Abstract
Background
In clinical practice, treatment sequences of biologicals are applied for active fistulising Crohn’s disease, however underlying health economic analyses are lacking.
Objective
The purpose of this study was to analyse the cost-effectiveness of different biological sequences including infliximab, biosimilar-infliximab, adalimumab and vedolizumab in nine European countries.
Methods
A Markov model was developed to compare treatment sequences of one, two and three biologicals from the payer’s perspective on a five-year time horizon. Data on effectiveness and health state utilities were obtained from the literature. Country-specific costs were considered. Calculations were performed with both official list prices and estimated real prices of biologicals.
Results
Biosimilar-infliximab is the most cost-effective treatment against standard care across the countries (with list prices: €34684–€72551/quality adjusted life year; with estimated real prices: €24364–€56086/quality adjusted life year). The most cost-effective two-agent sequence, except for Germany, is the biosimilar-infliximab–adalimumab therapy compared with single biosimilar-infliximab (with list prices: €58533–€133831/quality adjusted life year; with estimated prices: €45513–€105875/quality adjusted life year). The cost-effectiveness of the biosimilar-infliximab–adalimumab–vedolizumab three-agent sequence compared wit biosimilar-infliximab –adalimumab is €87214–€152901/quality adjusted life year.
Conclusions
The suggested first-choice biological treatment is biosimilar-infliximab. In case of treatment failure, switching to adalimumab then to vedolizumab provides meaningful additional health gains but at increased costs. Inter-country differences in cost-effectiveness are remarkable due to significant differences in costs.
Keywords
Key summary
In clinical practice, treatment sequences of biologicals are applied for active fistulising Crohn's disease, however underlying health economic analyses are lacking. The suggested first-choice biological treatment is bsIFX. In case of treatment failure, switching to adalimumab then to vedolizumab provides meaningful additional health gains but at increased costs.
Introduction
Fistulising Crohn’s disease (CD) can lead to significant impairment in health-related quality of life and imposes substantial burden both on patients and society.1,2 Fistulas are often difficult to treat and result in significant costs through the use of medication, surgical intervention and the need for hospitalisation. 3 Biological drugs has dramatically changed the treatment of CD. Randomised controlled trials (RCTs) established the clinical efficacy and safety of infliximab (IFX) for the treatment of fisultising CD, using fistula closure as primary endpoint.4,5 The scientific evidence for adalimumab (ADA) and vedolizumab (VEDO) is weaker as their effects were examined in subgroups of patients with fistulising CD in RCTs but only as secondary endpoints.6–11 IFX is the only biological drug that is specifically indicated for the treatment of adults with fistulising CD, but in clinical practice ADA and VEDO are also used to treat fistulising CD.12–15 Access to biologicals varies significantly between countries largely driven by budgetary constraints.16,17 Biological therapy and hospitalisation are the main cost drivers in CD, although studies focusing specifically on fistulising CD are scarce.1,2,18,19
Biosimilars, due to their lower price, have the potential to improve access to treatment and achieving substantial savings relative to the reference medical products. The first biosimilar monoclonal antibody, biosimilar-infliximab (bsIFX) was approved by the European Medicines Agency (EMA) in 2013 and U.S. Food and Drug Administration (FDA) in 2016 for the same indications as the originator molecule. 20 Although bsIFX is already used in many countries, few studies have dealt with its health economic aspects. Budget impact analyses suggest that significant savings can be achieved with bsIFX in CD.21,22
Three cost-utility studies of biological treatment in fistulising CD assessed the cost-effectiveness of IFX therapy in comparison with standard care from the third-party payers’ perspective.23–25 According to our knowledge, no health economic analyses with ADA, VEDO and bsIFX have been published yet. Inter-country comparisons are also lacking in the literature. More importantly, there is no cost-effectiveness evidence on the sequential use of biological treatment in fistulising CD. Health gains and costs of these treatment sequences have to be analysed and compared to identify the most cost-effective treatment strategy, particularly since the advent of including biosimilars.
The aim of our study was to analyse the cost-effectiveness of the different treatment sequences with available biologicals (IFX, bsIFX, ADA, VEDO), for the treatment of fistulising CD in nine European countries, namely in Belgium, France, Germany, Hungary, Italy, Spain, Sweden, the Netherlands and the UK.
Materials and methods
Model overview
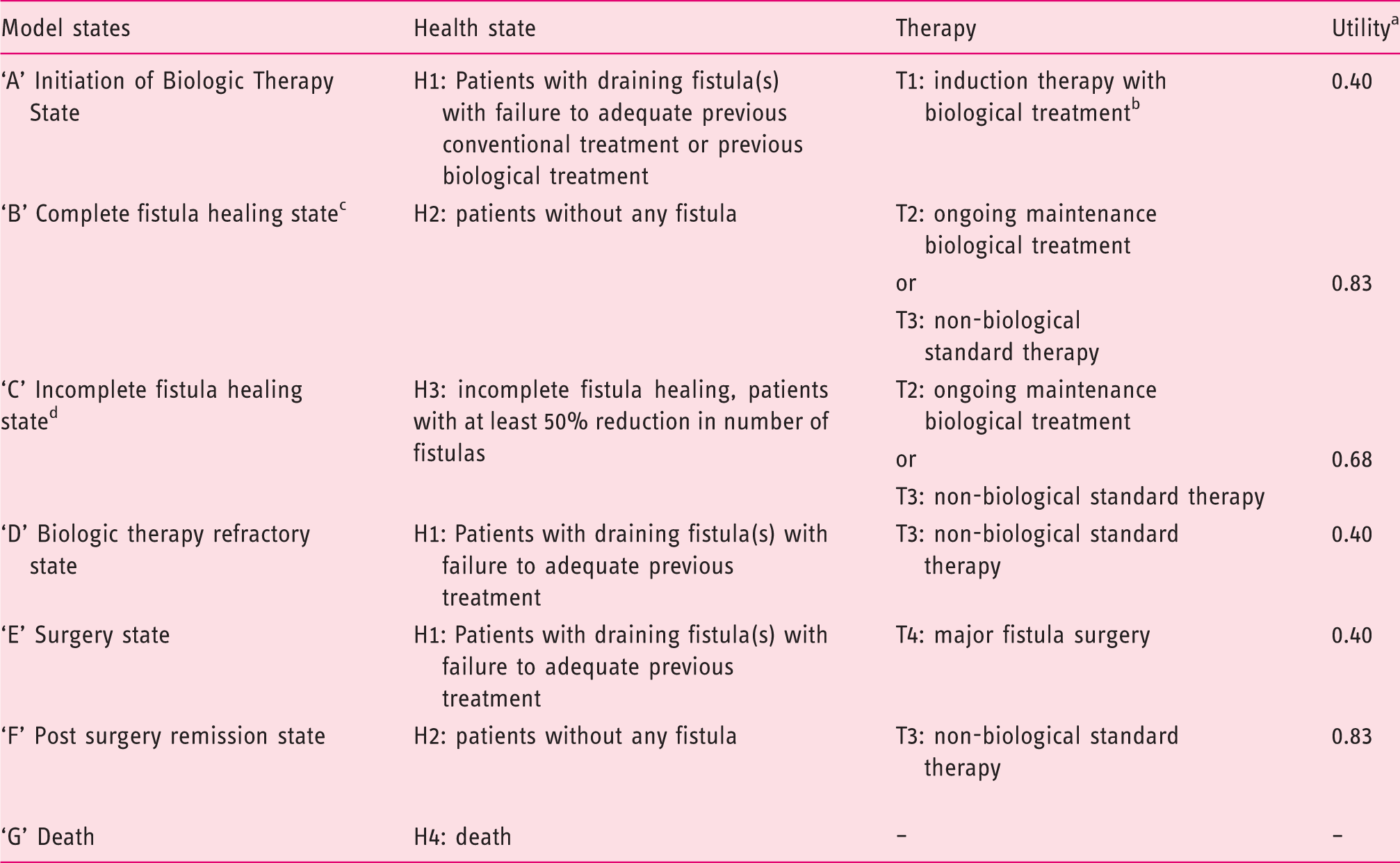
A Markov model was developed to analyse the cost-effectiveness of treatment sequences including bsIFX for the treatment of patients with active fistulising CD (Figure 1). Model states are described by the health states and the therapies (Table 1). Health states were defined by fistula healing related primary and secondary endpoints from RCTs.
The structure of the Markov model for fistulising Crohn’s disease (CD). Model states: health states, therapies applied and utilities. 30% of the patients have minor fistula surgery. Secondary end-point in the randomised controlled trial of infliximab by Present et al.,1999
4
and in the following studies with subgroup of fistulising CD patients: Hanauer et al., 2006 (CLASSIC I)
6
induction adalimumab study, Sandborn et al., 2007 (GAIN)
7
induction adalimumab study. Primary end-point in Sands et al., 2004 (ACCENT II)
5
maintenance infliximab study and secondary end-point in in the following studies with subgroup of fistulising CD patients: Colombel et al., 2009 (CHARM)
8
adalimumab maintenance study, Sandborn et al., 2013 (GEMINI)
9
maintenance study. Primary end-point in the randomised controlled trial of infliximab in Present et al., 1999
4
and secondary end-point in in the following studies with subgroup of fistulising CD patients Hanauer et al., 2006 (CLASSIC I)
6
induction adalimumab study, Sandborn et al., 2007 (GAIN)
7
induction adalimumab study.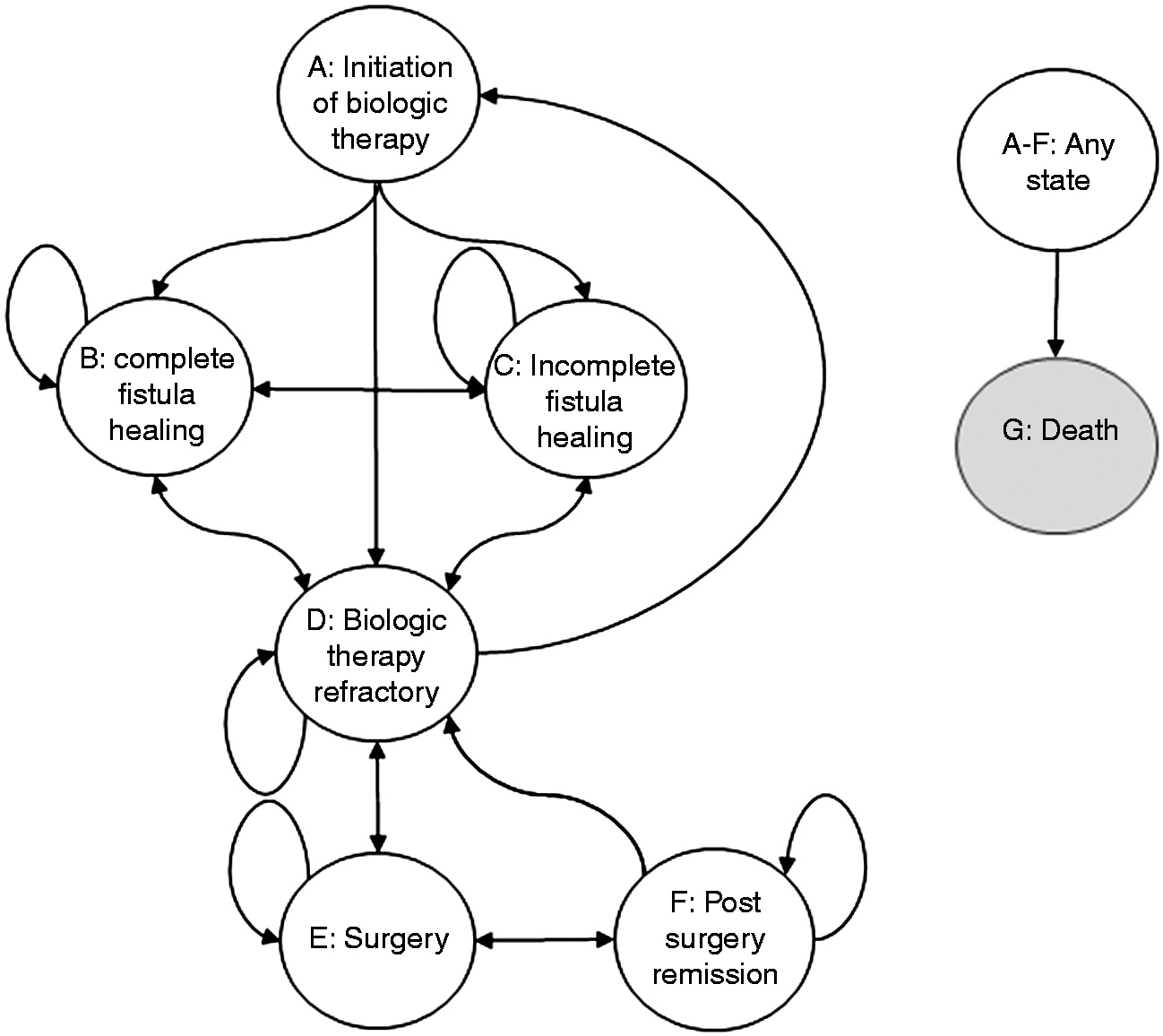
The model focuses solely on fistulising CD and does not take account of treatment effects on luminal CD symptoms (e.g. improvement of Crohn’s Disease Activity Index (CDAI) score).
At the end of each subsequent model cycle, patients either remained in the same model state or moved to a different one. The model runs in three-month cycles representing the course of the disease by time and the time horizon is five years. Patients could make transitions between model states at the end of a cycle. These movements are represented by the arrows on Figure 1.
In the cost-effectiveness model, first we compared single biological drug treatment with standard (non-biological) care. Then different treatment sequences of multiple biologicals were compared with each other. If more than one biological was included in a treatment sequence, it was assumed that all patients who get to the model state ‘D’ would immediately start with the subsequent biological therapy and moved to model state ‘A’. Once the patients went through all available biologicals, biological treatment could not be restarted again and they continued with non-biological standard care in the model state ‘D’ or moved to model state ‘E’. Patients receiving non-biological standard care could move from model state ‘D’ to model states ‘A’ or ‘C’, but this was allowed only once during the time horizon of the model. From model state ‘D’ patients could move to model state ‘E’. Patients with successful major fistula surgery moved to model state ‘F’. Patients with unsuccessful first surgery went through a second procedure, however a maximum of two major fistula surgeries was allowed during the time horizon of the model. Combining fistula surgery with biological treatment can have beneficial effects however, not all patients have surgery alongside the initiation of biological drug treatment.26–28 Hence, the model linked minor fistula surgery to the ‘Initiation of biologic treatment’ model state but assumed that this type of intervention applied to only 30% of the patient population.
Patients
The cost-effectiveness model was populated with patients with fistulising active CD who (a) had single or multiple draining abdominal and/or perianal fistulas at baseline; (b) who had not responded to conventional treatment (including antibiotics, drainage and immunosuppressive therapy); and (c) were eligible for biological treatment. These inclusion criteria correspond with the inclusion criteria of the only RCT designed specifically for fistulising CD, 4 with the fistulising subgroups of other RCTs in CD, as well as with the drug indications.5,8,9 We assumed that the average age of the patients was 40 years and the average weight was 65 kg (standard deviation (SD) = 16). 29 These figures are in line with the baseline characteristics of patients in the Present et al. study of 1999, 4 and other fistulising subgroups in RCTs.5,8,9
Comparison of treatment sequences
In the cost-effectiveness model we compared different treatment sequences including the available biologicals as well as non-biological standard care. Each treatment sequence could include a maximum of three different biologicals: IFX, bsIFX, ADA and VEDO followed by standard care. Each biological could appear only once in the treatment sequence. First, we calculated the cost-effectiveness of each biological drug (IFX, bsIFX, ADA and VEDO) followed by standard care compared with non-biological standard care. In the second step, we took the most cost-effective single biological and analysed the cost-effectiveness of adding a second biological to the sequence. In the third step, the most cost-effective two-item sequence was chosen and we explored the cost-effectiveness of adding a third biological.
Transition probabilities
Clinical efficacy data were derived from RCTs and were used to calculate transition probabilities. In the RCTs, fistulising CD-related inclusion criteria were typically the presence of single or multiple draining abdominal or perianal fistulas of at least three months’ duration as a complication of CD, concurrent therapies for CD in stable doses were permitted and patients were excluded from the study if they had a stricture or abscess for which surgery might be indicated. In RCTs where fistulising patients were only a subgroup of the sample, moderate or severe active CD (as measured by the CDAI score) was a criterion of inclusion. Patients were biological naive in some RCTs4,5 and have already had previous exposure to anti-tumour Necrosis Factor Alpha (anti-TNF-alpha) treatment in others.8,9,30
Transition probabilities between model states.

Other assumptions: Mortality: we assume different mortality rates for complete healing (OR = 1) and incomplete healing (OR = 1.266) and refracter (OR = 2.256) health states (based on: Lichtenstein et al. 2006).34 The source of Life Tables: EUROSTAT. Due to the lack of data we assume that the probability of moving from ‘Incomplete healing’ to ‘Complete healing’ model state is 0. Due to the lack of data for fistulising patients, we assume that the efficacy of the 2nd and 3rd biological treatment will decrease by 10% (regardless of the agent).
Costs and outcomes
The analyses were performed from the third-party payer perspective. IFX has patient weight-dependent dosing, the remaining portion of drug in the vial is either used (no wasting) or not (wasting) for the treatment of another patient. In the base case we considered no wasting for IFX and for bsIFX. Country-specific unit costs were considered based on official price lists and available sources to calculate health care utilisation costs.
Input data for the model: costs (€, 2015).
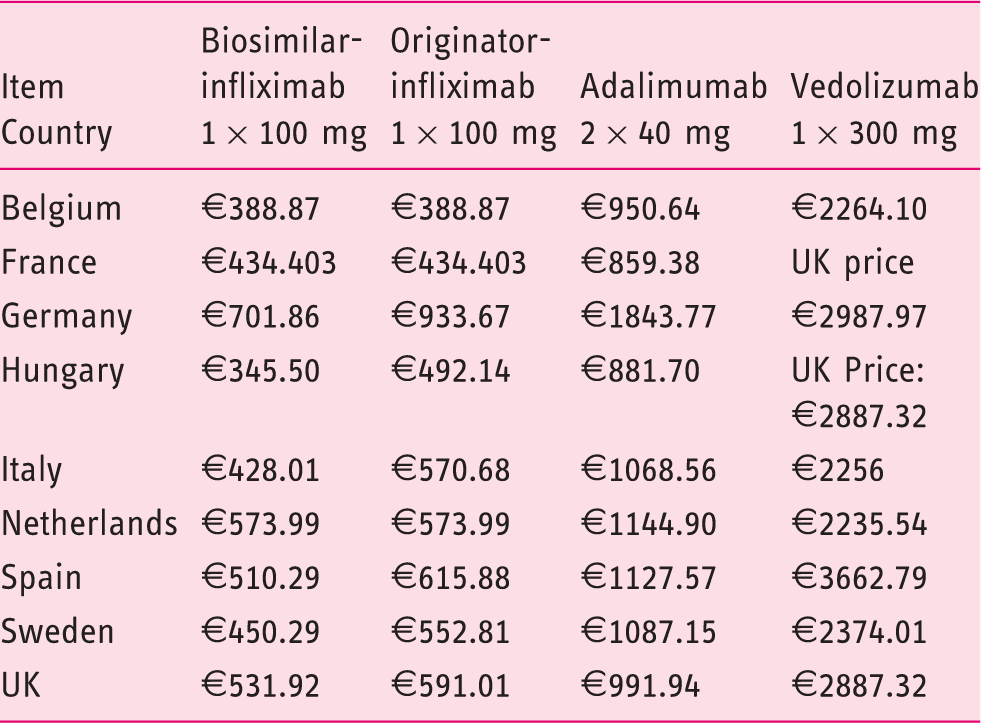
Conversions: Hungary €1 = 312 HUF; Sweden: €1 = 9.3 SEK; UK: €1 = £0.71.
Sources of biological drug costs:
Belgium: Rijksinstituut voor ziekte-en invaliditeitsverzekering. Pharmaceutical list prices (2016), (last accessed 18/05/2016).
France, https://www.vidal.fr (last accessed 18/05/2016).
Germany: Medipreis. Preisvergleich für Medikamente (2016), https://www.medipreis.de (last accessed 18/05/2016).
Hungary: National Health Insurance Fund Administration brutto price Országos Egészségbiztosítási Pénztár Publikus Gyógyszertörzs (1 June 2016). (last accessed 18/05/2016).
Italy: Medicina e Società (2016), http://www.medisoc.it/scheda-tecnica-infliximab/ (last accessed: 18/05/2016).
Netherlands: Medicijnkosten, http://www.medicijnkosten.nl (last accessed 18/05/2016).
Spain: http://a.nomenclator.org (last accessed 19/05/2016).
Sweden: Dental and Pharmaceutical Benefits Agency (TLV), http://www.tlv.se/In-English/medicines-new/ (last accessed 18/05/2016).
UK: British National Formulary (BNF), https://www.evidence.nhs.uk/ (last accessed 18/05/2016).
Administration costs for biologicals were considered only for infusions (bsIFX, IFX, VEDO) as the administration cost of subcutaneous injections (ADA) is usually minor. For standard care we considered the cost of sulfasalazine, 2000 mg/day and 20 mg prednisolone/day. In this patient population other per os drugs are also used such as azathioprine and/or antibiotics, which would result in similar costs. Both the items considered for the monitoring of patients and their frequency varied according to the treatments applied and also across countries. Monitoring included routine laboratory tests (blood cell count, liver and kidney function test, albumin, iron, ferritin, C-reactive protein, urine analysis, faecal calprotectin test, outpatient visit, chest x-ray, abdominal ultrasound, magnetic resonance and endoscopy (rectosigmoidoscopy and colonoscopy). Non-surgical hospitalisation and costs were considered only for 40% of patients in model state ‘D’. Informal care and transportation costs were not included.
Utility values were attached to each model state to calculate quality adjusted life years (QALYs) in cost-effectiveness analyses. Few data are available on the utility of health states of patients with fistulising CD.25,35 We applied utility weights presented by Lindsay at al. in 2008 25 (Table 1). Discounted costs and outcomes were calculated on a five-year time horizon.
Sensitivity analyses
The structural uncertainty of the model was evaluated using one-way sensitivity analysis varying parameters such as price, time horizon, patients’ age and weight, health state utilities, cost of surgery and inpatient care, discounting rates and wasting of remaining drugs in the vial. Productivity loss was not included in the base case analysis but was considered in the sensitivity analysis providing an assessment from the societal perspective. A temporary productivity loss of 20-day sick leave per year (multiplied by average gross daily wages of the countries) was considered for ‘A’, ‘D’ and ‘E’ model states. Parameter uncertainty (transitional probabilities, costs, utility weights, patients’ characteristics) was evaluated using probabilistic sensitivity analysis.
Results
Cost-effectiveness analyses (base case)
Cost-effectiveness results (third-party payer’s perspective, time horizon of five years, patients mean weight 65 kg, no wasting of remaining infliximab in the vial, utility weights from Lindsay et al., 25 results shown in €).
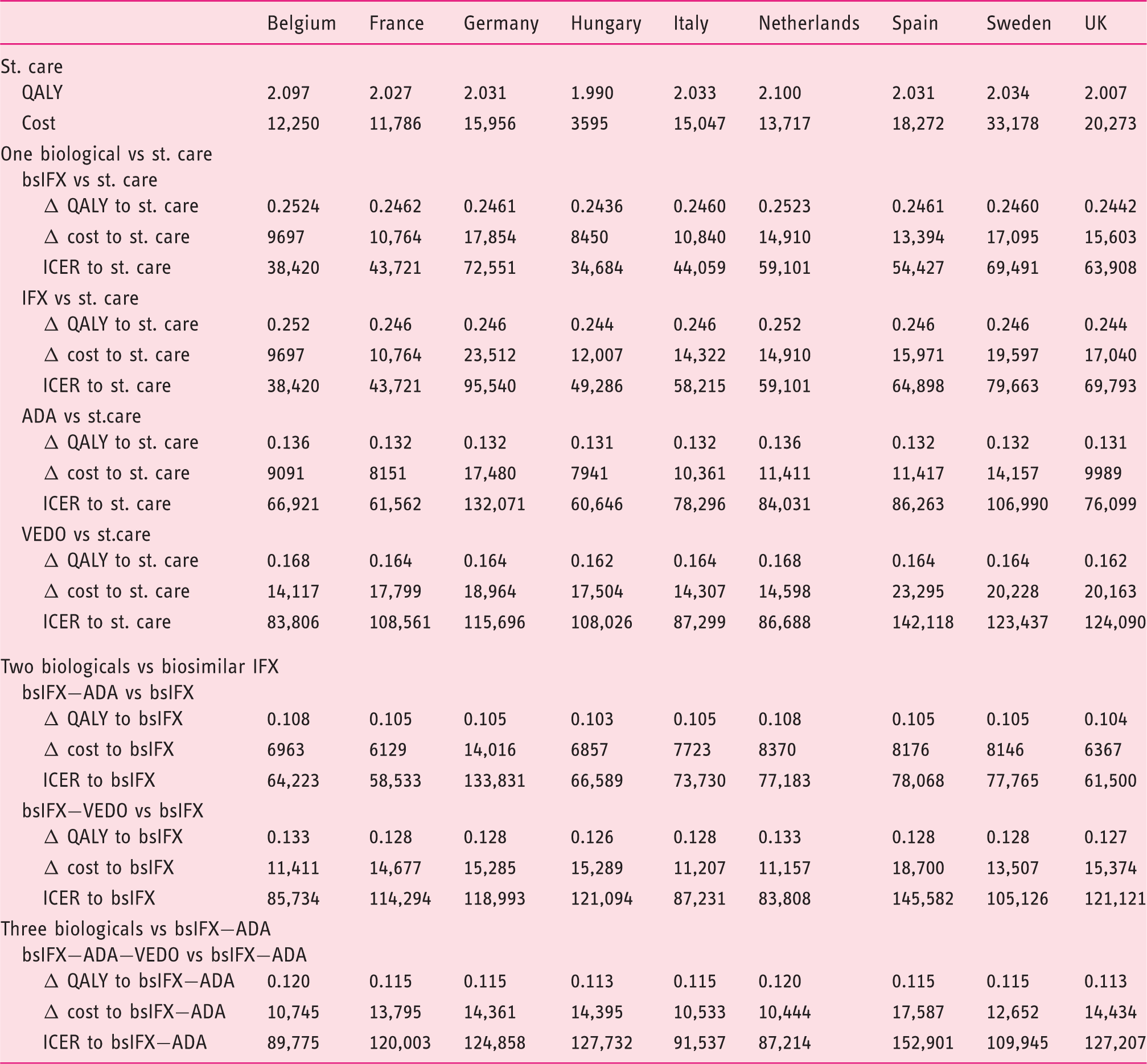
ADA: adalimumab; bsIFX: biosimilar-infliximab; ICER: incremental cost-utility ratio; IFX: originator-infliximab; QALY: quality adjusted life year; st. care: standard care; VEDO: vedolizumab.
Discount rates: equal discount rates were applied for both costs and effects in the following countries: France, Germany, Italy, Spain, Sweden 3.0%, Hungary 3.7%, UK 3.5%. For Belgium costs were discounted by 3% and effects by 1.5%, and for the Netherlands by 4.0% and 1.5%, respectively.
Single biologicals compared with standard care – list price
Given the same clinical efficacy and safety for IFX and bsIFX, additional QALY gains compared with standard care were equal and varied between 0.244–0.252 QALY across countries due to differences in mortality and discount rates. Lower additional QALY gains were observed for ADA and VEDO in all the nine countries, 0.113–0.136 and 0.162–0.168 QALY, respectively. In six countries where the list prices of bsIFX were lower than of IFX (Germany, Hungary, Italy, Spain, Sweden and the UK), the additional costs compared with standard care were lower for bsIFX (varied between €8450–€17854) than for IFX (varied between €9697–€23512), and the highest were for VEDO (varied between €14117–€23295). Germany, Italy and the Netherlands are exceptions where the additional cost of the VEDO scenario was slightly lower than IFX (Table 4). The additional cost of ADA compared with standard care varied between €7941–€17480. The ICER (compared with standard care) was the lowest for bsIFX in all the nine countries and varied between €34684–€72551/QALY. Due to the same list prices of bsIFX and IFX, the ICERs of these two drugs were identical in Belgium, France and the Netherlands. The ICERs were between €38420–€95540/QALY for IFX, €60646–€132071/QALY for ADA, and €83806–€142118/QALY for VEDO (Table 4).
Treatment sequences with two or three biologicals – list prices
Since bsIFX was the most cost-effective treatment as a one-agent treatment sequence, this was chosen as a comparator for treatment sequences with two and three biologicals. Adding a second biological to the treatment sequence after bsIFX provided additional QALY gains (0.103–0.133) compared with bsIFX alone but at larger additional costs (€6129–€18700). The bsIFX−ADA was the most cost-effective two-agent treatment sequence in all countries with ICERs (compared with bsIFX) between €58533–€133831/QALY. Germany was an exception where the bsIFX–VEDO sequence resulted in a lower ICER than the bsIFX–ADA sequence. The ICERs of the bsIFX–VEDO treatment sequence were between €83808–€145582/QALY (Table 4). Adding a third biological (VEDO) to the two-agent treatment sequence provided additional QALY gains (compared with bsIFX–ADA sequence: 0.113–0.120) but at larger additional costs (€10444–€17587). The ICERs varied between €87214–€152901/QALY. In Germany, adding ADA after bsIFX–VEDO sequence resulted in an ICER of 140666/QALY compared with bsIFX–VEDO.
Cost-effectiveness with estimated real prices
Cost-effectiveness results with price discounts of 30% for originator and biosimilar-infliximab (bsIFX) and 20% for adalimumab (ADA) (third-party payer’s perspective, time horizon five years, patients mean weight 65 kg, no wasting of remaining infliximab (IFX) in the vial, utility weights from Lindsay et al., 25 results shown in €).
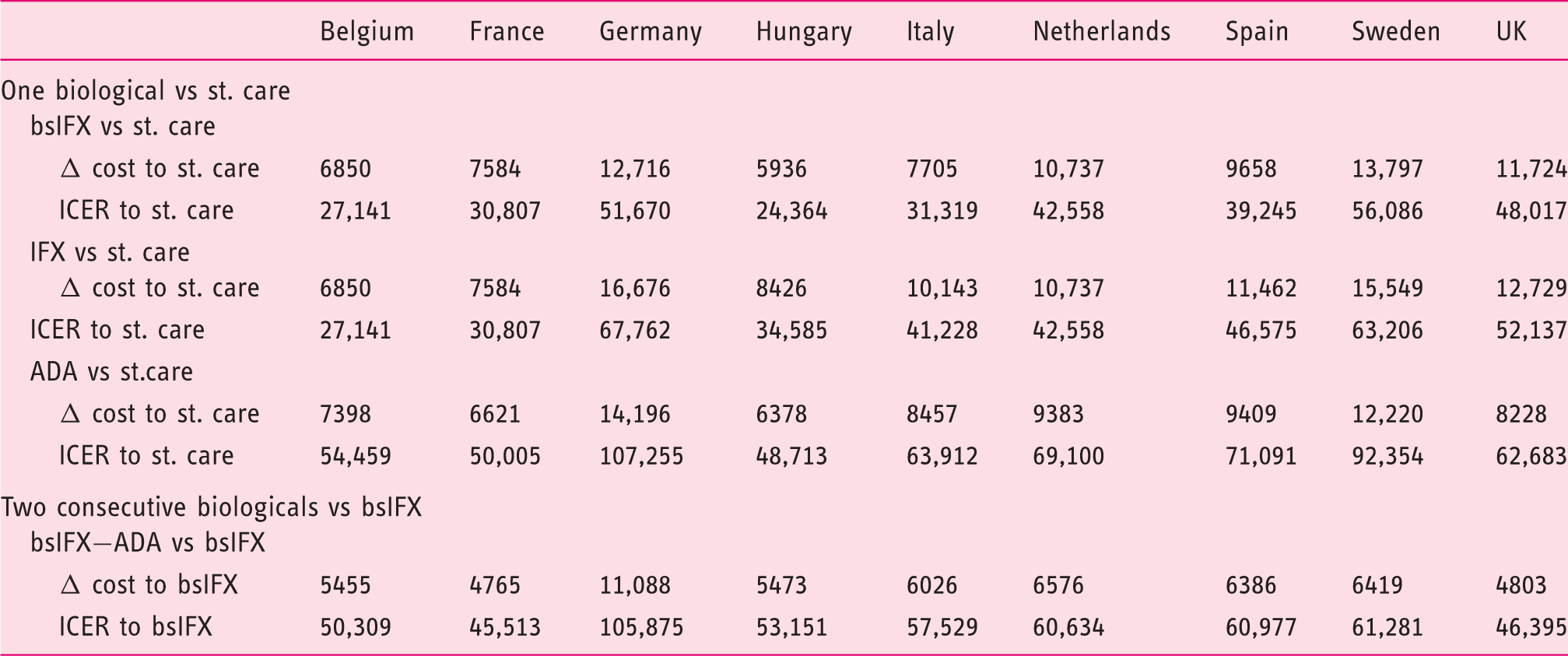
ADA: adalimumab; bsIFX: biosimilar-infliximab; ICER: incremental cost-utility ratio; IFX: originator-infliximab; QALY: quality adjusted life year; st.care: standard care; VEDO: vedolizumab.
Quality adjusted life year (QALY) gains are equal to results presented in Table 4 as only drug costs were modified. Vedolizumab price was not discounted hence no change in the vedolizumab versus standard care and the bsIFX-VEDO vs bsIFX scenario.
One-way sensitivity analysis
One-way sensitivity analysis for biosimilar-infliximab (bsIFX) – vs standard care.
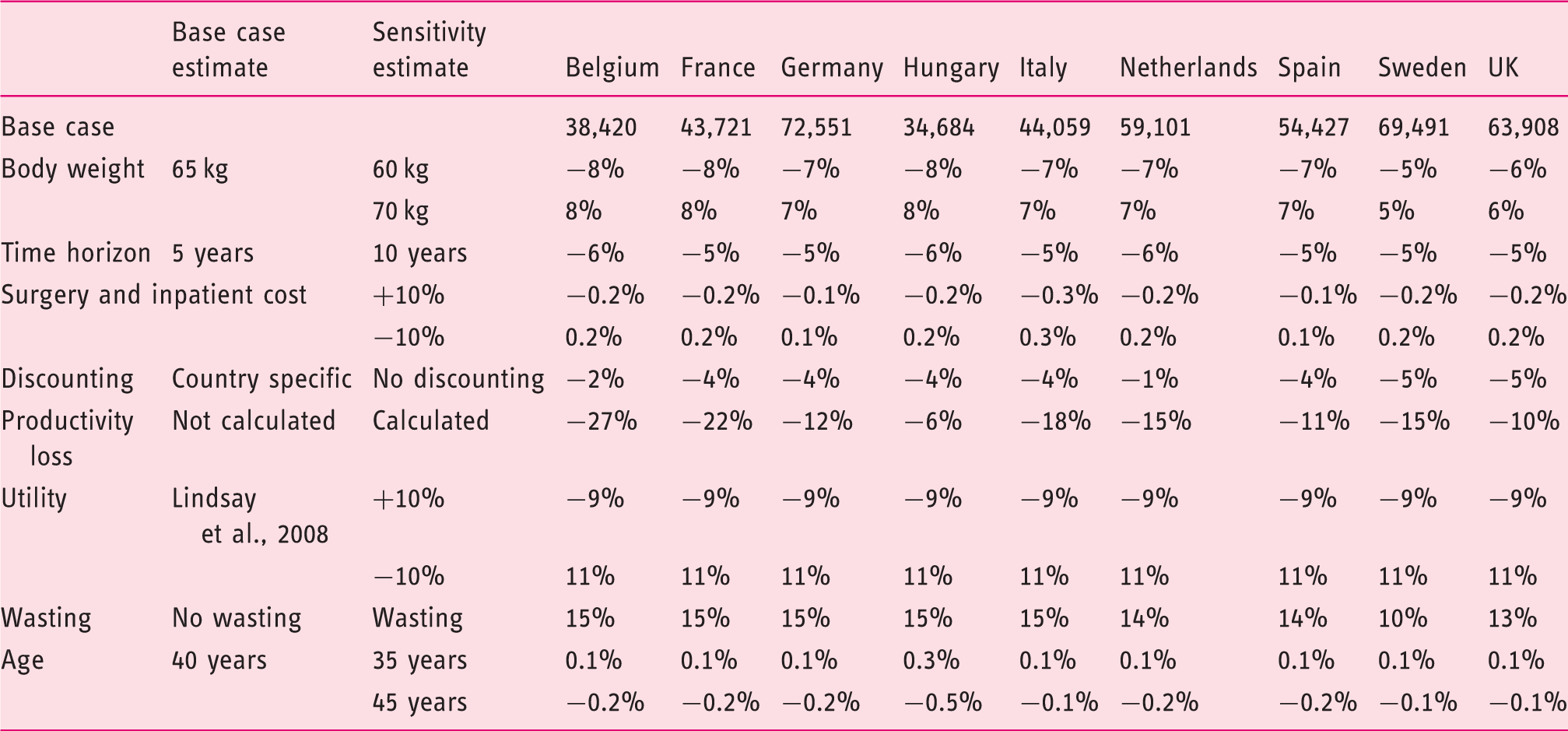
Regarding parameters relating to the weight dependent dosing of bsIFX and IFX, the ±5 kg change in patients’ weight resulted in a change of ±8% in ICERs. Inclusion of wasting of infliximab vials increased the ICERs by 10–15%.
Probabilistic sensitivity analysis (PSA)
Results were robust for the changed in input data and assumptions. For example in the UK, The 95% confidence intervals of the ICERs calculated by the simulations were €45788–€97677/QALY for bsIFX and €51081–€94182/QALY for IFX, both compared with standard care.
In 69% of the simulations, the ICER of bsIFX remained under a 2 × GDP per capita financial threshold in the UK. This was the case in 56% of the cases for IFX.
Discussion
In this study we assessed the cost-effectiveness of single biologicals and treatment sequences in comparison with non-biological standard care and with each other for adult patients with fistulising active CD in nine European countries. This is the first cost-effectiveness analysis with bsIFX, ADA and VEDO in fistulising CD, and also the first which assessed the cost-effectiveness of biological treatment sequences including two and three biological substances. Nine European countries were involved in the analyses which enables us to make cross-country comparisons. Also, this is the first study to analyse the cost-effectiveness of biologicals in fistulising CD not only from third-party payers but also from the societal perspective.
In scenarios including a single biological drug, bsIFX was the most cost-effective and VEDO was the least cost-effective treatment compared with non-biological standard care. In France, the Netherlands and in Belgium the cost-effectiveness of IFX and bsIFX were identical as the list prices of these two drugs were equal (Table 4). Starting treatment with bsIFX was not only the most cost-effective scenario but also IFX had the highest level of clinical evidence that supports this strategy.
As a second biological treatment following bsIFX, ADA provided better cost-effectiveness results compared with VEDO, except in Germany. Including VEDO as a third agent in the treatment sequence (after bsIFX and ADA) provides high cost-effectiveness ratios.
The use of bsIFX in the treatment sequences instead of IFX, reduces the ICERs to below a threshold of 2 × GDP per capita across all the nine countries. The introduction of bsIFX has made biological treatment more affordable, providing cost-savings, thus it improves access to biological therapy.
The cross-country differences in ICERs can be largely explained by the differences of the list prices of biologicals (i.e. higher biological prices resulting higher ICERs). The highest ICERs were observed in Germany (Table 4) due to the highest list prices (Table 3).
It is important to point out that real purchasing prices are substantially lower than list prices, but are not publicly available, and can vary even within a country due to local tenders. Based on personal communication, the price of bsIFX is about 50% lower than the list price of the originator product. Thus, we performed repeat calculations applying a 30% price decrease for bsIFX and IFX, and a 20% decrease for ADA list prices to get cost-effectiveness ratios which we assume to better reflect real world practice (Table 5). As a result, ICERs decreased by 19–30%, although the optimal treatment strategies remained the same. Using this approach in Germany, ADA became the most cost-effective treatment as a second treatment option after bsIFX.
Comparison with the available three cost-effectiveness studies in fistulising CD is hampered by methodological differences. Arseanau and colleagues (2001) evaluated IFX therapy in comparison with standard care on a one-year time horizon in the USA, and the ICER varied from $355,450 to $377,000/QALY. 23 Clarke and colleagues (2003) analysed IFX against standard care in the UK and the ICERs were between £82,000–£123,000/QALY. 24 Lindsay et al. (2008) found a somewhat lower ICER (£29,752 per QALY) with IFX compared with standard care in the UK on a five-year horizon. 25
This cost-effectiveness model has several limitations. The model movements represent clinical practice, however we applied some simplifications. We did not calculate with the effects of discontinuation of biological therapy, and assumed that spontaneous healing from the refractory state is only allowed once in the model. In addition, patients could go through surgery a maximum of two times during the full time horizon of the model. Nevertheless, the model results are robust despite these assumptions as they are identical in the active and comparator scenarios. The model focuses on fistulising CD and treatment effects on luminal CD symptoms were not captured. Specifically, we focused on endpoints related to fistula healing in the model and did not take into account the potential improvements in the underlying CD (e.g. improvements in the CDAI score)). Thus, giving conservative estimations, we probably underestimated the efficacy of therapy in case of patients where fistula closure was not achieved but underlying CD activity improved. In these cases larger health gains are achieved on the same costs, resulting better cost-effectiveness ratios.
Due to the lack of data in fistulising CD, we did not calculate with the effects of dose-increases. However, dose increase occurs in everyday clinical practice in fistulising CD.
Transition probabilities between model states are key points in cost-effectiveness modelling. Due to the scarcity of evidence from RCTs we had to apply some estimations. For fistula healing as a primary outcome in RCTs, there is only evidence for IFX. Evidence for ADA and VEDO comes from subgroup analysis of RCTs.
We used country-specific costs in the model. However we could not consider all intercountry variations due to shortage of local data (such as patients’ age, weight and efficacy), thus for some parameters we assumed no significant differences across the countries. Nevertheless, results of our sensitivity analysis suggest that in a country where the average weight of patients is +5 kg, the ICERs increase by about 8%. Results were much less sensitive to changes in the age of patients, ±5 years change results in −0.5% to +0.3% change in the ICERs. Moreover, health status and disease progression of patients initiating biological treatment in the real world might differ between jurisdictions due to variations in health care delivery structures, guidelines, clinical practice (e.g. surgical strategies, the level of multidisciplinary teamwork) and access (availability, affordability and acceptability) to biological treatment.16,36 These might result in some differences of effectiveness as well, however at a minor level compared with the overall treatment effects. Nevertheless, refinement of our results based on more precise, local evidence is to be encouraged. Our study revealed a remarkable deficiency of data in the literature on fistulising CD patients, a point we suggest should be considered in the development of CD registries and in publication strategies in the future.
In conclusion, as the number of biologicals is growing for fistulising CD the focus of cost-effectiveness analysis is moving to the comparison of treatment sequences. This is the first study which assessed the cost-effectiveness of biological treatment sequences in fistulising CD. We found that bsIFX was the most beneficial first treatment choice in all of the nine countries. Appling a second and third biological provides some additional health gain at a high cost. Significant differences were found in costs but only minor variations in health gains across countries.
Conclusion
In clinical practice, treatment sequences of biologicals are applied for active fistulising CD, however underlying health economic analyses are lacking. The suggested first-choice biological treatment is bsIFX. In case of treatment failure, switching to ADA then to VEDO provides meaningful additional health gains but at increased costs.
Footnotes
Conflicts of Interest
PB reports grants from Hospira a Pfizer Company during the conduct of the study; grants from Sager Pharma, grants from Egis Pharma, non-financial support from Astellas, outside the submitted work. LG received consultancy and lecturing fee from Egis Pharmaceutical, Hospira, Pfizer, Celltrion, Sandoz, Astellas. VB reports grants from Hospira a Pfizer Company during the conduct of the study; grants and personal fees from Egis Pharma, personal fees from Sager Pharma, outside the submitted work. ZV reports having received speakers' bureau fees from AbbVie and Takeda. SD has served as a speaker, a consultant and an advisory board member for Schering-Plough, Abbott Laboratories, Merck & Co., UCB Pharma, Ferring, Cellerix, Millenium Takeda, Nycomed, Pharmacosmos, Actelion, Alphawasserman, Genentech, Grunenthal, Pfizer, Astra Zeneca, Hospira, Novo Nordisk, Cosmo Pharmaceuticals, Vifor, and Johnson and Johnson. PMI has received consultancy and lecturing fees from AbbVie, Warner Chilcott, Ferring, Falk Pharma, Takeda, MSD, Janssen, Shire, Vifor, Pharmacosmos, Samsung Bioepis, VH2, Hospira, Genetech. LPB reports personal fees from Abbvie, Janssen, Genentech, Mitsubichi, Ferring, Norgine, Tillots, Vifor, Shire, Pharmacosmos, BMS, UCB-Pharma, Hospira, Takeda, Boerhinger-Ingelheim, Pfizer, Lilly, Celltrion, Merck, Therakos, Pilege, Biogaran, HAC-Pharma, Index-Pharmaceuticals, Amgen, Sandoz, Forward Pharma GmbH, Celgene, outside the submitted work. SS received consultancy and/or lecturing fees from AbbVie, Biogen, Celltrion, Celgene, Falk, Ferring, Janssen, MSD, Pfizer, Roche, Takeda and UCB. FR reports grants from Astellas, outside the submitted work. PLL reports having received consultancy and lecturing fees from AbbVie, Celltrion, Celgene, Ferring, Hospira, Mitsubichi Tanabe Pharma, MSD, Pfizer, Roche and Takeda. MP reports grants from Hospira a Pfizer Company during the conduct of the study; grants and personal fees from Egis Pharma, Merck, Pfizer, Sager Phama, outside the submitted work.
Funding
This work was supported by an unrestricted grant from Pfizer Hospira UK.
Ethics approval
This is a non-interventional study and ethical approval was not needed. Data were derived from published publicly available articles. No other data sources were used.
Informed consent
Not applicable as patients were not included.
