Abstract
Background
As small-bowel adenocarcinoma (SBA) is scarce, no standard systemic regimen in metastatic disease has been defined.
Objective
To obtain insights into the use and effects of palliative chemotherapy in patients with metastatic SBA in a population-based setting.
Methods
Data from the Netherlands Cancer Registry of patients with metastatic SBA between 2007 and 2016 were used (n = 522). For patients treated with palliative chemotherapy, differences in treatment regimens and survival were evaluated.
Results
Palliative chemotherapy was received by 38% of patients (n = 199). First-line combination chemotherapy was administered to 80% of patients, mainly CAPOX/FOLFOX. Single-agent chemotherapy mostly consisted of capecitabine. Second-line treatment, mostly irinotecan-based (58%), was prescribed to 27% of patients. Age 70 years or older was an adverse predictive factor for receiving first-line combination chemotherapy (odds ratio (OR) 0.2, 95% confidence interval (CI) 0.08–0.62) and second-line therapy (OR 0.3, 95% CI 0.10–0.72). Median overall survival with palliative chemotherapy was 9.3 months, compared with 3.0 months without. In subanalyses, patients who received only first-line treatment had a median overall survival of 5.6 and 7.0 months after single-agent and combination chemotherapy, respectively.
Conclusion
A minority of patients were treated with palliative chemotherapy. First-line treatment consisted predominantly of oxaliplatin-based combination chemotherapy, whereas second-line treatment was mainly irinotecan-based. Population-based median overall survival for selected patients treated with chemotherapy amounted to nine months.
Key points
Summarise the established knowledge on this subject:
Small-bowel adenocarcinoma (SBA) is scarce, comprising only less than 5% of all gastrointestinal tumours. Owing to its rarity, the role of palliative chemotherapy in patients with metastatic disease in daily practice has not been well established. Several phase II trials and retrospective studies showed that palliative chemotherapy could prolong overall survival in patients with metastatic SBA, but a standard systemic regimen has not been defined. Significant and/or new findings of this study:
Palliative chemotherapy is received by only a minority of 38% of patients with synchronous metastases of SBA. First-line palliative chemotherapy mainly consisted of CAPOX/FOLFOX, whereas second-line chemotherapy was mostly irinotecan-based. Population-based median overall survival with palliative chemotherapy was nine months, compared with three months with best supportive care. In subanalyses, patients who received only first-line treatment had a median overall survival of 5.6 and 7.0 months after single-agent and combination chemotherapy, respectively.
Introduction
Small-bowel adenocarcinoma (SBA) is a rare gastrointestinal tumour, accounting for only 2% to 3% of all malignant digestive tumours. In SBA patients, metastatic disease is a common phenomenon, as approximately 30% to 40% of patients presents with synchronous metastases.1–4 It has been hypothesised that these high metastatic rates are caused by nonspecific and atypical symptoms in patients and the lack of simple and reliable diagnostic tools to reach the small bowel. The prognosis of patients with metastatic SBA is poor, with a reported overall survival of four to 11 months.2,3
Due to the rarity of the disease, most data on palliative chemotherapy in SBA are derived from phase II trials and retrospective series of mainly high-volume or tertiary medical centres.5–7 These studies have demonstrated that chemotherapy prolongs overall survival in patients with metastatic SBA.4–15 However, in the absence of randomised controlled trials, a standard chemotherapeutical regimen has not been defined. Patients are usually treated with chemotherapeutical regimens which have been extrapolated from other gastrointestinal cancers. Several studies have demonstrated that combination chemotherapy consisting of a fluoropyrimidine and platinum compound seems to have the most beneficial effect on survival, with median overall survival rates ranging between 14 and 18 months.4,7,12,14,15 As a result, a frontline regimen of platinum-based combination chemotherapy has been suggested.7,16
Population-based data, which reflect daily practice and could be of help for clinicians to guide treatment decisions, are currently lacking. As large prospective clinical studies and randomised controlled trials are virtually impossible to conduct because of the rarity of SBA, this population-based study was performed to provide insight into daily-based chemotherapeutical treatment and its results in patients with synchronous metastatic SBA.
Material and methods
Data collection
Data were obtained from the population-based Netherlands Cancer Registry (NCR). The nationwide NCR covers all nearly 17 million inhabitants of the Netherlands and collects data on all newly diagnosed malignancies. Primary source of notification is the automated nationwide pathological archive (PALGA), accompanied by data from the National Registry of Hospital Discharge Diagnoses. Information on patient and tumour characteristics, diagnosis and treatment were routinely collected from medical records by specially trained administrators. In the NCR, the primary tumour stage was determined based on the tumour-node-metastasis (TNM) classification. In case the pathological TNM stage was missing, the clinical TNM stage was used. The anatomical site of the primary tumour and its metastases were recorded according to the third version of the International Classification of Diseases for Oncology (ICD-O).
For the present study, additional data were retrospectively collected by registry clerks of the NCR on systemic treatment regimens for patients treated with palliative chemotherapy for synchronous metastases of SBA (ICD-O code C17) diagnosed between 2007 and 2016. Synchronous metastases were defined as metastases diagnosed within three months after initial SBA diagnosis. Additional data comprised information on first-, second- and third-line systemic treatment regimens, including details and duration of chemotherapeutical and targeted agents. First-line systemic treatment was defined as the initial therapy with chemotherapeutical and/or targeted agents. If one of the agents of the initial therapy was discontinued, while other drug(s) were continued, it was still regarded as first-line treatment. Second- and third-line systemic treatment was defined as the adoption of a different treatment regimen, mostly because of failure of first-line therapy or unacceptable toxicities. In case of a rechallenge of a chemotherapeutical and/or targeted agent within three months or after maintenance therapy, we defined the therapy to be a next-line treatment. If the rechallenge occurred after three months, without maintenance therapy, the therapy was classified as the same-line treatment.
Statistical analysis
Descriptive statistics were used to describe patient and tumour characteristics of the study population. Differences in patient- and tumour characteristics between the treated and nontreated patients were analysed with a two-sided chi-squared test. In all analyses concerning palliative chemotherapy, only the patients receiving palliative chemotherapy with a known treatment regimen were included. First-, second- and third-line systemic treatment regimens were categorised according to the administrated number of chemotherapeutical agents into single-agent chemotherapy and combination chemotherapy, apart from the additional administration of targeted agents. Time to progression from the start of first- to start of second-line treatment and second- to third-line treatment was calculated and presented in months. Differences in time to progression from first- to second-line treatment between the patients receiving single-agent chemotherapy and combination chemotherapy were evaluated with the Wilcoxon rank-sum test. Univariable logistic regression, including the 95% confidence interval (CI), was used to assess the independent influence of all patients and clinical characteristics on the probability of receiving second-line treatment. If univariable analyses tended towards statistical significance (p < 0.20), the probability of receiving second-line treatment corrected for these potential prognostic factors was afterwards calculated with multivariable regression analyses. For the odds of receiving first-line combination chemotherapy, only multivariable logistic regression analyses were run.
Overall survival was calculated on mortality of any cause. Overall survival time was defined as the time from date of diagnosis to death or last follow-up date. Patients who were lost to follow-up, emigrated or still alive at 1 February 2018 were censored. Overall survival was estimated with the log-rank test and Kaplan-Meier analyses. Median overall survival was presented in months, with its corresponding 95% CIs. The median overall survival of treated patients was compared with patients receiving only best supportive care. Multivariable Cox proportional-hazards regression analyses were performed to identify prognostic factors for overall survival in patients treated with palliative chemotherapy. Hazard ratios (HRs) were presented with their corresponding 95% CIs.
The statistical package SAS Statistical Software (version 9.4, SAS Institute, Cary, NC, USA) was used to analyse the data. A two-sided p value of p < 0.05 was considered as statistically significant for all statistical tests.
Results
Patients
General characteristics of the total patient population according to palliative chemotherapy treatment (n = 522) and of patients treated with palliative chemotherapy according to first-line chemotherapy regimen (n = 187).
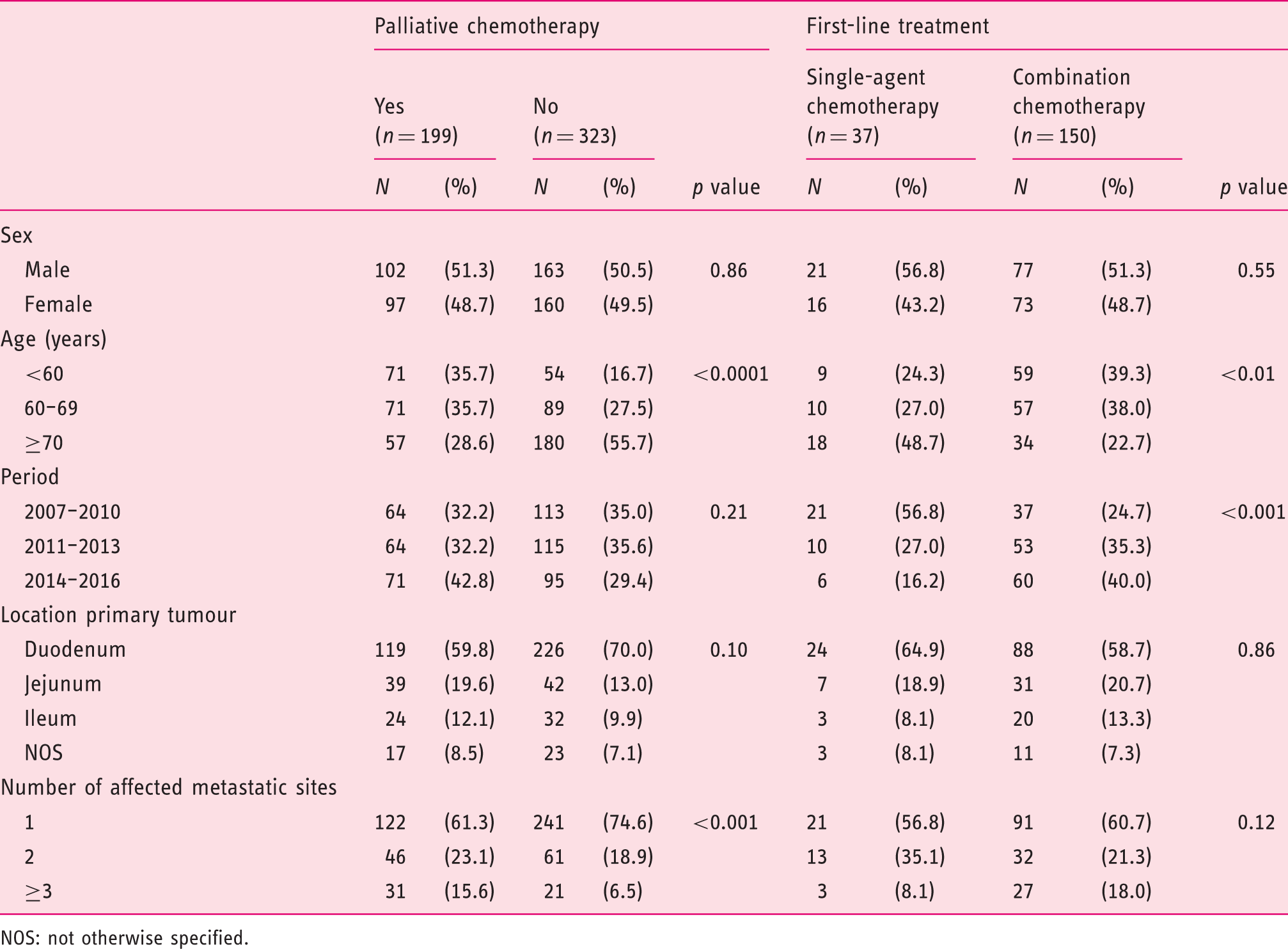
NOS: not otherwise specified.
In patients receiving palliative chemotherapy, the primary tumour was mainly located in the duodenum (60%), followed by the jejunum (20%) and ileum (12%). Patients presented with liver and peritoneal metastases in 63% and 36% of the cases, respectively. In 40% of patients multiple metastatic sites were diagnosed. Only two patients were diagnosed with Lynch syndrome. In 66 patients (35%) palliative interventions were performed, including surgical bypass and stenting, mostly in patients with a primary duodenal tumour (79%).
Chemotherapy regimens
The first-line chemotherapy regimens were mainly based on fluoropyrimidines (5-fluorouracil and capecitabine) and oxaliplatin (Figure 1). Combination chemotherapy was prescribed to 80% of patients, of whom 97% received an oxaliplatin-based doublet or triplet regimen, including capecitabine (CAPOX, n = 91), 5-fluorouracil (FOLFOX, n = 29) or epirubicin with capecitabine (EOX, n = 2). Single-agent chemotherapy was administered to 20% of patients, consisting of fluoropyrimidines in 92% of cases, mainly capecitabine (n = 32). The targeted agent bevacizumab was prescribed to 13% of patients (n = 25), most frequently in addition to combination chemotherapy. Other nonfrequently used first-line treatment regimens included epirubicin, cisplatin and capecitabine (ECC, n = 2), carboplatin with paclitaxel (n = 2), capecitabine with irinotecan (CAPIRI, n = 1) and oxaliplatin (n = 2) or irinotecan monotherapy (n = 1).
Prescribed chemotherapeutic regimens according to combination chemotherapy and single-agent chemotherapy in first-, second- and third-line treatment in metastatic small-bowel adenocarcinoma.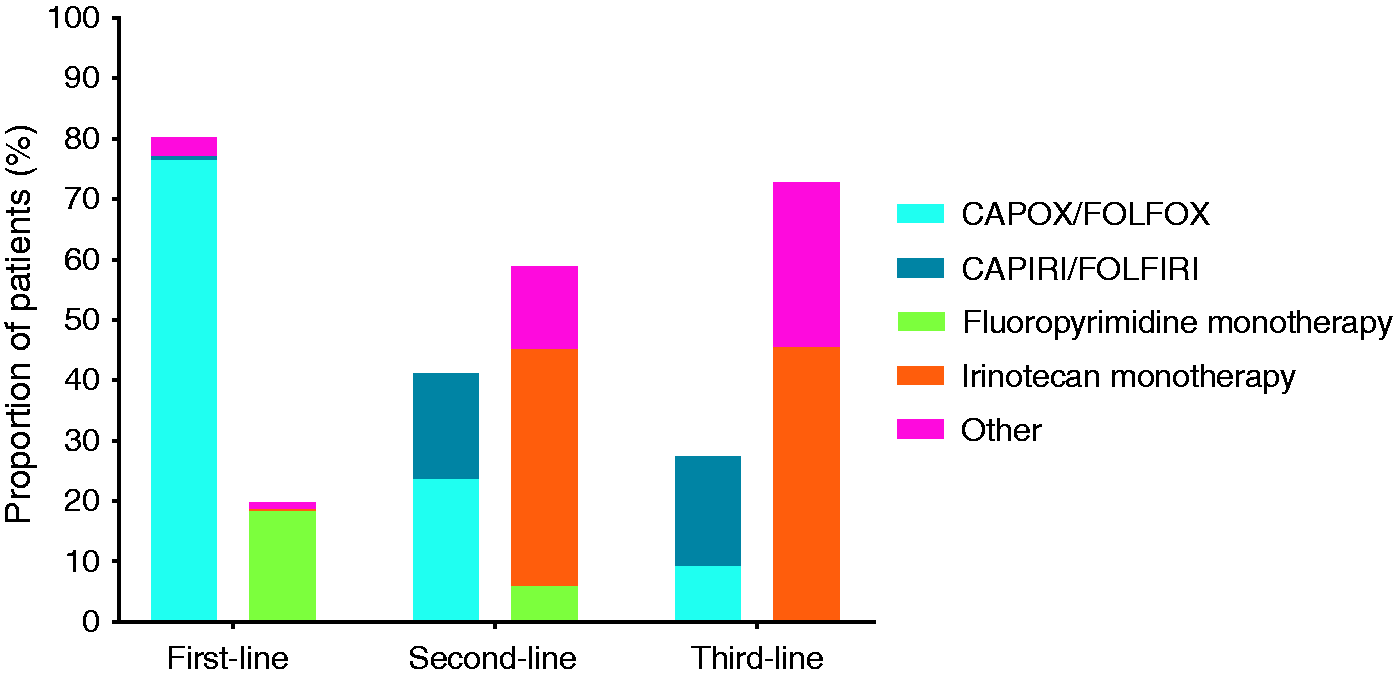
General characteristics and multivariable logistic regression modelling the odds of patients receiving combination therapy in first-line treatment (left) and general characteristics, univariable logistic regression and multivariable logistic regression modelling the odds of receiving second-line treatment compared with patients receiving first-line treatment (right).
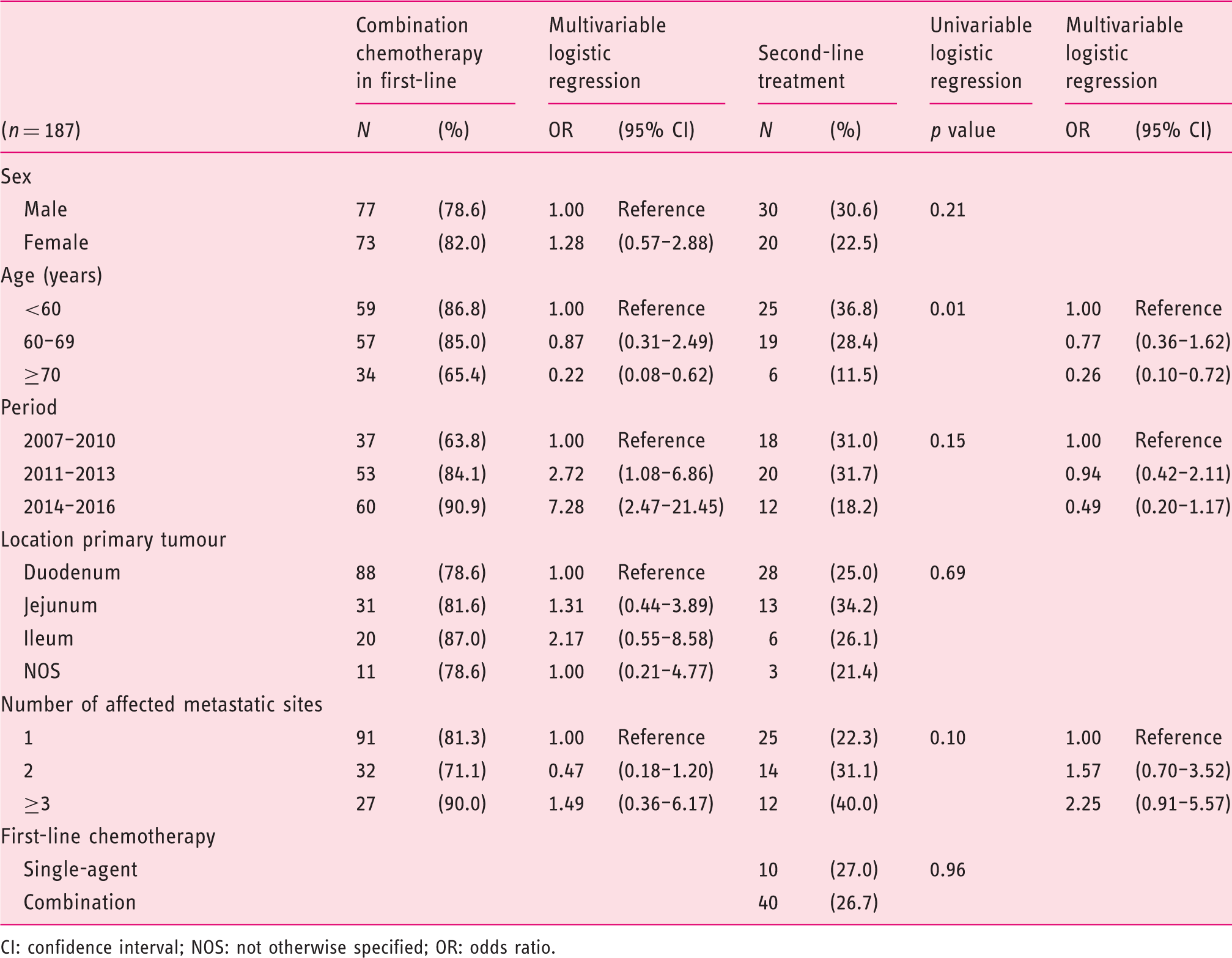
CI: confidence interval; NOS: not otherwise specified; OR: odds ratio.
Second-line systemic therapy was received by only 27% of patients (n = 50), mostly consisting of single-agent chemotherapy (58%) (Figure 1). Irinotecan was administered as a single-agent in 69% of patients (n = 20). Patients treated with combination chemotherapy, received an oxaliplatin-based (n = 12) or an irinotecan-based (n = 9) regimen with either capecitabine (CAPOX/CAPIRI) or 5-fluorouracil (FOLFOX/FOLFIRI). In seven patients who received a second-line oxaliplatin-based regimen, a rechallenge after use of oxaliplatin-based combination chemotherapy in first-line occurred. Epidermal growth factor receptor monotherapy was administered to two patients. In eight patients, bevacizumab was prescribed in addition to second-line chemotherapy.
Logistic regression analysis identified advanced age (≥70 years) to be inversely associated with the receipt of second-line treatment (OR 0.26, 95% CI 0.10–0.72) (Table 2).
Third-line systemic therapy was received by 11 patients (6%). Most third-line regimens were irinotecan-based (n = 8), consisting of irinotecan monotherapy (n = 3) or combination therapy of a fluoropyrimidine with irinotecan (CAPIRI/FOLFIRI, n = 3) (Figure 1). The targeted agent panitumumab was used in four patients (36%), either as monotherapy (n = 2) or in combination with irinotecan (n = 2).
Survival
The median overall survival for patients treated with palliative chemotherapy and a known systemic treatment regimen was 9.3 months, with a one-year survival rate of 39%. Patients receiving only best supportive care had a median overall survival of 3.0 months, with a one-year survival rate of 17%. In patients receiving only first-line treatment, the median overall survival of patients receiving single-agent chemotherapy was 5.6 months, compared with 7.0 months with combination chemotherapy, with one-year survival rates of 22% and 29%, respectively. The median time to progression from first- to second-line therapy was 7.7 months, which did not significantly differ between the single-agent chemotherapy and combination chemotherapy groups (p = 0.82).
Patients receiving second-line systemic therapy had a significant higher median overall survival, compared with those who received only first-line therapy, with observed median overall survival times of 15.2 and 6.8 months, respectively (p < 0.0001). In the 11 patients receiving third-line treatment, a median overall survival of 28.4 months was noted. Among this subgroup of patients, five out of 11 had a rechallenge of CAPOX/FOLFOX (n = 4) or capecitabine monotherapy (n = 1) in second-line treatment. The median time to progression from second- to third-line systemic therapy was 9.8 months.
Multivariable survival analyses for patients treated with palliative chemotherapy in synchronous metastatic small-bowel adenocarcinoma (n = 187).
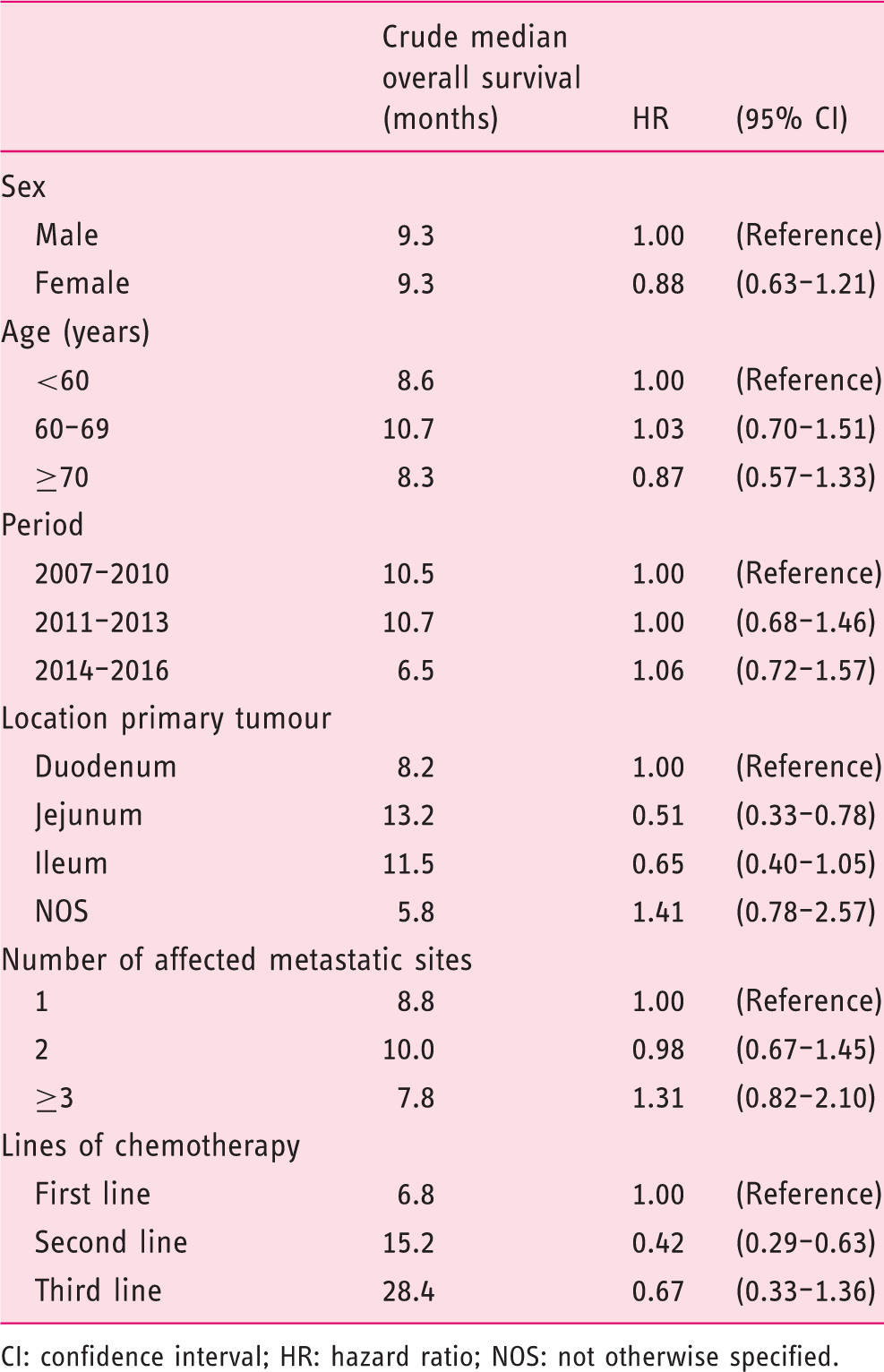
CI: confidence interval; HR: hazard ratio; NOS: not otherwise specified.
Discussion
This population-based study aimed to provide insight into the community-based use of palliative chemotherapy in patients with synchronous metastases of SBA. Palliative chemotherapy was administered to 38% of patients with metastatic disease. The vast majority of treated patients received an oxaliplatin-based combination regimen in first-line treatment with CAPOX/FOLFOX. Second-line therapy was administered to only 27% of patients, with mainly irinotecan monotherapy.
In first-line treatment, combination chemotherapy with mainly an oxaliplatin-containing regimen was most often prescribed, analogous to gastric cancer and colorectal cancer (CRC), for which fluoropyrimidines and platinum derivatives are the backbone of palliative chemotherapy.17,18 In the present study, only a small minority of patients with a primary duodenal tumour were classically treated as gastric cancer with triplet chemotherapy as ECC or EOX, probably as it was historically thought that duodenal tumours behave more like gastric cancer, whereas distal tumours share more similarities with CRC. 6 In recent years, several studies conducted on the efficacy of chemotherapy in advanced gastric cancer have favoured the use of oxaliplatin over cisplatin in terms of its toxicity profile and its noninferiority in overall survival, and have questioned a beneficial effect on survival from the contribution of anthracyclines as epirubin to an oxaliplatin-based combination regimen.18–20 As a result, both CAPOX/FOLFOX are a well-studied and effective first-line palliative treatment regimen in a variety of gastrointestinal cancers, including gastric cancer and CRC. These obtained results suggested a potential role for this regimen in metastatic SBA, because of the embryological derivation of the small bowel and the presence of some overlapping genomic alterations both with gastric cancer and CRC. 21 Other small retrospective and phase II studies conducted on the efficacy of CAPOX/FOLFOX in metastatic SBA showed improved survival rates in treated patients with tolerable toxicity.4,7,12,14,15
Combination chemotherapy in first-line treatment was increasingly administered over time, although all used cytotoxic agents were registered and available during the total study period in the Netherlands.22–24 Hypothetically, the obtained experience of clinicians with these cytotoxic drugs in more frequently encountered gastrointestinal cancers, and the obtained evidence of the beneficial effect of oxaliplatin-based regimens in phase II studies for metastatic SBA, could account for the increased use of combination chemotherapy over time.5,7,14
Second-line therapy was administered to only about one-quarter of patients, which is limited, even at a population-based level, as compared with patients receiving second-line treatment in metastatic disease of gastric cancer and CRC.25–27 Possibly, the lack of data for second-line chemotherapy in SBA could be accountable for these lower rates. Second-line treatment was mainly irinotecan-based, and 40% of patients received irinotecan monotherapy, which could have been influenced by the favourable results of second-line FOLFIRI in establishing disease control after failure of first-line platinum-based chemotherapy as reported in a French study. 28 The prescription of irinotecan-based second-line chemotherapy is in accordance with Dutch guidelines for second-line treatment for metastatic CRC, but not for gastric cancer. 29 Of note, the median time to progression between first- and second-line treatment is comparable for SBA and CRC.30–32
The median overall survival of six to nine months as found in the current study is lower than survival rates reported in other retrospective studies, although equivalent treatment regimens were used.4,11,12 In the present study, the median age was slightly higher and more patients had liver metastases compared with other retrospective studies. Moreover, no patients with locally advanced disease were included in the current study, whereas one study did include patients both with locally advanced and metastatic disease of SBA. 4 In comparison with patients in population-based studies treated with palliative chemotherapy for other gastrointestinal cancers, patients with metastatic SBA have a worse median overall survival than patients with metastatic CRC, whereas the survival of patients with metastatic SBA and gastric cancer is comparable.33,34
Patients who received first-line chemotherapy only had a higher overall survival of three to four months compared with patients receiving best supportive care. However, selection bias should be taken into account as patients treated with palliative chemotherapy were already selected by their treating physician, as for instance reflected in a lower median age. On a population-based level, a doubling of median overall survival was observed in patients receiving first-line combination chemotherapy.
The highly selected group of patients who were prescribed second-line or even third-line treatment had a median overall survival of almost 15 and 28 months, respectively. However, immortal time bias could have influenced these results and should be taken into account, since patients should be alive to receive further line chemotherapy. Moreover, considering nearly one-half of the patients treated with third-line therapy had a rechallenge of oxaliplatin doublet chemotherapy or capecitabine monotherapy in second-line treatment, it could suggest that these patients had a more indolent tumour behaviour or their tumours had a prolonged or increased chemosensitivity to oxaliplatin-containing combination chemotherapy. The impact of irinotecan on median overall survival in second- and third-line treatment needs to be studied in a larger subset of patients.
As already stated, selection bias is a potential drawback of the current study with regards to overall survival. Furthermore, detailed information on for instance performance status, nutritional status, comorbidities, disease-related symptoms and the extent of disease were lacking, since these data were not provided in the databases of the NCR or were often not noted in the patient records.
In conclusion, a minority of patients with synchronous metastases of SBA were treated with palliative chemotherapy in daily practice. First-line treatment consisted predominantly of oxaliplatin-based combination chemotherapy, whereas second-line treatment was mainly irinotecan-based. Population-based median overall survival for selected patients treated with chemotherapy amounted to nine months.
Footnotes
Acknowledgements
The authors thank the registration team of the Netherlands Cancer Registry for their dedicated data collection. We thank Nienke Bosman, Claudia van Leeuwen and Erica Masseling for their contribution with regard to the additional data collection.
Declaration of conflicting interests
I.H.J.T.H. received funding from the Dutch Cancer Foundation (KWF), as well as from Roche, all of which is not related to the presented research. The other authors have nothing to declare.
Funding
This work was financially supported by a grant of the Catharina Research Fund, which was used for the additional data collection, and had no involvement in relation to the presented outcomes.
Ethics approval
According to the Central Committee on Research involving Human Subjects, this type of study does not require approval from an ethics committee.
Informed consent
Informed consent was not required for the present study.
