Abstract
Tardive dyskinesia is a hyperkinetic movement disorder most often induced by chronic antipsychotic use. It can be challenging to treat, particularly when symptoms persist despite standard therapies such as vesicular monoamine transporter 2 inhibitors. We describe a 37-year-old male with schizoaffective disorder and persistent tardive dyskinesia involving oro-buccal-lingual and axial movements. Despite withdrawal of antipsychotics, a trial of deutetrabenazine, and high-dose tetrabenazine (150 mg/day for several weeks), symptoms persisted and significantly impaired critical care management. On hospital day 46, phenobarbital was initiated (dose and route here), resulting in rapid improvement of dyskinetic movements within 3 days. By hospital day 54, complete resolution of tardive dyskinesia was observed, and notably, symptoms did not recur after phenobarbital discontinuation. The patient maintained clinical improvement during long-term follow-up, with Abnormal Involuntary Movement Scale score improving to 2 at 18 months. This case illustrates a potential novel role for phenobarbital in refractory tardive dyskinesia, particularly when conventional therapies fail. The sustained response beyond the drug’s pharmacokinetic profile raises the possibility of a disease-modifying or prolonged therapeutic effect. Given phenobarbital’s activity on Gamma Aminobutyric Acid B receptors, its mechanism may involve modulation of GABAergic pathways implicated in the pathophysiology of tardive dyskinesia. Further investigation in clinical studies is warranted.
Introduction
Tardive dyskinesia (TD) is an iatrogenic hyperkinetic movement disorder characterized by involuntary, repetitive, and purposeless movements affecting the face, tongue, trunk, or extremities. It occurs most frequently in patients exposed to long-term dopamine receptor-blocking agents, particularly antipsychotics. 1 The prevalence of TD is estimated at approximately 20%–30% among patients receiving chronic antipsychotic therapy, with higher rates reported in elderly populations and those on prolonged treatment courses.2,3 Patients with TD experience significantly greater healthcare utilization and associated costs compared to those without the condition. 2 Clinical presentation typically involves oro-buccal-lingual movements (e.g., lip smacking, tongue protrusion, grimacing), as well as axial or limb choreoathetoid movements. 4 Complications may include functional impairment, psychosocial distress, aspiration risk, and difficulty managing critical illness due to persistent involuntary movements. 5
Management strategies for TD include withdrawal or reduction of the offending antipsychotic when feasible, pharmacologic therapy with vesicular monoamine transporter 2 (VMAT-2) inhibitors such as tetrabenazine or deutetrabenazine, and in severe cases, clozapine. Deep brain stimulation (DBS) is considered a last resort for patients with persistent, functionally disabling symptoms; however, it is not feasible for all patients due to cost, availability, comorbidities, and surgical candidacy. Despite these therapeutic options, some patients remain refractory to conventional treatment. 6 This case highlights a rare clinical scenario of treatment-resistant TD in a critically ill patient, in whom the introduction of phenobarbital led to marked clinical improvement. Phenobarbital, a barbiturate with GABAergic activity, is not routinely used in TD management, making its successful application in this case noteworthy. 7
Case report
We present the case of a 37-year-old White male with a past medical history notable for schizoaffective disorder and substance abuse (benzodiazepines), and tobacco abuse who arrived at our emergency department reporting abnormal movements persisting for 1 month. These movements comprised lip smacking, tongue protrusion, blinking, grimacing, smiling, choreoathetoid movements of the upper extremities, and rocking and twisting of the neck and trunk. The patient had been treated with long-term antipsychotic medication, and it was noted that the patient had been prescribed two antipsychotics with medication evaluation. The patient’s medications at the time of admission are further detailed in Table 1. Prior to admission, magnetic resonance imaging (MRI) revealed bilateral globus pallidus hyperintensities with no signs of stroke (Figure 1).
List of medications prior to admission.


MRI showing bilateral globus pallidus T2 hyperintensity lesions.
Upon admission, vital signs were remarkable for sinus tachycardia (pulse 102 beats/min). On physical exam, the patient was alert and fully oriented. The TD movements were noted. The remainder of the physical exam was unremarkable. Laboratory investigations revealed leukocytosis with a white blood cell count of 21,700/μL. Additionally, computed tomography (CT) of the chest revealed multifocal pneumonia, prompting initiation of antibiotic therapy with piperacillin-tazobactam and doxycycline. The sequence of events is depicted in Figure 2.
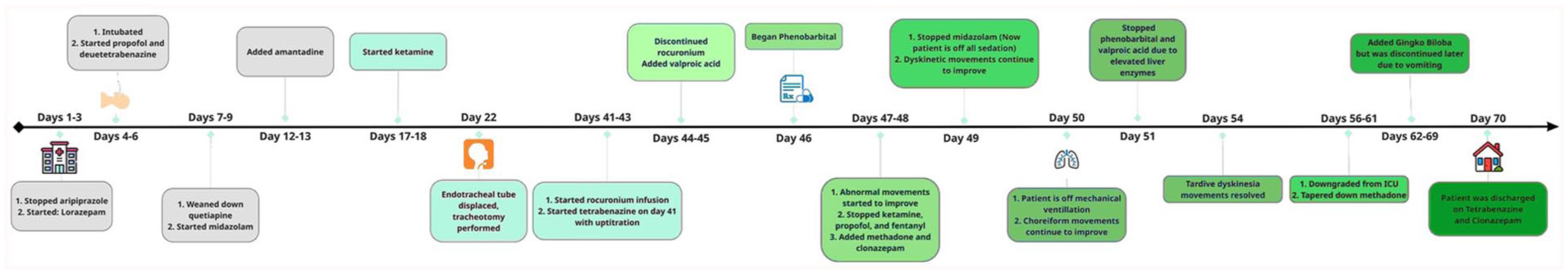
Timeline of events.
By hospital day 4, the patient was transferred to the intensive care unit (ICU) and intubated due to worsening respiratory status with an oxygen saturation of 73%. Norepinephrine was commenced for septic shock secondary to pneumonia, upon recommendations from the infectious disease service consulting with the critical care team. On the same day, the neurology service initiated treatment with deutetrabenazine with gradual uptitration, albeit without improvement in abnormal movements, necessitating the use of physical restraints to mitigate potential self-harm in the ICU.
Further evaluation via CT scan of the head revealed attenuation within the globus pallidus bilaterally. On hospital day 22, with ongoing difficulty in controlling TD, displacement of the endotracheal tube occurred, necessitating tracheostomy tube insertion.
Subsequently, on day 41, due to lack of availability and cost concerns, deutetrabenazine was transitioned to tetrabenazine after consultation with the pharmacy, ultimately reaching a maximum daily dose of 150 mg by hospital day 45. Additionally, on day 41, rocuronium was added to the sedative regimen to suppress TD movements and prevent tracheostomy tube displacement, albeit temporarily. It is worth noting that, despite high dosing of sedative and paralytic therapy, any decrease in dosage leads to worsening of movement, further destabilizing the patient.
On hospital day 46, the internal medicine team, following an extensive literature review, started a trial of oral phenobarbital to attempt to increase the recruitment of GABAergic neurons, in hopes of suppressing refractory TD movements. This was considered an unorthodox clinical approach, but the medical team recognized that the benefits outweighed the risks given the complicated ICU hospital course. Therefore, phenobarbital was initiated via nasogastric tube in hopes of suppressing TD.
By day 49, the teams observed significant improvement in dyskinetic movements: fewer types and reduced frequency of TD movements. By day 50, the patient regained consciousness and answered questions by nodding. The patient was successfully weaned off mechanical ventilation, transitioning to high-flow oxygen via the tracheostomy tube, which was later removed. Complete resolution of TD was observed by day 54.
However, at this time, due to elevated liver function tests, the neurology service discontinued the phenobarbital. Nevertheless, TD movements remained suppressed until the time of discharge. The patient was subsequently discharged to a skilled nursing facility for rehabilitation following the critical care hospitalization. Upon follow-up in the primary care clinic, the Abnormal Involuntary Movement Scale (AIMS) score was 13. The patient reported difficulties obtaining tetrabenazine from the pharmacy. Insurance issues were resolved by the patient and primary care provider, and the patient became more adherent to his medication. His AIMS score progressively continued to improve until the AIMS score was 2 at 18 months follow-up. A repeat MRI performed in the outpatient setting showed bilateral cystic changes in the globus pallidus.
Discussion
There is a focused differential diagnosis for this unfortunate event, which includes hypoxic injury to the globus pallidus, medication-induced TD, and stroke. Although the patient is an avid smoker, the bilateral nature of the injury, the patient’s relatively young age, the absence of diffusion-weighted MRI abnormalities, and the lack of other significant vascular risk factors make stroke less likely. Hypoxic injury remains a possible cause; however, the patient had been experiencing abnormal movements for a month prior to admission, which is inconsistent with an acute hypoxic insult. Given the chronicity of symptoms, the characteristic nature of the movements, and the patient’s use of dual antipsychotic therapy (both known to be associated with TD), this leads to TD emerging as the most likely diagnosis. This case illustrates a distinctive and challenging instance of TD that responded to phenobarbital after failing to improve with antipsychotic withdrawal, addition of a VMAT-2 inhibitor, and use of benzodiazepines.
The criteria for DBS in TD include severe symptoms that substantially impair functioning and persist for over a year, with inadequate response to pharmacological interventions such as clozapine or tetrabenazine at maximum tolerated doses for at least 4 weeks. 8
Our patient did not meet these criteria and was therefore not considered for transfer to another facility for DBS.
Pathophysiology of TD is implicated through different neuronal pathways. Although upregulation of D2 receptors resulting from long-term antipsychotic treatment is highly cited in the literature as the cause of TD, it is not the only contributor to TD. Gamma aminobutyric acid (GABA) neuronal dysfunction in the striatum, also resulting from long-term antipsychotic use, has been demonstrated in human and animal studies.8–11
Midazolam (a benzodiazepine), which acts on GABA neurons, should have helped this patient’s symptoms according to the GABAergic neuron dysfunction hypothesis.
GABA exerts its inhibitory effects through two receptor types. GABAA receptor, a ligand-gated chloride channel, that quickly hyperpolarizes the postsynaptic membrane, and GABAB receptor, a metabotropic receptor which generates slow inhibitory signals via G proteins and second messengers.10,12,13 Dysfunctions in GABAB receptors are linked to epilepsy, depression, addiction, cognition, and pain perception. 14 Midazolam activates primarily the GABAA receptor, which leads to the opening of chloride channels, causing an influx of chloride ions, which hyperpolarizes the neuron’s membrane. 15 As noted, midazolam primarily activates GABAA receptor. Phenobarbital, on the other hand, stimulates the GABAB receptors, potentially broadening the recruited GABAergic neurons. Our working clinical theory in this vexing case was to provide stimulation to GABAB receptors, which might alleviate the patient’s TD movements. Phenobarbital did indeed provide complete termination of the patient’s abnormal movements and subsequently allowed the team to withdraw all sedation (and switch intravenous midazolam to oral clonazepam). It is crucial to promptly control dyskinetic movements if they compromise the airway such as in this case.
This article with its description of refractory TD movements provides evidence that TD could result from dysfunction in both GABAA and GABAB receptors, in which stimulation of GABAA receptors by benzodiazepines alone is not sufficient to decrease TD movements.
Bobruff et al. 16 assigned 21 patients to receive either clonazepam or phenobarbital and found that clonazepam improves mainly orofacial dyskinesia, while phenobarbital had more noticeable effects in improving limbs and axial movements. 12 In this case, phenobarbital had favorable effects in reducing orofacial, limb, and axial movements. What is left, in this most challenging case, that still eludes contemporary evidence-based medical understanding is that subsequently, upon discontinuation of phenobarbital, the patient remained TD-free. Our bedside observations suggest a sustained therapeutic effect well beyond the duration of phenobarbital, which has a mean half-life of 5.1 days when taken orally. 13 The sustained resolution of TD following phenobarbital discontinuation suggests either that phenobarbital exerted a rapid, additive effect through GABAB receptors complementing benzodiazepines (which act on GABAA receptors) to control TD movements until tetrabenazine reached its full therapeutic effect, or that there is a possible disease-modifying or prolonged therapeutic benefit, or both, extending well beyond the pharmacokinetic properties of phenobarbital itself.
Conclusion
This case report highlights a unique approach in utilizing phenobarbital in the case of refractory TD, which fails to respond to pharmacological therapies in critically ill patients. Our work with this single challenging patient pushes us as bedside clinicians to recommend further clinical trials exploring phenobarbital as a novel treatment approach to refractory TD not responding to therapy.
Footnotes
Acknowledgements
None.
Ethical considerations
Ethical approval to report this case was obtained from (AdventHealth Risk Management Ethics Committee).
Consent to participate
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
