Abstract
Background
Protocols for treating recurrent Clostridium difficile infection (rCDI) through faecal microbiota transplantation (FMT) are still not standardised. Our aim was to evaluate the efficacy of different FMT protocols for rCDI according to routes, number of infusions and infused material.
Methods
MEDLINE, Embase, SCOPUS, Web of Science and the Cochrane Library were searched through 31 May 2017. Studies offering multiple infusions if a single infusion failed to cure rCDI were included. Data were combined through a random effects meta-analysis.
Results
Fifteen studies (1150 subjects) were analysed. Multiple infusions increased efficacy rates overall (76% versus 93%) and in each route of delivery (duodenal delivery: 73% with single infusion versus 81% with multiple infusions; capsule: 80% versus 92%; colonoscopy: 78% versus 98% and enema: 56% versus 92%). Duodenal delivery and colonoscopy were associated, respectively, with lower efficacy rates (p = 0.039) and higher efficacy rates (p = 0.006) overall. Faecal amount ≤ 50 g (p = 0.006) and enema (p = 0.019) were associated with lower efficacy rates after a single infusion. The use of fresh or frozen faeces did not influence outcomes.
Conclusions
Routes, number of infusions and faecal dosage may influence efficacy rates of FMT for rCDI. These findings could help to optimise FMT protocols in clinical practice.
Key summary
Faecal microbiota transplantation (FMT) is highly effective against recurrent Clostridium difficile infection (rCDI). However, there is still no clear evidence supporting the superiority of one working protocol over another. Routes of delivery, number of infusions and faecal dosage may influence efficacy of FMT for rCDI. These findings may be useful to optimise FMT protocols in clinical practice.
Background
Clostridium difficile infection has been recently increasing in incidence, severity, mortality and likelihood of recurrence, and represents a significant burden for healthcare systems worldwide. 1 A considerable body of evidence shows that faecal microbiota transplantation (FMT) is highly effective for the treatment of recurrent C. difficile infection (rCDI),2–4 and it has been recommended for this condition by both the European Society for Microbiology and Infectious Disease and the American College of Gastroenterology.5,6
Despite the development of guidelines on indications and methodology,7,8 and the establishment of stool banks, 9 FMT is still not a standardised procedure. Current protocols differ in several aspects, including route of delivery, the timing and number of infusions, and the quantity and quality (fresh or frozen material) of infusate. To date, there is still no clear evidence supporting the superiority of one protocol over another for the treatment of rCDI. In two previous meta-analyses, FMT was shown to be an effective treatment for rCDI, independently of preparation and route of delivery.4,10 Until recently, single-infusion FMT (SIF) has been commonly accepted to be a satisfactory option for the treatment of rCDI; however, multiple-infusion FMT (MIF) is demonstrating even higher cure rates than SIF.3,4,10–12 Taking into account the increasing worldwide burden of rCDI and the rising demand for rCDI therapies, the standardisation of protocols is urgently needed.
The aim of this meta-analysis was to evaluate the efficacy of different FMT protocols for rCDI, based on different routes of delivery, the number of infusions, and the quantity and quality (fresh or frozen faeces) of infusate, to let physicians offer the best approach to their patients in clinical practice, according to local facilities.
Methods
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist.

PICOS stands for: P = patient or population; I = intervention; C = comparison, control or comparator; O = outcome; S = study design.
Eligibility criteria
We considered eligible all original reports with the following characteristics: (a) inclusion of human subjects of any age treated with FMT for rCDI; (b) a working protocol offering multiple faecal infusions to patients if SIF failed to cure rCDI; and (c) clear reporting of efficacy outcomes after single faecal infusion and after overall infusions, respectively, after a minimum follow-up of 8 weeks. This last criterion has been included as evidence-based guidelines recommend that patients with CDI should be followed-up for at least 8 weeks after therapy (including FMT) to determine treatment response and address recurrence.5,6
Studies investigating other microbiota modulators than FMT (including synthetic microbiota suspensions or probiotics), as well as those including subjects receiving FMT for disorders other than rCDI, were excluded.
Information sources and search strategy
A literature search was performed using PubMed Central/Medline, Embase, SCOPUS, Web of Science (ISI) and the Cochrane Library, which were searched systemically for records up to 31 May 2017. Keywords included for the search are available as supplementary material. Database searches were supplemented with literature searches of reference lists from potentially eligible articles by three reviewers (G.I., M.M. and C.S.) to find additional studies.
Both randomised and nonrandomised studies were considered, without year-span restriction. For randomised controlled trials (RCTs), we collected only data from the FMT arm. We excluded case reports, case series involving less than 10 subjects and studies presented only as abstracts at symposia, as well as studies published in other languages than English. Both paediatric and adult subjects were included. The bibliographies of relevant papers (based on title and abstracts) were handsearched. If needed, authors were contacted and asked for clarifications or missing information about their findings.
Study selection
Two investigators (G.I. and M.M.) independently reviewed and checked titles and abstracts of all retrieved studies. Studies fulfilling the eligibility criteria were selected for analysis. In the case of doubt, full texts of articles were reviewed. A third author (G.C.) arbitrated in all cases of a lack of agreement.
Data extraction and quality assessment
Data extraction and quality assessment are included as supplementary material.
Data synthesis and statistical analysis
Data synthesis and statistical analysis are included as supplementary material.14–18
Results
Study selection and characteristics of included studies
Figure 1 shows the flow diagram of study selection. Fifteen studies, published between 2012 and 2017, were included for the final analysis,4,11,19–31 including two RCTs,4,11 nine retrospective case series19–22,24–27,29 and four prospective case series.23,28,30,31 Most were single-centre studies and three were multicentre studies.11,20,25 One RCT
3
was not considered within the final analysis as its cohort was included in a further paper.
31
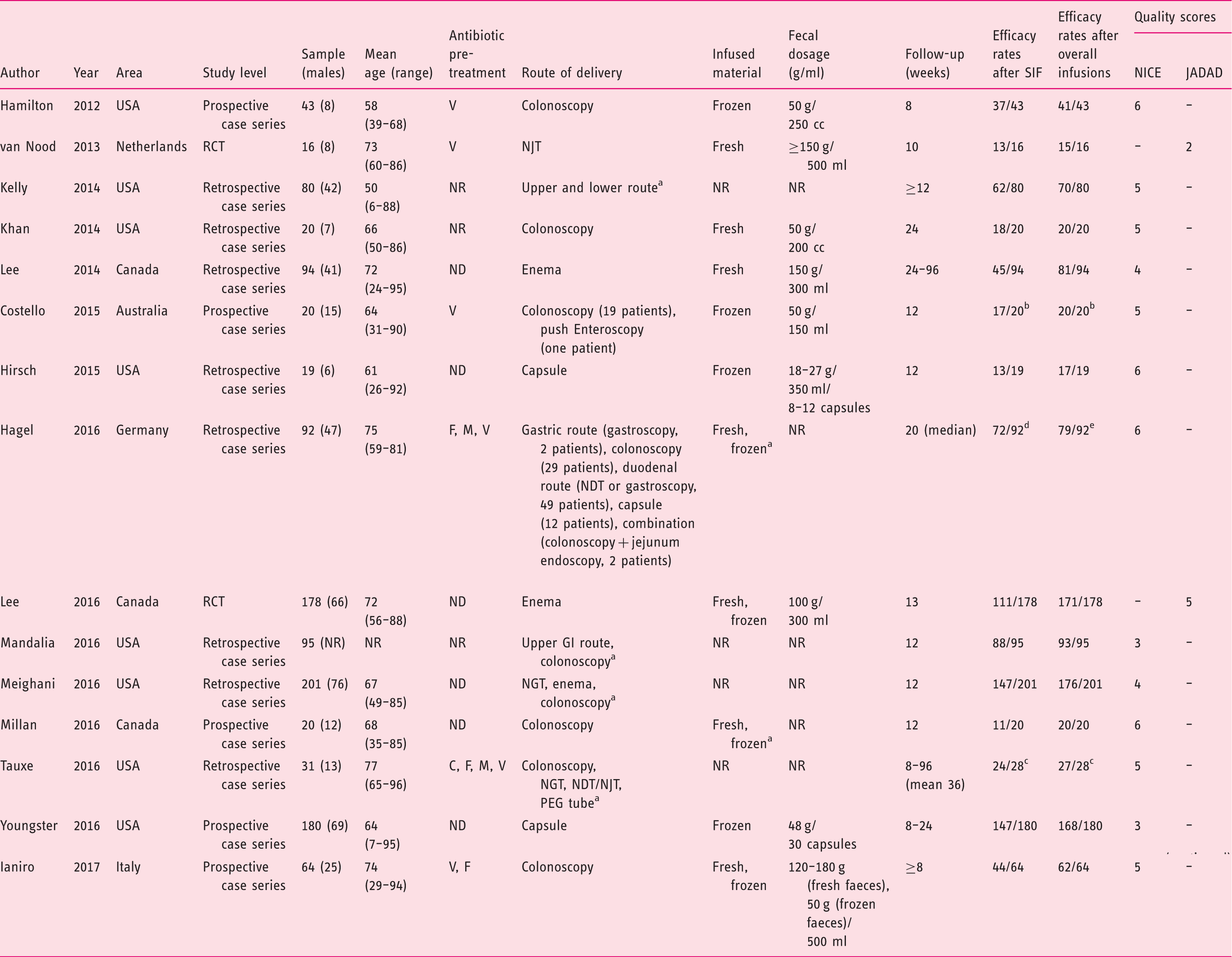
A summary of included studies with individual quality assessment is available in Table 2. Eight studies were carried out in the United States of America,19–21,24,26,27,29,30 three in Canada,11,22,28 three in Europe4,25,31 and one in Australia.
23
Finally, a visual assessment of funnel plots (Supplementary Figure 1) and the Egger’s test for publication bias (SIF p = 0.54; overall infusions p = 0.09) showed no evident risk of having missed studies from the literature.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram of the search process. Summary of included studies. C: clindamycin; F: fidaxomicin; GI: gastrointestinal; M: metronidazole; ND: not detailed; NDT: nasoduodenal tube; NGT: nasogastric tube; NJT: nasojejunal tube; NR: not reported; PEG: percutaneous endoscopic gastrostomy; RCT: randomized controlled trial; SIF: single-infusion faecal microbiota transplantation; V: vancomycin. Separate data are not available. All failures after single-infusion faecal microbiota transplantation occurred when colonoscopy was used as route of delivery; all further faecal infusions were performed by colonoscopy. Three patients from the original cohort of 31 subjects did not receive further faecal microbiota transplantation after failure of first procedure, and were removed from the analysis. Gastric delivery = 2/2; colonoscopy = 24/29; duodenal/jejunal delivery = 35/49; capsule = 10/12; combination = 2/2. Colonoscopy = 28/28 (one successful secondary response was obtained by combination faecal microbiota transplantation - colonoscopy + jejunum endoscopy, and was excluded); duodenal/jejunal delivery = 36/49; capsule = 11/12.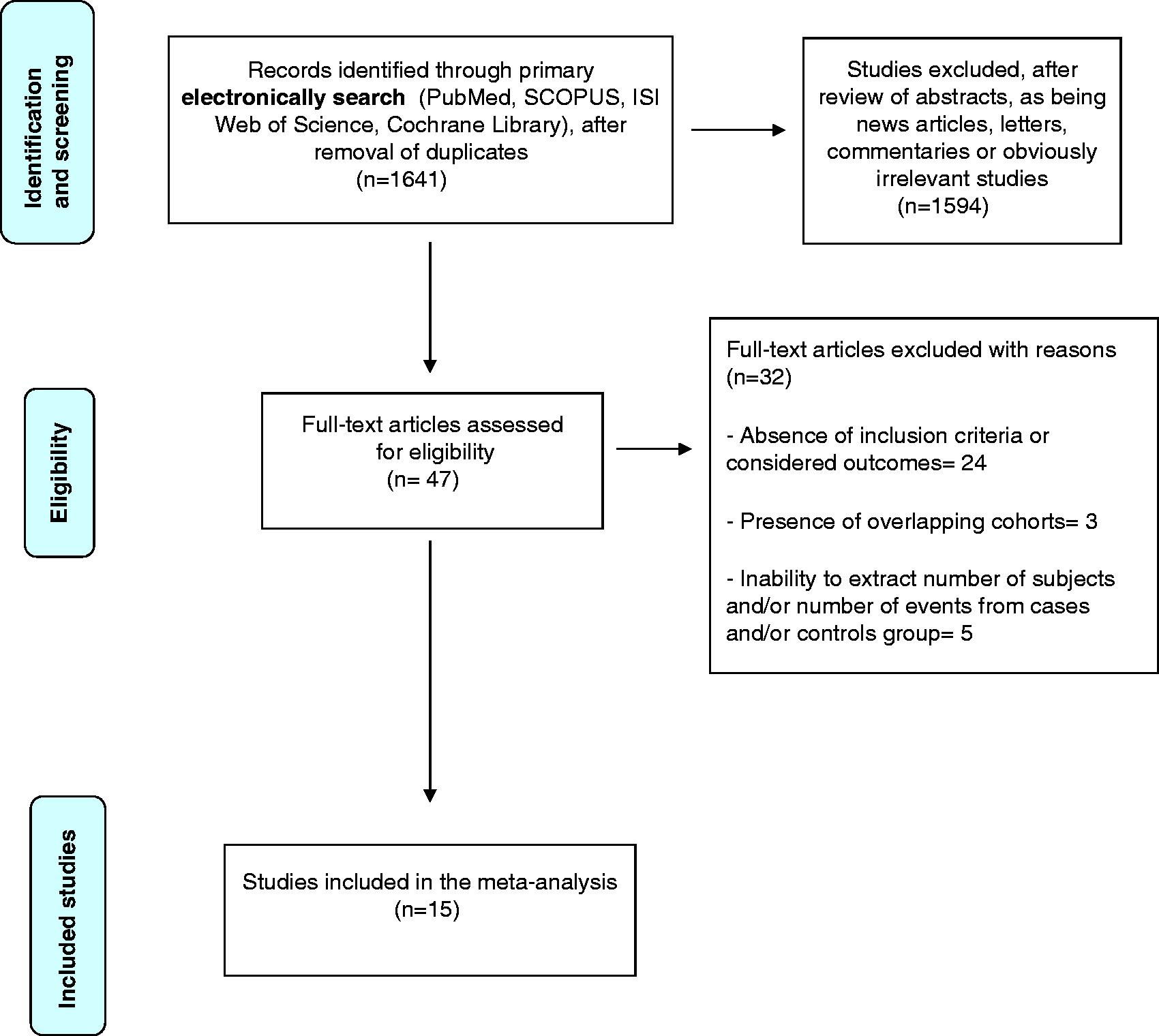
Characteristics of patients and of FMT protocols
Characteristics of patients and of FMT protocols are included as supplementary material.
Efficacy outcomes of FMT
Efficacy outcomes of FMT are summarised in Figure 2 and Figure 3. Overall pooled estimates of efficacy rates were, respectively, 76% (95% confidence interval (CI) = 69–82%) for SIF, with considerable heterogeneity among studies (I2 = 83.5%, 95% CI = 73.7–88.5%), and increased to 93% (95% CI = 90–95%) for overall infusions, with lower but still substantial heterogeneity among studies (I2 = 61.5%, 95% CI = 21.8–76.7%).
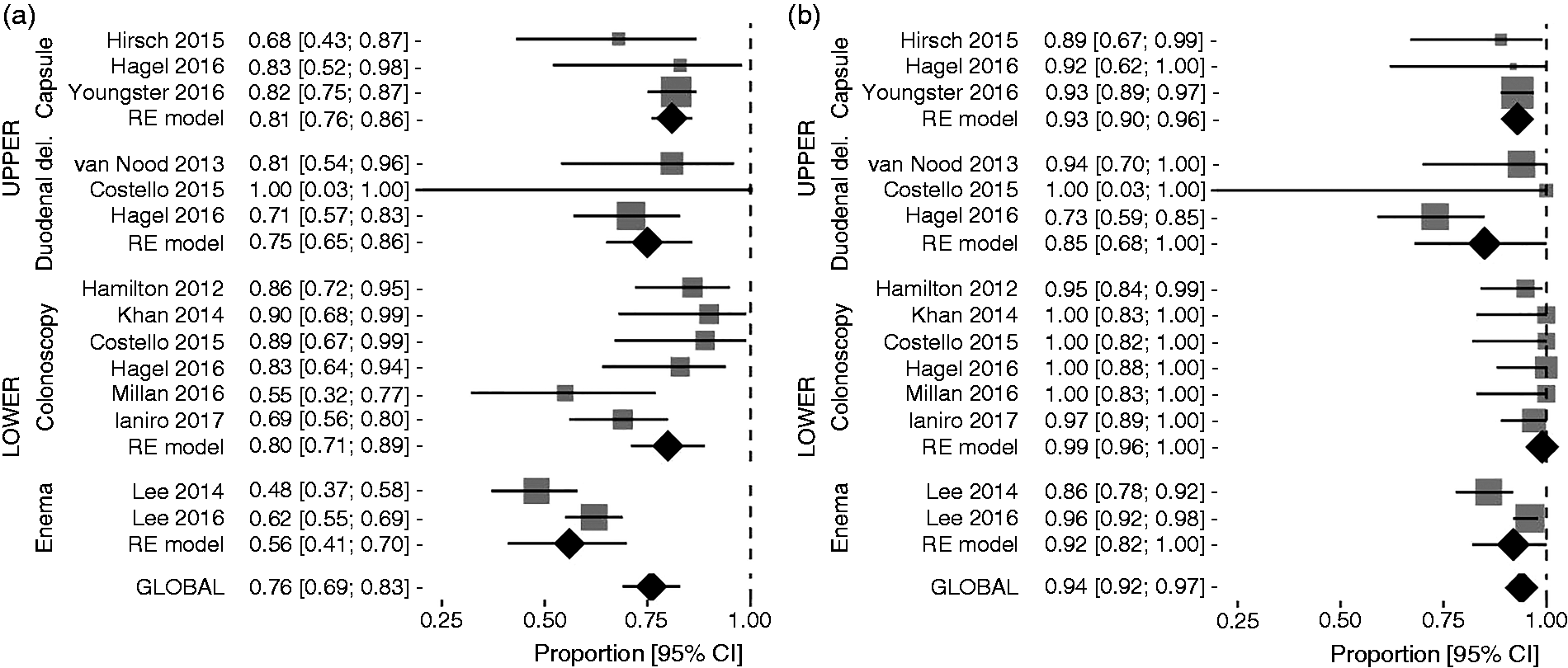
Proportion meta-analysis plot of Clostridium difficile infection resolution rates for single-infusion faecal microbiota transplantation (a) and overall infusions (b). Proportion meta-analysis plot of Clostridium difficile infection resolution rates for single-infusion faecal microbiota transplantation (a) and overall infusions (b) according to different routes of delivery.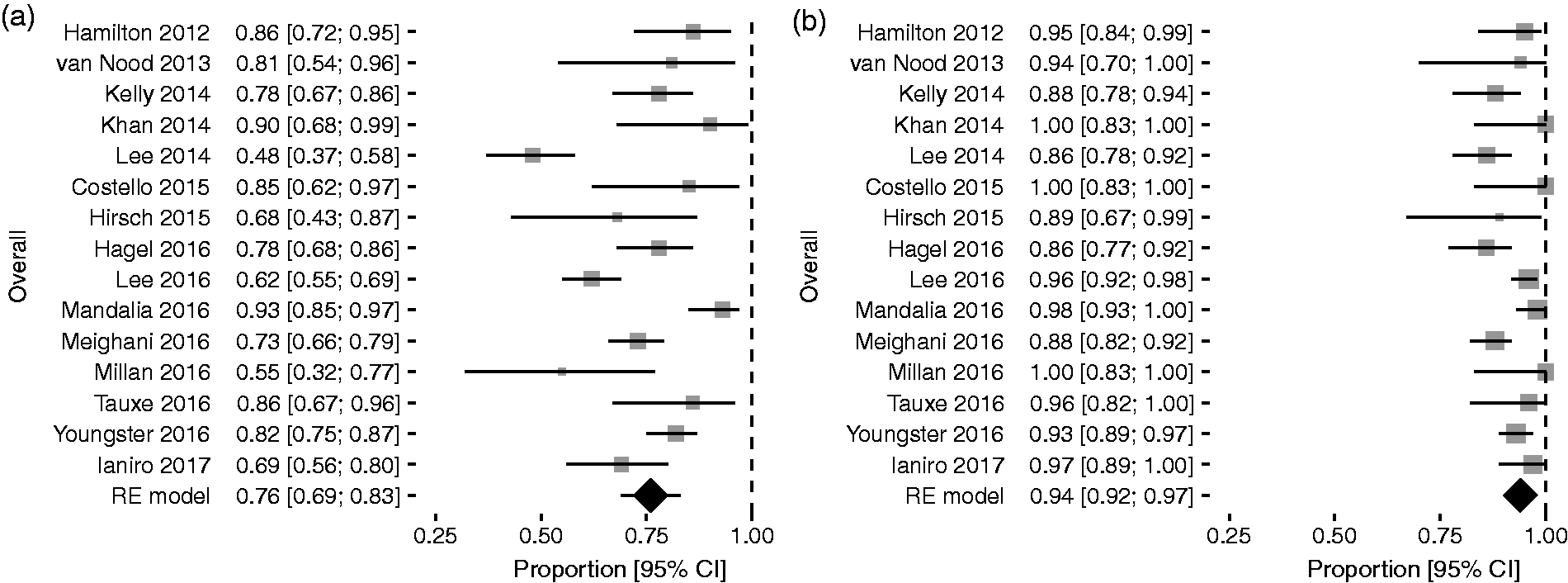
Upper route of delivery
Among patients receiving FMT through upper routes (seven studies, including nasogastric tube, nasojejunal tube, upper endoscopy/push enteroscopy and capsule), the efficacy rate of SIF was 79% (95% CI = 74–83%) without heterogeneity among studies (I2 = 0%, 95% CI = 0–64.1%), while that achieved by overall infusions was 88% (95% CI = 78–95%), with substantial heterogeneity (I2 = 60.5%, 95% CI = 0–83,1%).
Capsule
Capsule FMT showed 80% resolution rate with SIF (95% CI = 75–85%) (Figure 3a) and 92% after overall infusions (95% CI = 88–96%) (Figure 3a), with no heterogeneity determined among studies in both analyses (I2 = 0% for both of them).
Duodenal delivery
Duodenal delivery accounted for the lowest difference between efficacy rates achieved by SIF (73%, 95% CI = 62–83%, without heterogeneity (I2 = 0%)) (Figure 3(a)) and overall infusions (81%, 95% CI = 65–93%, with moderate heterogeneity (I2 = 35.8%)) (Figure 3b).
Lower route of delivery
Patients treated by lower routes of delivery (eight studies, including enema and colonoscopy) experienced similar rCDI resolution rates to those treated with upper routes after SIF, as the pooled estimate of efficacy rate was 72% (95% CI = 61–82%) with considerable heterogeneity (I2 = 82.3%, 95% CI = 62.9–89.3%). However, the efficacy rate of overall infusions performed by lower route (96%, 95% CI = 92–98%, with substantial heterogeneity (I2 = 54.8%, 95% CI = 0–77.7%) was higher than that of upper-route MIF.
Colonoscopy
When we analysed efficacy outcomes according to different routes of delivery, colonoscopy was the most effective route, as cure rates were, respectively, 78% (95% CI = 68–87%) with substantial heterogeneity (I2 = 60.7%, 95% CI = 0–81.9%) after SIF (Figure 3a) and 98% (95% CI = 95–99%), without heterogeneity (I2 = 0%, 95% CI = 0–61%) after overall infusions (Figure 3b).
Enema
Efficacy rates of enema FMT after SIF (56%, 95% CI = 41–69%) (Figure 3a) were almost doubled by the use of multiple faecal infusions (92%, 95% CI = 79–98%) (Figure 3(b)), with considerable heterogeneity among studies at both analyses (I2 = 80.8% for SIF and 87.6% for overall infusions, respectively).
Quality of included studies
The quality of included studies did not appear to influence the efficacy outcomes as they were comparable, both for SIF and for overall infusions between high-quality studies (SIF = 75%, 95% CI = 68–81%; overall infusions = 93%, 95% CI 90–96%) and low-quality studies (SIF = 77%, 95% CI = 62–89%; overall infusions = 93%, 95% CI = 87–97%).
Faecal material
The type of infused material did not influence efficacy outcomes. Frozen faeces achieved 77% resolution rate (95% CI = 68–85%) after SIF and 94% resolution rate (95% CI = 91–94%) overall, and fresh faeces obtained 69% resolution rate (95% CI = 55–82%) after SIF and 94% resolution rate (95% CI = 88–98%) after overall infusions, respectively. The additional subgroup analysis for routes of delivery was possible only for studies using lower routes of delivery, without showing any significant difference, neither for frozen (SIF = 77%, 95% CI = 68–85%; overall infusions = 94%, 95% CI = 91–96%) nor for fresh faeces (SIF = 69%, 95% CI = 55–82%; overall infusions = 94%, 95% CI = 88–98%).
Meta-regression results
Univariate logistic regression analysis was used to explore and explain potential sources of heterogeneity among the studies.
Meta-regression analyses for single-infusion faecal microbiota transplantation.
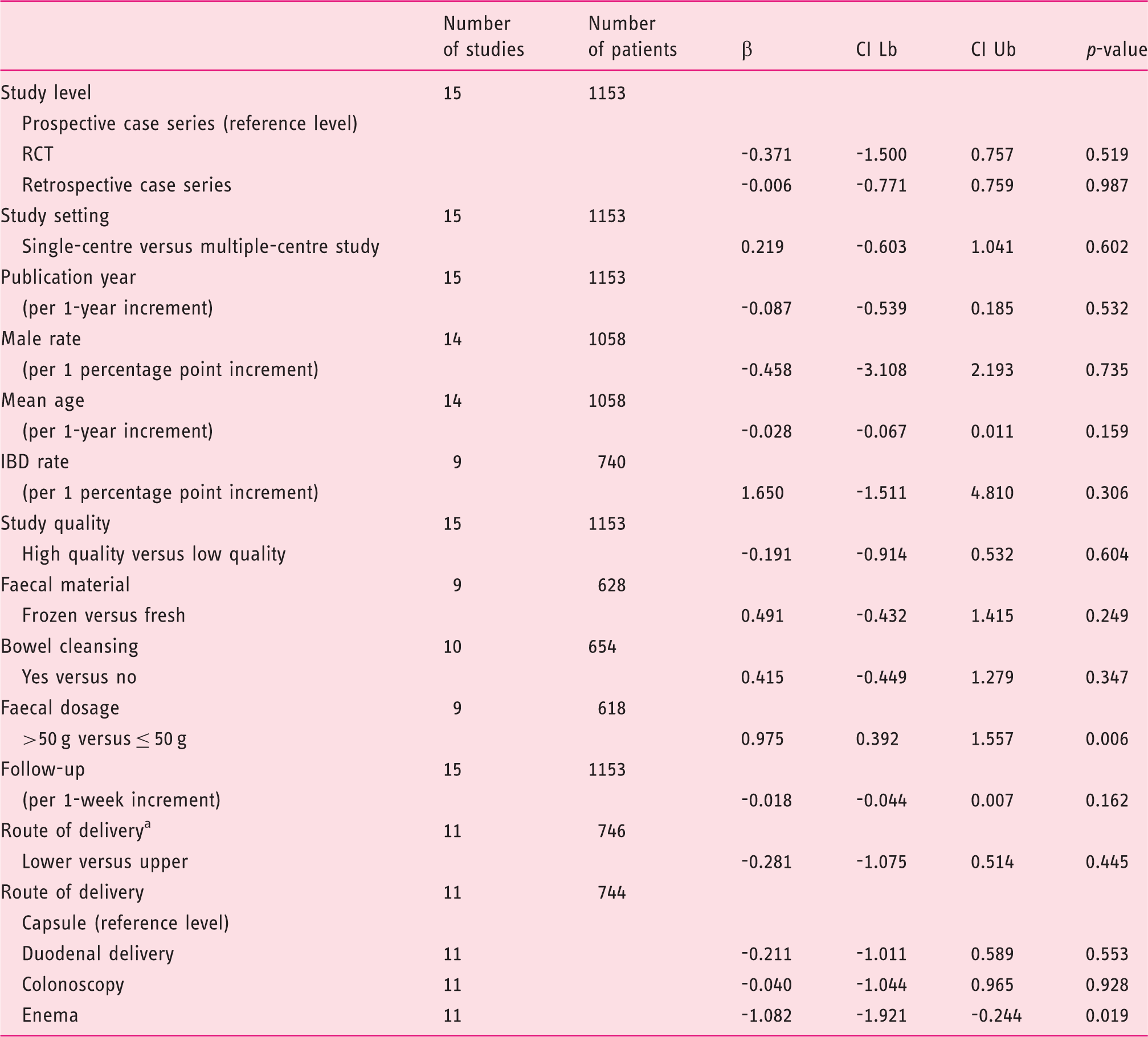
CI Lb: confidence interval lower bound; CI Ub: confidence interval upper bound; IBD: inflammatory bowel disease; RCT, randomized controlled trial.
Upper routes include: capsule and duodenal delivery. Lower routes include colonoscopy and enema.
Meta-regression analyses for overall infusions.
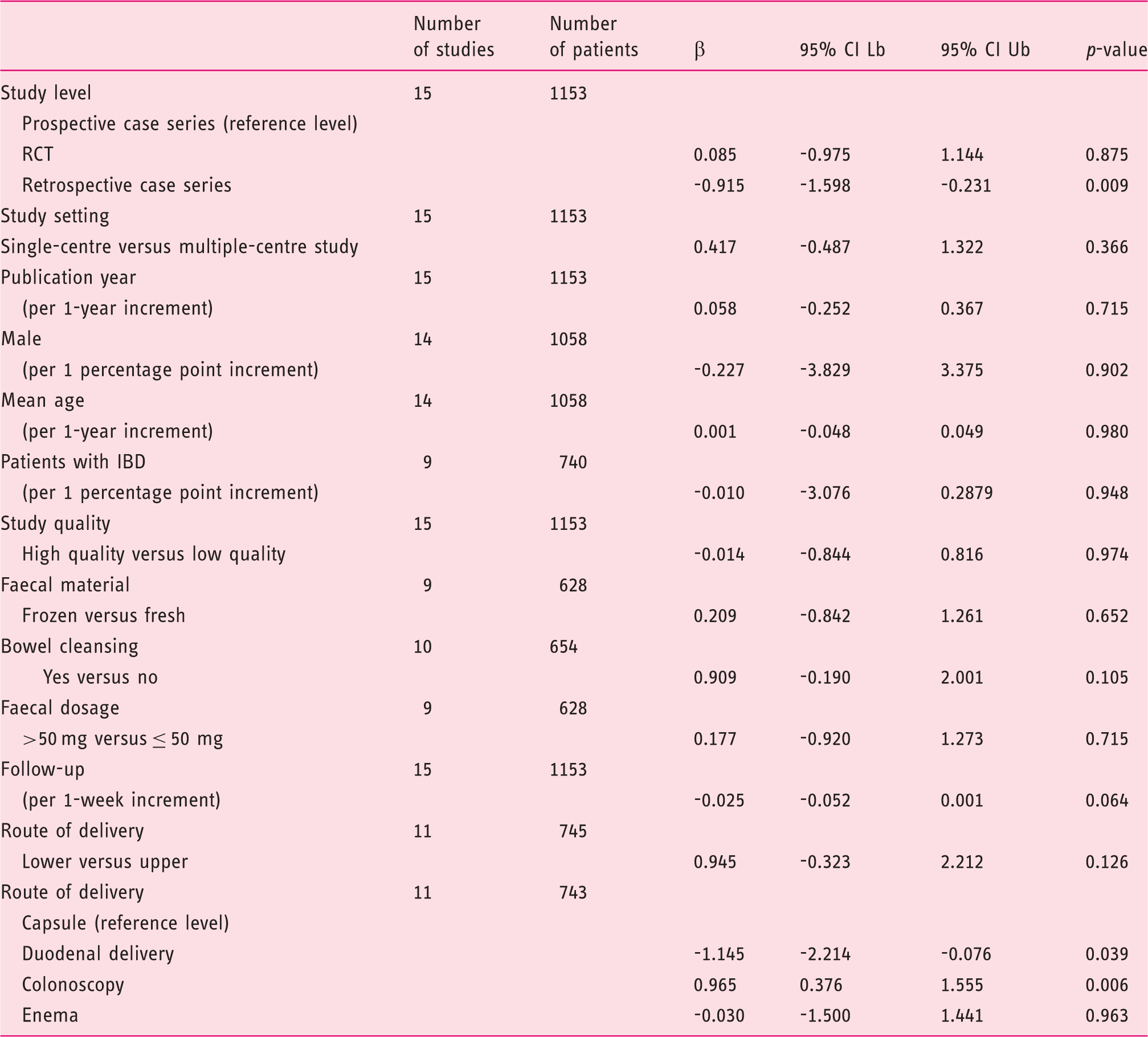
CI Lb: confidence interval lower bound; CI Ub: confidence interval upper bound; IBD: inflammatory bowel disease; RCT, randomized controlled trial.
Discussion
This meta-analysis shows that although overall efficacy rates of FMT for the treatment of rCDI are impressive (overall response 93%, 95% CI = 90–95%), they appear to be influenced by several characteristics of working protocols.
In our study, the rCDI resolution rates (85–90%) observed in previous systematic reviews and meta-analyses4,10 can only be confirmed when the overall number of infusions was evaluated, but not after SIF, for most routes of delivery.
Both the subanalysis for different routes and the meta-regression analysis led to interesting results. First, multiple infusions increased the efficacy rates of FMT overall and in each different subgroup. This result is expected, and matches another observation of this study, that is the significant association between the use of low faecal dosages (≤50 g) with lower efficacy rates after SIF (p = 0.006), but not after overall infusions (p = 0.715). Both these findings point out the importance of providing a sufficient biomass to restore a healthy microbiota, either by infusing a large amount of faeces in one time or by repeating infusions.
Moreover, the efficacy rates of FMT and the efficacy gap between SIF and overall infusions changed according to different routes of delivery.
The duodenal delivery (including nasoduodenal/nasojejunal tube, upper endoscopy and enteroscopy) was associated with lower efficacy rates (p = 0.039) at overall analysis. Moreover, it accounted for the least increase of efficacy between single (73%) and overall infusions (81%). These results could explain the lower use of the duodenal route in our analysis and worldwide. However, duodenal FMT is significantly more effective than standard antibiotic therapy, and this route was successful even in treating severe clinical pictures of CDI. 32 Therefore, it is still difficult to find evidence for a definitive recommendation.
In our study, capsule FMT was found to be highly effective (80% after SIF, 92% overall). This result was recently confirmed in an RCT showing that single treatment with capsules is not inferior to colonoscopy SIF in preventing rCDI. 33 Its minimal invasiveness makes it the most suitable route to disseminate FMT. However, its widespread use is still curbed by its cumbersome preparation process. Moreover, current capsule FMT protocols include a high number of capsules needing to be swallowed for a single 2-day course of treatment.25,33 Future strategies to disseminate this approach may rely on the development of more specialist FMT centres, or other enterprises, equipped for the production and dispatch of capsules, as well as the improvement of treatment protocols, for example to decrease the number of capsules required for an effective single dose.
Overall cure rates of enema (92%, 95% CI = 79–98%) were almost twice those of enema-SIF (56%, 95% CI = 41–69%). Moreover, at meta-regression analysis, enema was associated with lower efficacy rates after single infusion (p = 0.019), but not overall (p = 0.963). These results confirm findings reported in a recent RCT, where enema-SIF obtained comparable CDI resolution rates to vancomycin therapy, 34 and cannot be related, at least in our analysis, to faecal dosage, because all included enema studies used at least 100 g of faeces per infusion. This observation suggests that other protocol details, including the colonisation of the whole colon (not provided by enema), can influence efficacy rates of FMT, and also that enema FMT protocols may a priori include repeated faecal infusions.
Colonoscopy was associated with higher efficacy rates (98%, p = 0.006) at overall analysis. Although this technique is invasive and could be unsuitable for critically ill patients, it allows the infusion of large volumes of faeces throughout the whole colon, as well as being able to identify some risk factors for FMT failure, such as pseudomembranous colitis or inadequate bowel preparation. 31
Finally, the type of infused material (frozen or fresh faeces) appeared not to influence efficacy outcomes of FMT, as already found in a large RCT. 11
This is, to our knowledge, the first meta-analysis to delve into the efficacy of different FMT protocols for the treatment of rCDI. In previous meta-analyses,4,10 lower faecal delivery was more effective than upper faecal delivery. By contrast, in our study, we found no significant differences between the two modalities at meta-regression analysis. This discrepancy could be explained by the inclusion of different studies in each meta-analysis. However, we showed that FMT protocols could differ significantly in their efficacy rates. Therefore, pooling together different routes based on the upper or lower delivery may not be appropriate.
We acknowledge that some of our findings should be handled cautiously, because of several limitations. First, we were not able to evaluate known risk factors for FMT failure (e.g. severe CDI, inadequate bowel preparation, etc.) in the different studies, as individual data were not always available. Additionally, most studies were of retrospective design, therefore requiring that associated risks of selection and recall bias should be considered. At meta-regression analysis, retrospective studies were also significantly associated with lower overall efficacy rates (p = 0.009), suggesting that the risk of an information bias, as well as that of a selection bias, could not be excluded.
Included studies differed each other with regard to inclusion criteria, antibiotic pre-treatment, dosage and quality of infusion material, route of delivery and follow-up length, limiting the application of our results to new populations and settings. In particular, in two studies,12,22 antibiotics were continued between different procedures, potentially increasing efficacy rates of FMT. Additionally, in some studies, we were not able to retrieve data on relevant components of working protocols, such as the amount and the type of faecal material used. This finding confirms data from a recent systematic review, which has recently identified that most FMT studies poorly reported details of methodological protocols. 35
We are also aware that the results for the enema subgroup, for the duodenal subgroup and for the capsule subgroup should be treated cautiously, as the analysis was based on only two studies or three studies.
Moreover, through the present meta-analysis, we were able to evaluate only the repetition of faecal infusions as a therapeutic option to treat FMT failures. Other suggested approaches to manage relapses after FMT include antibiotic treatment alone (with a theoretical preference for fidaxomicin), provided that the initial diagnosis of CDI was correct. 36 Although evidence is lacking, the use of a different donor may be also considered.
Finally, although we found no evident risk of having missed studies from the literature, we cannot exclude the possibility that a publication bias could exist in the reported literature.
In conclusion, this meta-analysis shows that routes of delivery, number of infusions and faecal dosage may influence the effectiveness of FMT for rCDI. Our findings could be useful for the design of effective standardised treatment approaches, which should be tailored according to local facilities and the needs of each patient.
Supplemental Material
Supplemental material for Efficacy of different faecal microbiota transplantation protocols for Clostridium difficile infection: A systematic review and meta-analysis
Supplemental material for Efficacy of different faecal microbiota transplantation protocols for Clostridium difficile infection: A systematic review and meta-analysis by Gianluca Ianiro, Marcello Maida, Johan Burisch, Claudia Simonelli, Georgina Hold, Marco Ventimiglia, Antonio Gasbarrini, Giovanni Cammarota and the CENSUR Working Survival Group in United European Gastroenterology Journal
Footnotes
Acknowledgements
The authors sincerely thank Tony Bruns, Dina Kao, Stefan Hagel, Karel Madsen, Sam Costello, Maria Vehreschild and Ilan Youngster for their precious help in providing details of their published studies.
Author contributions
GI and GC conceived and designed the study protocol. GI, MM and CS performed the literature search. GI and MM performed the study selection, data extraction and the quality assessment. GC arbitrated on the study selection in all cases of a lack of agreement between GI and MM. JB and MV performed the statistical analysis. GI, MM, JB, GH, AG and GC interpreted the data. GI, MM, JB, MV, GH and GC wrote the original draft. All authors revised the draft critically for important intellectual content and approved the final version of the paper, including the authorship list.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Ethics approval
Ethics approval was not necessary for this article.
Informed consent
Informed consent was not necessary for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
