Abstract
Background and aims
Apparent aspiration is a notable adverse event during gastrointestinal endoscopy under sedation (GIES), but data about inapparent aspiration are scarce. Generally, particularly older patients are at higher risk of suffering from adverse events.
Objective
The objective of this article is to determine the risk of pneumonia, lower respiratory tract infection (LRI) and systemic inflammatory activation after GIES, especially in patients of at least 65 years.
Methods
The retrospective case-control study included 250 patients undergoing GIES and assigned age-, gender- and time of performance-matched controls without invasive procedure or sedation (ratio 1:1).
Results
On day 3 patients of advanced age presented with both pneumonia and LRI more often (2.6 vs. 0.0%, p = 0.041 and 7.8 vs. 2.5%, p = 0.034, respectively). In general, several inflammatory parameters increased significantly after GIES (i.e. white blood cell count (increase of ≥ 25%) 18.6 vs. 6.9%, p < 0.001), leading to more frequent antibiotic treatment (6.8 vs. 1.6%, p = 0.004). The effects were less pronounced on day 7.
Conclusions
Patients of advanced age carry an increased risk of pneumonia and LRI after GIES. Patients are generally more likely to feature inflammation and to receive antibiotic treatment.
Keywords
Key summary
Summarize the established knowledge on this subject
Aspiration is a notable adverse event during gastrointestinal endoscopy under sedation, but data about inapparent aspiration are scarce. Patients of advanced age are at higher risk to experience adverse events. Inflammatory reactions are common after gastrointestinal endoscopy. Gastrointestinal endoscopy is associated with pneumonia and lower respiratory tract infections in patients of advanced age.
What are the significant and/or new findings of this study?
Introduction
Esophagogastroduodenoscopy (EGD) and colonoscopy are frequently performed diagnostic and therapeutic endoscopic procedures of the gastrointestinal tract. 1 An estimated 15 million colonoscopies were performed in the United States of America (USA) in 2012. 2 The number of EGDs is even higher. In Germany, for instance, where a routine colonoscopy screening program is implemented, the number of EGDs exceeds that of colonoscopies by nearly twofold. 3 More than 90% of gastrointestinal endoscopies are performed under sedation4–8 to reduce the patient’s anxiety, pain and inconvenient experiences, as well as improving its quality and outcome.4,9–12
Therefore, in addition to common risks associated with gastrointestinal endoscopy itself, such as perforation, bleeding and abdominal discomfort,1,13–15 adverse events related to sedation have to be considered,16,17 among which cardiovascular and pulmonary events predominate.1,12,18,19 The attenuation of protective airway reflexes and reduction of the tone of the upper and lower esophageal sphincters,1,11,18,20–22 the patient’s position during endoscopy, 23 insufflation of air or fluid during colonoscopy 24 and the provocation of the urge to gag during EGD may contribute to regurgitation and aspiration of gastric contents. Since patients undergo EGD and colonoscopy without airway management, aspiration of gastric contents plays a decisive role in various studies. Agostoni et al. 12 showed apparent aspiration to occur in 0.16% of colonoscopies and 0.19% of EGDs under sedation in a prospective database of 17,999 procedures, but did not report the incidence of pneumonia. Cooper et al. 20 supported these findings in a population-based study by showing the relevance of sedation in a large cohort of 165,527 diagnostic colonoscopies: The risk of aspiration pneumonia identified by diagnosis codes varied from 0.10% without to 0.14% with sedation.
On the other hand, patients undergoing EGD experienced respiratory symptoms, such as coughing, shortness of breath and other respiratory adverse events within 24 hours more often in up to 5.3% of cases. 18 Since coughing during endoscopy has been attributed to an increased risk of aspiration-related postprocedural infection, 19 respiratory infections might be underreported. Additionally, elderly patients are determined not only as a high-risk group for gastrointestinal adverse events following colonoscopies, 25 but are also more likely to develop hospital-acquired pneumonia. 26
Hence, this retrospective study aimed to evaluate the frequency of pneumonia and lower respiratory tract infections (LRIs) and the development of surrogate parameters such as the development of C-reactive protein (CRP) and white blood cell (WBC) count, pulmonary infiltration and initiation of antibiotic treatment after routine endoscopy under sedation in general and in older patients in particular.
Materials and methods
Study population
Patients undergoing EGD alone or both EGD and colonoscopy under sedation during a hospital stay of at least three days between June 2005 and May 2015 were thought eligible for a retrospective age-, gender- and length of hospital stay-matched case-control study (see Table S-1, supplementary material). Laboratory parameters had to be available before (d0) gastrointestinal endoscopy under sedation (GIES) as well as three (d3) and/or seven days (d7) after endoscopy. Age- (± 2 years), gender- and time-matched (± 10%) inpatients who had neither obtained any invasive procedure nor sedation served as controls in a ratio of 1:1. The identification of eligible patients from the clinic’s electronic data system was realized by the inclusion criteria mentioned previously.
Exclusion criteria were defined as elevated inflammation parameters (WBC > 10,000/µl or < 4000/µl, CRP > 2 mg/dl), preexisting antibiotic treatment, preexisting pneumonia and diseases or conditions accompanied by increased risk of aspiration (see Table S-1, supplementary material). Additional exclusion criteria for the control group included any type of sedation, endoscopy (including bronchoscopy and transesophageal echocardiography) or surgery.
Endoscopy and sedation
All endoscopies were performed at the university hospital Berufsgenossenschaftliches Universitaetsklinikum Bergmannsheil gGmbH. Sedation was administered as non-anesthesiologist-administered sedation, according to national and international guidelines.4,10 Midazolam mono, propofol mono or a combination of both were used. Midazolam was usually given only at the beginning, while propofol was administered in boluses throughout the examination. The patient was monitored by a person who had the patient’s sedation as his or her sole task. Clinical data were obtained from the original endoscopy and sedation reports and the clinic’s electronic data system.
Objectives
The primary objective was the development of pneumonia as defined in national and international guidelines27–29 in general, and particularly in patients of at least 65 years of age. Development of LRI, as defined by the Centers for Disease Control and Prevention, 29 elevation of inflammation parameters (WBC and CRP, respectively), development of fever (≥ 38.3℃), initiation of antibiotic treatment, newly diagnosed pulmonary infiltration in X-ray or positive pathogen detection in blood cultures or sputum were regarded as secondary objectives. All objectives were collected on days 3 and/or 7.
Statistics
Statistical analysis was performed using SPSS, version 23.0 (SPPS Inc, USA). Arithmetic mean was used for evaluating metric variables and standard deviation for determining measures of variation. Nominal data were stated as absolute and relative frequencies.
Nominal variables and the dichotomous primary and secondary objectives were compared using the chi-squared (χ2) test. Metric variables were analyzed by using a two-sample t test and analysis of variance (ANOVA, subgroup analysis). Binary logistic regression was used to detect associations between potential risk factors and dichotomous objectives.
Results
Demographic data.
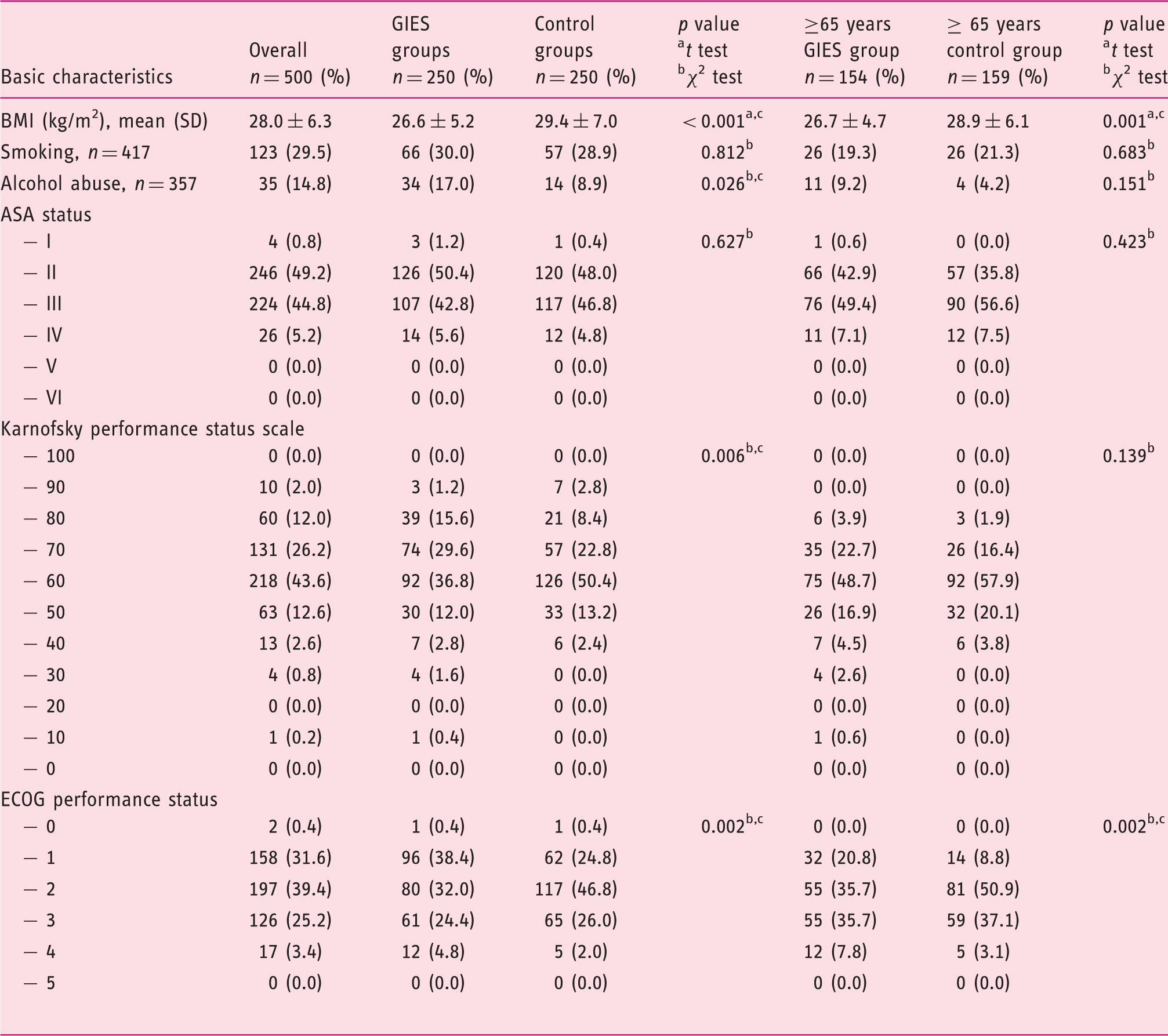
BMI: body mass index; ASA: American Society of Anesthesiologists; GIES: gastrointestinal endoscopy under sedation; GIES groups: esophagogastroduodenoscopy (EGD) only group (EoG) + EGD plus colonoscopy group (EpCG); control groups: EGD only control group (Eo-CG) + EGD plus colonoscopy control group (EpC-CG); ECOG: Eastern Cooperative Oncology Group. cSignificant with p < 0.05.
Characterization of endoscopy and sedation
Sedation characteristics.
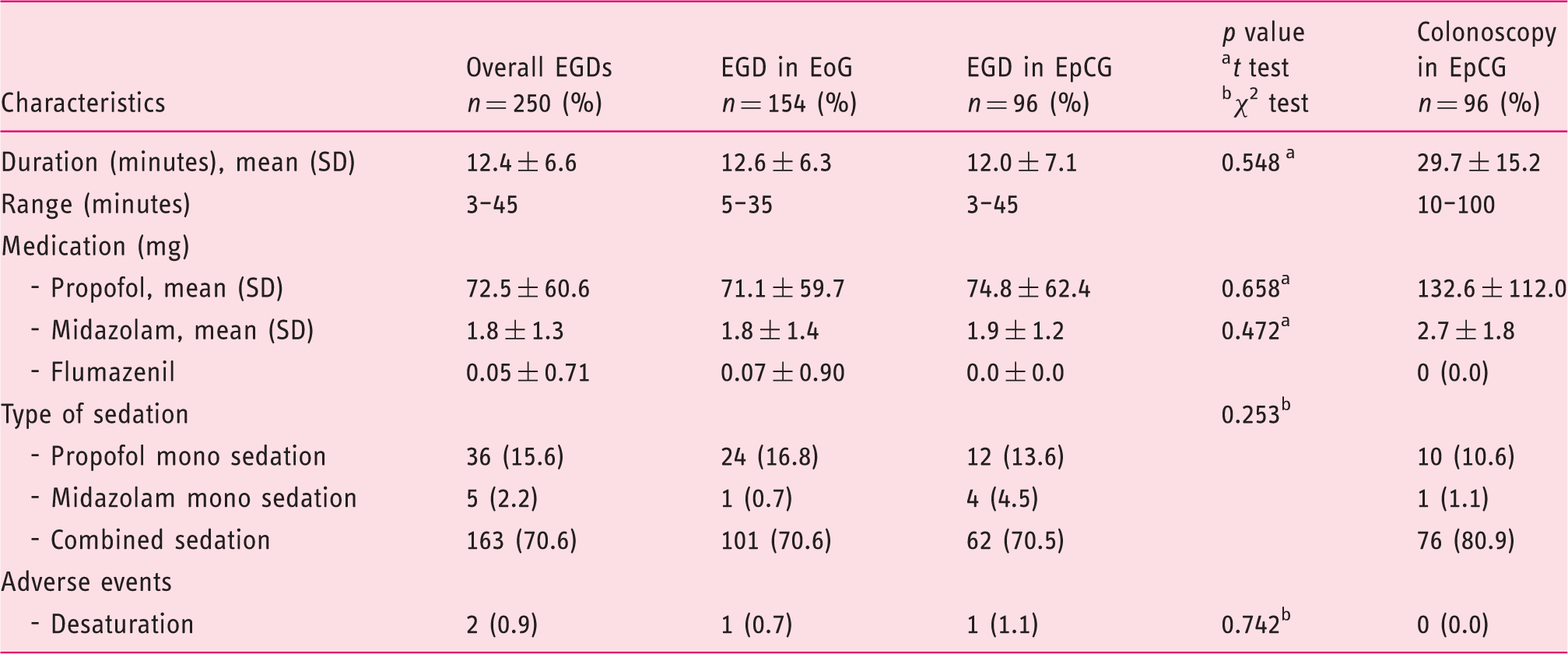
EoG: esophagogastroduodenoscopy (EGD) only group, EpCG: EGD plus colonoscopy group.
The mean duration of colonoscopy was 29.7 ±15.2 minutes. As with EGD, most colonoscopies were performed under combined propofol and midazolam sedation (n = 76, 80.9%). Propofol mono sedation was again the second choice (n = 10, 10.6%). No adverse events were recorded for colonoscopy. Detailed information is quoted in Tables S-6 and S-7, supplementary material.
Pneumonia and LRI
Pneumonia, lower respiratory tract infection, fever, antibiotic therapy, pulmonary infiltration, pathogen detection.
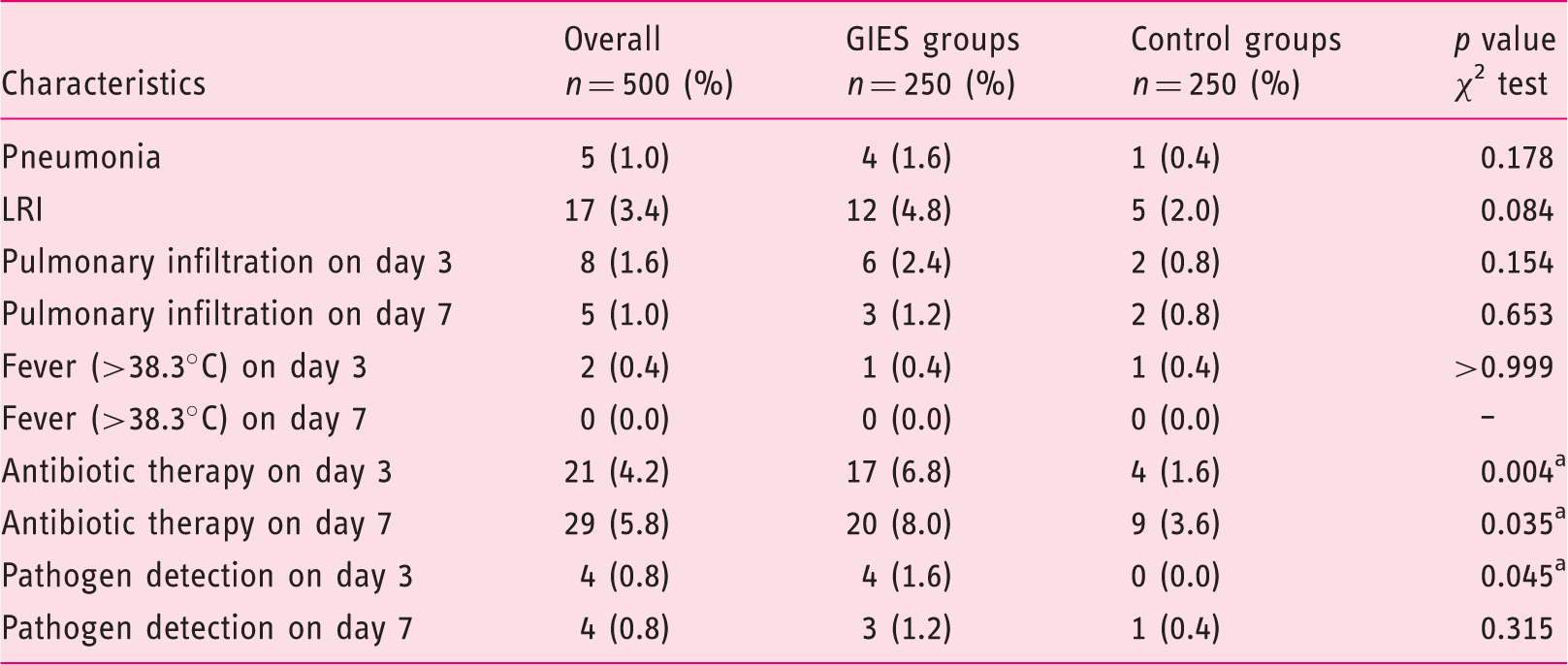
GIES: gastrointestinal endoscopy under sedation; GIES groups: esophagogastroduodenoscopy (EGD) only group (EoG) + EGD plus colonoscopy group (EpCG); control groups: EGD only control group (Eo-CG) + EGD plus colonoscopy control group (EpC-CG). aSignificant with p < 0.05. Pulmonary infiltration was detected by chest X-ray. Pathogen detection included blood and sputum.
Inflammation parameters
Although within reference range, the CRP and WBC on d0 differed significantly, with slightly higher values in the control group (see Table S-5, supplementary material). However, significant differences regarding the development of CRP ensued three days after the endoscopy with higher values in the GIES group (see Table S-8 and S-9, supplementary material). The results for day 7 varied markedly concerning the frequency of CRP > 2 mg/dl and the increase of CRP of at least 25% with a CRP level of > 1 mg/dl. Higher values and numbers were again detected in the GIES group among all parameters.
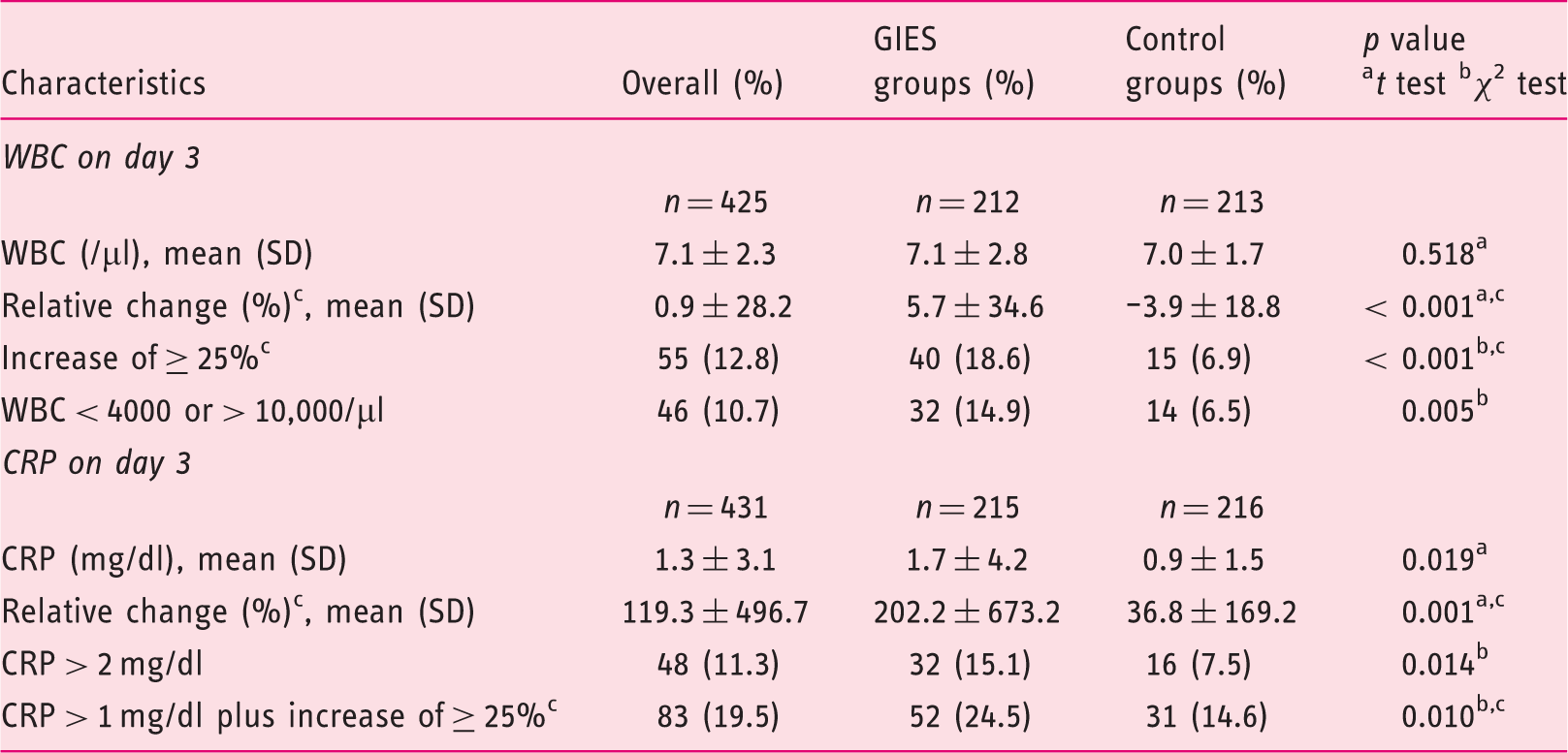
Similar differences were found for the WBC on day 3. The relative change of WBC, the frequency of WBC increase of at least 25% and WBC of < 4000 or > 10,000/ µl were significantly higher in the GIES group. Further details are outlined in Table 4.
Pneumonia and inflammation in the elderly
Pneumonia (n = 4 (2.6%) vs. 0 (0.0%), p = 0.041, χ2 test) and LRI (n = 12 (7.8%) vs. n = 4 (2.5%), p = 0.034, χ2 test) in patients of at least 65 years of age occurred significantly more often in the GIES group. Similar effects were found for antibiotic treatment (n = 13 (8.4%) vs. n = 3 (1.9%), p = 0.008, χ2 test), pathogen detection (n = 4 (2.6%) vs. 0 (0.0%), p = 0.041, χ2 test), the level of CRP (2.16 vs. 1.09 mg/dl, p = 0.022, t test), the frequency of CRP level > 2 mg/dl (n = 27 (20.5%) vs. n = 11 (8.4%), p = 0.005, χ2 test) and the increase of WBC of at least 25% (n = 24 (17.9%) vs. n = 11 (8.2%), p = 0.018, χ2 test) on day 3. The difference regarding pulmonary infiltration was not significant. Further details are outlined in Tables 4 and 5; additional information can be found in Tables S-10, S-11 and S-12 of the supplementary material.
Pneumonia and lower respiratory tract infection (LRI) in general, in older patients and referring to the gastrointestinal endoscopy under sedation (GIES) group (%). Development of inflammatory parameters on day 3. GIES: gastrointestinal endoscopy under sedation; GIES groups: esophagogastroduodenoscopy (EGD) only group (EoG) + EGD plus colonoscopy group (EpCG); control groups: EGD only control group (Eo-CG) + EGD plus colonoscopy control group (EpC-CG); WBC: white blood cell count; CRP: C-reactive protein. bSignificant with p < 0.05. cRelative change and increase were calculated from values prior to endoscopy. Pneumonia, lower respiratory tract infection, pulmonary infiltration, antibiotic therapy and pathogen detection in the elderly (≥ 65 years) on day 3. GIES: gastrointestinal endoscopy under sedation; GIES groups: esophagogastroduodenoscopy (EGD) only group (EoG) + EGD plus colonoscopy group (EpCG); control groups: EGD only control group (Eo-CG) + EGD plus colonoscopy control group (EpC-CG). aSignificant with p < 0.05. Pulmonary infiltration was detected by chest X-ray. Pathogen detection included blood and sputum.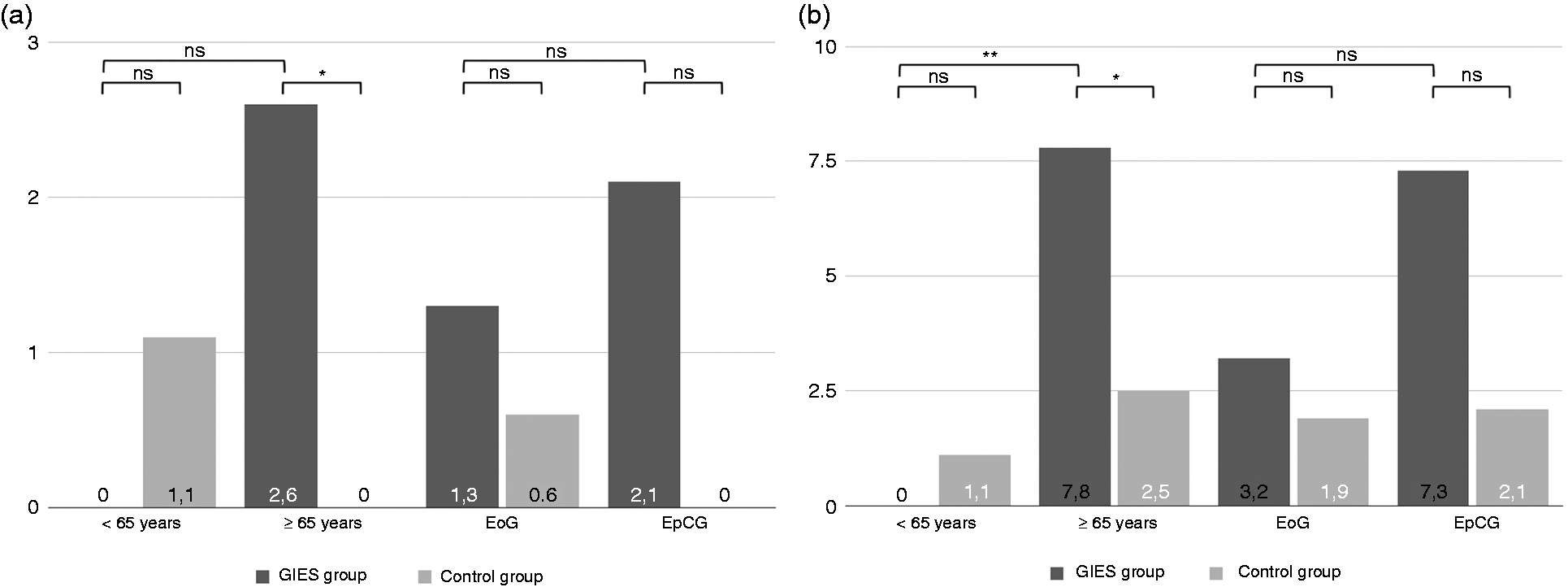

Influence of endoscopic approach
The EGD only group (EoG) and EGD only control group (Eo-CG), as well as the EGD plus colonoscopy group (EpCG) and EGD plus colonoscopy control group (EpC-CG), respectively, did not differ significantly regarding pneumonia, LRI, pulmonary infiltration, fever and pathogen detection on days 3 and 7 (details are outlined in Table S-13, supplementary material). Despite this, antibiotic treatment was more common in EpCG compared to EpC-CG on day 3 (n = 10 (10.4%) vs. n = 1 (1.0%), p = 0.005, χ2 test) and 7 (n = 10 (10.4%) vs. n = 2 (2.1%), p = 0.017, χ2 test), while EoG and Eo-CG did not vary markedly. Similarly, CRP criteria and the WBC indicated a significantly higher inflammatory reaction in EpCG compared to EpC-CG, especially on day 3. The EoG and Eo-CG did not differ markedly. Further details are outlined in Table S-14 and S-15 of the supplementary material.
Risk factor analysis
A multiple logistic regression model considering the whole cohort revealed significant effects for age (p = 0.024, odds ratio (OR) = 1.066 (95% confidence interval (CI95%) = 1.009–1.127)), body mass index (BMI) (p = 0.030, OR = 1.095 (CI95% = 1.009–1.189)), American Society of Anesthesiologists (ASA) status (ASA 1 and 2 were combined to low-grade comorbidity, ASA 3 and 4 to high-grade comorbidity) and endoscopy to predict the frequency of LRI with an OR of 3.378 (CI95% = 1.043–10.938) for endoscopy (p = 0.042) and 3.740 (CI95% = 1.015–13.775) for higher ASA grades (p = 0.047). The duration of endoscopy did not influence the risk of LRI. The same model regarding pneumonia did not reveal any significant effects, but did so for the development of inflammatory parameters such as CRP > 2 mg/dl.
BMI (p = 0.028, OR = 1.104 (CI95% = 1.011–1.206)) and endoscopy (p = 0.019, OR = 4.725 (CI95% =1.297–17.212)) were determined as relevant risk factors for LRI in the cohort of elderly patients. Again, no significant effects were found for the occurrence of pneumonia.
The manner of endoscopy did not influence the risk of pneumonia or LRI in a model considering the GIES group.
Discussion
A registry study rarely associated GIES with aspiration pneumonia, 20 but, as subsequently demonstrated, respiratory symptoms occur in more than 5%, 18 and even 20% of patients with apparent aspiration while obtaining EGD present with pulmonary infiltration immediately after endoscopy. 30 Notwithstanding, older patients are at particularly higher risk of developing endoscopy-related adverse events5,20,25 and nosocomial pneumonia. 26 The results of this retrospective observational study provide evidence that patients of at least 65 years of age have an increased risk of developing LRI and pneumonia after GIES.
The cohort of inpatients with indication of GIES has been carefully selected; all patients had to stay at least three days and laboratory results for CRP and WBC had to be available. Hence, a certain bias might have occurred. On the other hand, all cases have been thoroughly reviewed; pneumonia and LRI had been confirmed as standardized and defined in international guidelines.27–29 Under these assumptions, patients of advanced age developed pneumonia in 2.6% and LRI in 7.8% after GIES. Both infections proved statistically significant in comparison to a control group of age-, gender-, and length of hospital stay-matched inpatients (figure 1). Furthermore, we found several surrogate parameters, such as the initiation of antibiotic treatment and pathogen detection, to be significantly increased in patients who had GIES, although no significant differences could be found for pulmonary infiltration.
Inflammatory parameters
While pneumonia and LRI occurred more frequently in the elderly, the overall GIES group developed an inflammatory response indicated by both WBC and CRP levels that was not observed within the control group. Albeit the controls showed slightly higher values prior to the endoscopy, several inflammatory markers increased significantly after GIES, but not in the controls. Although the cohort of younger inpatients showed a similar increase, almost all cases of pneumonia and LRI occurred in the group of elderly inpatients. The weakened immune system in elderly people may be to blame for this effect.31,32
Influence of endoscopic procedure
All patients in the GIES group underwent EGD and about 38% received an additional colonoscopy. Neither direct comparison nor the regression model revealed an increased risk of the combination group to develop pneumonia (2.1 vs. 1.3%, p = 0.146) or LRI (7.3 vs. 3.2%, p = 0.631). These findings coincide with other studies. 18 We were not able to form a proper group of patients who underwent colonoscopy only because screening colonoscopy is only rarely performed in inpatients. Therefore, we cannot weigh the effect of a single colonoscopy. Furthermore, we could not distinguish whether the inflammatory reaction is related to the endoscopy itself or the bowel preparation that has been demonstrated to rarely induce aspiration. 33
Timing of inflammatory response
The effects mentioned previously concerning pneumonia, LRI, inflammation parameters and other surrogate parameters can be seen clearest on day 3 after GIES, while the effects blur on day 7. We therefore conclude that day 3 is the proper point to record signs of respiratory infections, particularly pneumonia and LRI, within the scope of gastrointestinal endoscopy.
Strengths and limitations
Definitions are sometimes incorrectly used in daily routine practice and under study conditions. 34 We therefore reviewed all cases and used the terms pneumonia and LRI as standardized and defined in international guidelines.27–29 Each of the patients in the GIES group was compared with a precisely age-, gender- and length of hospital stay-matched control patient. All controls were identified by searching through samples of patients with suitable admission diagnoses and without any procedure under sedation. Consequently, we could not achieve equal groups concerning comorbidities and medication. Because of the necessity of obtaining laboratory values on day 3 or at least day 7, our study population, both the GIES group and the control group, consisted mostly of inpatients who were potentially sicker and may have had more blood samplings than the average patient, which might have led to a certain bias. Moreover, we concentrated on endoscopies carried out under sedation, as most patients request. 35 The retrospective design itself is associated with certain limitations: primary lack of data acquisition, such as chest X-ray or bowel cleansing, and endoscopy-related adverse events, laboratory values past the procedure or missing detailed sedation parameters.
Conclusion
In summary, inpatients in advanced age carry an increased risk of developing pneumonia, LRI and systemic inflammation after gastrointestinal endoscopy, particularly combined EGD and colonoscopy under sedation. The need for antibiotic treatment compared to the matched controls indicates the clinical relevance of these findings. Therefore, endoscopists should be aware that older inpatients bear an increased hazard for pneumonia and LRIs. Nonetheless, additional studies should prospectively evaluate the risk of respiratory infections, the underlying pathomechanisms and risk factors in a larger and unselected population.
Supplemental Material
Supplemental material for Gastrointestinal endoscopy under sedation is associated with pneumonia in older inpatients—results of a retrospective case-control study
Supplemental Material for Gastrointestinal endoscopy under sedation is associated with pneumonia in older inpatients—results of a retrospective case-control study by Christopher M Kollmann, Wolff Schmiegel and Thorsten Brechmann in United European Gastroenterology Journal
Footnotes
Acknowledgments
We gratefully thank Claudia Dillmann for the preselection of eligible patients and the staff of the Department of Gastroenterology and Hepatology, especially Michaela Meyburg, for their support throughout the study.
Declaration of conflicting interest
None declared.
Ethics approval
This study was approved by the institutional review board of the Ruhr-University Bochum (registry number 15-5323, 24 July 2015).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Informed consent was obtained from all patients before endoscopy. The study protocol conforms to the ethical guidelines of the Declaration of Helsinki.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
