Abstract
We present a case of a 16-year-old boy with asthma referred to pediatric gastroenterology for nausea and chronic cough. Esophagogastroduodenoscopy and bronchoscopy revealed parakeratosis/hyperkeratosis in the esophagus and nontuberculous mycobacteria pulmonary infection, respectively. Presumed reflux as a unifying cause for his symptoms was confirmed with intra-esophageal pH monitoring, and he was successfully treated with a proton pump inhibitor and a four-drug antimicrobial regimen.
Keywords
Introduction
Esophageal parakeratosis and hyperkeratosis are relatively rare findings, especially in children. Likewise, nontuberculous mycobacteria (NTM) is also uncommon in immunocompetent individuals. This is a unique case of an adolescent male with asthma and chronic cough who had characteristic findings of esophageal hyperkeratosis with NTM pulmonary infection as well as pulmonary Aspergillus. This case highlights a rare constellation of findings connected to long-term inhaled and systemic corticosteroid use in the setting of reflux and demonstrates the importance of coordinated multidisciplinary care in complex pediatric patients.
Case report
A 16-year-old male with a history of asthma, eczema, deviated septum, appendectomy, and recurrent acute otitis media requiring tympanostomy tube placement at the age of 2 years was referred to our Pediatric Aerodigestive Clinic for a half-year history of moderate persistent asthma, chronic cough, and gastrointestinal symptoms including nausea and constipation. As part of his asthma maintenance regimen, he had been taking high-dose inhaled corticosteroid/long-acting-beta-2 (ICS-LABA) medications for more than 9 years with adequate control of his asthma symptoms. He developed a prolonged cough with hemoptysis following an upper respiratory infection a few months prior to our evaluation and was initially placed on a 5-day oral prednisone course and azithromycin by his pulmonologist. The cough persisted and was accompanied by fatigue. He was subsequently prescribed dexamethasone. In terms of his gastrointestinal symptoms, he had intermittent nausea and reflux and reported feeling acid coming up in his throat. There was no improvement noted following proton pump inhibitor (PPI) therapy with lansoprazole 15 mg once daily. He also reported chronic constipation and frequent flatulence.
As part of his diagnostic workup, he underwent esophagogastroduodenoscopy (EGD) and bronchoscopy. The EGD demonstrated white-appearing linear furrows in the mid and distal esophagus (as shown in Figure 1) and esophagitis in the distal esophagus. Biopsies were collected in the proximal, mid-, and distal esophagus. Histopathology (as demonstrated in Figure 2(a) and (b)) showed patchy acute inflammation throughout the whole esophagus and focal parakeratosis/hyperkeratosis in the proximal and mid-esophagus. No increased eosinophils were identified in the squamous mucosa at any of the esophageal levels, no fungal forms were identified. Bronchoscopy demonstrated a moderately inflamed airway, mild cobblestoning of the trachea and carina, a small amount of bright yellow, thick mucus in the distal airway, and severely edematous turbinates. Bronchoalveolar lavage (BAL) cultures grew both Mycobacterium chelonae and Mycobacterium avium complex (MAC) at 2 weeks.

Endoscopic finding of white-appearing linear furrows in the mid and distal esophagus.
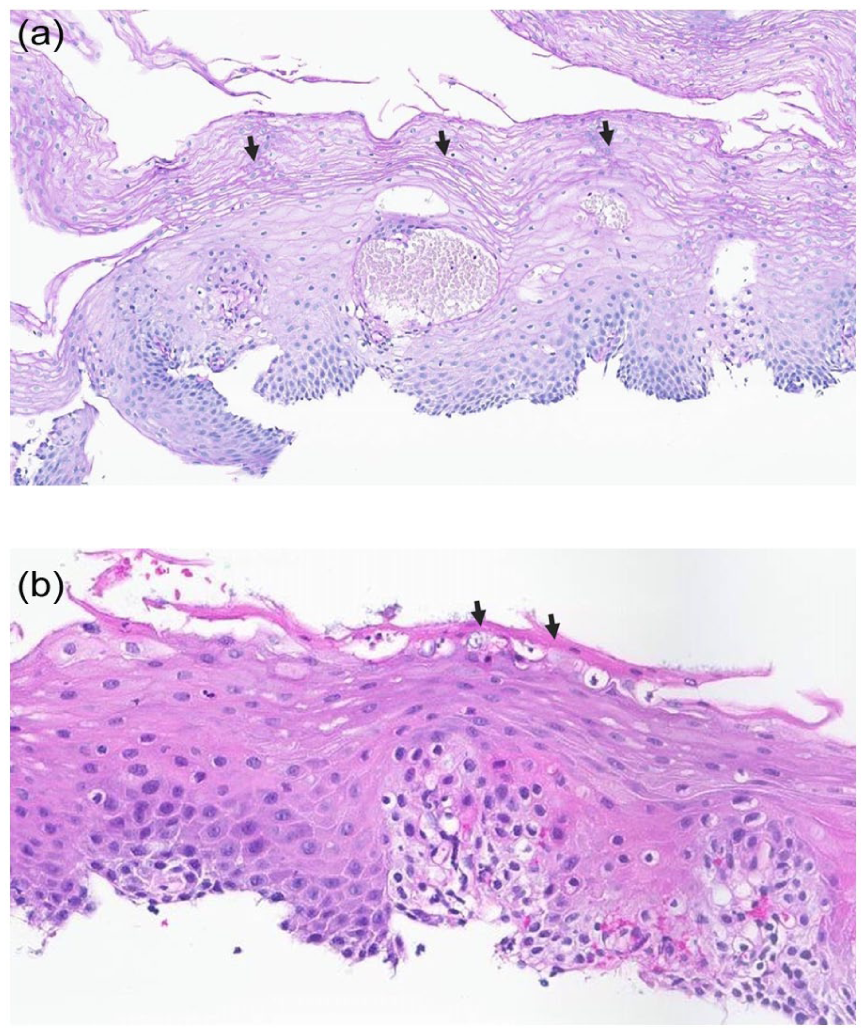
(a) Esophageal squamous mucosa with parakeratosis in the superficial aspect (black arrows; hematoxylin and eosin stain). (b) Esophageal squamous mucosa with a thin layer of hyperkeratosis appearing as a dense eosinophilic layer on the surface of the epithelium (black arrows; hematoxylin and eosin stain).
Chest computed tomography showed no hyperexpansion or bronchial wall thickening. Gastroesophageal reflux was presumed to be a contributor and was confirmed with intraesophageal pH monitoring which revealed esophageal acid exposure at a degree of 14.3% (normal: <5%) and frequency of 51 reflux episodes within a day. His PPI therapy was optimized to pantoprazole 40 mg once daily with subsequent improvement in gastrointestinal symptoms. The patient did not want to take antibiotic therapy without confirmation that the NTM was not a contaminant. Therefore, repeat EGD and bronchoscopy were performed 2 months later. EGD appeared normal and repeat biopsies showed resolution of initial pathology. Bronchoscopy showed a significantly improved airway with no signs of inflammation or cobblestoning. Despite these findings, BAL again grew M. chelonae and MAC at 2 weeks. Fungal cultures also revealed growth of Aspergillus fumigatus at this time.
After confirmation of infection with NTM, the patient was started on a 4-drug regimen for 8 weeks (Clarithromycin 1000 mg, Rifampin 600 mg, Ethambutol 1600 mg, Linezolid 600 mg, each three times weekly). He completed the treatment with remarkable symptom improvement without any major side effects. The patient was also evaluated for underlying immunodeficiency during his treatment course. His T cell function and IgA were found to be normal. A third and final bronchoscopy was performed following treatment and BAL was negative.
Discussion
Esophageal parakeratosis and hyperkeratosis are rare findings, especially in children. Taggart et al. reported that hyperkeratosis involves ~2% of esophageal biopsies in adult patients. 1 Hyperkeratosis is known as a complication of gastroesophageal reflux disease and alcohol/tobacco consumption, and is related to squamous neoplasms, especially when not associated with Barret esophagus/adenocarcinoma.1,2 Of note, our patient’s hyperkeratosis was most likely related to underlying gastroesophageal reflux, the diagnosis of which was confirmed by intraesophageal pH monitoring. Hyperkeratosis is also reported to have an association with the aberration of fat-soluble vitamins, including vitamin A, vitamin B, and zinc deficiency, and vitamin E excess.1,3–6 Our patient was tested and had normal micronutrient levels.
Two case reports of esophageal hyperkeratosis in adults with gastroesophageal reflux showed persistent histopathologic changes of hyperkeratosis for many years despite PPI therapy.2,7 However, in our case, hyperkeratosis disappeared within 2 months after treatment of gastroesophageal reflux. It is unclear if this rapid resolution is atypical as some of the cases described in adults only comment on symptom improvement rather than endoscopic follow-up. As noted, our patient also had a history of asthma. Interestingly, the diagnosis of esophageal hyperkeratosis has also been reported in an adult patient with asthma. 8 While this is most likely a spurious association, it may warrant further investigation. Esophageal hyperkeratosis has also been reported in the setting of esophageal eosinophilia, though it seems this is more likely related to underlying Gastroesophageal Reflux Disease (GERD) rather than eosinophilic esophagitis.9–11
This case was also combined with NTM (M. chelonae and MAC) and pulmonary Aspergillus lung infections. NTM is a group of ubiquitous environmental bacteria that can be found in soil, dust, and water. 10 MAC is the leading cause of NTM disease in immunosuppressed patients, while M. chelonae is one of the rapidly growing mycobacteria (RGM). 11 Pulmonary disease due to RGM is very rare and known to be resistant to standard antituberculosis medications. 12 One study showed an increased risk of NTM infection in adult asthmatic patients, particularly those who are older and have used longer duration and higher doses of inhaled corticosteroids compared with controls. 13 Furthermore, oral prednisone use was reported to be eight times higher among NTM cases than in controls. 14 Fayos et al. reported that NTM and Aspergillus pulmonary co-infection is rare but tends to occur in patients with underlying lung disease such as asthma and those who have used inhaled or systemic corticosteroids. 15 Additionally, gastroesophageal reflux disease is associated with a 3.4-fold increased risk of NTM pulmonary disease. 16 Gastroesophageal reflux disease likely raises the risk of NTM pulmonary disease given micro aspiration, which leads to continuous destruction of the protective airway barrier, leading to a propensity and risk for NTM infection.17,18 Additionally, NTM can be microaspirated and deposited into the lung, where it can then cause infection of epithelial cells. 19 Our patient had asthma treated with both inhaled and systemic steroids, which increased his propensity for NTM pulmonary disease. Furthermore, he also had gastroesophageal reflux which may have further accelerated his risk of NTM infection due to micro aspiration. A four-drug regimen was selected in this patient to ensure effective coverage of both M. chelonae and MAC. This minimizes risk of macrolide resistance and enhances likelihood of microbiological clearance in the setting of dual NTM and Aspergillus infection. Long-term follow-up for this patient included repeat bronchoscopy which confirmed negative BAL cultures posttherapy. Patient continues undergo close clinical monitoring for recurrence.
Conclusion
In conclusion, esophageal parakeratosis and hyperkeratosis are rare pathological findings, especially in children. Clinicians should consider the associations of gastroesophageal reflux and nutritional deficiencies when hyperkeratosis is detected. Moreover, for patients who are immunocompromized or those being treated with steroids, the potential for NTM pulmonary infection should be recognized.
Footnotes
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and was ethically approved by the IRB Ethics Committee of Cedars Sinai (#STUDY00003030) on Oct 20, 2023.
Consent to participate
The participant and the legally authorized representative provided written informed consent prior to participating.
Consent for publication
Informed consent for publication was provided by the participant and the legally authorized representative.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author, [LJW], upon reasonable request and with appropriate IRB approval.
