Abstract
Adalimumab, a tumor necrosis factor-alpha inhibitor, is widely used for chronic plaque psoriasis and psoriatic arthritis. While cutaneous adverse effects are known, perforating dermatosis is rare and poorly understood. A 34-year-old woman with psoriasis and psoriatic arthritis developed acquired perforating dermatosis after switching from adalimumab biosimilar GP2017–CTP17. She presented painful, ulcerated plaques on the thighs, gluteal area, and elbows. Histopathology confirmed the diagnosis. The biosimilar drug was discontinued and a 4-week course of systemic corticosteroids led to complete resolution. Both conditions were later managed with methotrexate and ixekizumab. Perforating dermatosis following anti-tumor necrosis factor is rare and underreported with adalimumab. No other known triggers (e.g., diabetes and renal failure) were present. Hypothesized mechanisms include fibronectin dysregulation and advanced glycation end accumulation, disrupting keratinocyte function. Perforating dermatosis should be recognized as a rare adverse effect of tumor necrosis factor-alpha inhibitors. Early recognition and discontinuation may prevent progression. Further studies are needed to clarify pathogenesis.
Keywords
Introduction
Adalimumab is a tumor necrosis factor-alpha (TNF-α) inhibitor biologic agent 1 approved for the treatment of several inflammatory immune-mediated diseases such as chronic plaque psoriasis, hidradenitis suppurativa, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and uveitis.1,2
Opportunistic infections, reactivation of latent tuberculosis, demyelinating diseases, and lupus-like syndrome have been reported as safety concerns related to adalimumab.3–5 The most common cutaneous adverse events induced by adalimumab include injection-site reactions, paradoxical psoriasis, eczematous dermatitis, urticaria and angioedema, areata alopecia, and vasculitis.6–9 Less common cutaneous adverse events have been occasionally reported, including sweet-like neutrophilic dermatosis and perforating dermatosis.10–12
The case of a 34-year-old woman who developed perforating dermatosis while receiving adalimumab biosimilar CTP17 for the treatment of plaque psoriasis and psoriatic arthritis is presented.
Case report
A 34-year-old Caucasian woman was referred to the outpatient clinic of the University Hospital of Verona for the acute onset of diffuse erythematous plaques with concentric hyperkeratosis and ulceration localized on the right thigh, the right gluteal area, and the elbows, associated with pain and difficulties in walking (Figure 1). The patient was affected by chronic plaque psoriasis and psoriatic arthritis and had been successfully treated with adalimumab biosimilar GP2017 for 2 years, which was later switched to CTP17 for non-medical reasons. The acute skin reaction occurred 3 days after switching to adalimumab biosimilar CTP17. The patient did not report any comorbidities, known allergies, or was not taking other medications apart from the adalimumab biosimilar. No other symptoms were reported. The skin lesions were biopsied, and the histological examination revealed epidermal hyperplasia, occasionally with pseudoepitheliomatous, featuring an ulcerated cup-shaped depression or funnel-like invagination forming a “transelimination channel” filled and covered by a plug consisting of parakeratotic keratin, neutrophils, and basophilic debris mixed with a fragment of eosinophilic elastic fibers expelled from the dermis. The acanthotic epidermis at the periphery appeared covered by both a parakeratotic stratum with an absent granular layer and a compact orthokeratosis with accentuation of the granular layer. A florid lymphoplasmacytic infiltrate was observed in the reticular dermis, with scattered neutrophils, eosinophils, and some microvacuolated histiocytes. These findings were consistent with a diagnosis of acquired perforating dermatosis (Figure 2). Fungal, bacterial, and mycobacterial cultures were negative.

Erythematous plaques with concentric hyperkeratosis on the right thigh and gluteal area.
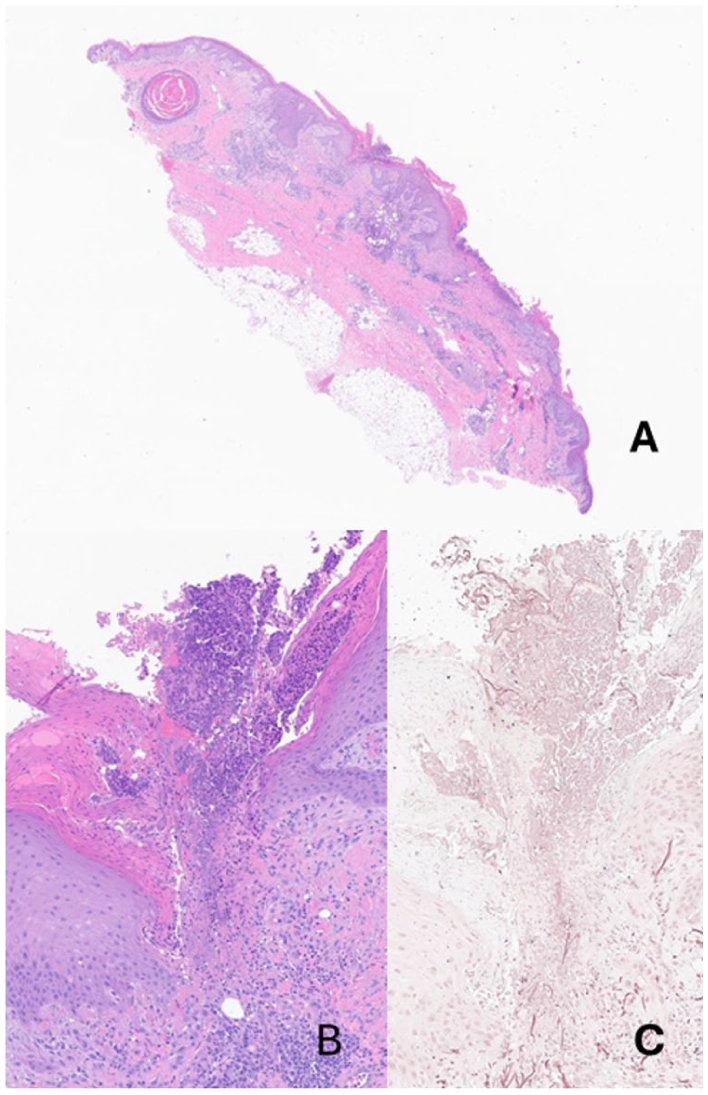
(a) Full-thickness skin section of the lesion (Hematoxylin and Eosin, ×0.9). (b) High-power view of the transepidermal elimination (Hematoxylin and Eosin, ×10). (c) High-power view of the transepidermal elimination (Orcein, ×20).
Consequently, adalimumab biosimilar CTP17 was suspended, and a 4-week course of systemic therapy with corticosteroids was prescribed. After approximately 8 weeks, the ulcers re-epithelialized, achieving complete resolution of the lesions. (Figure 3).
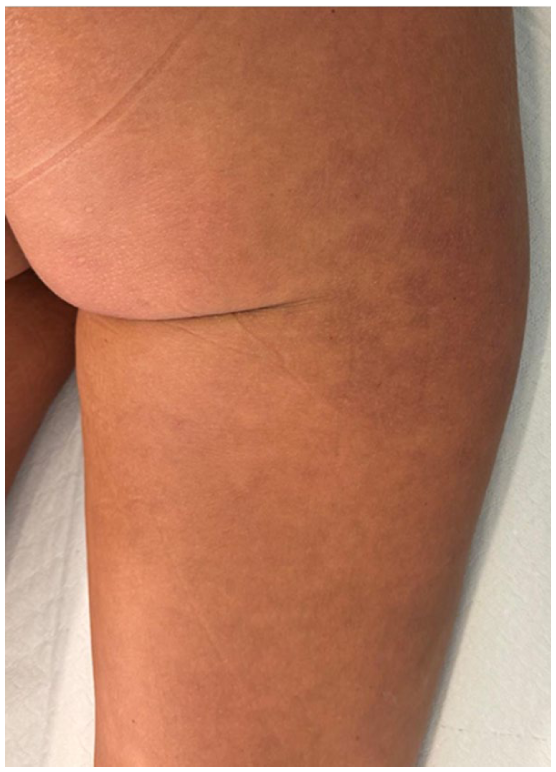
Clinical presentation after 8 weeks from adalimumab withdrawal with complete resolution of the lesions.
After 4 months, PsA recurred with tenosynovitis of the hands and feet bilaterally. The patient was referred to the consultant rheumatologist, who first prescribed methotrexate at a 12.5 mg/week dose. Since no benefits on the clinical manifestations and symptoms were reported, ixekizumab 80 mg every 4 weeks was combined with methotrexate, achieving better control of symptoms after 6 months of therapy.
Discussion
A case of acquired perforating dermatosis in a young female patient with psoriasis and psoriatic arthritis receiving adalimumab biosimilar CTP-17, which fully resolved after its withdrawal and a 4-week course of systemic corticosteroids, was presented.
Despite some levels of uncertainty, the hypothesis on the possible culprit of the disease in our patient was the adalimumab biosimilar. The patient developed acquired perforating dermatosis after switching from adalimumab biosimilar GP2017 to CTP17. No other possible triggers were identified, including trauma, diabetes, 13 chronic renal failure, 14 and lymphoma. 15 The patient also did not report any comorbidities beforehand.
Another case of perforating folliculitis was seen in a patient with a 10-year history of psoriasis. In this case, the authors hypothesize that, by treating psoriasis with topical agents (inhibiting cell replication), the patient changed the balance in favor of keratinization, particularly in the infundibulum, resulting in perforation. 16
Other cases of perforating dermatosis associated with TNF-α inhibitors have been reported.11,12
In a case series by García-Malinis et al., 12 five patients treated with infliximab for rheumatoid arthritis (2 patients), ankylosing spondylitis (2 patients), and ulcerative colitis (1 patient) developed acquired perforating dermatosis.
Gilaberte et al. 11 reported a case of perforating folliculitis that occurred in a patient affected by rheumatoid arthritis and several comorbidities, who had been treated with infliximab for 5 months. The perforating folliculitis resolved after the discontinuation of the drug, yet recurred after the administration of etanercept. In both cases, perforating dermatosis was attributed by the authors to TNF-α inhibitors.
Our case differs from those reported in the literature as it occurred in a patient with psoriasis and psoriatic arthritis, and it was caused by adalimumab.
Acquired perforating dermatosis was also associated with other biological drugs. In particular, Alhadlg et al. 17 depicted a case of risankizumab-induced perforating dermatosis in a patient treated for psoriasis. Other cases have been triggered by bevacizumab and natalizumab, yet no exact pathogenesis has been proposed. 18
In the attempt to find the mechanisms by which adalimumab triggers perforating dermatosis, hypotheses regarding the compromise of extracellular matrix (ECM) were identified in the literature, particularly in the role of fibronectin.
TNF-α has been implicated in the correct development of ECM through its action on fibronectin. Fibronectin plays a role in normal epithelial cell signaling, locomotion, and differentiation. A study has demonstrated that TNF-α directly inhibits fibronectin production and stimulates its degradation through the action of several metalloproteinases.19–22 Thus, by inhibiting TNF-α, fibronectin levels rise, inducing epidermal perforation.19–22
Patients with perforating dermatosis also presented overexpression of the advanced glycation end products. Literature shows that higher levels of these molecules may interfere with keratinocyte growth and migration. This might lead to abnormalities in the physiological ECM proliferation, differentiation to a normal skin structure, and development of perforating dermatosis.23–26
Despite these hypotheses, future research will be needed to better investigate the pathogenesis of perforating dermatosis, focusing on its correlation with biological drugs.
Footnotes
Consent to participate
The patient consented to publish the case report and images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
