Abstract
The unprecedented progress in the science and clinical investigation of psychedelic medicine will require those in healthcare leadership and the legislative policy arena to conceptualize how future reforms, policy creation, and clinical practice should occur to broaden access to these agents while simultaneously maximizing effectiveness and mitigating harm. The pharmacy profession has surprisingly had little engagement on this front. This article provides a perspective commentary and overview of potential future strategies in legal reform, professional regulatory authority policy creation, and pharmacy operations regarding the psychedelic agents’ psilocybin and methylenedioxymethamphetamine, using Canada as a national case study.
Introduction
The general awareness and excitement surrounding psychedelics as potential “breakthrough treatments” for a range of mental health disorders and associated psychiatric situations have perhaps never been higher in the Western world (Aday et al., 2020; Basky, 2021; Lamkin, 2022). This is evidenced by a PubMed search wherein just the word “psychedelic” alone demonstrated that the 3 years with the highest number of published records, starting from 1957, were 2020, 2021, and 2022 (Hadar et al., 2023; Plesa and Petranker, 2022). Additionally, in Canada, news stories and media coverage have further excited the public consciousness as to their potential as treatments for severe depression and anxiety, post-traumatic stress disorder, obsessive–compulsive disorder, and addiction (to name only the most prominent indications for which these agents have been studied) (Bloomberg, 2022; CBC News, 2022; Global News, 2022).
The purpose of this paper is to propose a future pharmaceutical access policy and operational framework approach whereby pharmacists, as medication experts and trusted overseers of patient medication safety, can be enabled in the future medical distribution of these agents using Canada's federalist legislative model as an example. This is based on the potential for the continued accumulation of research data to develop favorably in the direction of legislative changes toward their access and broader acceptance in society. Many believe this to be a possible eventuality given the promising early evidence combined with the recent access to psychedelic medicine by a small number of patients through Health Canada's federal special access program (Udoh, 2022).
The discourse that follows shall be confined to psilocybin and 3,4-methylenedioxy-n-methylamphetamine (MDMA) due to these agents having the most recent and cumulative development of supportive evidence for their safe and effective clinical use relative to other psychedelic agents such as lysergic acid diethylamide (LSD) or the more culturally based entheogenic use of mescaline (from peyote cactus) and dimethyltryptamine (DMT) – from Indigenous Amazonian Ayahuasca brew (among other sources) (Carhart-Harris and Goodwin, 2017; Kisely et al., 2022). This is not to disqualify the potential for these latter three compounds as emerging psychedelic treatments in their own right (Agin-Liebes et al., 2021; Argento et al., 2019; Krebs and Johansen, 2012; Rossi et al., 2022) Rather, it is to emphasize a step-wise, conservative approach to psychedelic medicine access which prioritizes the two agents with the largest amount of positive controlled trial evidence that fit the regulatory and methodological clinical trial standards of the 21st century. 1 A non-partisan approach where the thoughtful application of the scientific evidence to health regulatory affairs would presumably be a principled diplomatic solution for bridging party lines on Canadian public healthcare legislation and policy toward progressive reforms for the benefit of all Canadians.
The role of the recreational distribution of psychedelics through pharmacies has also been addressed in the past, with some preliminary survey data showing some intuitive preference for this model by university-educated UK citizens (James et al., 2018). However, this issue shall not be taken up here. The core focus of this article is instead on regulated medical access only and does not address the broader socio-political debate topics of legalization and decriminalization of psychedelics. 2
A brief overview defining psychedelics, describing their pharmacology, and concisely reviewing the current state of the evidence will proceed first to establish a background to make this topic more accessible to lay-persons in the Canadian public as well as academics, policymakers, government officials, healthcare providers, and patients who may take interest in these drugs. I shall then proceed to the primary purpose of this paper—a proposal for future legislative, policy, and operational considerations for broadening pharmacy access to psychedelic medicine.
Basic overview of therapeutic mechanisms
The classical “hallucinogenic” psychedelics (i.e., LSD, mescaline, psilocybin, and DMT) produce altered states of consciousness characterized by profound sensorial, emotional, and perceptual changes that are widely believed to be attributable mainly to their complex serotonergic pharmacology mediated by partial agonism at G-protein-coupled 5-HT2A receptors (Kyzar et al., 2017; Ray, 2010). MDMA, while not being a “hallucinogen” per se and rather an “entactogen” (based on its propensity to release serotonin and dopamine from nerve terminals in the central nervous system) is more widely recognized for its utility in eliciting emotional change over visual or perceptual distortion (Green et al., 2003). Nonetheless, both of these compounds given their potent mind-altering effects and overlap in a clinical presentation have justified their mutual categorization by organizations as being “psychedelics” (Multidisciplinary Association for Psychedelic Studies, 2022; Psychedelic Association of Canada, 2022). While the topic of detailed psychopharmacology is taken up in extensive detail elsewhere, it is important to recognize the distinctiveness of these drugs in their potential to elicit rapid-onset synaptic plasticity (compared to antidepressants) and disruption of the brain's “default mode network” (Banks et al., 2021; Ly et al., 2018). Such effects may explain how patients overcome mental health challenges when psychedelics are used in the therapeutic context of the appropriate set, setting, and post-sessional psychological intervention (Majić et al., 2015). The “peak” subjective experiences and “afterglow” phenomena on mood and mental outlook have been identified as potential explanations of differing therapeutic mechanisms that may correspond to brain state changes that may be sustained by psychological integration via talk-psychotherapy (Majić et al., 2015).
Status of evidence for psychedelic-assisted psychotherapy
Psilocybin
Since the 2000s, psilocybin (from “magic mushrooms” of various biological genera) has seen a resurgence of clinical research interest with small-scale, controlled trials showing positive results in treatment-resistant depression, anxiety, obsessive–compulsive disorder, alcohol dependence, tobacco dependence, and end-of-life “existential” palliative care (Bogenschutz et al., 2015; Carhart-Harris et al., 2016; Griffiths et al., 2016; Grob et al., 2011; Johnson et al., 2014; Moreno et al., 2006; Ross et al., 2016). The results of these trials have typically shown a reduction in symptom scores from validated clinical assessment scales which is often sustained for months. Nevertheless, these trials have had minimal participants, are unblinded (an ethical and logistical dilemma given the nature of these compounds), and occur in tightly regulated environments that may be challenging to replicate exactly in real-world practice. A 2022 systematic review by The Canadian Network for Mood and Anxiety Treatments, demonstrating the scarcity of high-quality evidence for major depressive disorder, suggests that psilocybin is far from a panacea for the treatment of depression at this stage (Rosenblat et al., 2023). While safety also remains a relevant concern, a recent thorough evaluation of the medical literature reports that psilocybin appears safe enough for clinical practice (Roscoe and Lozy, 2022). In summary, more evidence is needed overall before the United States Food and Drug Administration (FDA), Health Canada, and other federal drug regulators grant federal licenses to the pharmaceutical industry for widespread production, carefully vetted medical marketing, and controlled distribution of psilocybin into healthcare settings.
3,4-methylenedioxy-n-methylamphetamine
MDMA is a compound with a long-standing cultural legacy as “ecstasy”; the “love drug” that predominated in the musical rave culture of past decades (Passie and Benzenhöfer, 2016). It has been clinically re-purposed for various psychiatric conditions with promising, early results in recent years (Sessa et al., 2019). Its unique serotonergic pharmacology and mostly non-hallucinogenic mental effects appear to be conducive to social–emotional learning and cognitive reprocessing (Regan et al., 2021). This has distinguished it from the other classical psychedelics despite its similar utility in the monitored psychotherapeutic setting. MDMA has been most researched in recent years in its potential to aid in the emotional reconsolidation of traumatic memories with the possibility of accomplishing “fear extinction” in post-traumatic stress disorder patients (Bahji et al., 2020; Feduccia and Mithoefer, 2018). Additional phase 3 trials that are currently underway or recently completed are all that seem to currently stand between the FDA and MDMA's entry into the psychiatric clinic (Ot’alora G et al., 2018). Preliminary investigation on MDMA as a treatment for depression and anxiety has produced some positive results that have not been free of scrutiny or criticism from detractors who caution against substantiated safety concerns from earlier literature relating to dose, chronic use, and neurotoxicity (Parrott, 2014; Patel and Titheradge, 2015; Schenk and Highgate, 2021; Schenk and Newcombe, 2018).
Current legislative framework for psychedelic drugs—toward future reform
All Western nations carry some restrictive or punitive legislation on psychedelic drug use in society. At an international level, the United Nation's 1971 Convention on psychotropic substances set a precedent among 71 delegate nation-states (including Canada, the United States, and the United Kingdom) by categorizing psychedelic drugs as Schedule 1 substances with special provisions (United Nations, 1971). Schedule 1 substances are those “with no currently accepted medical use and a high potential for abuse” (United Nations, 1971). Canada has continued to abide by this international treaty by enabling psilocybin to be accessed only for recently approved research purposes and through Health Canada's special access program (Health Canada, 2022). However, recent demand has resulted in the creation of a special class exemption to the federal Controlled Drugs and Substances Act for psilocybin and MDMA in 2022 (Government of Canada, 2022).
Evaluating the current legislative and regulatory control of psilocybin and MDMA for medical purposes in Canada requires a contextual understanding of the relevant international, federal, and provincial statutes, treaties, and policy frameworks to sufficiently raise awareness about how these legal documents may operate in an obsolete fashion in light of the emerging evidence of benefit for these drugs. Table 1 provides a simplified overview of the most relevant international and federal treaties and statutes that currently operationalize the enforceable Canadian legal environment surrounding the psychedelic drug.
Key international and federal legal documents relevant to psychedelic drugs in Canada.

MDMA: 3,4-methylenedioxy-n-methylamphetamine; CDSA: Controlled Drugs and Substance Act.
Federal drug scheduling considerations
In Canada, psilocybin and MDMA are scheduled differently from one another within the framework of the Controlled Drugs and Substances Act (CDSA). MDMA is treated as a Schedule 1 agent and psilocybin is a Schedule 3 agent. The difference in scheduling guides sentencing for indictable offenses such as production, possession, trafficking, or smuggling. Amendments to the CDSA for scheduling purposes are necessary only in relation to law enforcement proceedings. Amendments to broaden medical access would presumably not contradict the scheduling other than for the symbolism implicit in the respective scheduling and what it says about each drug's respective public safety profiles.
Food and Drug Act approval process
Substances that fall under the purview of the CDSA obviously require a higher level of oversight in the scientific investigation and marketing authorization process. The bureaucracy of Health Canada is vast and, as such, is not easily navigable for those outside government or pharmaceutical industry. Before the pre-requisite technical reports and clinical data collection that fulfills the standard package criteria for a New Drug Submission (NDS), additional applications for conducting clinical trials, importation, and/or production of the drug on Canadian soil may be required. The first marketed MDMA and psilocybin products would receive significantly more regulatory scrutiny with subsequent entry products often requiring lesser data such as bioavailability and formulation-specific information in the form of an abbreviated NDS. At the time of writing, however, a number of clinical trials are already underway for both psilocybin and MDMA in Canada (Table 2). Successful results from these trials are necessary before proceeding to the proposed healthcare quality and patient access approach outlined in this paper.
Canadian trials for psilocybin and MDMA in adults.*

PTSD: post-traumatic stress disorder; MDD: major depressive disorder; MDMA: 3,4-methylenedioxy-n-methylamphetamine.
*As of January 2023.
CDSA regulatory reform
The simplest and most expected legislative reform solution would be the creation of a new regulation, under the CDSA, specifically for psychedelic compounds. Given the unique use of these agents in psychiatric practice, there are a number of logistical considerations for patient safety that legislators should account for in drafting such federal regulations. For instance, one special consideration for the distribution of psychedelic medicine to the pharmacy or other dispensing practitioners would be the need for formal registration with wholesale pharmaceutical distributors for establishing ordering privileges for pharmacies that meet the criteria to dispense these agents. Similar regulations to the CDSA exist in regard to benzodiazepines, narcotics, and controlled drugs such as amphetamines. These regulations offer important legal provisions for both pharmacists and practitioners. As such, a similar regulation format for psychedelics could be a welcome solution if or when the first products enter the prescription drug market. Further considerations for policymakers and legislators are outlined in the following section.
Proposed policy and coordinated collaborative care approach for pharmacy and psychiatry
The potential pathway for amendments outlined in the previous section would foreseeably enable the pharmacy profession to participate in a collaborative fashion with psychiatrists and other mental health specialists in the clinical care of patients before and after (but not typically “during”) their psychedelic therapy sessions. Federal legislation changes coordinated with appropriate consultations with representatives of the pharmacy profession would ensure that the implementation and scale-up of pharmaceutical psychedelic dispensing and associated patient care services would occur optimally as intended with sufficient political support from the psychiatric profession.
An interprofessional approach to the establishment of collaborative care models should be investigated and negotiated between the psychiatric and pharmacy professions. While this is beyond the scope of the present paper, a number of collaborative care models that have been previously utilized with success in other treatment areas could be explored and potentially researched in the future as it relates to interprofessional psychedelic patient care between qualified pharmacists and psychiatrists (Bardet et al., 2015).
Of note, other health professions beyond psychiatry, including midwives and nurses, are advocating for their involvement in psychedelic treatment (Denis-Lalonde and Estefan, 2020; Stein et al., 2022). I shall first demonstrate in the following section that pharmacy is even more rationally placed for a future where these medicines are utilized for psychopharmacologic mental healthcare. After this, I shall elaborate on how the profession can begin to embark on a process to ensure prudent regulatory reforms occur with the subsequent operational and practice-based changes at local levels to make this a reality for Canadian patients.
Why pharmacy? Reasons and evidence for psychedelic involvement
Compared to the medical profession and psychiatry in particular, pharmacy has had a minimal representation in the literature and media regarding psychedelic drug therapy. It is therefore important to convince the public, other professionals, policymakers, and government officials (beyond the obvious logical flow of reasoning that “psychedelic drugs” are “drugs” and that pharmacists “dispense drugs”) of the benefits of having pharmacists involved in the psychedelic treatment process. There are at least four major reasons that pharmacists should have oversight or involvement in the provision of psychedelic treatment for mental health.
The primary reason is the pharmacists’ essential duty to provide quality pharmaceutical care dispensing services by ensuring that the psychedelic prescription properly fulfills each of the criterion domains of the Indication, Effectiveness, Safety and Adherence (IESA) framework for pharmaceutical care practice (Cipolle et al., 2012; Nelson et al., 2021). A pharmacist is properly placed to scrutinize the psychedelic prescription to screen for the appropriate agent and dose and to ensure that drug–drug interactions and disease–state interactions are fully accounted for. The adherence side of drug therapy problems concerning psychedelics would often not be a factor given the typical single intended occasioned witnessed dose at the psychedelic session itself. However, with more research being conducted on regular (non-psychedelic effect) use in the form of “micro-dosing,” the role of the pharmacist in community out-patient monitoring and adherence assessment would once again become relevant (Polito and Stevenson, 2019).
The second reason is the opportunity for clarifying expectations and improving understanding and communication between psychiatrists and the patient prior to the psychedelic session. While the occasion for a corrective understanding for patients after psychiatric consultation may be minimal regarding psychedelics (given the trained expertise of psychiatrists in soliciting informed consent), it remains a possibility that pharmacists may enhance patient knowledge or understanding prior to the psychedelic session by successfully engaging with patients to address questions or concerns that may remain after the issuance of a psychedelic prescription (Kelly et al., 2014). Pharmacists are often cited as being the most accessible healthcare provider and so their availability to act in the capacity of a local, trusted, information resource for the patient and, when appropriate, their family or caregivers is not to be understated (Tsuyuki et al., 2018; Valliant et al., 2022). In regard to community mental health, past patient surveys have shown generally positive reception when community pharmacists become involved (Black et al., 2009).
The third reason for pharmacists to be involved in psychedelic drug treatment is to prevent all liability from falling on the medical profession. Psychedelics would be otherwise unique insofar as that essentially all other medication use, with perhaps the exception of emergency room encounters and operative procedures, are overseen by a pharmacist. This is true whether they be in hospital order sets for patients in a hospital ward, ambulatory patients in the community, or prescriptions for nursing home residents. Given that the clinical psychopharmacologic use of psychedelics should always be a carefully planned drug treatment with adequate preparation, it is questionable why the eventual route for normalized access would be any different from other prescription medications for ambulatory, community-dwelling patients (Figure 1).
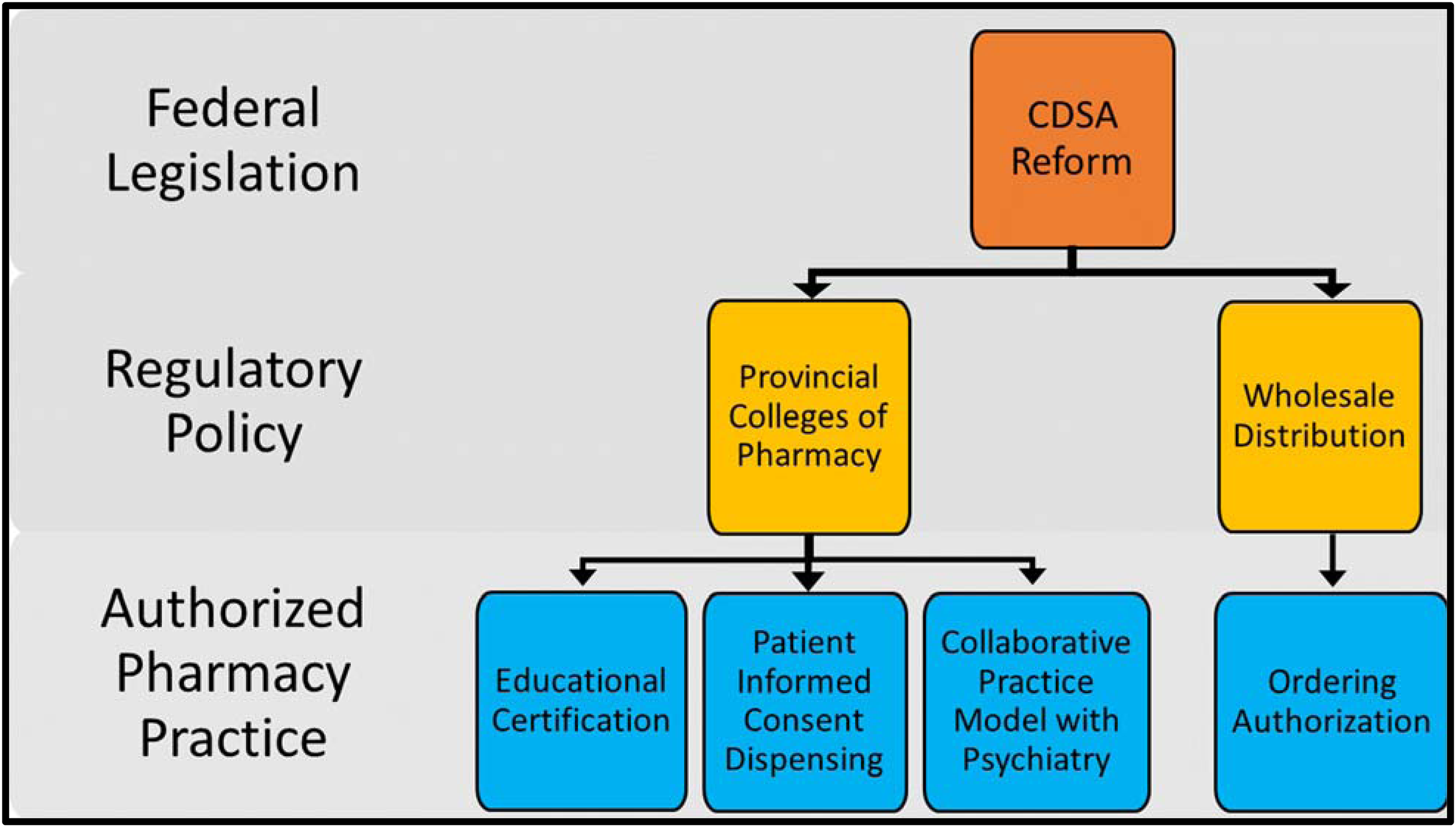
Proposed Strategy for Patient Access to Psychedelics in Canadian Community Pharmacies.
The final reason to engage pharmacists in the psychedelic space is that there is already pre-existing evidence that, when pharmacists become involved in collaborative mental healthcare, in both in-patient and out-patient settings, they have a positive impact on various outcomes as concluded by a number of systematic reviews (Bell et al., 2005; Davis et al., 2020; Finley et al., 2003; Ng et al., 2022).
Regulatory training requirements for licensed pharmacy participation
Even with the previously reviewed legislative reformation approach to improving medical access to psychedelics through pharmacy, there is much that regulatory authorities self-governing the pharmacy profession can do to further establish high standards of practice and expectations among pharmacists who are involved in this area of patient care. One of the obvious approaches that would incorporate a high level of expected collaboration and quality of pharmaceutical care is to require pharmacists to have additional expertise in psychiatry or to take a comprehensive training program on psychedelics that would be tailored to their unique position and role in society as medication dispensing healthcare providers. The Board Certification in Psychiatric Pharmacy (BCPP) credential designation, offered by the American College of Clinical Pharmacy's Board of Pharmacy Specialities, is one such rigorous post-graduation certification route that ensures a high level of expertise in the psychopharmacologic management of complex mental health patients (Dopheide et al., 2022). Another similar credentialing process exists in the UK through the College of Mental Health Pharmacy (College of Mental Health Pharmacy, 2022). On the other hand, such a training requirement to dispense these agents would likely severely limit access to psychedelics among Canadian patients living in remote locations because, at the time of writing, there were only 42 BCPP pharmacists out of a workforce of ∼42,500 pharmacists in Canada (Board of Pharmacy Specialties, 2022; Canadian Pharmacists Association, 2022). Furthermore, many of them may work in psychiatric in-patient or collaborative mental health primary care settings and thus are not employed within a traditional community dispensary setting. 3 As an alternative, it is worth considering a number of other psychedelic educational training programs that currently exist or whose content may be adaptable for the role of the pharmacist (Cusimano, 2022; Psychedelic Support, 2022).
A potentially more acceptable (and certainly less politically restrictive) method for engaging the pharmacy profession properly in such a patient care practice would be for the National Association of Pharmacy Regulatory Authorities (NAPRA) to establish more accessible training requirements and practice expectations for the safe and effective utilization of the community pharmacy workforce in psychedelic dispensing (NAPRA, 2023). In concept, this would not be dissimilar to what NAPRA has published recently in the form of model standards of practice documents to guide other domains relating to pharmacy practice (National Association of Pharmacy Regulatory Authorities, 2022a, 2022b). It would then be the subsequent responsibility of each provincial pharmacy regulatory authority to transfer the NAPRA guidance within the context of their own provincial jurisdiction. Potential considerations that may inform such guidance from pharmacy regulatory authorities are provided in the following subsection.
Policy operations approach governing pharmacy practice and dispensing
Patients who come to the pharmacy with a psychedelic prescription would reasonably expect a different experience of medication access compared to the average prescription or prescription refill for any other kind of medicine. They should be made aware upfront by their prescribing psychiatrist of which local pharmacies are authorized to dispense their prescription and what may be needed from them to receive the prescription at the pharmacy. As with all controlled drug prescriptions, identity verification is of paramount importance for responsible dispensing and limiting diversion.
Currently, licensed pharmacists follow a strict protocol in coordination with wholesalers to account for all narcotic and controlled drugs that are used as prescription medication. This is to legally abide by the Controlled Drugs and Substances Act. Should a separate psychedelic regulation come into force (as described in the previous section) there would need to be a clear and verifiable registration protocol for pharmacists to become authorized to order psychedelics from a licensed Canadian pharmaceutical wholesale distribution company. For example, all licensed pharmacists are a priori able to order narcotic medicines in Canada with a pharmacist license. These privileges can be rarely revoked via a punitive disciplinary process at the regulatory level should problems arise. I propose that the default ordering status for a licensed pharmacist would be ineligibility for ordering psychedelic drugs. It would be only after satisfactory proof is provided that a pharmacist has completed the requisite training that the granting of ordering privileges for psychedelic agents would become available to them. An additional requirement that may be optionally imposed could be for a collaborative practice arrangement to be in place between a psychiatrist and an ordering pharmacy. A wholesale distributor may thereby require a template letter from a prescribing psychiatrist to further demonstrate the benefit or need for having a pharmacist with training on psychedelic medicine available for patient care and medication access.
Given the profound mental effects of psychedelics at psychoactive “trip” doses, care would be needed to ensure that the prepared prescription package for the patient to receive is secure and that others involved in the patient's care can be assured that it was not significantly tampered with prior to the supervised psychedelic session. This is an important operational consideration for the protection of the public and should be the responsibility of the pharmaceutical industry when producing various strengths of psychedelic “trip” dose formulations (Bunn, 2019; Lockhart and Paine, 1996). Such a product would be dispensed unopened to the patient with the seal intact such that the pharmacy staff would void the integrity of the dose should they break the seal. The dispensed psychedelic, with the prescription label appended to the product, would remain unopened until in the supervised clinical setting.
Given the stigmatizing (but not unfounded) historical reputation of psychedelics as “psychotomimetics,” any preventive approaches to limit vulnerable persons from taking another individual's prescribed psychedelic is needed for the general protection of the public (Nichols and Walter, 2021). The required use of tamper-resistant packaging in conjunction with the need for patients themselves or a pre-authorized agent (such as another clinician or trusted family member) to sign an informed consent document, at the time of transaction and prescription pick-up, would be a further safeguard against the irresponsible use of the agent in the period between its dispensing and its witnessed consumption at the supervised site of the psychedelic therapy session. It would be beyond the scope of this paper to suggest or otherwise outline criminal offense liability should these conditions be knowingly breached by patients. However, such consideration must be at least weighed carefully by federal and provincial legislators.
Another public harm reduction approach would be to place dispensing time limitations based on the date of the session itself (that perhaps should be legally required to be indicated on the prescription—at least for psychedelic psychotherapy doses). As an example, pharmacists may not be authorized to dispense the agent outside of a tightly specified time window before the psychedelic psychotherapy session. The date of the scheduled psychedelic session would thus need to be specified on the prescription itself. This would prevent the psychedelic medicine from being kept in an unsecured location (i.e., at the patient's home or elsewhere) for an extended period and limit the possibility of improper access by others who may attempt to break the tamper-proof packaging, thus placing themselves at risk of an adverse psychoactive event or place the intended recipient of the therapy at the legal consequence. However, a provision of this kind should also warrant sensitive policy consideration of potential exemptions to enable equitable access to psychedelics for geographically remote communities and minority populations (i.e. Canada's indigenous population) (Thrul and Garcia-Romeu, 2021). A final consideration, to make matters more complex, is the continuing focus of new research on virtual care and how these technologies (along with the emergence of associated advocacy agendas) may eventually intersect with psychedelic distribution and treatment (Camp et al., 2020). The development of virtual care infrastructure for remote communities, when combined with highly secured mail-order distribution of prescription psychedelics, might enable the right balance to be eventually struck between public health, medication safety, and health equity for all persons throughout Canada's vast geographic expanse.
Conclusion
In this paper, I have attempted to provide a robust pathway informed by legal, policy, operational, and clinical considerations to present a future vision whereby Canadian patients, psychiatrists, pharmacists, and other mental healthcare experts work collaboratively toward high-quality psychedelic treatment.
The complex politics of psychedelics is further made unpredictable by the still rapidly emerging scientific and clinical evidence regarding their use. This remains the fundamental limitation of this paper insofar as that projecting one's vision into the future always yields the risk of miscalculation in the nuances of any topic. Nevertheless, it is hoped that such a proposal, when considered in light of other past policy proposals, can at least be informative for future public policy discussion or debate regarding the proper placement of psychedelic medicine and its access in Canada (Haden et al., 2016; Mocanu et al., 2022).
In conclusion, members of the pharmacy profession should become and remain engaged with the development of policies and processes related to psychedelic treatment at least in preparation for the possibility that it may impact their own practices or patients in the future. Policymakers and health professionals outside of the pharmacy profession should remember the opportunistic placement of community pharmacies for scaled-up distribution of psychedelic medicine as well as the interprofessional role that community pharmacists play in the care of community-dwelling patients with conditions amenable to treatment with psychedelics.
Footnotes
Acknowledgements
The author acknowledges and thanks the Multi-disciplinary Association for Psychedelic Studies (MAPS-Canada) and Dr.’s Gregory Kos and Carolyn Weiss for their engagement and interest during the writing of this work. He also acknowledges the Psychedelic Pharmacists Association (![]() ) for their relevant work in this area and his late connection with them immediately prior to publication.
) for their relevant work in this area and his late connection with them immediately prior to publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
Research Ethics Board approval and/or patient consent was not sought as it was irrelevant to this work.
