Abstract
A 2019 study revealed more than 80 products available online with implied benefits to alcohol hangover. Since then, several developments may have influenced the market. In order to gauge the extent to which this may have changed in the ensuing 5 years, two market evaluations (25 March 2023 and 7 November 2024) were conducted via the online web shop www.amazon.com, using the search term “hangover treatment.” In 2023, there were 38 different hangover products available. The most common dosage forms were capsules (14) and tablets (6), and most frequently reported ingredients were dihydromyricetin (DHM, 52.6% of products), vitamin B1 (42.1%), vitamin B12 (42.1%), sodium (42.1%), and vitamin B6 (39.5%). Only one product contained N-acetyl L-cysteine. Exactly half of the marketed products (19, 50.0%) made illegal disease modification claims. The second market evaluation (7 November 2024) revealed 46 hangover products. The most common dosage forms were capsules (18) and transdermal patches (13), and the most frequently reported ingredients were vitamin B6 (56.5% of products), vitamin C (54.3%), vitamin B1 (50.0%), and vitamin B12 (50.0%). Also, DHM (47.8%) and milk thistle extract (47.8%) were popular. In conclusion, over the 5 years to 2024 the US hangover product market has dramatically changed, including a significant reduction in the number of marketed hangover products. DHM, vitamins, and minerals remained the most popular ingredients of hangover products. Illegal disease claims were made by 41.3% of the marketed products. Of concern, the efficacy to prevent or reduce alcohol hangover has not been demonstrated for any product on the US hangover market.
Introduction
The alcohol hangover, defined as the combination of negative mental and physical symptoms, which may be experienced the day after a single episode of alcohol consumption, starting when blood alcohol concentration approaches zero (Verster et al., 2020), is the most frequently reported negative consequence of alcohol consumption (Verster et al., 2009). There is an ongoing debate about whether hangover treatments should be developed per se (Išerić et al., 2024), as there may be a risk of overindulgence when an effective hangover treatment would be available. However, most research into this possibility does not support this premise. For example, a 2017 study among n = 1837 Dutch students revealed that a relatively small minority (13.4%) currently deliberately moderated their alcohol consumption to prevent hangovers while also reporting that the availability of an effective hangover treatment would increase their alcohol consumption (Mackus et al., 2017). Most students (71.6%), however, reported that the availability of an effective hangover treatment would not alter their drinking behavior. The data suggested that while they wish to enjoy an evening out including the consumption of alcohol, participants also wish to wake up refreshed the next day and participate fully in society, unhindered by any hangover effects (Mackus et al., 2017). Although more research is needed beyond student samples, the outcomes of this study suggest that there is a clear need for an effective and safe hangover treatment.
Market research confirms that there is a growing demand for hangover treatments (Grand View Research, 2024), with many hangover products being sold via the Internet. However, most of these hangover products have not been evaluated in double-blind, placebo-controlled trials in humans. Those products that have been investigated either lack efficacy, or have limited efficacy by selectively counteracting only a subset of hangover symptoms (e.g. reducing headache) (Pittler et al., 2005; Verster and Penning, 2010; Jayawardena et al., 2017; Roberts et al., 2022). Regarding individual ingredients, low-quality evidence for efficiency was found for clove extract, tolfenamic acid, pyritinol (vitamin B6), red ginseng, and Korean pear juice (Roberts et al., 2022). However, further research is needed to confirm their efficacy. Taken together, while there are many hangover products marketed, scientific support for their efficacy and safety is usually lacking.
In December 2019, before the start of the 2019 coronavirus (COVID-19) pandemic, Verster et al. (2021a) evaluated the US hangover product market. To this extent, the authors searched the website www.Amazon.com for available hangover treatments in the USA. The market evaluation retrieved 82 different products, which were all dietary supplements. Regarding the mode of administration, most products were capsules (46); other products were tablets (10), powders (10), drinks (9), and transdermal patches (5). The hangover products contained 215 different ingredients. These often included vitamins and minerals, and a variety of “natural” ingredients (e.g. plant extracts and herbs). A summary of the 20 most popular ingredients is presented in Table 1. The five most popular ingredients were vitamin B1, vitamin B6, milk thistle extract (Silymarin), dihydromyricetin (DHM), and N-acetyl L-cysteine (NAC).
Top 20 most reported ingredients of hangover treatments sold on US Amazon per 14 December 2019.

Also includes DHM from hovenia dulcis, Japanese raisin tea, vine leaf tea extract, and ampelopsis grossedentara leaf.
According to the US Food and Drug Administration (FDA), alcohol intoxication meets the definition of disease (Code of Federal Regulations, 2024). FDA considers the alcohol hangover as a sign or symptom of alcohol intoxication and therefore also treats the alcohol hangover as a disease (FDA, 2020a). According to their regulations, only FDA-approved registered drugs are allowed to make disease claims (FDA, 2000; FDA, 2002). Disease claims include statements that a product mitigates, treats, cures, or prevents a disease, or if explicit or implicit claims are made that a product has an effect on the characteristic signs or symptoms of a disease (Code of Federal Regulations, 2024). As dietary supplements are not registered as drugs, they are prohibited from making disease-related claims (FDA, 2000; FDA, 2002). However, dietary products are allowed to make general function and structure claims (e.g. “helps to maintain proper immune function”). Despite these clear regulations, the 2019 market evaluation revealed that 53 of 82 products (64.6%) made illegal disease claims. An exception is made by FDA for hangover products that fall under the 1991 Overindulgence Monograph (FDA, 1991a), which describes orally administered drug products for relief of symptoms associated with overindulgence in food and drink for over-the-counter (OTC) human use, and the related monographs on OTC antacid drug products (FDA, 1991b), OTC stimulant drug products (FDA, 1991c), and OTC internal analgesic, antipyretic, and antirheumatic drug products (FDA, 1991d). These monographs conclude that specific combinations of OTC drugs that are recognized by FDA as safe can be used for the treatment of hangovers. These comprise products that contain a combination of either (1) an OTC antacid (e.g. aluminum hydroxide) and an OTC analgesic (i.e. aspirin or acetaminophen), or (2) an OTC analgesic (i.e. aspirin or acetaminophen) with an OTC stimulant (i.e. caffeine). These two combination products are permitted for hangover relief and FDA allows disease claims for these products. Interestingly, FDA did not request any clinical studies to demonstrate the efficacy of these combination products (FDA, 1991a), and up to now such studies have not been conducted or made public. In the 2019 market evaluation, four aspirin and caffeine combination products were marketed but there were no antacid and analgesic combination products.
Finally, dietary supplement products are not allowed to include OTC or prescription drugs as ingredient. FDA considers products that do include registered drugs as new drugs that require evaluation of efficacy and safety and FDA approval before marketing. An example of an ingredient that is not allowed to be included in dietary supplements is NAC, which in 1963 was approved by the FDA as a drug (FDA, 2022). Notwithstanding this, the 2019 market evaluation revealed that 45.1% of the 82 available hangover products in the United States included NAC as ingredient.
Since December 2019, several developments may have had a significant impact on the US hangover product landscape. On 11 March 2020, the World Health Organization (WHO) declared COVID-19 a worldwide pandemic (WHO, 2020), and infection rates in the United States rose rapidly (CDC, 2025). During the COVID-19 pandemic, in several US states lockdown periods or other measures to spread the virus were enforced, including the closure of venues where alcohol is regularly consumed (e.g. restaurants, bars, and clubs), and major events were many people gather were either canceled (e.g. concerts) or continued without attendants (e.g. sports games) (Haffajee and Mello, 2020). While these developments may have limited the accessibility to alcohol, the literature on alcohol consumption during the COVID-19 pandemic in the USA shows mixed results (Merlo et al., 2025). Whereas most studies reported a decrease or unchanged alcohol consumption (Avery et al., 2020; Knell et al., 2020; Capasso et al., 2021; Jaffe et al., 2021), other studies reported an increase in alcohol consumption (Grossman et al., 2020, Salerno et al., 2021; Weerakoon et al., 2021). The mixed results are most likely due the fact that most studies comprised convenience samples that are not representative for the general US population. More reliable data comes from US alcoholic beverage sales statistics. These statistics show a steady yearly rise in alcoholic beverage sales in the USA before the COVID-19 pandemic. Whereas off-premise alcoholic beverage sales continued to grow during the COVID-19 pandemic, on-premise sales showed a 27% reduction from March to September 2020 (Castaldelli-Maia et al. 2021; Lee et al., 2021). Other data revealed that higher viral incidence was linked to both lower alcohol purchases and consumption (Moskatel and Slusky, 2023). Taken together, the COVID-19 pandemic was associated with changes in both alcoholic beverages sales and alcohol consumption in the USA. These changes may have impacted the demands for hangover treatments. It is likely that, with a possible reduction of revenues during the COVID-19 pandemic, companies with relative small capital resources (e.g. startups) may have discontinued. Second, since 2020 FDA has sent several warning letters to companies that made disease claims regarding hangover reduction (FDA, 2020a; FDA, 2020b; Voelker, 2020). The warning letters underline that FDA treats the alcohol hangover as a disease, and that products with claims of preventing or mitigating hangovers should be registered drugs. Third, FDA also sent warning letters to companies that included NAC as ingredient of their hangover product (FDA, 2020a; FDA, 2020b). Fourth, in 2022, the WHO adopted the definition of the alcohol hangover (Verster et al., 2020) and now lists the alcohol hangover as separate child entity of alcohol intoxication, a disease, in the 11th edition of the International Classification of Diseases (ICD-11) (Išerić et al., 2024). According to this classification, hangover treatments should be registered drugs. Finally, increasing scientific evidence has been published showing that popular ingredients of the hangover products that are marketed in the USA are not effective in preventing or reducing hangovers. Except for vitamin B6 (Khan et al., 1973), there is no scientific evidence from double-blind placebo-controlled clinical trials in humans that vitamins have any effect of the presence and severity of the alcohol hangover. Scientific evidence for the efficacy of or milk thistle extract (Silybum marianum) or products that fall under the 1991 Overindulgence Monograph (e.g. aspirin and caffeine combinations) is also lacking. Further, recent double-blind placebo-controlled studies found that both NAC (Podobnik et al., 2024) and DHM (Verster et al., 2021b; Lee et al., 2024) do not prevent hangovers, nor do they reduce overall hangover severity.
Together, these developments may have had a significant impact of the alcohol hangover treatment market in the USA. To evaluate this, two new searches at www.Amazon.com were conducted. The first evaluation was conducted in March 2023 and the second evaluation in November 2024.
Methods
Similar to the 2019 market evaluation (Verster et al., 2021a), a search for “hangover treatment” was conducted via the online web shop www.Amazon.com. The delivery postal code was set at 10001 (New York, USA). A first search was conducted on 25 March 2023. A second search was conducted 7 November 2024.
To be included in the analysis, the retrieved product's use should be related to alcohol hangovers. To evaluate this, the www.Amazon.com website (text, package, and product pictures) were checked. Excluded were irrelevant products (e.g. books and movies) and products with no referral to alcohol, hangover, or recovery from a night out (e.g. common pain medication, vitamin supplements for daily use, and sports drinks for rehydration purposes). In ambiguous or unclear cases, if available the company's website was consulted to verify the use of the product. Duplicate products were excluded. This included when the same product was sold in different package sizes, or when the same product was marketed in different flavors (e.g. strawberry or orange flavor).
For each product, the dosage form (delivery format) was recorded (e.g. capsules, tablets, powders, drinks, or transdermal patches). The ingredients and their amounts per serving were determined from the website, package, or nutrition label on the product. Only “active” ingredients were listed, that is, those on the nutrition label or part of the “proprietary blend.” Inactive ingredients, such as flavors, colors, stabilizers, preservatives, and fillers were not considered. The amount of the ingredients, if reported, were compared with the Dietary Recommended Allowance (DRA) levels, that is, the amount of a nutrient recommended per day for American healthy adults (Ross et al., 2011; Oria et al., 2019a). In addition, the amounts of ingredients were compared with the tolerable upper intake level (UL), that is, the highest level of daily nutrient intake that is likely to pose no risk of adverse health effects to adults (Oria et al., 2019b).
To determine whether a company made disease claims, the website text and product pictures were evaluated. Referral to hangover in the product name, package, or product description, and or terms referring to recovery from illness such as “cure,” “treat,” “correct,” “prevent,” or similar were regarded as a disease-related claim (FDA, 2000; FDA, 2002). Descriptions of the alcohol hangover without explicitly naming the condition (e.g. “to feel better the morning after a night out” or “feel better after drinking” were allowed and not considered a disease claim. In addition, products were checked for the inclusion of registered drugs as part of their ingredients (e.g. NAC).
Results
The first follow-up market evaluation was conducted on 25 March 2023. The search retrieved 428 items. After the removal of irrelevant products and duplicates, the number of available hangover products on www.Amazon.com was 38, a reduction of 53.7% compared to the 2019 market evaluation. The most common dosage forms were capsules (14), followed by tablets (6), powders (5), and drinks and liquids (3). Table 2 gives an overview of the top 20 most popular ingredients of the hangover products that were marketed on 25 March 2023.
Top 20 most reported ingredients of hangover treatments sold on US Amazon per 25 March 2023.

Also includes DHM from hovenia dulcis, Japanese raisin tea, vine leaf tea extract, and ampelopsis grossedentara leaf.
The DRI of vitamin B6 is 1.3 mg for males aged 19–50 and 1.7 mg for males aged 51–70.
The DRI of vitamin B6 is 1.3 mg for females aged 19–50 and 1.5 mg for females aged 51–70.
The DRI of magnesium is 400 mg for males aged 19–50 and 420 mg for males aged 51–70.
The DRI of magnesium is 310 mg for females aged 19–50 and 320 mg for females aged 51–70.
Abbreviations: DRA: Dietary Recommended Allowance; UL: tolerable upper intake level; ND: not determined.
The most frequently reported ingredients were DHM (52.6% of products), vitamin B1 (42.1% of products), vitamin B12 (42.1% of products), sodium (42.1% of products), and vitamin B6 (39.5% of products). Compared to the 2019 market evaluation, on 25 March 2023 only one product contained NAC as ingredient. New in the March 2023 listing were L-cysteine (8 out of 38 products) and ginger (6 out of 38 products). For vitamins and minerals, it is mandatory to report the nutrition value per serving, and the majority of products adhered to this (See Table 2). Much less often reported were the nutrition values per serving for natural ingredients (e.g. DHM and milk thistle). The latter is not required by FDA and companies often presented these ingredients as part of the proprietary blend, without mentioning the amount per serving. For 9 vitamins and 2 minerals, there were products that exceeded the DRA, and the UL was exceeded once for vitamin B3 and vitamin B9. Three products comprised the combination of aspirin and caffeine. According to the 1991 Overindulgence Monograph (FDA, 1991a), these products are allowed to make disease claims and refer to the alcohol hangover. This is not allowed for the other 35 dietary supplements. A review of the website text, products, and packages revealed that 19 hangover products (50.0%) made illegal disease claims.
The second market evaluation was conducted on 7 November 2024 and retrieved 79 items. The number of available hangover products on www.Amazon.com was 46, a reduction of 44% compared to the 2019 market evaluation, but a 21.1% increase compared to the 2023 market evaluation. Of the 82 hangover products marketed in 2019, only 14 products (17.1%) were still marketed in 2024. The most common dosage forms were capsules (18) and transdermal patches (13), followed by tablets (5), powders (3), drinks and liquids (3), pebbles (2), gummies (1), and gel (1). Table 3 gives an overview of the top 20 most popular ingredients of the hangover products that were marketed in the USA on 7 November 2024.
Top 20 most reported ingredients of hangover treatments sold on US Amazon per 7 November 2024.
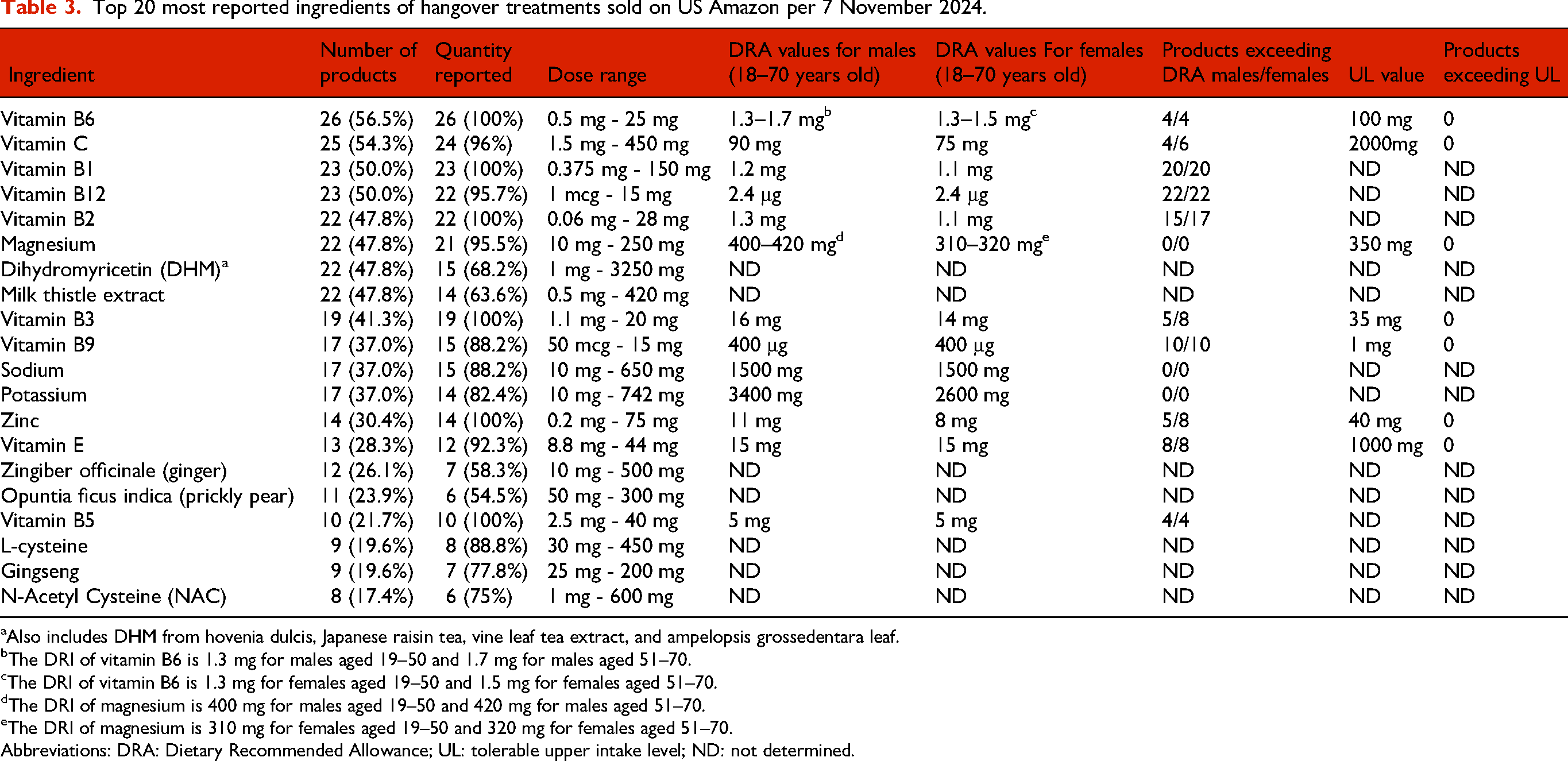
Also includes DHM from hovenia dulcis, Japanese raisin tea, vine leaf tea extract, and ampelopsis grossedentara leaf.
The DRI of vitamin B6 is 1.3 mg for males aged 19–50 and 1.7 mg for males aged 51–70.
The DRI of vitamin B6 is 1.3 mg for females aged 19–50 and 1.5 mg for females aged 51–70.
The DRI of magnesium is 400 mg for males aged 19–50 and 420 mg for males aged 51–70.
The DRI of magnesium is 310 mg for females aged 19–50 and 320 mg for females aged 51–70.
Abbreviations: DRA: Dietary Recommended Allowance; UL: tolerable upper intake level; ND: not determined.
The most frequently reported ingredients were vitamin B6 (56.5% of products), vitamin C (54.3% of products), vitamin B1 (50.0% of products), and vitamin B12 (50.0% of products). DHM (47.8%) and milk thistle extract (47.8%) remained popular ingredients. Compared to the 2019 market evaluation, on 7 November 2024 eight products (17.4%) contained NAC as ingredient. Three products comprised the combination of aspirin and caffeine. For 9 vitamins and 1 mineral (zinc), there were products that exceeded the DRA, but for none of the hangover products the UL was exceeded. A review of the website test, products, and packages revealed that 19 of the marketed hangover products (41.3%) made illegal disease claims.
Discussion
Over the past 5 years, the US hangover product market has dramatically changed. First, compared to the 2019 market evaluation, the number of marketed hangover treatments in 2024 has been significantly reduced by 44%. Of the 82 hangover products marketed in 2019, only 14 products (17.1%) were still available in 2024. Regarding dosage forms, capsules remained the most frequently used form. However, in 2024, transdermal patches were much more frequently used (13 products, 28.9%) compared to 2019 (5 products, 6.1%).
Second, a search of the scientific literature revealed that there is no proof of efficacy and safety from double-blind, placebo-controlled clinical trials in humans for any of the products available at the 2024 US hangover market (Pittler et al., 2005; Verster and Penning, 2010; Jayawardena et al., 2017; Roberts et al., 2022). This may in part be due to the fact that FDA does not require these studies for dietary supplements. In fact, according to the current FDA regulations, the citation of publications of studies demonstrating efficacy in the treatment of hangover is also considered a disease claim (FDA, 2000; FDA, 2002). The lack of supportive scientific evidence for hangover products is of concern.
Third, the percentage of products containing NAC reduced from 45.1% (37 products) in 2019 to 2.6% (1 product) in 2023, but rose again to 17.4% (8 products) in 2024. These changes may be caused by various factors which were not assessed in the current study, such as increased market demand for NAC-containing products, or the inconsistent enforcement by FDA (i.e. no recent warning letters were sent to companies with NAC-containing hangover products). NAC is transformed into L-cysteine and then into glutathione. All are antioxidants that theoretically could reduce the presence and severity of the alcohol hangover. However, a recent study revealed that NAC does not prevent or reduce hangovers (Podobnik et al., 2024). Further, a review concluded that the alcohol hangover is not caused by dehydration, and that blood glutathione concentrations were unrelated to hangover severity (Mackus et al., 2024). Another study reported that L-cysteine was effective in reducing hangover severity (Eriksson et al., 2020), but the results were based on one-sided statistical analyses. However, when applying the correct two-sided analyses, the effect of L-cysteine did not significantly differ from placebo (Benson et al., 2021).
Fourth, hangover products continue to make illegal disease claims. In 2023, about half of the hangover products made illegal disease claims. In 2024, illegal disease claims were made for 41.3% of the hangover products. In 2024, disease-related claims were presented in a less direct manner, and often these were not made on the product itself, the package, or the website text. Here, only indirect references to alcohol hangovers were made by using descriptions such as “for a better tomorrow,” “post-party pills,” “next morning support,” “feeling better after drinking,” or “pre-party natural defense.” Explicit mentioning that the product was effective against the alcohol hangover was most often done in the accompanying video messages that described the product or explained its usage.
The US hangover product market is dynamic, with new products regularly replacing other products. The developments over the past 5 years suggest that the US hangover product market will experience significant additional changes in near future. The FDA has been warning companies that made illegal disease claims or included registered drugs (e.g. NAC) as ingredient of their dietary supplement. These actions will likely be continued, as the FDA treats the alcohol hangover as a disease. This status is supported by the inclusion of the alcohol hangover as separate child entity of alcohol intoxication in the ICD-11 (Išerić et al., 2024). Given these developments, it will be increasingly difficult to market a dietary supplement as hangover treatment. Products that comprise the aspirin and caffeine combination are allowed to make claims (FDA, 1991a) and are therefore likely to remain present on the hangover product market. In addition, it is reasonable to assume that the future hangover product market will be dominated primarily by FDA-approved OTC drugs. Finally, with regard to developing new hangover products, cultural differences in consumer preferences are important to take into account. Zijlstra et al. (2025) recently compared the hangover product markets of the United Kingdom, Australia, and Japan and found significant differences between the three countries in both the popularity of different types of ingredients (e.g. vitamins and minerals, medicines, versus natural ingredients) and the preferred dosage forms (e.g. tablets, powders, drinks, or transdermal patches).
Hangover product ingredients such as vitamins and minerals are generally recognized as safe (GRAS) by FDA, and DRA and UL for their use has been established. However, much less is known about the safety of natural ingredients such as hovenia dulcis (a source of DHM) and milk thistle extract. There are no DRA and UL levels for natural ingredients and it is not known to what extent these natural ingredients interact with other ingredients of the hangover product (Basaran et al., 2022), or with prescription medicines a consumer may take (Chaachouay, 2025). For example, the co-use of natural ingredients could result in synergistic, additive, or antagonistic effects, considerably altering the pharmacokinetics and pharmacodynamics of medicines (Chaachouay, 2025). Therefore, it is in the interest of consumers that scientific studies are conducted to demonstrate the efficacy and safety of all marketed hangover products.
The DRA and UL values in table 2 and 3 refer to daily intake of vitamins and minerals from all sources. Therefore, the amounts included in the hangover products are consumed in addition to the amounts that are already present in the daily diet of the consumer. The DRA represents the highest daily intake level that is likely to pose no risk of adverse health effects, whereas intake levels above the UL increase the risk of adverse effects. It is important to take into account that these levels are valid for most individuals of the general population. However, they may be different for individuals with underlying health conditions or those who use medications.
Possible adverse effects that arise from occasionally exceeding the DRA or UL for vitamins are usually temporary, and not result in chronic health conditions. However, even though exceeding the DRA is not necessarily harmful, precautions should be taken into consideration, since the intake via hangover products adds to the daily intake via daily diet, which combined could then exceed the UL. In 2023, one hangover product exceeded the UL for vitamin B3 (niacin/nicotinic acid), and 8 out of 19 vitamin B3 containing products exceeded the DRA. Typical side effects of high vitamin B3 intake include flushing, hypotension, and peptic ulcers. (Parsons, 1960; Wilkin et al., 1982; Hrubša et al., 2022). Another product exceeded the UL of vitamin B9 (folate), and all 10 vitamin B9 containing products exceeded the DRA. Excess vitamin B9 intake in the specific form of folic acid has been associated with serious health conditions, including vitamin B12 deficiency (Naderi and House, 2018), neurological damage (Molloy et al., 2009), and an increased risk of prostate cancer (Wien et al., 2012). Although in 2024 none of the hangover products had ingredients that exceeded the UL, for several hangover products the amounts of various B-vitamins, vitamin C, and zinc exceeded the DRA levels.
To conduct the market evaluations, www.Amazon.com was chosen, as this is the biggest online shopping site were Americans buy health products (Chevallier, 2024). However, this could be viewed as a limitation, since there are other places where people can also buy hangover treatments, including other online platforms, directly from company websites, supermarkets, gas stations, and venues where alcohol is sold (e.g. bars and clubs). Although we did not investigate this alternative market, it is reasonable to assume that the most popular products sold via these alternative ways to a large extent will comprise the same products as available via www.Amazon.com.
In conclusion, over the past 5 years, the US hangover product market has dramatically changed, including a significant reduction in the number of marketed hangover products. While DHM, milk thistle extract, vitamins and minerals remained the most popular ingredients of hangover products, the popularity of NAC declined. However, despite FDA warning letters and guidelines, 8 products (17.4%) still included NAC as an ingredient. Illegal disease claims were made by 41.3% of the marketed products. Of concern, the efficacy to prevent or reduce alcohol hangover has not been demonstrated for any product on the US hangover market.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
Over the past 36 months, JV has received held research grants from Danone and Inbiose and acted as a consultant/expert advisor to Eisai, KNMP, Med Solutions, Mozand, Red Bull, Sen-Jam Pharmaceutical, and Toast!. JV has received travel support from Sen-Jam Pharmaceutical and owns stock from Sen-Jam Pharmaceutical. AS has acted as a consultant/expert advisor to Bayer, Coca Cola, Danone, Delica Therapeutics, GlaxoSmithKline, Givaudin, Liquid IV, Mars-Wrigley, Naturex, Nestlé, McCormick, Metavate Consultancy, PepsiCo, Pfizer, Pharmavite, REVIV, Sanofi, Verdure Sciences, and Wörwag Pharma; and in the past 36 months Scholey has held research grants from Abbott Nutrition, Arla Foods, the Australian Research Council, Bayer, BioRevive, DuPont, Fonterra, GlaxoSmithKline, the High Value Nutrition Fund, the National Health and Medical Research Council, Nutricia-Danone, Sanofi and Wörwag Pharma. AS is Chief Scientific Officer for Ārepa Nootropics and is on the Scientific Advisory Board of Sen-Jam Pharmaceutical. He has stock from Sen-Jam Pharmaceutical and Ārepa Nootropics. He has received travel support from Vitafoods and ILSI. MZ and EI have received travel support from Sen-Jam Pharmaceutical. The other authors have nothing to declare.
Data availability statement
The data is available from the corresponding author upon reasonable request.
