Abstract
Background
Fecal incontinence is a disabling condition affecting up to 20% of women.
Objective
We investigated fecal incontinence assessment in both inflammatory bowel disease and non-inflammatory bowel disease patients to propose a diagnostic approach for inflammatory bowel disease trials.
Methods
We searched on Pubmed, Embase and Cochrane Library for all studies on adult inflammatory bowel disease and non-inflammatory bowel disease patients reporting data on fecal incontinence assessment from January 2009 to December 2019.
Results
In total, 328 studies were included; 306 studies enrolled non-inflammatory bowel disease patients and 22 studies enrolled inflammatory bowel disease patients. In non-inflammatory bowel disease trials the most used tools were the Wexner score, fecal incontinence quality of life questionnaire, Vaizey score and fecal incontinence severity index (in 187, 91, 62 and 33 studies). Anal manometry was adopted in 41.2% and endoanal ultrasonography in 34.0% of the studies. In 142 studies (46.4%) fecal incontinence evaluation was performed with a single instrument, while in 64 (20.9%) and 100 (32.7%) studies two or more instruments were used. In inflammatory bowel disease studies the Wexner score, Vaizey score and inflammatory bowel disease quality of life questionnaire were the most commonly adopted tools (in five (22.7%), five (22.7%) and four (18.2%) studies). Anal manometry and endoanal ultrasonography were performed in 45.4% and 18.2% of the studies.
Conclusion
Based on prior validation and experience, we propose to use the Wexner score as the first step for fecal incontinence assessment in inflammatory bowel disease trials. Anal manometry and/or endoanal ultrasonography should be taken into account in the case of positive questionnaires.
Introduction
Fecal incontinence (FI) is defined as the involuntary loss of liquid or solid stool. 1 It represents not only an embarrassing and humiliating symptom for patients, but also a serious social problem impacting considerably on everyday life. 2 FI was traditionally thought to affect about 2–10% of the general population and to increase in incidence with age.3,4 However, more recent population-based studies5–7 have revealed an incidence of 18.8% in adult women in the USA. Although 18.8% might seem a high prevalence, in patients with inflammatory bowel disease (IBD) FI occurs in an even higher percentage of patients. 8 A study by the British National Crohn’s and Colitis UK Organization 8 including over 3000 subjects showed that 74% of IBD patients reported at least one episode of FI in their life. The increased rate of FI in IBD patients is probably due to predisposing factors such as perianal disease, invasive perianal surgical approach, liquid stools and secretory diarrhoea caused by bile acid malabsorption.9–12 The pathophysiology of FI is very complex and may depend on different structures, including pelvic floor, anal sphincter muscles and both the voluntary and autonomous nervous system. 13 Furthermore, the diagnosis of FI is not easy as patients are reluctant to report this embarrassing symptom, treating physicians often do not investigate it, and a clear and validated approach to diagnose it is lacking.5–7,14 A combination of different tests may be necessary to diagnose FI and to attribute the incontinence to a specific aetiology. Available options include self-reported questionnaires, anorectal manometry, electromyography (EMG), defecography, endoanal ultrasonography, magnetic resonance imaging (MRI), computed tomography (CT) and pudendal nerve terminal motor latency (PNTML) assessment. 15 Tools such as the validated Wexner score, 15 Vaizey score 16 and fecal incontinence quality of life (FIQL) questionnaire 17 assess FI measuring severity and impact on quality of life. Anorectal manometry allows us to measure resting and squeeze pressure, duration of the voluntary contraction and length of the anal canal. 18 Endoanal ultrasonography is recommended to investigate sphincteric lesions as a preoperative assessment, while physiology tests and other imaging procedures provide additional anatomical and functional data. 18 A correct FI evaluation is essential to obtain an early diagnosis of disease and to ensure a rapid treatment. Our aim was to investigate FI assessment in both patients with or without IBD in order to clarify the diagnostic approach for this disabling condition and to propose an algorithm for IBD trials.
Methods
This systematic review has been developed according to the guidance specified in the Cochrane Handbook and the preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines.19,20
Search strategy
The review consists of two parts: FI assessment in non-IBD patients and FI assessment in IBD patients. We searched for published studies on Pubmed, Embase and the Cochrane Library starting from January 2009 to December 2019 in order to investigate the literature evidence of the past 10 years. The following MESH terms alone or matched to ‘IBD’, ‘inflammatory bowel disease’, ‘UC’, ‘ulcerative colitis’, ‘CD’ and ‘Crohn’s disease’ were used: ‘fecal incontinence’, ‘anal incontinence’, ‘assessment’, ‘evaluation’, ‘diagnosis’, ‘tool’ and ‘investigation’. All studies on adult patients reporting data on FI assessment were included in our review. Reviews, meta-analyses, guidelines and editorials were excluded. Our search focused on full-text articles published in English, but relevant abstracts were evaluated. The careful analysis of the reference lists of the included studies allowed us to identify additional studies.
Selection process and data extraction
Two authors (FD and CG) independently screened all titles and removed duplicates and non-fitting papers. Subsequently the articles that met the inclusion criteria were evaluated by two authors (FD and CG) to define their eligibility. Any disagreement between investigators was discussed with the co-authors (SD and LPB) until consensus was reached. Two reviewers (FD and CG) extracted the following data from each study: name of the first author, publication year, study design, field of research (e.g. gastroenterology, surgery, gynaecology, etc.), patient cohort (number, sex, mean age, type of bowel disease) and FI assessment (diagnostic tools, used investigations). In the case of non-specified data, we indicated it as ‘NS’.
Quality of studies
The Newcastle–Ottawa Scale (NOS) score 21 was used to measure the quality of non-randomised studies, while in randomised clinical trials the Jadad score 22 was adopted. High quality studies were defined as NOS score of 6 or greater or Jadad score of 3 or greater, respectively. Two authors (FD and CG) graded the studies independently and disagreements were discussed with a third author until their resolution.
Results
Search results
The flow chart of the search process is shown in Figure 1. Patient demographics and main characteristics of non-IBD and IBD studies are reported in Tables 1 and 2, respectively. A total of 1727 articles were investigated (Pubmed: 1022; Embase: 426; Cochrane Library: 279). Three hundred and fifty-five papers were eligible for inclusion after the elimination of duplicates and screening of all titles. After careful evaluation of full-text papers 27 studies were excluded: 15 studies23–37 did not evaluate FI assessment, six manuscripts38–43 were just protocols, five were paediatric studies,44–48 and one trial 49 had an inappropriate study design. Finally, 328 studies were included in our review: 306 studies50–355 on non-IBD patients and 22 studies on IBD patients.8,356–376 Overall, 102,460 patients were enrolled in non-IBD studies (66.7% women) and 8289 in IBD studies (63.9% women), with the number of patients per study ranging from six to 47,714. Among non-IBD trials there were 137 prospective cohort studies,48,50–53,58,59,61,62,64,68–72, 74–76,79–81,93,103,104,107,110–113,119,120,124–126,128,131,135,137, 142–145,149,151,153,157–163,166,168,169,171–174,176–182,187,190,192, 197,198,202,204,207,211–214,216,218,219,221,222,224,225,231,234,235, 238,240,242,244,245,247–251,253,259,261–263,267,269,273,275,277,280, 281,285,288,290–293,298–300,303,304,306,308,312,315,317,323,325,327–331,333,340,343,346 78 retrospective cohort studies,51,57, 69,75,84,88,90–93,98,99,107,108,110,116,117,120,124,129,131,134,138, 140,141,148,150,152,154,158,167,169,177,185–188,190,193,198,201,205, 210,211,219,229,230,239,243,245,254,256,258,259,262,274,276,278,280,281, 285,286,288,289, 291, 299,311,315,337,339–341,344, 346,347,349,350, 355 76 randomised clinical trials,58,65,67,68,79,80,85–87,96,97,100–104,111,118,119,123,125,132,135,136,142,143,156,157,166,172,191,195–197, 203,207,212,217,225,228,231,232,234,235,238, 241,248,257,260,267,268,270, 272,273,284,296–298,303,304,307,309,312,321–324,328,334,336,338,343, 351–354 nine cross-sectional56,59,62,89,222,266,316,318,320 and six case–control studies.94,149,202,208,313,326 In IBD trials, instead, there were 11 retrospective cohort studies,8, 359,360,362,365–368,371,372,374 10 prospective cohort studies356–358,361,363,364,370,373,375,376 and one cross-sectional study. 369 Overall, studies on FI involved 16 different research areas: the most frequent were surgery, gynaecology and gastroenterology (179,51,52,54,59–64,66–73,75,80, 81,83,84,88,89,97–99,105,106,109,110,114,115,120,124–127,129–132,135–139, 141,148,154–156,160–163,165–167,170,174–181,183,184,188,193–196,198–208, 210–215,219,223–229,234–238,240–245,247–253,256,258–260,262–264,266, 267,269–273,275,276,278,279,282,284–286,292,295–297,299,302–308,310, 311,314–316,319,321,324,327–330,332–336,340,341,344,345,349,350,352–355 45,53,56,79,93,112,113,116–118,121,133,140,142–144,146,147,149, 151–153,169,171,173,182,186,187,222,254,255,283,287–291,293,298,312, 313, 317,318,325,331,342 and 32 studies,50,65,76–78,82,90–92,94–96,103, 107,122,123,172,216–218,257,277,280,281,301,309,338,343,347,348,362,376 respectively). Overall, 172/252 non-randomised studies (68.2%) had a NOS score of 6 or greater and 51/76 randomised studies (67.1%) had a Jadad score of 3 or greater (see Supplementary Tables 1 and 2).
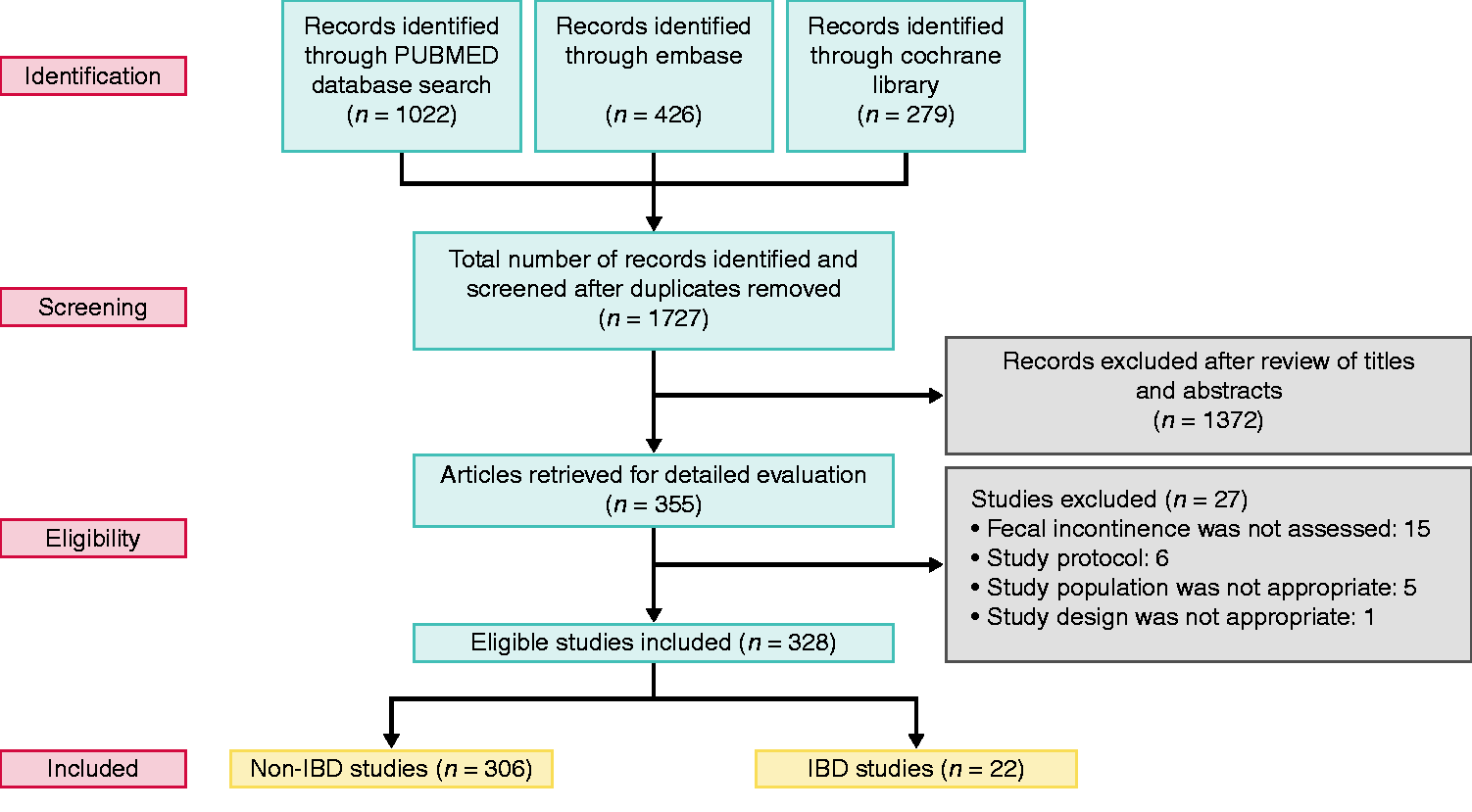
Flow chart of the search process.
Fecal incontinence assessment in non-IBD patients.
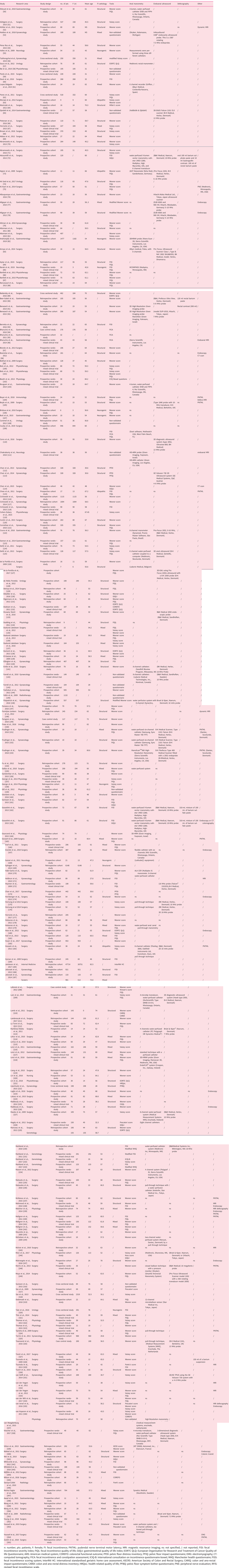
n: number; pts: patients; F: female; FI: fecal incontinence; PNTML: pudendal nerve terminal motor latency; MRI: magnetic resonance imaging; ns: not specified; /: not reported; FISI: fecal incontinence severity index; FIQL: fecal incontinence quality of life; GIQLI: gastrointestinal quality of life index; EORTC QLQ: European Organisation for Research and Treatment of Cancer Quality-of-life Questionnaire; AMS: American medical system; COREFO: colorectal functional outcome; PFDI: pelvic floor distress inventory; PFIQ: pelvic floor impact questionnaire; EMG: electromyography; CT: computed tomography; FICA: fecal incontinence and constipation assessment; ICIQ-B: international consultation on incontinence questionnaire-bowel; MHQ: Manchester health questionnaire; FISS: fecal incontinence scoring system; InterRAI HC: international standardised geriatric home care assessment; ASCRS: American Society of Colon and Rectal Surgery; CARIQ: colon and ano-rectal impact questionnaire; CARDI: colon and ano-rectal distress inventory; HRQoL: health-related quality of life; PFBQ: pelvic floor symptom bother questionnaire; PNE: peripheral nerve evaluation; RFIS: revised fecal incontinence scale.
Fecal incontinence assessment in IBD patients.
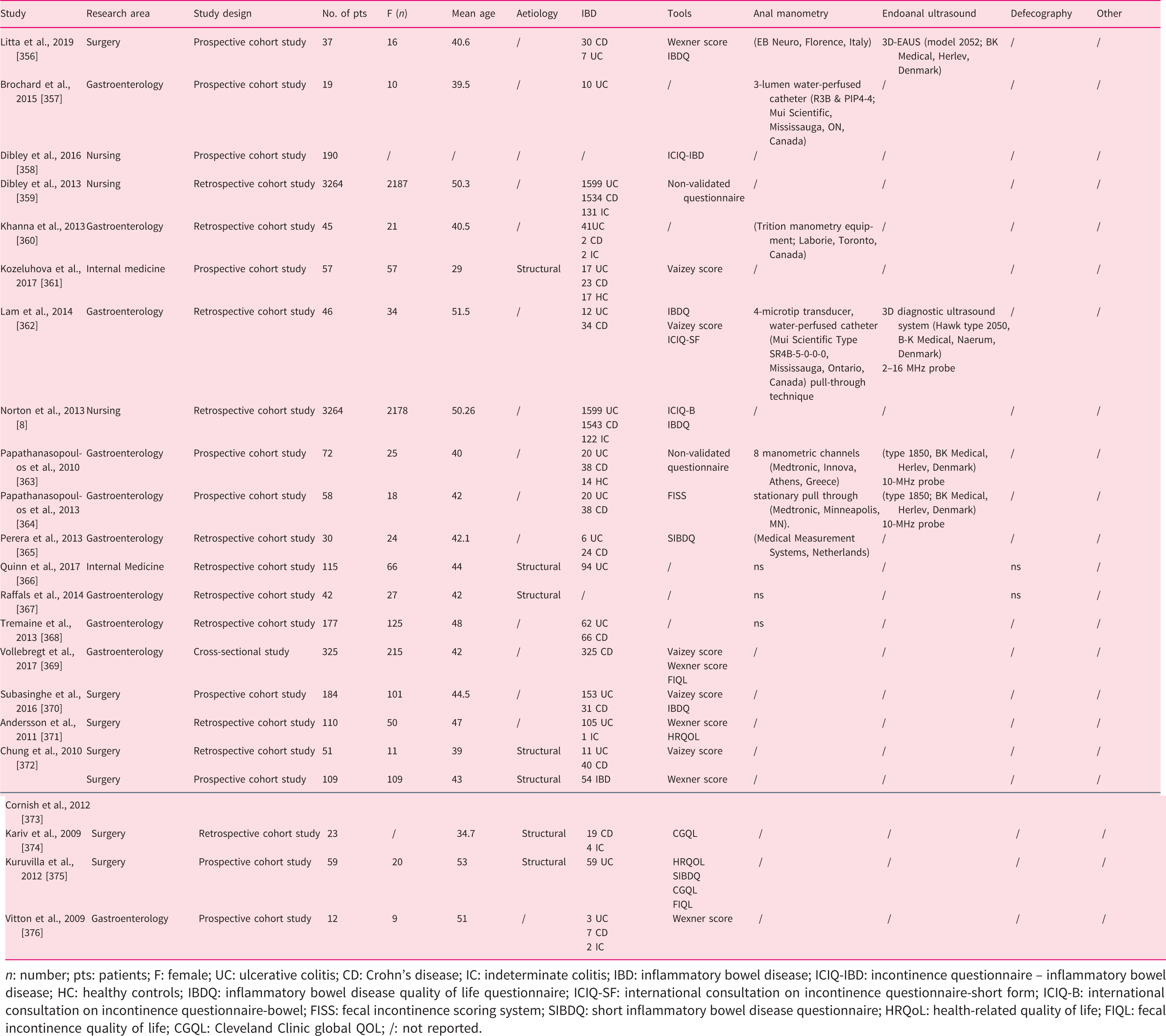
n: number; pts: patients; F: female; UC: ulcerative colitis; CD: Crohn’s disease; IC: indeterminate colitis; IBD: inflammatory bowel disease; ICIQ-IBD: incontinence questionnaire – inflammatory bowel disease; HC: healthy controls; IBDQ: inflammatory bowel disease quality of life questionnaire; ICIQ-SF: international consultation on incontinence questionnaire-short form; ICIQ-B: international consultation on incontinence questionnaire-bowel; FISS: fecal incontinence scoring system; SIBDQ: short inflammatory bowel disease questionnaire; HRQoL: health-related quality of life; FIQL: fecal incontinence quality of life; CGQL: Cleveland Clinic global QOL; /: not reported.
Fecal incontinence assessment in non-IBD patients
FI was assessed using specific questionnaires in all studies, except for four trials.168,213,274,280 Twenty-nine different questionnaires were applied: the most widely adopted were the Wexner score (187 studies),50,52,54,55,59,61,63,66,69–71,73–83,86–92,97–99,101,103–107,110,114,116,117,120–123,126–128,130,131,133,135–139,141,144,146–156,160,164–167,169,170,174,177–179,182,194–203,205–208,210–212,214, 215,217–220,223–226,229,234–239,241–245,247–253,256,259,261,264, 265,270–273,276,278,279,281,284–287,289,291–296,298–306,308–311,314, 315,319,321,323–326,328,330,333–336,339–341,347,349,350,353,354 FIQL questionnaire (91 studies),50,62,67,73–75,79,84–88, 100,101,104,106,109,115,121,123–125,127–129,131,134–136,139,147,151, 152,155,157,158,161,165,166,170,172,179,181,183,190,208,209,212,214, 217,218,235,240–242,249,255–257,260–263,265,269,270,273,279,283,288, 292–297,302,303,309,311,314,319,321,322,324,333,334,338,349,350,355 the Vaizey score (62 studies)51,56,63,65,67,68,70,71,81,100, 101,119,124,125,135–137,157,158,175,176,180,183,189–192,196,204,209, 216,227,232,246,255,266,268,269,277,279,282,290,297,300,303,307,309, 311–313,315,318,322,327,331–333,335,336,338,348,352 and fecal incontinence severity index (FISI; 33 studies).42,60,72, 84,85,93,109,115,118,140,161–163,171,172,181,184,186,187,230–232,238, 240,256,260,308,314,320,329,342,344,355 In 13 studies (4.2%)53, 58,65,108,111,142,143,145,159,316,337,343,351 non-validated questionnaires were used. Anal manometry was used in 126 studies (41.2%)50,53,55,57,61,65,72,73,77,78,80,82,83, 85,86,91,92,95,96,98,103–107,109,111,115,122,123,125,127,134,141,142, 146,151–153,155,162–165,167,168,170,174–176,178,179,183,191,193,199–201,204,206,213,217,218,223,227,228,230,234,236,237,239,240,244–251, 253,256,258,260,261,264,268–270,274,276–278,280,281,285,290,293,294, 296,298,299,301,302,304–306,310,314,319,324,326,332–334,337,338,347, 349–351,353–355,362,376 for the evaluation of FI: eight-channel water-perfused manometries were the most common systems (in 22 studies).61,83,122,125,141,142, 146,170,183,199,200,227,251,253,258,261,264,285,296,298,302,353 Endoanal ultrasonography was performed in 104 studies (34.0%).53,65,72,73,76–78,80,83,86,90,92,104,105,109,110,113, 115,122,125,127,133,141,142,146,151–153,155,157,162,163,165,167,172–176,178,179,183,191,193,200,201,204,213,225,227,230,238–240,243–246, 248–253,258,261–263,266,268,269,272,273,276,278–281,284–286,290,291, 294,298,301,302,306,310,314,326,327,329,331–334,338,340,349,351,354, 355,362 In 26 studies (8.5%)52,72,90,91,98,105,109,148,162, 163,167,223,237, 245,247,293,306,308,326,328,334,335,340,344,346,354 the FI diagnostic approach included defecography. Other methods for FI evaluation implied PNTML investigation (23 studies),104,105,115,151,153,165,183,204,237, 239,240,248,258,268,269,277,286,293,294,302,314,324,354 MRI (20 studies),52,96,104,111,148, 171,238,239,245,262,274,285,293,306,308, 314,329,332,333,335 endoscopic procedures (18 studies),77, 78,98,163,167,174,180,223,226,238,239,247,301,306,308,314,335,340 EMG (five studies),191,206,260,294,354 CT scan (two studies),98,114 peripheral nerve evaluation (PNE; two studies),75,297 CT colonography (two studies),163,308 gastrointestinal transit time study (one study) 340 and fecoflowmetry (one study). 299 FI was diagnosed with one technique in 142 (46.4%) studies (questionnaires in 141 studies51,54,56,58–60,62–64,66–71,74,79,81,84,87–89,93,94,97,99–102,108,112,116–121,124, 126,128–132,135–140,143–145,147,149,150,154,156,158–161,166,169,177, 181,182,184–190,192,194–198,202,203,205,207,208,210–212,214–216,219–222,224,229,231–233,235,241,242,254,255,257,265,267,271,275,282,283, 287–289,292,295,300,303,307,309,311–313,315–318,320–323,325,330,336, 341–343,345,348,352 or anal manometry in one study), 168 with two techniques in 64 trials (20.9%),50,55,57, 61,75,76,82,85,95,103,106,107,110,113,114,123,133,134,157,164,170–173, 180,199,213,217,218,225,226,228,234,236,243,252,256,259,263,264,266, 270, 272–274,279, 280,284,291,296,297,304, 305,319,327, 328,331,337,344, 346, 347,350,353,376 and with three or more combined methods in 100 studies (32.7%).52,53,65,72,73,77,78,80,83,86,90–92,96,98, 104,105,109,111,115,122,125,127,141,142,146,148,151–153,155,162,163,165, 167,174–176,178,179,183,191,193,200,201,204,206,223,227,230,237–240, 244–251,253,258,260–262,268,269,276–278,281,285,286,290,293,294,298,299, 301,302,306,308,310,314,324,326,329,332–335,338,340,349,351,354,355,362
Fecal incontinence assessment in IBD patients
In 17 trials (77.3%)8,356,358,359,361–365,369–376 FI was evaluated using specific questionnaires. Ten different tools were adopted. The Wexner score, the Vaizey score and the inflammatory bowel disease quality of life (IBDQ) questionnaire were the most frequently used tools (in five (22.7%),356,369,371,373,376 five (22.7%)361,362,369,370,372 and four (18.2%)8,356,362,370 studies, respectively). In two studies (9.1%)359,363 non-validated questionnaires were applied. Anal manometry was used in 10 studies (45.4%) for FI assessment. Data concerning anal manometry were not specified in three studies (13.6%),366–368 while in seven studies (31.8%)356,357,360,362–365 singularly different systems were used. Anal ultrasound was performed in four studies (18.2%)356,362–364 using Bruel and Kjaer medical probes in all cases. Defecography was used in two studies (9.1%).366,367 However, no additional data were specified regarding diagnostic evaluation. In 15 studies (68.2%) the diagnosis of FI was obtained only with one technique (questionnaires in 12 studies8,358,359,361,369–376 and anal manometry in three studies).357,360,368 In three trials (13.6%)365–367 two techniques were used, while in four papers (18.2%)356,362–364 more than three methods were combined.
Discussion
We summarised all tools and investigations for FI diagnosis of the past 10 years in both IBD and non-IBD patients, including over 300 papers. Questionnaires represented the main technique for FI evaluation. They were used in almost all non-IBD studies and in over 75% of IBD trials. The most frequent tools in descending order of use were the Wexner score, FIQL, the Vaizey score and FISI for non-IBD patients and the Wexner score, the Vaizey score and the IBDQ for IBD patients. Patients’ evaluation included anal manometry in about 40% of cases in both groups, while endoanal ultrasound was adopted more in non-IBD patients than in IBD patients (34.0% vs. 18.2%, respectively). Defecography was performed in a similar percentage of studies in both examined groups (8.5% vs. 9.1%), while investigations on anal physiopathology (such as PNTML and EMG) and morphological studies (such as MRI, CT scan and endoscopy) were used only in a small number of non-IBD patients. The difference in FI assessment between IBD and non-IBD patients could be due to several reasons. The management by many specialists, from the surgeon to the gastroenterologist to the gynaecologist, etc. could justify the different approaches. Moreover, in IBD patients the involuntary loss of feces could be attributed to the underlying disease rather than a concomitant FI related to other causes, determining not only an underestimation of the problem but also a less comprehensive approach. A recent systematic review and meta-analysis by Gu et al. 377 analysed the prevalence, pathophysiology and treatment of FI in IBD patients, reporting questionnaires and anal manometry as the main instruments for FI diagnosis. No additional tool was used to reach the diagnosis of FI. However, questionnaires are based on subjective responses of the patients and they could determine evaluation bias. Some psychometric properties, 101 such as sensitivity, validity, reliability and responsiveness are measured to overcome this limitation. The sensitivity and validity of four scores (Pescatori score, Wexner score, Vaizey score and American medical systems score) were assessed in a small prospective study 16 including 23 patients showing a high and significant correlation of all scores with the clinical impression of physicians. A prospective randomised clinical trial 101 compared the Vaizey score, Wexner score and FIQL scale in order to evaluate their responsiveness and interpretability. All instruments showed adequate external responsiveness (Pearson correlation (r)=0.60, r=0.44 and r=0.48, respectively) and interpretability (minimally important change (MIC)=20%, MIC=17.5% and MIC=33.8%, respectively) although none reached high psychometric levels. The main strength of our review is the large number of included studies, providing a summary of the evidence on FI diagnostic approaches in the past 10 years. Furthermore, to our knowledge it is the first paper that is specifically designed to investigate FI assessment, comparing the approach between IBD and non-IBD patients. However, there are some limitations. First of all, the marked heterogeneity of the studies, which included different scientific areas and took into account several patient populations and evaluation methods. Second, the study design of our work did not allow us to obtain sufficient data to compare the accuracy of the diagnostic tools and only direct comparative studies between the different methods will allow us to overcome this gap. Third, no distinction between ulcerative colitis and Crohn’s disease in IBD trials was provided. Fourth, only studies published in the past 10 years were included in our systematic review representing a possible bias for result interpretation. There is no globally accepted consensus on the FI diagnostic approach. The neuro-urological guidelines 378 state that the initial FI assessment should be based on a careful patient history using standard instruments such as Wexner and Vaizey scores. Digital rectal examination is considered an essential component for FI diagnosis, while the role of physiological and radiological tests is not established and their execution depends on the presence of alarm symptoms (e.g. weight or blood loss) or comorbidities. 378 The National Institute for Health and Care Excellence (NICE) guidelines, 379 instead, suggest clinical examination as a first step and a combination of anorectal physiology tests and endoanal ultrasonography (or MRI if ultrasonography is not available) in patients with FI who are candidates for surgical treatment. These data, as also indicated by our review, emphasize the lack of standardization in FI assessment. In some hospitals, colorectal units have been developed in order to manage complex FI cases. A retrospective study by Saunders et al. 380 analysed the impact of the colorectal unit on the rate of adverse events and functional outcomes. A multidisciplinary management resulted in reduced rates of post-treatment complications and in higher rates of functional outcomes compared to the pre-colorectal unit management. 380 However, the team consisted only of a surgeon, gastroenterologist, psychologist and a specialised nurse; some specialists who have a fundamental role in the management of patients with FI were not consulted. Currently, several tools have been developed to assess FI but there is no solid evidence on FI evaluation in the field of IBD. The Wexner and Vaizey scores are generic tools that can be applied to different research areas, including IBD. They are widely used in clinical practice as they are simple, quick and reliable. 381 The Wexner score is mainly focused on symptoms related to anal sphincter impairment and assesses the presence of solid, liquid, or gas incontinence. 381 The Vaizey score is based on the Wexner score and includes two additional parameters: defecation urgency and the use of constipating medication. 381 However, they do not evaluate patients’ quality of life and were developed without any input from patients, ignoring patients’ views. The International consultation on incontinence questionnaire-inflammatory bowel disease (ICIQ-IBD) is a new initially validated questionnaire, specifically structured for IBD patients. 358 It is based on both expert and patient inputs including a quality of life measure. Nevertheless, the ICIQ-IBD is hardly utilised, due to its complexity and lack of full validation. 381 To date, no score is perfectly fit for FI evaluation in IBD patients and data on the accuracy of FI diagnostic tools in the field of IBD are lacking, preventing definitive conclusions from being drawn. The Wexner score could be used, as the most appropriate for historical comparison, until a better tool is developed. The ongoing International Organization for the Study of Inflammatory Bowel Disease (IOIBD) consensus on endpoints for disease modification will determine which tools should be recommended for FI assessment in IBD patients. Therefore, based on the data of our work we propose to use the Wexner score as a first step for assessment of IBD patients with suspected FI. Other investigations should be performed only in the case of doubt and after multidisciplinary evaluation, including a team of experts consisting of a gastroenterologist, surgeon, gynaecologist, urologist, physiologist, neurologist, radiologist and psychologist. They should define the best diagnostic approach and guide therapeutic decisions, allowing better patient management and avoiding FI under/overestimation and misdiagnosis.
Conclusion
Fecal incontinence is a common and disabling condition in patients regardless of whether or not they have IBD. A validated and globally accepted algorithm to diagnose FI has not yet been defined. A detailed history, including the characteristics of the feces and the type and frequency of incontinence, is necessary promptly to identify red flag symptoms. In the past 10 years questionnaires have been the most frequently used tools for FI evaluation. Based upon validation and global researcher preference, we propose to start FI assessment of IBD patients with the Wexner score. Any future instrument development might best be achieved by interdisciplinary international collaboration. Anal manometry and/or endoanal ultrasonography should be considered when questionnaires are suggestive of fecal incontinence.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620943699 - Supplemental material for Tools for fecal incontinence assessment: lessons for inflammatory bowel disease trials based on a systematic review
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620943699 for Tools for fecal incontinence assessment: lessons for inflammatory bowel disease trials based on a systematic review by Ferdinando D’Amico, Steven D Wexner, Carolynne J Vaizey, Célia Gouynou, Silvio Danese and Laurent Peyrin-Biroulet in United European Gastroenterology Journal
Footnotes
Author contribution
FD wrote the manuscript with support from CG. SD critically reviewed the content of the paper and supervised the project. CV and SW performed critical appraisal of the data and critical revision of the paper. LPB conceived the study, contributed to the critical interpretation of the results and supervised the project. All authors discussed the results and contributed to the final paper.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FD declares no conflicts of interest. CG declares no conflicts of interest. SDW reports consulting fees from Medtronic, TiGenix, Regentys, Karl Storz Endoscopy America, Intuitive Surgical Innovations, LifeBond, and Stryker. He has received royalties for intellectual property licence from Medtronic, Intuitive Surgical Innovations and Karl Storz Endoscopy America. He has received stock options for consulting from LifeBond, NeatStitch and novoGI. He holds stock options from Pragma, Regentys, CRH Medical and Renew Medical. He is the managing member of Unique Surgical Innovations LLC, the company which has licensed the intellectual property responsible for his personal royalty payments from Karl Storz Endoscopy America, Intuitive Surgical Innovations and Medtronic. CJV is a consultant, speaker and educator for Medtronic Inc. and an educator for THD. She holds stock options from Renew Medical. SD has served as a speaker, consultant and advisory board member for Schering-Plough, AbbVie, MSD, UCB Pharma, Ferring, Cellerix, Millenium Takeda, Nycomed, Pharmacosmos, Actelion, Alphawasserman, Genentech, Grunenthal, Pfizer, Astra Zeneca, Novo Nordisk, Cosmo Pharmaceuticals, Vifor and Johnson&Johnson, Nikkiso Europe GMBH, Theravance. LPB has served as a speaker, consultant and advisory board member for Merck, Abbvie, Janssen, Genentech, Mitsubishi, Ferring, Norgine, Tillots, Vifor, Hospira/Pfizer, Celltrion, Takeda, Biogaran, Boerhinger-Ingelheim, Lilly, HAC-Pharma, Index Pharmaceuticals, Amgen, Sandoz, Forward Pharma GmbH, Celgene, Biogen, Lycera, Samsung Bioepis and Theravance.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
