Abstract
Background
Diverticular disease of the colon is frequent in clinical practice, and a large number of patients each year undergo surgical procedures worldwide for their symptoms. Thus, there is a need for better knowledge of the basic pathophysiologic mechanisms of this disease entity.
Objectives
Because patients with colonic diverticular disease have been shown to display abnormalities of the enteric nervous system, we assessed the frequency of myenteric plexitis (i.e. the infiltration of myenteric ganglions by inflammatory cells) in patients undergoing surgery for this condition.
Methods
We analyzed archival resection samples from the proximal resection margins of 165 patients undergoing left hemicolectomy (60 emergency and 105 elective surgeries) for colonic diverticulitis, by histology and immunochemistry.
Results
Overall, plexitis was present in almost 40% of patients. It was subdivided into an eosinophilic (48%) and a lymphocytic (52%) subtype. Plexitis was more frequent in younger patients; and it was more frequent in those undergoing emergency surgery (50%), compared to elective (28%) surgery (p = 0.007). All the severe cases of plexitis displayed the lymphocytic subtype.
Conclusions
In conclusion, myenteric plexitis is frequent in patients with colonic diverticular disease needing surgery, and it might be implicated in the pathogenesis of the disease.
Keywords
Introduction
Colonic diverticular disease (CDD) represents a significant socioeconomic burden and an increasingly common indication for outpatient visits and hospitalization. 1 CDD is and its complications are the final result of a complex interaction between exposure to a low-fiber diet, possible genetic influences, the coexistence of other bowel diseases and the impact of medicine use. 2 Thus, it is not surprising that the researchers were interested in focusing on this topic by investigating both the clinical3,4 and pathophysiologic 5 aspects. The latter, in particular, have received relatively little attention in the past, whereas in recent years there is a renewed interest in the basic mechanisms that underlie the clinical grounds for this disease. This is also due to the fact that it is often possible to have available full-thickness samples of colonic tissue from CDD patients, since the number of surgical procedures performed on these patients worldwide is still consistent. 6
Concerning the pathophysiologic basis of the disease, there is evidence that CDD patients often display abnormalities in colonic neuromuscular function; 5 with dysfunction involving elastosis of the taeniae coli, 7 the collagen content of the viscus’ wall, 8 the neurotransmitter and neurotrophic system,9,10 the muscular component11–13 and the enteric nervous system (ENS).14–16 All these abnormalities probably act in a synergistic manner to cause at least some of the symptoms that are related to the colonic motor dysfunction often observed in these patients.17,18
In a previous study, we documented that patients undergoing emergency surgical procedures for CDD display myenteric plexitis (i.e. infiltration of the myenteric plexus by inflammatory cells, represented by lymphocytes or eosinophils) 19 ; however, this was a relatively small study carried out in a single center.
Because myenteric plexitis may be present in inflammatory bowel diseases 20 and is claimed as an important predictive factor for surgical relapses in other pathological conditions, such as Crohn’s disease,21,22 we carried out a multicenter study in a large group of CDD patients whom underwent surgery, in order to investigate the actual frequency of myenteric plexitis in these subjects.
Materials and methods
Patients and controls
We retrieved resection specimens of patients with colonic diverticulitis from four archival pathology laboratories in Italy (in Brescia, Genova, Perugia and Sassari) and one in Switzerland (Liestal). Patients were subdivided in two groups, i.e. patients undergoing emergency surgery for purulent/fecal peritonitis, resulting from free perforation of a diverticulum (Hinchey Stage III–IV, 23 severe disease according to Ambrosetti classification 24 and patients undergoing elective surgery after either the third or fourth attack of diverticulitis 25 or for sigmoid stenosis, due to recurrent episodes of diverticulitis. 26
We obtained control samples from the proximal resection margin of 15 patients (seven women and eight men; age range 44–83 years) undergoing left hemicolectomy for non-obstructing cancer. These patients were not constipated nor colon-dilated. Control specimens were taken at least 3 cm from the resection margin, from tumor-free areas.
Ethical considerations
Because this was a retrospective study, no individual patient identification was involved and no study-driven clinical intervention was performed; therefore, no ethical approval was necessary.
Methods
Archival resection samples from the proximal resection margins of patients and controls were always analyzed according to a standard protocol. The proximal resection margin was chosen, in order to have a homogeneous standard and to avoid architectural distortions, due to the inflammatory process of diverticulitis. We obtained full-thickness sigmoid samples from formalin-fixed tissue and obtained transversal sections after paraffin embedding and processing for both conventional histological hematoxylin and eosin (H&E) staining and immunohistochemistry (IHC). We assessed the presence of T lymphocytes by IHC, using the monoclonal mouse anti-human CD3 antibody (Dako Cytomation, Carpinteria, CA, USA) at a dilution of 1:40.
Data analysis
Slides from patients and controls were centralized to Brescia, Italy, and then coded and interspersed by a pathologist, to be read by a second pathologist, blinded to the groups (elective, emergency and control groups). The presence of inflammatory infiltrate in the myenteric plexuses (plexitis) was evaluated on both the H&E and CD3-stained slides, and defined as the presence of one or more inflammatory cells (particularly eosinophils or lymphocytes, as these were the only cell types we had identified in our previous study) 19 as penetrating or appositioned to, or within an enteric ganglion; and this was graded based on the appearance of the most severely inflamed ganglion in the available tissue, according to previously-described criteria.19–21 For each ganglion, plexitis was considered as mild in the presence of four or less inflammatory cells, moderate if there were four to nine cells present, and severe if > 10 cells were found.19,21
Statistical analysis
Statistical evaluation was carried out by the chi square test and the Kendall rank correlation coefficient, 27 where appropriate; values of p < 0.05 were chosen for rejection of the null hypothesis.
Results
Demographic and clinical variables and presence of myenteric plexitis in patients with CDD
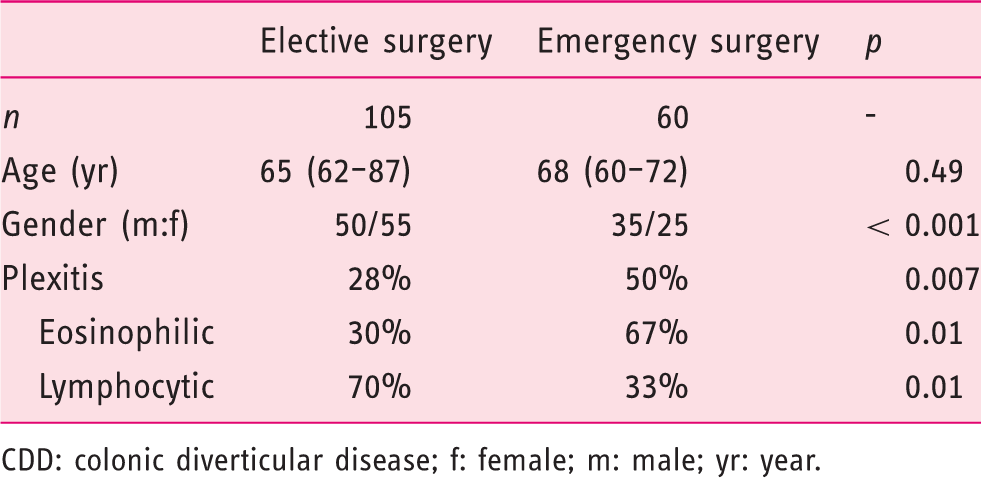
CDD: colonic diverticular disease; f: female; m: male; yr: year.
Overall, myenteric plexitis was found in 60 (36%) patients and was further subdivided into eosinophilic in 29 (48%) patients and lymphocytic in 31 (52%) patients (p = not significant), according to the cell types penetrating the ganglia. No other inflammatory cells (neutrophils, plasmacytes or mast cells) were identified in such a setting.
In terms of demographic variables, no relationship was found between plexitis and gender; whereas plexitis was inversely correlated with age, in patients undergoing emergency surgery (tau = –18; p = 0.03).
By looking at the association with the surgical approach, we found that plexitis was present in 28% of patients undergoing elective surgery and 50% of those undergoing emergency surgery (p = 0.007). In the first group, there were 30% eosinophilic and 70% lymphocytic plexitides; and in the second group, there were 67% eosinophilic and 33% lymphocytic plexitides, respectively.
With respect to the grading of results, we found that myenteric plexitis was mild in 37% of cases (all eosinophilic), moderate in 20% of cases (one-half eosinophilic and one-half lymphocytic), and severe in 43% of the cases (all lymphocytic). We show representative features in Figure 1. In contrast, none of the controls displayed myenteric plexitis.
Representative images of mild ((a) eosinophilic and (b) lymphocytic) and severe ((c) eosinophilic and (d) lymphocytic) myenteric plexitis in patients with CDD. (a) and (c) are H&E stained, at 40x the original magnification; (b) and (d) have the CD3 stain, at 40x the original magnification.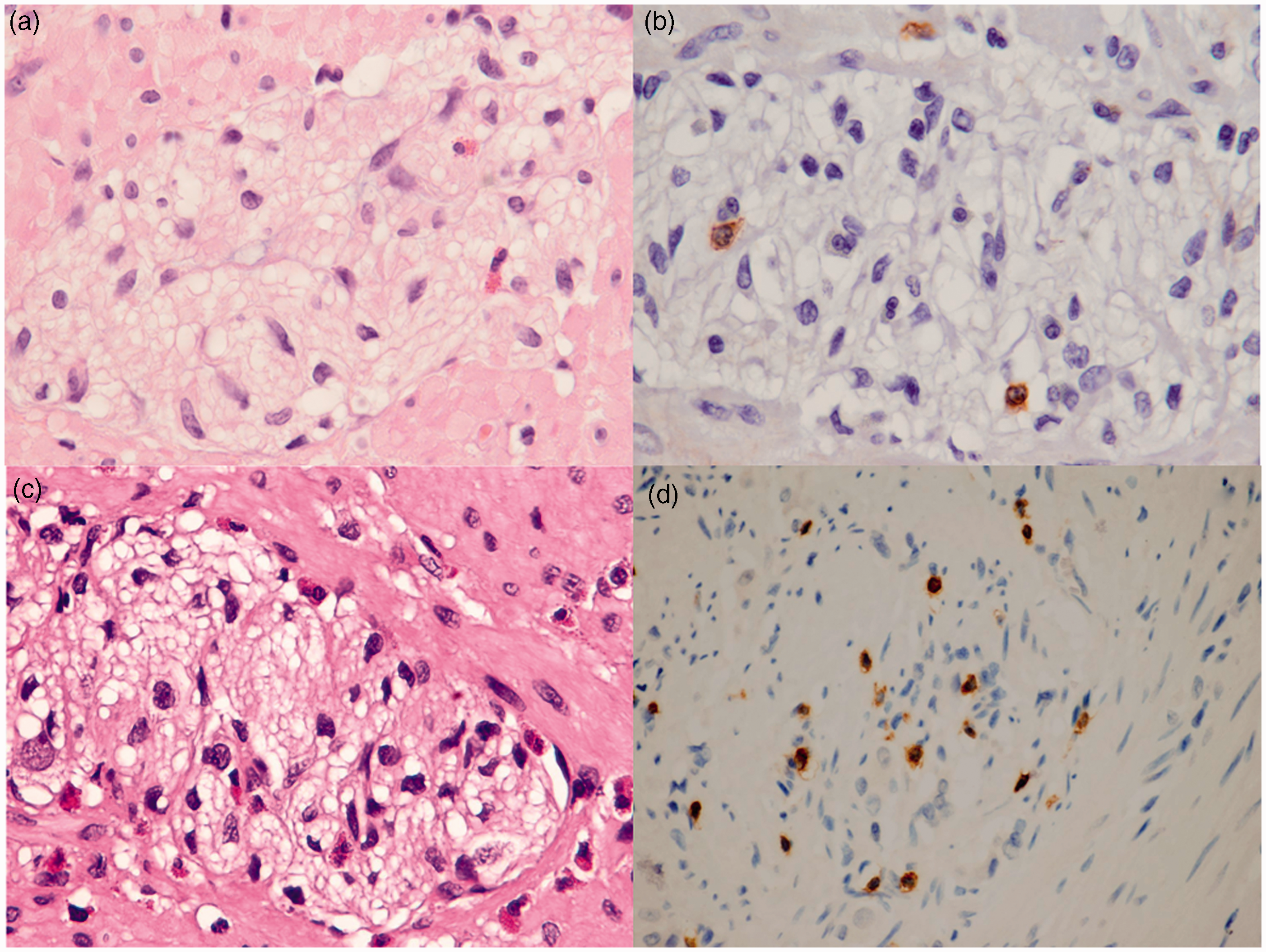
Discussion
Some pathophysiologic mechanisms implicated in the pathogenesis of CDD
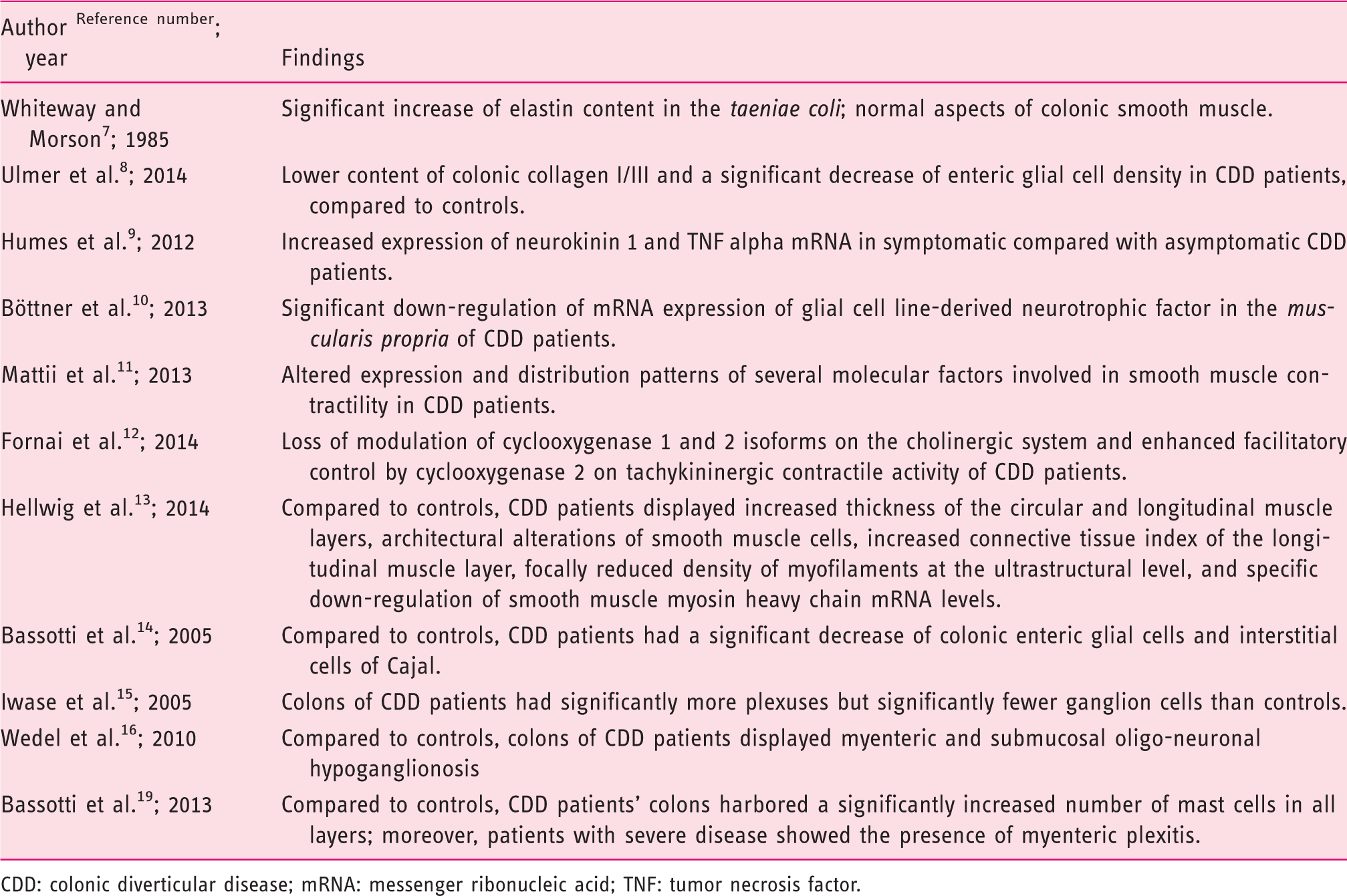
CDD: colonic diverticular disease; mRNA: messenger ribonucleic acid; TNF: tumor necrosis factor.
Myenteric plexitis, also known as myenteric ganglionitis, is the infiltration of the myenteric plexus by inflammatory cells, which is a pathologic finding that has been occasionally described in several conditions, often characterized by abnormal motility of the gut, such as: encephalomyelitis, 28 paraneoplastic neurologic syndromes, 29 viral infections, 30 connective tissue disease, 31 miscellaneous pediatric diseases, 32 severe irritable bowel syndrome (IBS) 33 and intestinal dysmotilities, 34 in addition to inflammatory bowel diseases.20–22 More recently, we reported the presence of myenteric plexitis also being in patients with CDD undergoing emergency surgery. 19
In the present study, which was performed in a large-sized cohort of patients undergoing both emergency and elective surgery for CDD, the prevalence of plexitis was about 40%. We observed that plexitis was more frequent in patients undergoing emergency surgery (50% versus 28%) than elective, and that the emergency surgical procedures were more frequent in younger subjects. Also that plexitis was specifically due to eosinophilic or lymphocytic infiltration of the myenteric ganglia, with the latter being more severe, compared to the effects of the eosinophilic infiltrate.
Analogous with reports in other conditions that are characterized by intestinal dysmotility, infiltration by inflammatory cells of the neural structures that are mainly involved in the control of colonic motor activity might be hypothesized as responsible for the abnormal colonic motility that is frequently documented in CDD.5,17,18 Thus, myenteric plexitis might be considered as an organic marker for some ‘functional’ gut disturbances, including CDD. Indeed, there is presently evidence that several so-called functional disorders may actually be underlined by organic pathologies (low-grade mucosal inflammation, abnormalities of the enteric nervous system, etc.).35,36 These abnormalities may be responsible for at least some of the symptoms these patients complain of. Moreover, some pathophysiologic similarities between CDD and IBS might explain the symptom overlap that is sometimes present in these two conditions. 19
Concerning the cell types involved in plexitis, these were represented by eosinophils and lymphocytes, as is also reported in most of the studies on this topic. This kind of enteric inflammation may be considered as a primary, and not a secondary phenomenon; because in the latter instance, one would expect a granulocytic infiltrate. Moreover, as the infiltrates were restricted to eosinophils and lymphocytes, it could be hypothesized that the presence of a Type 2 (Th2) cell reaction might be associated with maladaptive Th2 inflammation. 37
The presence of eosinophils in such a setting appears particularly intriguing. In fact, eosinophils interact directly with T cells and interface directly and indirectly with the ENS, contain numerous inflammatory mediators and toxic granule proteins; and they also secrete multiple cytokines, chemokines and neuroactive chemicals. 38
A causal relationship between this cell type and intestinal dysmotility has been hypothesized. 39 This relationship is based on experimental animal studies that show intestinal smooth muscle hypercontractility associated with increased cytokine expression. 40 Thus, it is not surprising that immune cell infiltration of the myenteric plexus is thought to be of pathophysiologic relevance, in patients with functional gastrointestinal disorders. 38
Although it could be argued that plexitis (especially in patients with peritonitis) might be a consequence, and not a pathophysiologic cause (or, more likely, a contributing cause) of colonic diverticulitis, we feel that this is unlikely, because this finding was located quite distant from the main inflammatory location/perforation, and more importantly, it was found even in patients without any overt peritonitis.
Our study shows some limitations. Due to its retrospective nature, it was designed to obtain only limited information; we were, therefore, unable to correlate our pathological findings with laboratory data. However, we feel that our finding of myenteric plexitis in almost one-half of CDD patients undergoing surgery is quite interesting, as also is the fact that plexitis was more frequent in younger subjects that were being operated on as an emergency.
Further studies are obviously needed to obtain more information on this: To try to correlate the clinical, laboratory and pathological findings; as well as to establish whether patients with plexitis are more prone to disease relapses.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
The authors declare that there is no conflict of interest.
