied in two completed clinical trials: PURSUIT induction and PURSUIT maintenance. 4 , 5 In the second trial, a continuous clinical response (CCR) through week 54, that is, maintenance of a clinical response through week 54 among golimumab-induction responders, was adopted as the primary end point, and this achieved in 47.0% of patients receiving 50 mg golimumab and in 49.7% of receiving 100 mg golimumab compared to 31.2% receiving placebo. 5 Long-term open-label follow-up confirmed a good profile of effectiveness up to four years, more evident among patients with CCR at 54 weeks. 6 , 7 To date, few long-term real-life data have been reported, showing highly variable persistence on golimumab therapy in some cohorts, and particularly reduced in patients pluri-exposed to anti-tumour necrosis factor alpha (TNF-α) drugs and treated with the fixed dose of 50 mg during maintenance therapy.8–12
The aims of this study were to investigate the mid- and long-term outcomes of patients with UC treated with golimumab in real life and to explore potential predictors for these outcomes.
Methods
We performed an observational retrospective/prospective study in which consecutive patients who started golimumab therapy between May 2014 and December 2015 at 29 Italian centres, affiliated with the Italian Group for the study of Inflammatory Bowel Disease (IG-IBD), were enrolled. All patients had a prospectively designed standardised follow-up until December 2017.
In Italy, to guarantee the prescribing appropriateness, the Italian Medicine Agency (Agenzia Italiana del Farmaco (AIFA)) has instituted a computerised database system for several drugs, including golimumab, accessible to physicians and mandatory to finalise the prescription both at the beginning of and during maintenance treatment. Therefore, accessibility criteria and follow-up visits scheduled every eight weeks, requiring a clinical assessment through partial Mayo Score (PMS), 13 are standardised for all patients on treatment with golimumab. Accordingly, we adopted a prospectively planned follow-up protocol, with a shared common database mirroring the AIFA registry, to enrol patients and to follow them up until December 2017.
According to the current European-approved golimumab label, 2 all patients received golimumab induction with 200 and 100 mg at weeks 0 and 2, respectively, followed by 50 or 100 mg every four weeks, depending on their weight (≥80 kg or <80 kg). Patients were not allowed to increase the dose in case of partial response after the induction or loss of response.
The collected baseline data included: sex, age, weight, height, body mass index (BMI), duration of UC, extension of UC according to the Montreal classification, 14 clinical and endoscopic activity, previous therapies (both conventional and biological), the date of the first golimumab dose and concomitant therapies. Baseline and follow-up clinical and endoscopic activities were determined according to PMS and endoscopic sub-score, respectively. 13 Concomitant medications, new prescriptions during follow-up, the tapering of steroids and timing of treatment discontinuation were left to the investigators’ evaluation.
The primary end point of our study was to evaluate the long-term persistence on golimumab therapy due to sustained clinical benefit.
Secondary analyses looked for (a) proportion of patients achieving clinical remission at week 54; (b) CCR through week 54 among patients with a clinical response after induction; (c) rate of surgery for medical refractory UC; (d) effectiveness of treatment in sparing steroids among patients taking steroids at baseline; and (e) proportion of patients achieving endoscopic remission.
A clinical response was defined as a reduction in the PMS of at least two points and a decrease of at least 30% from the baseline score, with a decrease of at least one point on the rectal bleeding subscale or an absolute rectal bleeding score of 1 or 0. Clinical remission was defined as a PMS of two or lower and no sub-score higher than one. We adopted the same definition of CCR through week 54 previously reported, even though the interval between each clinical assessment was set every eight weeks . 5 Endoscopic examinations were mandatory at week 54, but could be anticipated according to clinical judgement. Endoscopic remission was defined as an endoscopic Mayo sub-score of 0 or 1. For patients undergoing two or more endoscopic assessments during the study, the last one was considered for the evaluation of endoscopic remission.
Reasons for golimumab discontinuation were categorised as: primary failure, defined as the absence of a clinical response at week 8; secondary failure, defined as a relapse of clinical symptoms during maintenance treatment requiring physicians’ interventions; and others, including intolerance or adverse events, lost to follow-up and pregnancy.
All adverse events that occurred from the beginning of golimumab treatment to the date of withdrawal or last follow-up visit on therapy were recorded and categorised as adverse events of interest (AEI) if requiring medical intervention/hospitalisation and/or treatment discontinuation (temporary or permanent).
Statistical analysis
Data were described using means with standard deviation (
Ethical considerations
The protocol was approved by the ethics committee of the coordinator centre (Fondazione Policlinico Universitario A. Gemelli IRCCS-Università Cattolica del Sacro Cuore, Roma, Italy, protocol 1462, 26/01/2017) and of all participating centres. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution's human research committee. Written informed consent was obtained from each patient included in the study.
Results
Patient population
A total of 173 patients were included. Baseline patients’ characteristics are summarised in Table 1. According to AIFA eligibility criteria, all patients had moderate to severe active disease, and all of them had showed an inadequate response or had a contraindication to steroids. In particular, 137 (79.2%) patients were steroid dependent, and 27 (15.6%) were refractory according to IG-IBD definitions. 15 The remaining nine patients had contraindications to steroid therapy. At baseline, 60 (34.7%) patients were on concomitant steroid therapy; 52 (30.1%) and 36 (20.8%) were taking oral steroids at weeks 8 and 14, respectively. A total of 131 (75.7%) patients weighed <80 kg and thus received 50 mg every four weeks as a maintenance dose; the remaining 42 (24.2%) weighed ≥80 kg and thus received 100 mg every four weeks. A total of 111 (64.2%) patients had been previously exposed to thiopurines, and 92 (53.2%) patients had been previously exposed to at least one anti-TNF agent: 52 (30.1%) to infliximab, six (3.5%) to adalimumab and 34 (19.7%) to both.
Baseline patient characteristics.
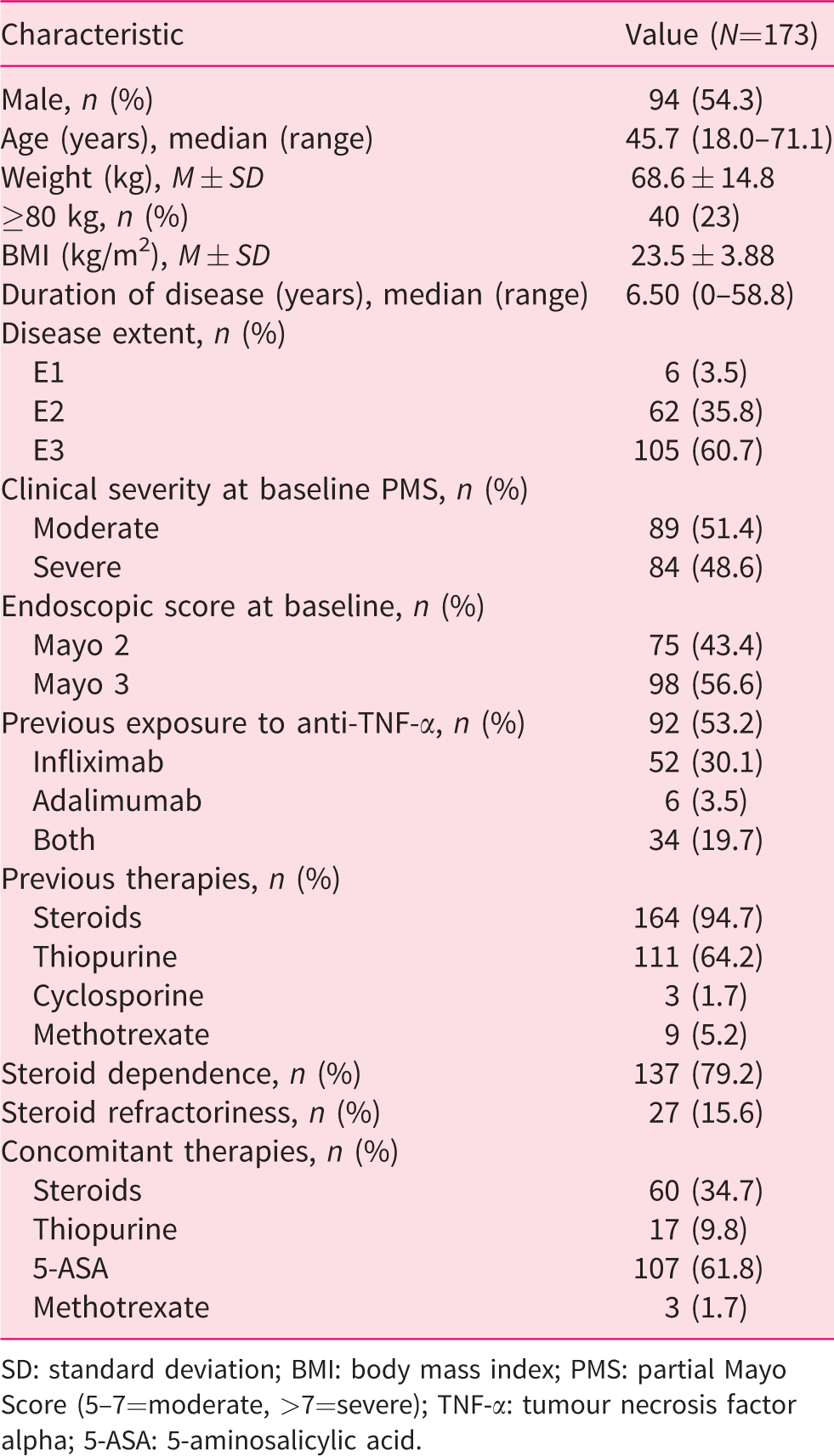
SD: standard deviation; BMI: body mass index; PMS: partial Mayo Score (5–7=moderate, >7=severe); TNF-α: tumour necrosis factor alpha; 5-ASA: 5-aminosalicylic acid.
Persistency on golimumab therapy
The median time on golimumab treatment was 52 weeks (range 4–142 weeks). The cumulative probability of maintaining golimumab treatment was 47.3% and 22.5% at 54 and 108 weeks, respectively (Figure 1). Overall, 126 (72.8%) patients withdrew from golimumab therapy after a median of 31.5 weeks (range 4–126 weeks). Reasons for discontinuation were primary failure in 51 (40.5%) patients, secondary failure in 51 (40.5%) patients and other causes in 24 (19.1%) patients. Among the 102 patients who withdrew from treatment due to failure, 65 (63.7%) were anti-TNF-α experienced compared to 37 (36.3%) who were naïve (
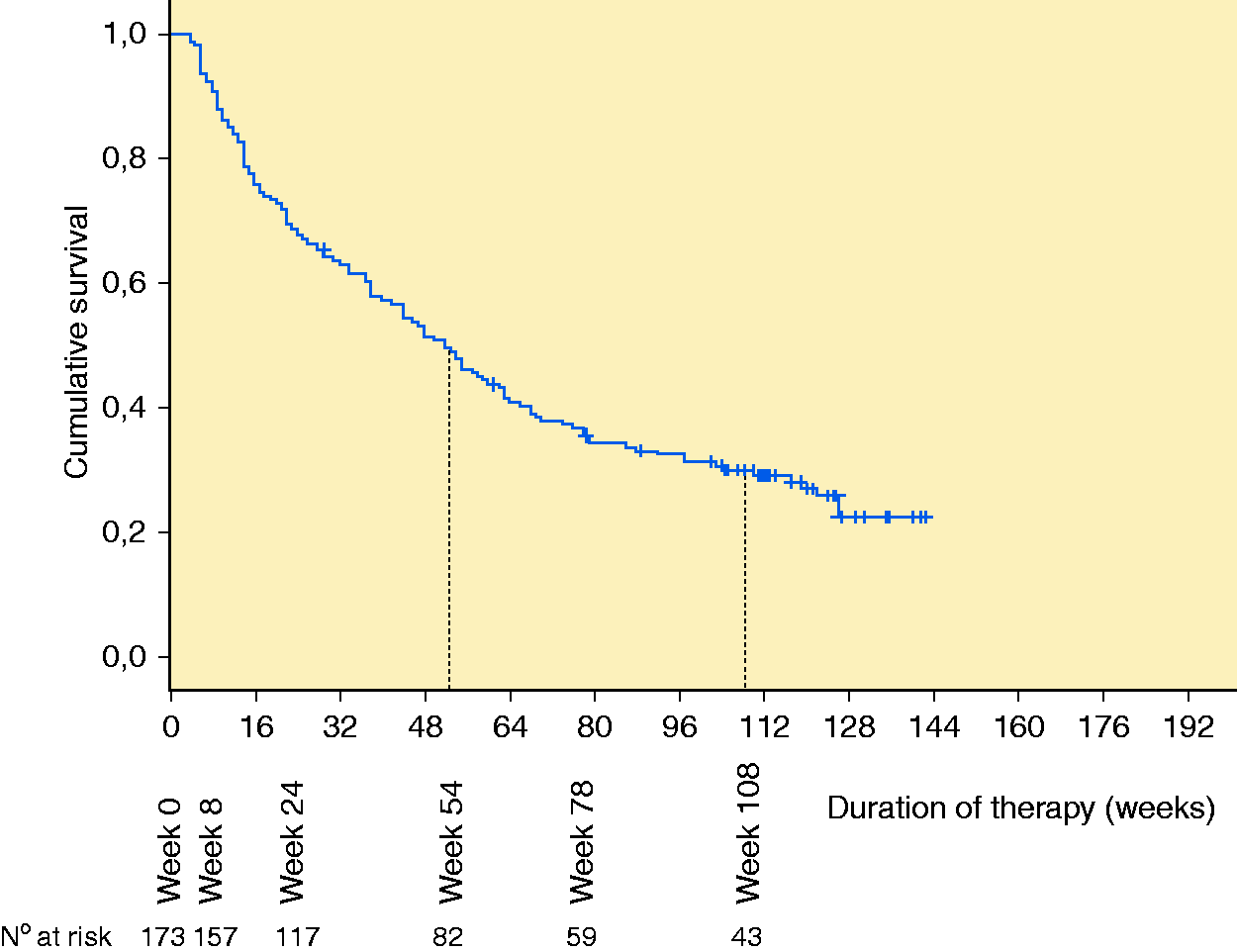
Cumulative probability of maintaining golimumab treatment.
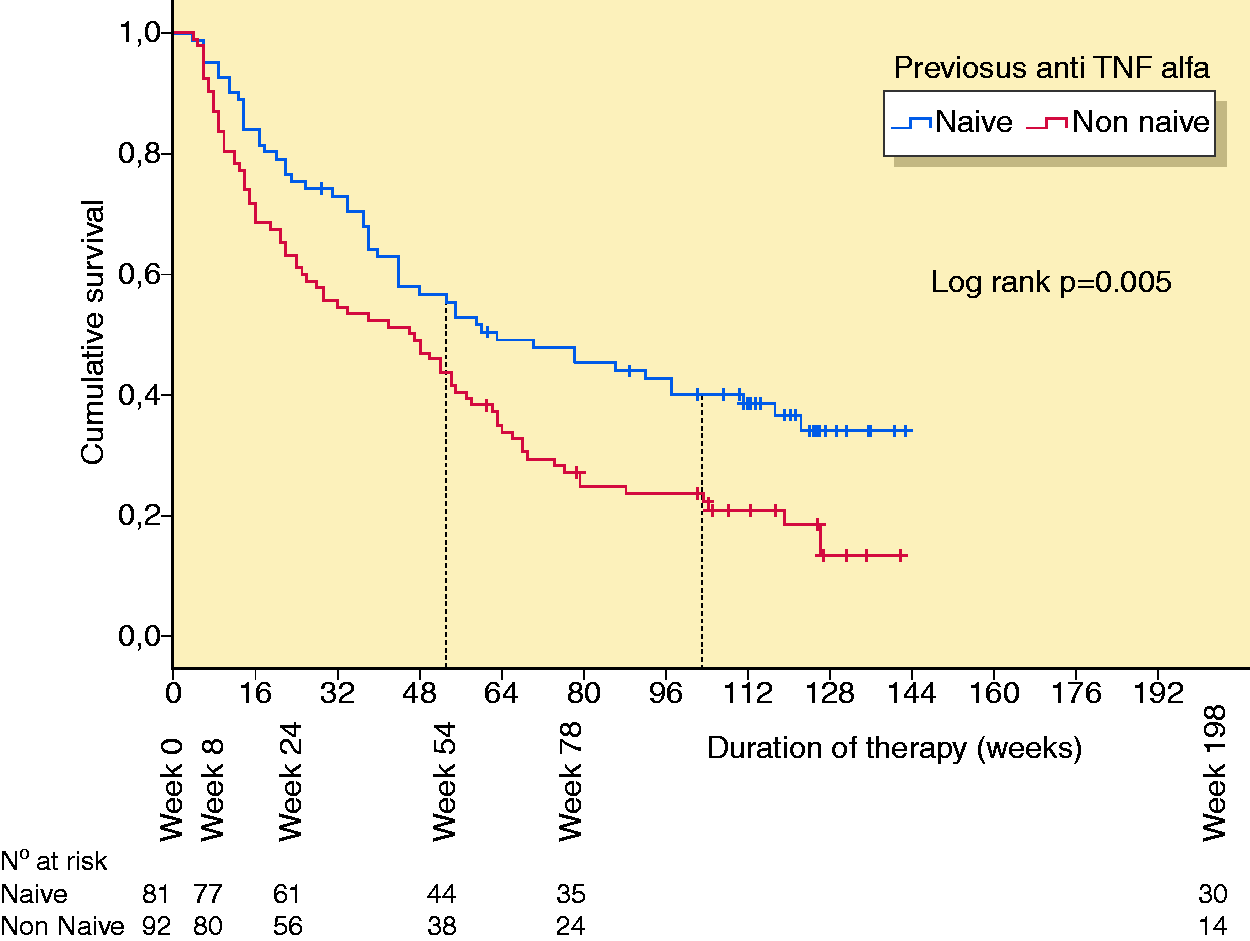
Cumulative probability of maintaining golimumab treatment. Patients split between those who were anti-tumour necrosis factor alpha naïve and those who were anti-tumour necrosis factor alpha experienced.
Results of binary logistic regression for persistence on golimumab therapy in 173 UC patients.
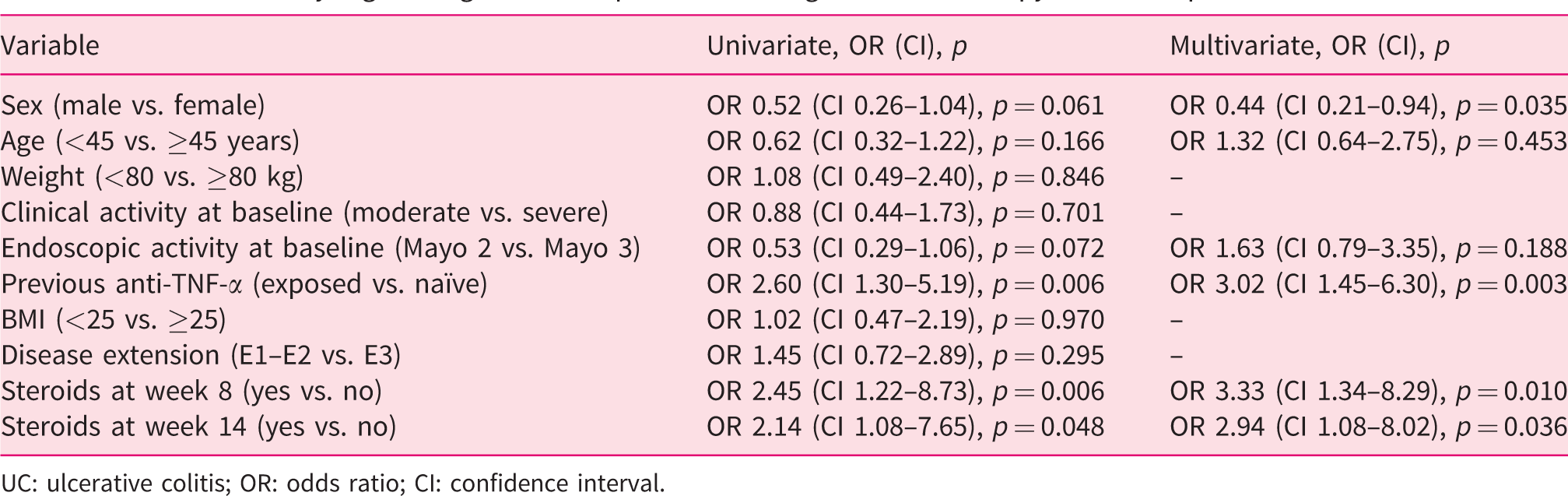
UC: ulcerative colitis; OR: odds ratio; CI: confidence interval.
Secondary outcomes
Among 124 patients in clinical response after induction, 65 (52.4%) maintained CCR through week 54. Clinical remission at week 54 was recorded in 40 (23.1%) patients. Among the 83 patients still on therapy after one year, CCR through week 54 was associated with a lower likelihood of golimumab discontinuation throughout the subsequent year of therapy (23% with CCR vs. 61% without CCR,
Twenty-two (12.7%) patients underwent total colectomy due to medical refractoriness after a median time of 28 weeks (range 11–92 weeks) from golimumab initiation. Of these, 20 (90.9%) were anti-TNF-α experienced. Sixty (34.7%) patients were taking steroids at baseline: 36 (60%) were able to withdraw corticosteroids within 30 weeks. Among the remaining 24 patients, 21 (87.5%) withdrew from golimumab therapy during follow-up.
At least one follow-up endoscopy was performed in 119 (68.8%) patients after a median of 54 weeks (range 8–122 weeks) from starting golimumab. Endoscopic remission was reported in 44/119 (36.9%) patients.
Golimumab safety
Twenty-six AEI were reported by 21 (12.1%) patients. The most frequent AEI were infections (eight patients, 4.6%). Four patients had respiratory infections, one patient had acute gastroenteritis and one patient had genitourinary infection. Two patients experienced opportunistic infections: one experienced cytomegalovirus reactivation, and another was diagnosed with oropharyngeal candidiasis. The last two patients were on concomitant steroid therapy. Six (3.4%) patients developed skin manifestations (two psoriasis and four eczematous dermatitis). Four patients showed allergic reactions: one reaction at the injection site, and three diffuse skin rashes. One patient was diagnosed with oral condyloma, and one with basal-cell carcinoma. Sixteen patients discontinued golimumab due to an AEI: five infections (three respiratory, one genitourinary and one candidiasis), six skin manifestation, four allergic reactions and one basal-cell carcinoma.
Discussion
This study focused on the long-term clinical effectiveness and safety of a large cohort of 173 patients with moderate to severe active UC treated with golimumab. Most of our patients (60.7%) had extensive colitis, and more than a half (53.2%) had already been exposed to at least one anti-TNF-α agent. In our cohort, the median follow-up on golimumab therapy was 52 weeks (range 4–142 weeks), and the cumulative probability of maintaining golimumab treatment due to sustained clinical benefit was 47.3% and 22.5% at 54 and 108 weeks, respectively. These figures are different from other real-world experiences, showing around up to 60% of persistence at week 54. 8 , 11 However, the higher frequency of golimumab discontinuation in our study could be partially explained by the impossibility of escalating to 100 mg early in patients with a primary non-response or partial response during the maintenance phase. Most of our patients (75.7%) were in fact maintained with golimumab 50 mg because of their weight (<80 kg). We recorded a primary failure rate of up to 40.5% and 30% golimumab withdrawal within the first 14 weeks.
A post hoc analysis of the PURSUIT trial showed that up to 28.1% of week 6 non-responders who were escalated early to golimumab 100 mg achieved a clinical response at week 14. Moreover, after one year, these late responders achieved similar clinical and endoscopic outcomes compared to early responders. Pharmacokinetic data showed that early week 6 non-responders had half the golimumab serum concentrations compared to early week 6 responders. 16 Indeed, in their recent work, Magro et al. found that week 6 golimumab serum levels were positively correlated with clinical, endoscopic and histological remission, thus reinforcing the idea that early dose escalation could reduce the rates of primary non-response. 17
In our cohort, naive patients were more likely to maintain golimumab therapy because of a sustained clinical benefit compared to anti-TNF-α exposed patients. It should be noted that in about 37% of patients who were anti-TNF-α experienced, golimumab was used as a third-line treatment after failure of infliximab and adalimumab. This situation has already been shown to be associated with a worse outcome compared to first- or second-line utilisation. 8 Therefore, the use of golimumab should be advised at most after the failure of first-line TNF-α therapy. Therapeutic drug monitoring could help physicians to determine the most suitable therapeutic option in case of a loss of response to anti-TNF-α drugs, including switching within the class for patients with a high titre of neutralising anti-drug antibodies or, conversely, out of class for patients with a ‘pharmacodynamics escape’ (trough levels within the therapeutic range with negative anti-drug antibodies). 18
Most patients (79.2%) included in our study were steroid dependent. For such patients, golimumab was expected to provide a clinical improvement by exerting a steroid-sparing effect as well. Among those who were taking steroids at baseline, the inability to discontinue them after 8 and 14 weeks of golimumab therapy was indeed associated with a higher rate of treatment discontinuation. Accordingly, we might suggest that in clinical practice, patients on golimumab therapy who still need steroids after two to three months or, similarly, require an early reintroduction should be revaluated for a therapeutic change.
CCR through week 54 was observed in 65 (52.4%) patients comparable to those reported in the clinical trial. 5 Achieving CCR was associated with a higher rate of long-term persistence on golimumab therapy. Moreover, none of the CCR patients underwent colectomy in the subsequent year.
The outcome of CCR, introduced for the first time in the PURSUIT study, also represents a potential goal for the treatment of UC patients in clinical practice, since it is based on the concept of tight monitoring of patients and of targeting continuous disease control. 19 Even though the evidence supporting that uncontrolled inflammation causes structural bowel damages are limited in comparison with Crohn’s disease, 20 UC shows features of a progressive disease, including the proximal extension and the developing of structuring or functional disorders. 21 , 22
Finally, the overall safety profile of golimumab was confirmed to be good, consistent with those reported in other real-life experiences and of other anti-TNF-alpha drugs.8–12, 16 No new safety concerns about golimumab emerged during our two years of follow-up.
Our study has some limitations: as described above, including the impossibility of adapting the dose in patients with a partial or lack of response, but also a lack of data on inflammation markers (e.g. C-reactive protein, faecal calprotectin). Conversely, the strengths of our study are the follow-up of up to two years (median 52 weeks, range 4–142 weeks), predefined standardised intervals between each clinical visit and homogeneous assessments of clinical and endoscopic activities. Moreover, we reported, for the first time to our knowledge, data on CCR in the real-life setting and its correlation with a more favourable long-term outcome.
In conclusion, golimumab may be considered as an effective and safe treatment option in UC patients, with higher rate of retention in therapy for biological-naïve patients and for those who are able to discontinue steroids early. CCR could potentially represent a target to pursue in clinical practice in order to improve disease control.
Footnotes
Acknowledgements
Ennio Sarli provided statistical consulting.
Declaration of conflicting interests
The authors declare the following conflicts of interest: D.P. received speaker fees from AbbVie, MSD, Takeda, Janssen and Pfeizer. G.P. received consultancies fees from Alphasigma. M.A. received consulting fees from Nikkiso Europe and lecture fees from Janssen, Abbvie and Pfizer. M.C. served as an advisory board member for AbbVie, MSD and Takeda Pharmaceuticals, and received lecture grants from AbbVie, MSD, Chiesi and Takeda Pharmaceuticals. M.D. received lectures, board and/or congress fees from Abbvie, Pfizer, Takeda, Mundipharma, Janssen, MS&D, SOFAR, Ferring and Chiesi. M.D.G. received speaker fees from Abbvie. F.R. acted as consultant for Janssen, Abbvie, Takeda, MSD and Amgen, and participated in a speaker’s bureau sponsored by Abbvie, Janssen, Takeda, Ferring, MSD, Sofar and Chiesi. A.A. received consulting and/or advisory board fees from AbbVie, Allergan, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Celltrion, Ferring, Janssen, Lilly, MSD, Mylan, Pfizer, Samsung Bioepis, Sandoz and Takeda; lecture and/or speaker bureau fees from AbbVie, Amgen, Biogen, Ferring, Janssen, MSD, Mitsubishi-Tanabe, Nikkiso, Pfizer, Sandoz, Samsung Bioepis and Takeda; and research grants from MSD, Pfizer and Takeda. All the other authors have no conflict of interest to declare.
Ethics approval
The protocol was approved by the ethics committee of the coordinator centre (Fondazione Policlinico Universitario A. Gemelli IRCCS-Università Cattolica del Sacro Cuore, Roma, Italy, protocol 1462, 26/01/2017) and of all participating centres. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from each patient included in the study.
