Abstract
When abdominal pain presents with peripheral eosinophilia, allergic and infectious causes should be considered. However, diagnostic dilemmas are frequent when symptoms do not match expected patterns of common etiologies. In this report, we present the case of a 13-year-old male who presented with diffuse abdominal pain and peripheral eosinophilia. Though the initial assessment suggested an infectious cause, the infectious workup was negative. A broad workup, including imaging, laboratory studies, and endoscopy, was performed. Targeted endoscopic biopsies revealed extensive eosinophilic gastrointestinal disease as the underlying pathophysiologic cause. A broad differential and extensive workup is often crucial for accurate diagnosis of abdominal pain with hypereosinophilia.
Keywords
Introduction
Abdominal pain in adolescents has a broad differential, ranging from functional disorders to inflammatory and infectious etiologies. This case highlights the importance of considering a rare diagnosis, eosinophilic gastrointestinal disease (EGID), in patients with unexplained or unresolving abdominal pain and eosinophilia.
Presentation 1
A previously healthy 13-year-old male presented to the emergency department (ED) with a 7-day history of stabbing right lower quadrant pain and a 1-day history of pre-syncopal episodes that occurred several times daily, associated with resolved non-bloody, non-bilious emesis and non-bloody diarrhea 5 days before presentation. In the ED, he was well-appearing with a heart rate of 88, blood pressure of 113/73, respiratory rate of 22, and temperature of 36.8°C. On exam, he had diffuse abdominal tenderness, most notably in the right lower quadrant.
Laboratory studies and imaging
Complete blood count (CBC) demonstrated a white blood cell count (WBC) of 9480 cells/μl, red blood cell count (RBC) 4,990,000 cells/μl, Hgb 14.5 g/dl, and hematocrit 41.1%. Computed tomography (CT) with contrast of his abdomen/pelvis showed a normal appendix but suggested diffuse colitis (see Table 1).
Laboratory study results from multiple hospital presentations.

CT: Computed tomography; WBC: White blood cell count; RBC: Red blood cell count.
Disposition
He was discharged with presumed viral gastroenteritis.
Presentation 2
Ten days after his initial presentation, he returned to the ED with worsening abdominal pain. His exam demonstrated persistent mild tenderness to deep palpation in the right lower quadrant.
Laboratory studies and imaging
Labs demonstrated an increased WBC count with eosinophilia compared to the prior CBC; WBC 22,000 cells/μl with 46% eosinophils and absolute eosinophils 10,070 cells/μl. Urinalysis, erythrocyte sedimentation rate, and C-reactive protein were all normal. Repeat CT abdomen/pelvis showed “Diffuse bowel wall thickening involving the entire small/large bowel, gastric antrum, and distal esophagus, which appears increased as compared to prior. Unchanged diffuse peritoneal fat stranding, likely edema/inflammation/infection” (Figure 1). Other lab studies performed at this visit included testing for viral and bacterial stool pathogens, which ultimately were negative (see Table 1).
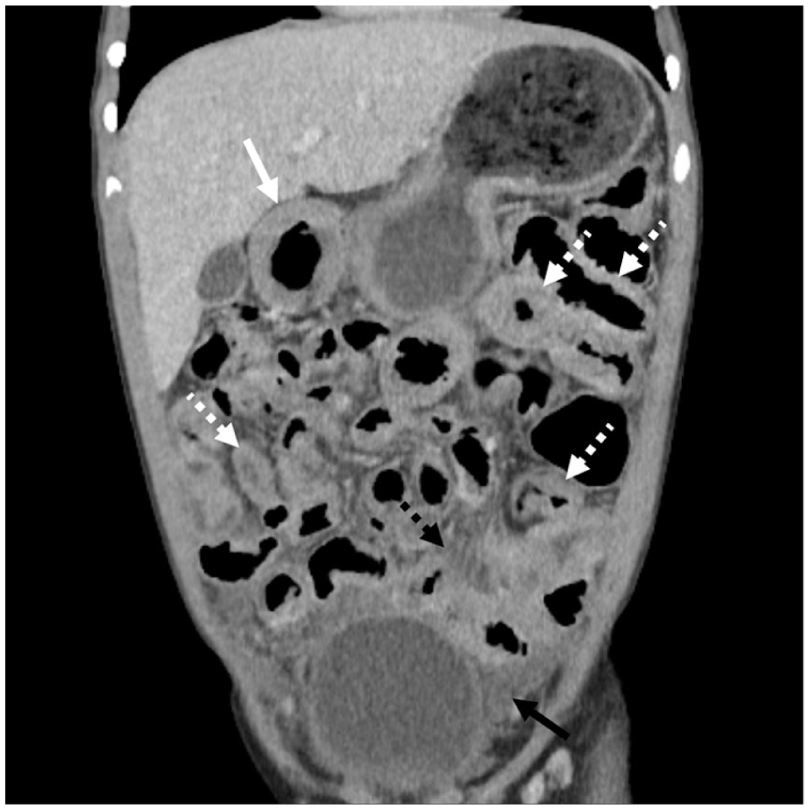
Coronal contrast-enhanced CT scan of the abdomen and pelvis in this 13-year-old patient shows numerous regions of varying degrees of nonspecific small bowel wall thickening (dotted white arrows) as well as moderate wall thickening of the gastric antrum (solid white arrow).
Disposition
Given the worsening abdominal pain and colitis, along with the unusual CBC and differential, the patient was admitted for additional evaluation and subspecialty consultation.
Differential diagnosis
While the etiology of his peripheral eosinophilia, abdominal pain, colitis, enteritis, gastritis, and distal esophagitis was unknown at the time of admission, the differential diagnosis included parasitic infections, fungal infections, inflammatory bowel disease, EGID, hypereosinophilia syndrome, neoplasia, and atopic disease.
Inpatient course
After admission, additional social and exposure history was obtained. The family lived on a farm with various animals, including rabbits, cows, donkeys, and horses. He frequently interacted with these animals and played in the dirt multiple times per week. One of the family rabbits had recently been ill with conjunctivitis. Otherwise, they reported no new exposure to illness or medication.
Multiple specialty services were consulted. Infectious Disease (ID) recommended repeat stool ova & parasites testing (times three), HIV,
Presentation 3
Approximately 3 days after discharge, he presented to the ED with inability to tolerate oral intake, vomiting, profuse watery diarrhea, severe abdominal pain, and weight loss. On exam, he had diffuse abdominal tenderness, especially periumbilical and epigastric. Labs were significant for WBC 17,600 cells/μl with 27.2% eosinophils (see Table 1). Given his ongoing emesis and worsening symptoms, he was admitted for management of dehydration and further specialty evaluation.
Inpatient course
The patient was reevaluated by the ID team, who recommended empirical treatment with metronidazole and albendazole for symptom improvement. While
On day three of admission, all infectious labs were officially negative, but his symptoms had significantly worsened, so GI elected to perform esophagogastroduodenoscopy and colonoscopy with biopsies. Initial pathology results showed eosinophilic gastritis, enteritis, and colitis. Per the pathology report, “findings raise concern for multi-site eosinophilic gastrointestinal disease (EGID),” suggesting a diagnosis of EGID. He started intravenous steroids, and eosinophilia decreased from 40.2% to 8.1%. He was discharged on prednisone and follow-up with GI and allergy as an outpatient was scheduled. These specialists monitored his CBC and slowly tapered his prednisone. His eosinophils returned to normal levels after his prednisone taper.
For this patient who presented with abdominal pain, hypereosinophilia, and gastritis, enteritis, and colitis on imaging, biopsies confirmed the diagnosis of eosinophilic gastrointestinal disease (EGID). This patient’s symptoms improved after treatment with corticosteroids.
Discussion
In this case, we discuss the diagnosis and ongoing management of a 13-year-old male who initially presented with abdominal pain and later developed hypereosinophilia. This case presented a diagnostic dilemma with a delay in determining an accurate diagnosis, as his initial presentation suggested more common diagnoses, given the abrupt onset of his symptoms and the external exposure to potential zoonotic diseases, leading ID to recommend empiric treatment with antibiotics and antifungals pending stool staining and cultures. For an adolescent male presenting with abdominal pain, the differential diagnosis is broad, including infectious, structural, metabolic, hematologic, genitourinary, and neoplastic etiologies. Further complicating things, abdominal pain can be caused by systemic infections (such as influenza) or infections in organs not contained within the abdomen. 1 In one study, the most common cause of patients presenting to one ED with abdominal pain was constipation. 2 Astute clinicians must remain systematic and thorough in their evaluations and avoid diagnosis anchoring. A thorough history and physical examination can narrow the differential, though advanced imaging and lab work are often required. In this case, the finding of hypereosinophilia narrowed the differential diagnosis considerably, making the most likely diagnoses infectious, neoplastic, or primary eosinophilic disorders. Though there are a multitude of etiologies underlying eosinophilia in children, this patient’s abdominal pain and imaging findings pointed to a parasitic gastrointestinal infection or EGID being the most likely unifying diagnosis. 3 Extensive laboratory testing and imaging ruled out infection, and biopsy findings were most consistent with a diagnosis of EGID.
EGIDs are food antigen-driven inflammatory conditions characterized by aeroallergen sensitization and concurrent atopic disease, resulting in the infiltration of eosinophils into intestinal tissue.4,5 This inflammation occurs throughout the GI tract, including in the esophagus (eosinophilic esophagitis (EoE)), stomach (eosinophilic gastritis (EoG)), small intestine (eosinophilic enteritis (EoN)), or colon (eosinophilic colitis (EoC)). EGID was first described in the early 1990s, and its incidence has continued to increase.4,6 EoE has the highest prevalence of all EGIDs at 34.4–52/ 100,000 people in the United States.7,8 Distal EGIDs are far less common. The prevalence of EoG is 6.3/100,000, EoN is 8.4/100,000, and EoC is 3.3/100,000. 8
EGIDs can be diagnosed at any age (including infancy), but more recent case series have shown that the 3rd–5th decade of life is most common. 9 Younger patients’ symptoms are often related to food allergies and present as feeding intolerance, vomiting, fussiness, and poor weight gain when introducing new foods. 10 However, symptoms vary based on the location and severity of eosinophilic infiltration. Chronic esophageal inflammation can eventually lead to narrowing of the esophagus. 10 Eosinophilic inflammation is not known to cause strictures outside the esophagus. 11 If limited to mucosal involvement, then distal EGID symptoms include abdominal pain, vomiting, diarrhea, and malabsorption, which can progress to GI bleeding or protein-losing enteropathy. 11 If eosinophils extend to or beyond the muscularis layer, obstructive symptoms like vomiting, abdominal pain, and abdominal distension may occur.9,11 If serosal involvement is present, a hallmark is eosinophil-predominant ascites driving abdominal distention and peritonitis.9,11
Diagnosis of EGIDs is endoscopy (both esophagogastroduodenoscopy and colonoscopy) with tissue biopsy. EoE has specific diagnostic criteria, 12 while the diagnostic criteria for EoG are still evolving. 13 The intensity of eosinophilic infiltration past the upper GI tract is quite variable, so distal EGID does not have standardized diagnostic criteria which contributed to the diagnostic challenges of our patient with multi-site EGID. Currently, there are no noninvasive diagnostic techniques and no proven blood-based biomarkers that accurately detect intestinal eosinophilia or response to treatment. Allergy testing, including food antigen-specific immunoglobulin E (IgE) testing, has a low positive predictive value in identifying dietary triggers that would increase EoE susceptibility. 14
Treatment options vary based on the location and extent of eosinophilic infiltration. Initial treatment options focus on dietary changes to identify and eliminate antigen-producing substances. The most prominent allergens include milk, egg, soy, wheat, nuts/tree nuts, and fish/shellfish.5,8,12,15 In severe cases, elemental diets can be considered. 16 However, these restrictive diets make adherence difficult. Thus, medications have been implemented to induce disease remission. Common attempted treatments include proton pump inhibitors and topical corticosteroids. 8 Swallowed fluticasone propionate and budesonide slurries have both resulted in significant improvement in eosinophil counts and dysphagia symptoms. Formulations of these topical corticosteroids have been developed to ease use and improve esophageal delivery. 15
New biologic treatments continue to emerge, with ongoing studies evaluating their safety and efficacy. However, most of these biologic agents have been developed for treatment of EoE, leaving a therapeutic gap for patients with multi-site EGID. Dupilumab, a human monoclonal antibody directed against IL-4 receptor alpha component, has been shown to have benefits in EoE. Weekly subcutaneous administrations have been shown to alleviate symptoms and improve histologic outcomes. Other biologics with ongoing trials include RPC4046, a monoclonal antibody against IL-13, Antilomab, an antibody to a surface receptor found on human eosinophils and mast cells, and Benralizumab, a monoclonal antibody against IL-5 receptor alpha. In early phase trials, these biologics have shown histologic improvement for EoE.15,16 Severe cases of stricture or bowel obstruction are treated with surgical interventions. 17
Conclusions
In conclusion, this case illustrates the challenges of reaching a timely and accurate diagnosis when a child presents with abdominal pain. The initial clinical picture, marked by abrupt symptom onset and potential zoonotic exposure, made more common infectious causes seem likely and appropriately prompted empiric antimicrobial therapy. However, the persistence of symptoms despite treatment emphasized the importance of maintaining a broad differential diagnosis and involving multiple specialties early in the evaluation. Ultimately, endoscopy and biopsy were critical in confirming the diagnosis of EGID, a rare but important cause of abdominal pain in children. This case highlights the need for vigilance in atypical presentations, recognition of key diagnostic clues such as eosinophilia and gastrointestinal inflammation, and the value of coordinated follow-up to guide effective treatment and improve outcomes.
Footnotes
Authors’ note
This case report was not part of a clinical trial.
Ethical considerations
Ethics committee and IRB approval are not required per institutional standards for a case review as parental consent and patient assent were obtained.
Consent to publication
The authors certify that patient and parental written informed consent was obtained for use of images and for publication of this case. Patient identifiers were removed for patient protection.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
