Abstract
Background
Current methods to establish the diagnosis of intestinal tuberculosis are inadequate.
Objectives
We aimed to determine the clinical features of intestinal tuberculosis and evaluate inflammatory biomarkers in intestinal as well as pulmonary tuberculosis.
Methods
We recruited 38 intestinal tuberculosis patients, 119 pulmonary tuberculosis patients and 91 controls with functional gastrointestinal disorders between October 2009 and July 2012 for the investigation of clinical features, C-reactive protein (CRP), faecal and serum calprotectin. Faecal calprotectin ≥200 µg/g was used as a cut-off to determine intestinal inflammation of clinical significance. Three patient categories were established: (a) pulmonary tuberculosis and faecal calprotectin <200 µg/g (isolated pulmonary tuberculosis); (b) pulmonary tuberculosis and faecal calprotectin ≥200 µg/g (combined pulmonary and intestinal tuberculosis); (c) isolated intestinal tuberculosis.
Results
Common clinical features of intestinal tuberculosis were abdominal pain, fatigue, weight loss and watery diarrhoea. Intestinal tuberculosis patients had elevated median CRP (10.7 mg/l), faecal calprotectin (320 µg/g) and serum calprotectin (5.7 µg/ml). Complete normalisation of CRP (1.0 mg/L), faecal calprotectin (16 µg/g) and serum calprotectin (1.4 µg/ml)) was seen upon clinical remission. Patients with combined pulmonary and intestinal tuberculosis had the highest levels of CRP (53.8 mg/l) and serum calprotectin (6.5 µg/ml) and presented with signs of more severe disease.
Conclusion
Calprotectin analysis reveals intestinal tuberculosis in patients with pulmonary tuberculosis and pinpoints those in need of rigorous follow-up.
Keywords
Background
Tuberculosis (TB) is an old disease which still harms mankind more than any other infectious disease. 1 Intestinal TB (ITB) is a common form of extra-pulmonary TB in India, the aetiologic agent being Mycobacterium tuberculosis (M.tb). Disease manifestation depends on the site of lesion in the gastrointestinal tract, although general signs such as fever, weight loss, cachexia and diarrhoea and symptoms of abdominal pain and malaise are usually present.2–8 However, the presentation and pathological findings of ITB may vary and be non-specific, and the disease may easily be confounded with other gastrointestinal diseases. 9
Gastrointestinal endoscopic examination plays a key role in the diagnosis of ITB. However, endoscopy with biopsies, histopathological investigation and advanced molecular laboratory work-up are costly and require highly qualified staff. This also applies to complementary diagnostic modalities such as computer tomography, magnetic resonance imaging and explorative laparotomy. Expensive interferon-γ release assays can be used to screen for TB in non-endemic settings, but have proved rather unspecific for detection of active TB in endemic areas. Co-existing malnutrition and helminth infections may further decrease the sensitivity of these assays. 10 All of the above require resources which are not easily available in economically deprived TB-endemic areas. Unfortunately, inexpensive diagnostic modalities such as acid-fast staining of biopsies or sputum, Mantoux tuberculin skin tests, M.tb cultures and chest X-rays have shown poor diagnostic performance in ITB.2,4–6 Consequently, prescribing a course of empiric anti-tuberculous chemotherapeutic drug treatment (ATT), with clinical re-evaluation of the diagnosis after 2 months, still seems to be the diagnostic method of choice in many TB-endemic regions.3,4,8
Calprotectin is a complex of myeloid-related proteins (MRP 8/14) released from neutrophilic granulocytes and monocytes/macrophages during activation or cell death. 11 Faecal calprotectin (FC) is routinely measured in patients with inflammatory bowel disease to monitor relapse of disease and/or effect of treatment. 12 Serum calprotectin (SC) has been found elevated in various inflammatory diseases such as rheumatoid arthritis, systemic lupus erythematosus, bacterial infections including sepsis and active pulmonary TB (PTB).13,14
The aim of this study was to describe the initial disease characteristics of ITB in Southern India and to evaluate calprotectin in ITB and PTB.
Materials and methods
Demographic and biometric variables of controls, pulmonary TB patients (PTB), intestinal TB (ITB) patients and patients with combined PTB and ITB (PITB)
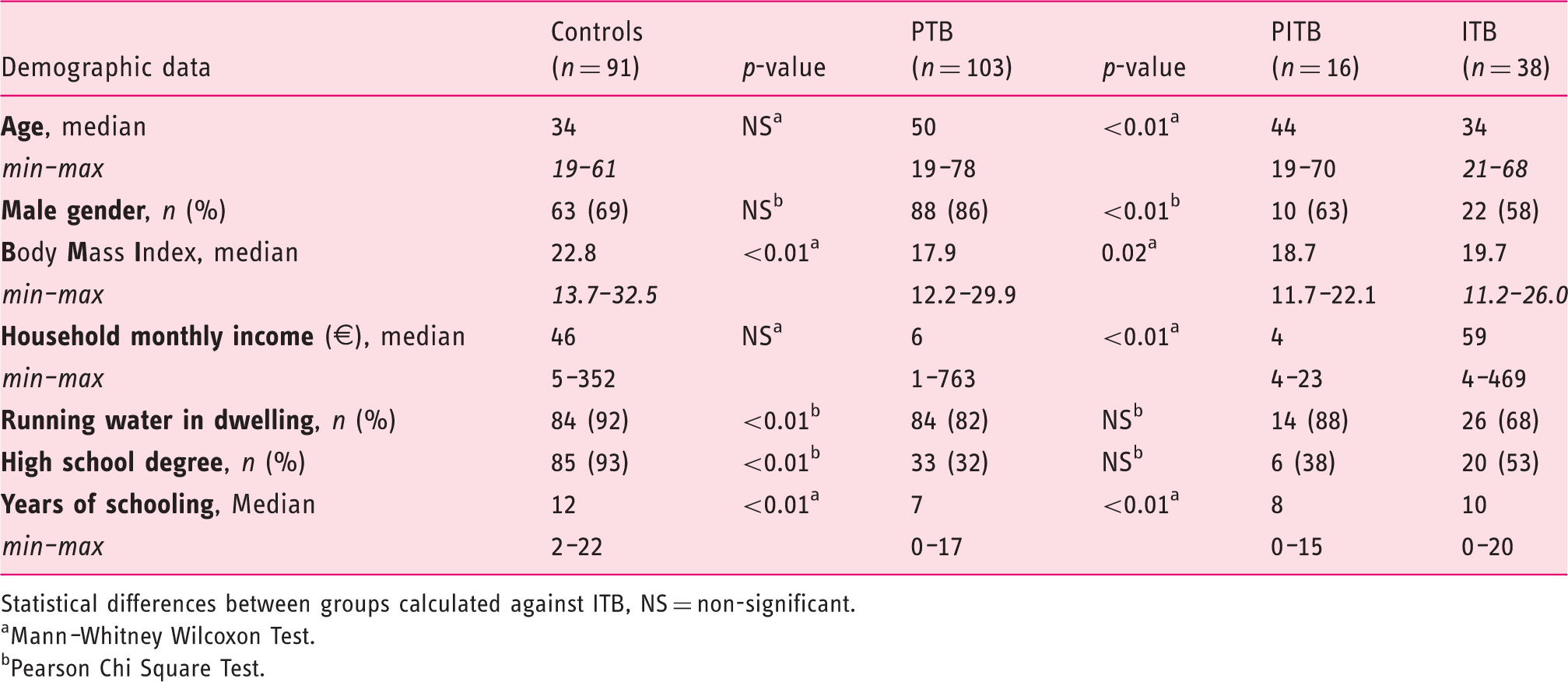
Statistical differences between groups calculated against ITB, NS = non-significant.
Mann–Whitney Wilcoxon Test.
Pearson Chi Square Test.
Exclusion criteria were age below 18 years, co-existing malignancy or Human Immunodeficiency Virus infection. Demographic information and disease-associated signs and symptoms were recorded in electronic case record forms prior to physical examination and further investigations. Abdominal pain and/or diarrhoea were classified as the ‘first appearance of disease’ in the patient history.
Blood samples were drawn for serum C-reactive protein (CRP) and SC analyses in parallel with the collection of faeces spot samples for FC analysis at baseline and on follow-up. Faecal aliquots and separated blood serum vials were stored at −20℃ until analysis. As recommended by the local laboratory, CRP <6.0 mg/l was regarded as normal. FC and SC were analysed with ELISA technology using EK-CAL and MRP 8/14 kits respectively, according to the manufacturer’s recommendations (Bühlmann Laboratories AG, Basel, Switzerland). For SC, cut-off levels are yet to be established and levels depend on the underlying condition. 13 For FC, a threshold of 200 µg/g was used as a cut-off to predict intestinal inflammation of clinical importance.15,16
In addition, 91 subjects with functional gastrointestinal disorders such as irritable bowel syndrome or functional dyspepsia were consecutively recruited as controls during the same period of time. Similarly, 119 patients with smear-positive PTB were recruited as TB controls, and these were further divided into two sub-groups based on their FC levels. Thus, three separate categories of TB patients were established: (a) patients with smear-positive pulmonary disease and FC <200 µg/g, hereafter designated ‘PTB’; (b) patients with smear-positive pulmonary disease and FC ≥200 µg/g, hereafter defined as combined pulmonary and intestinal TB, designated ‘PITB’; and (c) patients with primarily intestinal disease as per modified Paustian’s criteria, 17 hereafter designated ‘ITB’.
Statistics
As data were skewed, the continuous variables were described with median and inter-quartile range (IQR), while categorical variables were listed as counts and percentages. Crude differences between the groups were assessed with χ2 or Fischer’s exact tests for the categorical variables and Mann–Whitney Wilcoxon tests for the continuous variables. P < 0.05 was considered statistically significant to differentiate between the groups. Statistical Package for the Social Sciences software Version 18.1 was used to perform all analyses. The choice of statistical methods and interpretation of results were assisted by a biomedical statistician.
Ethics
The study was approved with respect to the Helsinki declaration by the The Ethical Committee of Sree Gokulam Medical College and Research Foundation, Trivandrum, India. Written informed consent was obtained from every participant.
Results
Laboratory variables of controls, pulmonary TB patients (PTB), intestinal TB (ITB) patients and patients with combined PTB and ITB (PITB)
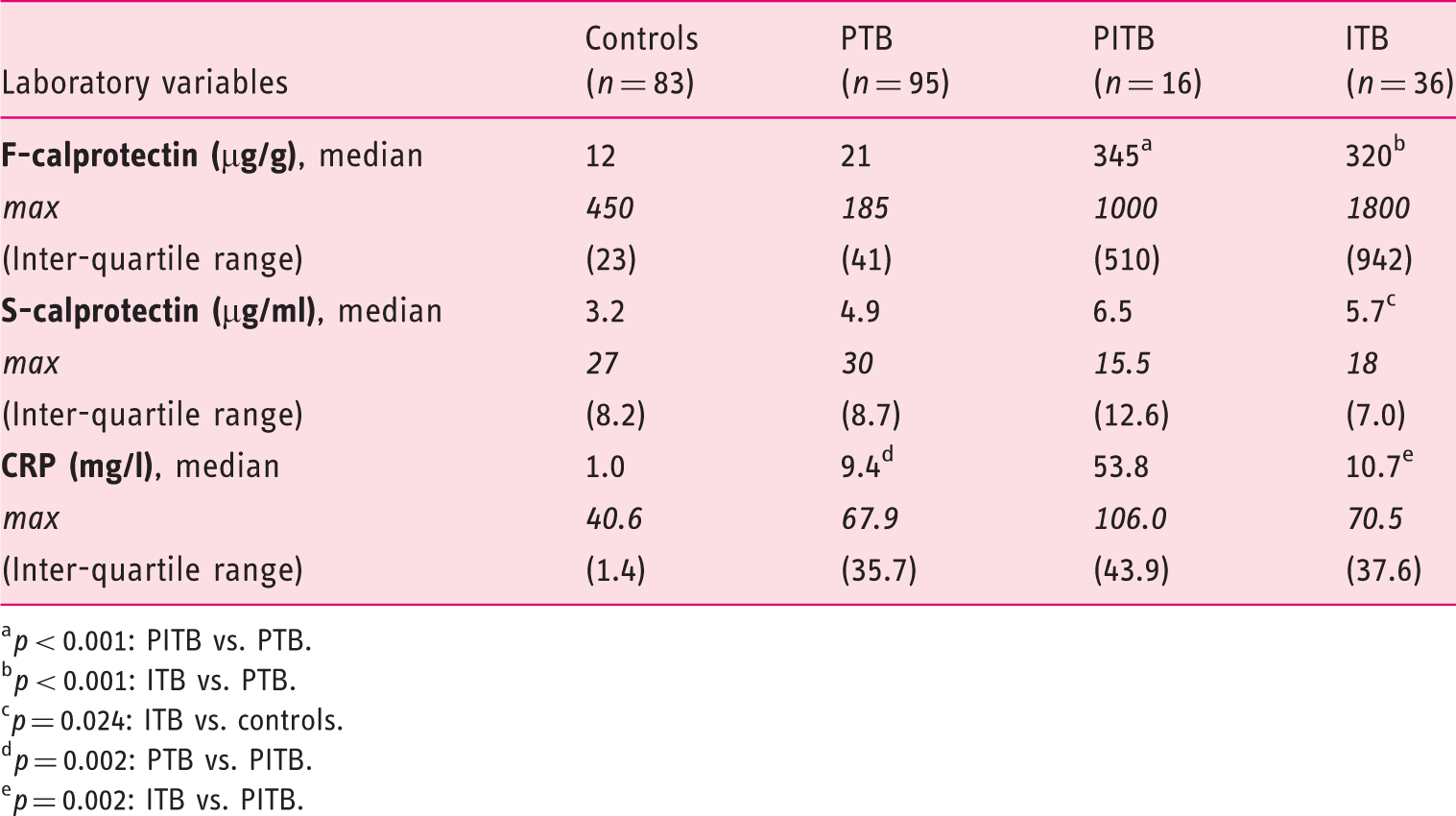
p < 0.001: PITB vs. PTB.
p < 0.001: ITB vs. PTB.
p = 0.024: ITB vs. controls.
p = 0.002: PTB vs. PITB.
p = 0.002: ITB vs. PITB.
Both the ITB and the PITB groups showed significantly elevated median FC levels when compared with the PTB group and the controls, respectively (Table 2, Figure 1(a)). Although median SC levels were higher in all TB groups compared with the controls, only the ITB group showed significantly elevated median SC (Table 2). The highest median SC was recorded in the PITB group. Median CRP was significantly elevated in all TB groups compared with the controls. Furthermore, median CRP was significantly higher in the PITB group compared with the PTB and the ITB groups, respectively (Table 2, Figure 1(b)).
(a) Faecal calprotectin and (b) CRP levels in tuberculosis.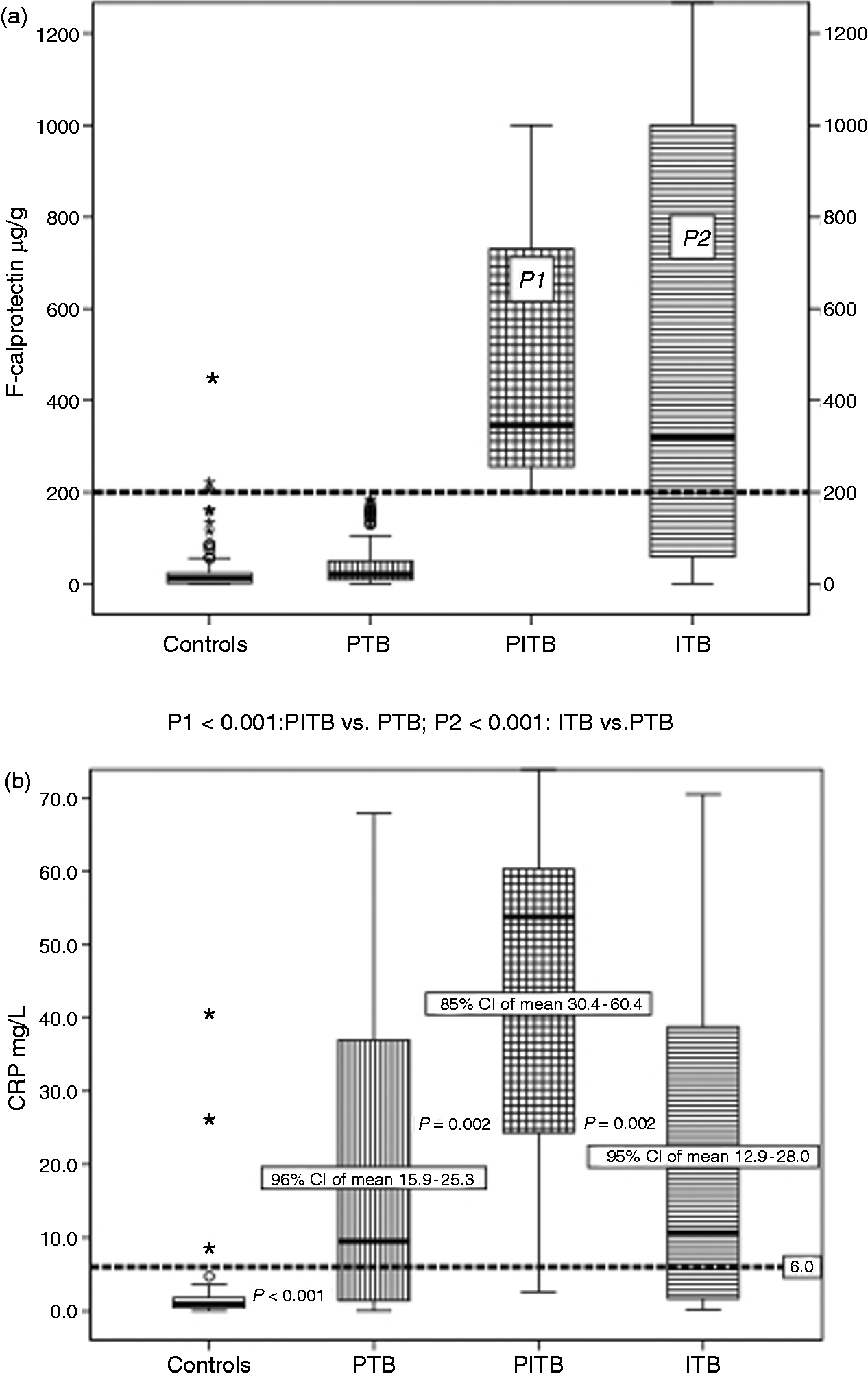
The majority of ITB patients reported of abdominal pain and fatigue as their leading symptoms (Figure 2). With regards to signs of disease, weight loss, watery diarrhoea and cachexia were most commonly observed (Figure 2). Low-grade fever was found in half of the patients. One-third presented with abdominal mass on physical examination. The median duration of symptoms prior to diagnosis was 6 months, but a considerably longer disease course was observed in some of the patients. The distribution of symptoms and signs was equal between patients with short and long courses of disease, respectively. Yet, more patients with long-standing disease reported fatigue and presented with abdominal mass on physical examination (data not shown).
Intestinal tuberculosis: disease-associated features in 38 patients.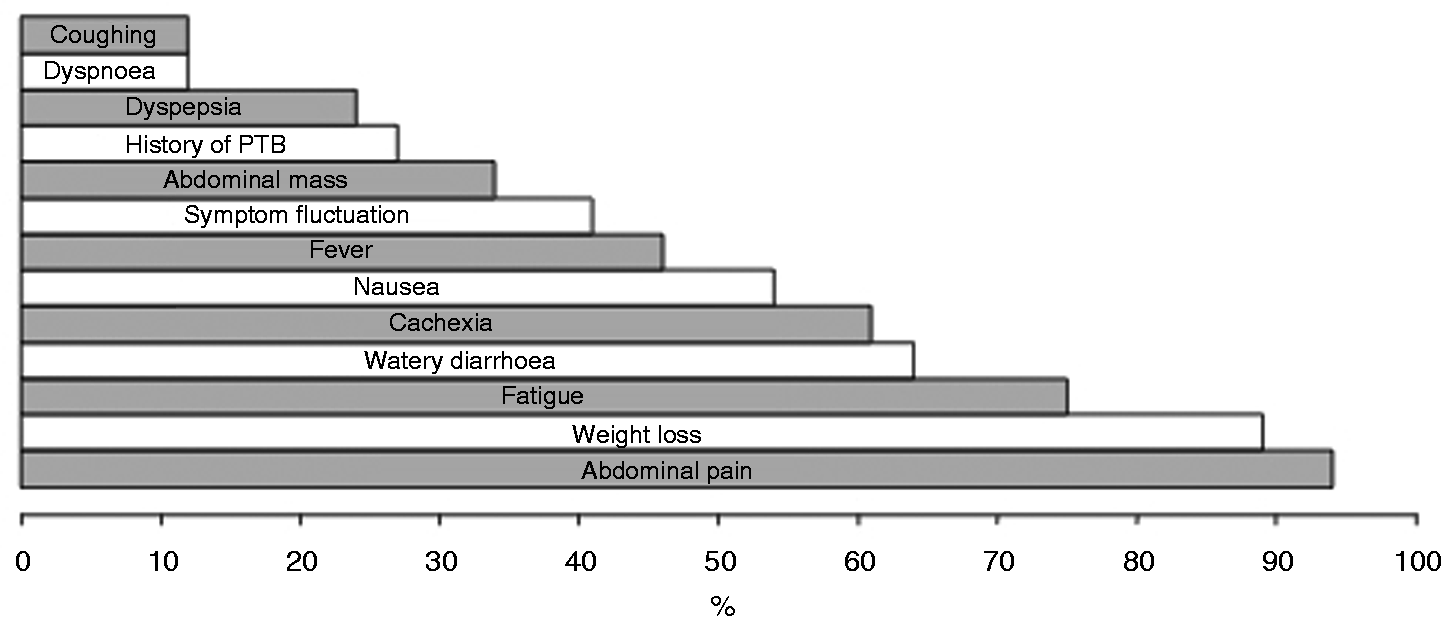
Of our patients, 59% reported that their symptoms were constant. However, disease manifestation did not differ significantly from those with a more fluctuating course of symptoms.
Five ITB patients had PTB and one-quarter of the ITB patients reported previous exposure to PTB either in their own or in their family history. Typically, ITB patients with PTB also reported coughing and daily tobacco smoking.
There was no statistical significant difference between men and women with regards to the distribution of signs and symptoms (data not shown). Of socioeconomic variables, the average monthly household income was 59€. Two-thirds of the patients lived in households with running water. The average educational level was 10 school years and half of the patients had completed a high school degree or higher (Table 1).
The distribution of signs and symptoms commonly seen in TB is described in Figure 3 Distribution of disease characteristics in tuberculosis.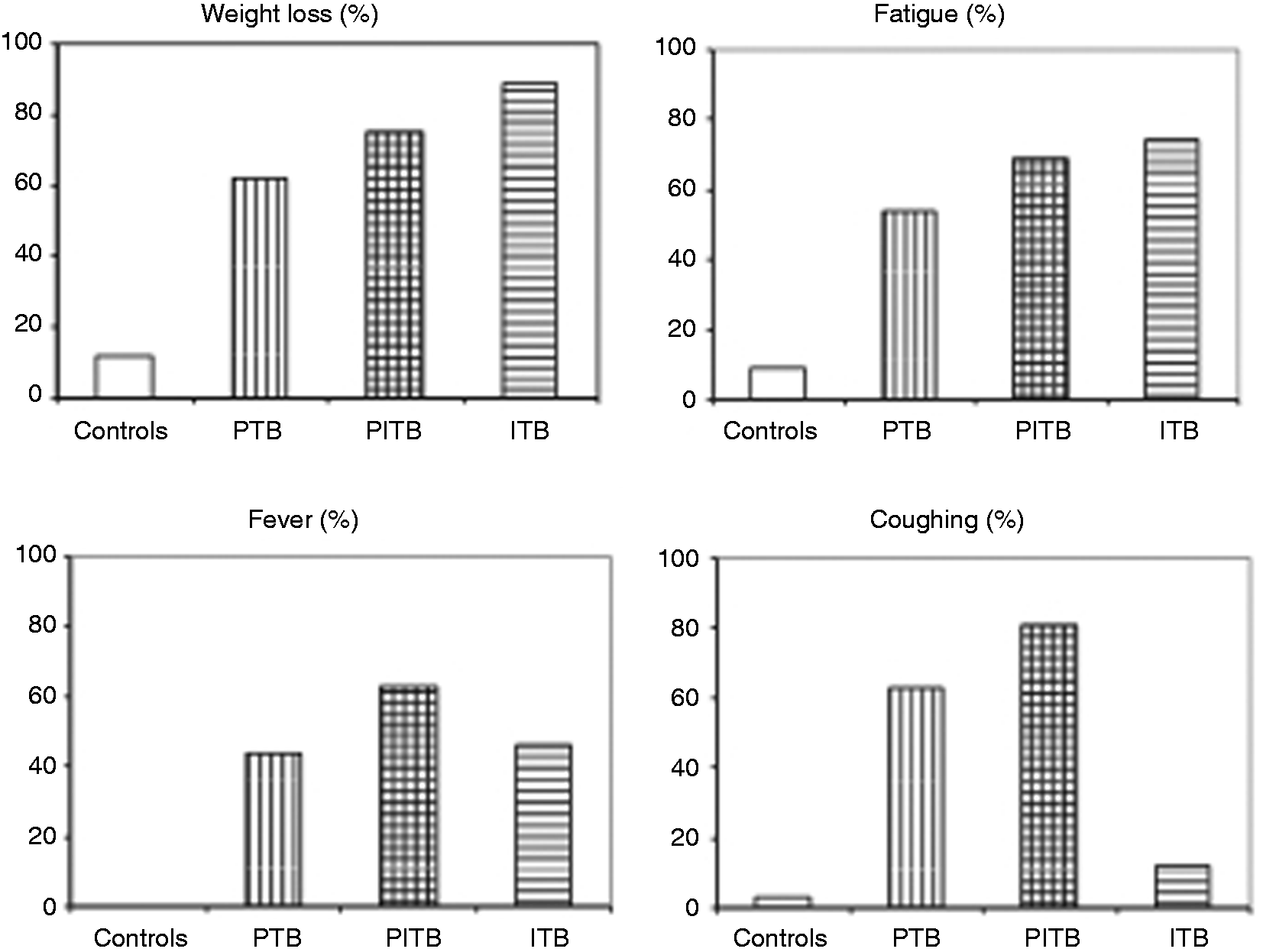
Discussion
Recently, a correlation was found between calprotectin levels, the number of neutrophils, the levels of neutrophil-attracting chemokines and the extent of lung inflammation in patients with active PTB. 14 Also, the pro-inflammatory cytokine interleukin-17, which mediates calprotectin release and neutrophil accumulation, has been found upregulated in peripheral blood mononuclear cells of ITB patients. 18 In this study, we demonstrated high calprotectin levels in patients with ITB. Clinical remission and a consistent decrease in CRP, SC and FC levels were observed on follow-up after ATT, suggestive of a positive treatment response. Endoscopic normalisation was observed in the six patients who agreed to undergo follow-up endoscopic investigations. The remaining patients, although in clinical and biochemical remission, refused follow-up endoscopy. Calprotectin could become a surrogate marker for remission in this patient category, similar to its application in inflammatory bowel disease. 19 Adherence to follow-up is crucial to assure an adequate treatment outcome and to decrease the risk of M.tb dissemination. This could be achieved by offering simple, non-invasive and affordable screening tools.
Median FC was significantly elevated in the PITB group as well as in the ITB group, suggestive of comparable pathology. Conversely, FC was normal in the PTB patients and not differing from the median FC in the controls. Median SC was moderately elevated in the PITB group, suggestive of systemic inflammation. Although the highest SC levels were detected in the PITB group, SC did not allow for discrimination between the patient categories. Larger study populations are needed to investigate the role of SC in TB.
By being a chronic granulomatous disease of the gut, Crohn’s disease (CD) resembles ITB in pathophysiological, histological and even immunological ways. 9 Therefore, considering the lack of previous studies on FC in ITB, we anticipated similar results in ITB as those reported in studies on FC in CD. There is an ongoing debate with regards to which FC threshold levels should be used as a cut-off to determine intestinal inflammation of clinical significance in CD. Previously, a cut-off level of 200 µg/g was suggested to predict endoscopically active disease, 20 whereas lately a cut-off level of 250 µg/g has been suggested.16,21,22 Just recently, an even higher cut-off level of 274 µg/g was suggested to determine endoscopic remission. 15 Also of interest, a cut-off of 150 µg/g has been suggested to determine organic intestinal disease in a primary health care setting. 23 Different FC kit manufacturers operate with different calprotectin antigens, thereby causing considerable inter-assay variability. The latter studies established FC with kits from the same manufacturer as in our study.15,23 Based on the aforementioned studies we chose an FC cut-off level of 200 µg/g to rule out intestinal inflammation of clinical significance in PTB.
Interestingly, some studies have shown a higher increase in the incidence of extra-pulmonary TB than of pulmonary TB.3,24 Whether the rise is a result of increased awareness or because of specific virulence factors in certain M.tb strains is unknown. Regardless, PTB patients with co-existing gastrointestinal TB lesions have been described as more seriously ill than patients with isolated PTB. 25 However, according to the case definitions of TB established by the World Health Organisation, these patients are classified as PTB. We question whether PTB cases with concomitant TB infestation in other organs should be described as such, in order to receive appropriate follow-up investigations. Furthermore, the evaluation of transmission mechanisms, treatment outcomes and mortality rates of the various types of TB may be facilitated if patients with combined TB infections are reported as such to the central TB registries.
The importance of distinguishing PITB as a more severe form of TB is supported by several findings. First, when asked about their levels of energy, three-quarters of the ITB patients confirmed that they felt deprived of energy since the onset of disease. Previous reports on TB patients’ perception of disease are scarce, but the few ones published have shown that during the course of disease and treatment half of all PTB patients feel deprived of energy.26,27 Fatigue could be related to the release of systemic inflammatory mediators in response to TB itself, but also due to ITB-associated anaemia and/or diarrhoea with malabsorption. We found that the proportion reporting of fatigue increased in the following order: PTB–PITB–ITB, and a significant difference was seen between the PTB and the ITB groups.
Second, we recorded lower body weight and body mass index in the ITB patients compared with the controls with functional gastrointestinal disorders. Leptin, an appetite-stimulating hormone produced in adipocytes, has been shown to decline in cachectic TB patients. 28 Compared with the PTB patients, a substantial number of the ITB patients were cachectic. This could reflect reduced energy uptake and additional mechanisms of weight loss, i.e. malabsorption of nutrients and/or decreased intestinal transit time. 29
Third, CRP levels may be elevated in both pulmonary and extra-pulmonary active TB.30,31 A small study on TB-diseased children found an inverse relationship between CRP and body mass index and between leptin levels and CRP, with poorer prognosis in patients with the highest CRP levels. 32 We recorded a five-fold increase in CRP and lower body weight in the PITB group compared with the other groups, suggestive of a more severe disease.
Finally, fever was reported in two-thirds of the PITB patients. In contrast, less than half of the patients in the two other TB groups had fever. Previously, low-grade fever has been described as frequent in ITB,7,33,34 while others have described it as less frequent.2,4,35 Coughing was mostly observed in the PITB patients, indicating a more active pulmonary disease.
Therefore, a more severe form of TB should generally be suspected in patients with PITB, although the severity of disease may vary between patients. As a ‘proof of concept’ of the association between PTB, high FC and suspected intestinal pathology, one of our high FC PTB patients showed ulcerohypertrophic lesions in the ascending colon upon endoscopy. Unfortunately, the remainder of the PITB patients refused lower gastrointestinal endoscopy. We believe that distinguishing patients with severe TB is important because they require more rigorous follow-up. The combination of CRP and FC analysis could be useful supplements in the initial investigation and follow-up of this patient category. International guidelines on case registration of PTB are perhaps not uniform, and may need refinement according to the population and setting in question. This especially counts for smear-negative TB patients where the diagnosis is the crux and tools for detecting active TB are missing. Calprotectin could prove as a useful adjunct in this regard as raised levels indicate active infection.
A median symptom duration of 6 months prior to diagnosis was found in another recent prospective study on ITB from India. 33 However, most studies from South and East Asia describe a symptom history of 1–2 years prior to diagnosis.2,4 However, these were retrospective and exclusively conducted in tertiary care centres. Nevertheless, the most common features detected were consistent with the literature: abdominal pain, weight loss, cachexia and diarrhoea.2,4,5,33
Only the minority of the ITB patients had concurrent PTB, similar to other studies.2,4,6 Rather than indirect infestation of bacilli first harboured in the lungs, we therefore suggest that the intestines are primarily infested by mycobacteria directly acquired from oral ingestion. The challenge of curbing the TB epidemic could partly be due to the relatively long-lasting and rather silent hideaway of M.tb in the intestinal lymphatics.
Our study has some limitations. First, the small number of 38 ITB patients only allows for crude statistical analyses, and the comparison of our results with previous studies may be difficult. Second, the study has some missing data as a complete record of all study variables was difficult to achieve, thus limiting statistical analyses. Third, because sampling and preservation of blood serum for SC analysis was performed at four different centres, variations in sample handling may have occurred. Even so, all technicians followed pre-defined standard operating procedures and only minimal alterations in SC concentrations were expected. We believe that our study is of clinical relevance because the data were population based and collected from routine clinical practice in secondary and tertiary care centres. To our knowledge, this is the first study which describes calprotectin in ITB. To further assess calprotectin in patients in clinical remission, future studies should aim at strict adherence to follow-up investigations.
In summary, FC may be used as a biomarker in the diagnostic work-up of ITB. FC analysis in PTB patients with abdominal symptoms is a simple and non-invasive tool to reveal ITB co-infection and pinpoints patients in need of a more rigorous follow-up. Also, calprotectin may be used in the evaluation of treatment response after ATT.
Footnotes
Funding
This work was supported by The South-Eastern Norwegian Regional Health Authority [grant number 2011132].
Acknowledgements
G Larsson would like to thank the staff at the Population Health and Research Institute in Trivandrum, India, with special regards to Mrs. Suja, for handling and preparing patient samples and filing the data. Special thanks also to investigator Dr. VG Mohan Prasad at Coimbatore centre for recruiting patients, and to Dr. B Holm and Mrs. AM Tangen at Lovisenberg Diaconal Hospital for allowing the initiation and financial support of this study. Thanks to Ms. MC Småstuen for assisting on the choice of statistical methods. Dr. PC Klepp is acknowledged for proofreading and thorough support during the study. Calprotectin analyses were performed with kits provided without cost by Bühlmann Laboratories AG, Basel, Switzerland. Dr. A Røseth kindly advised on the analysis of calprotectin.
