Abstract
Hepatic epithelioid angiomyolipoma is a rare mesenchymal liver tumor within the perivascular epithelioid cell tumor family, known for its uncertain malignancy and potential for aggressive behavior. Clinical presentation, laboratory findings, and imaging results lack specificity, making pathological examination critical for definitive diagnosis. This case report presents a 61-year-old male patient with a single tumor in the right liver lobe, measuring approximately 101 mm × 99 mm. Initial imaging misdiagnosed the mass as hepatocellular carcinoma. Following multidisciplinary treatment discussions and subsequent tumor resection, histopathology and immunohistochemistry confirmed hepatic epithelioid angiomyolipoma. Accurate diagnosis of hepatic epithelioid angiomyolipoma is challenging, relying on histopathological examination and immunohistochemistry as diagnostic standards. Continuous monitoring of patients with hepatic epithelioid angiomyolipoma over an extended period is deemed essential. This report highlights the positive impact of multidisciplinary treatment in diagnosing and managing hepatic epithelioid angiomyolipoma.
Introduction
Angiomyolipoma (AML) is a benign mesenchymal tumor commonly found in kidneys; it belongs to the perivascular epithelioid cell tumor (PEComa) family, with a higher incidence in women. 1 Hepatic epithelioid angiomyolipoma (HEAML), first documented in 1976, 2 is rare and composed variably of blood vessels, smooth muscle, and adipose cells. 3 It has been associated with tuberous sclerosis, an autosomal dominant genetic disorder. 4 Although rare, HEAML presents an uncertain malignant potential and aggressive behavior. 5 Fewer than 600 cases have been reported globally, with most publications comprising case reports or small case series. 6 Due to its low incidence, atypical clinical presentation, and nonspecific laboratory and imaging characteristics, HEAML is frequently misdiagnosed as other hepatic tumors, particularly hepatocellular carcinoma (HCC). Most HEAML lesions exhibit immunoreactivity toward melanocytic markers (Human Melanoma Black-45 [HMB-45] or Melan-A or both) and smooth muscle markers (smooth muscle actin (SMA) or desmin or both). Therefore, histopathological examination and immunohistochemistry of biopsy or operational specimens are essential for a definitive diagnosis of HEAML. 7
The rarity of HEAML, combined with its low preoperative diagnosis rate and indeterminate malignant potential, presents considerable diagnostic and therapeutic challenges, contributing to ongoing debate within the medical community. This report presents the case of a 61-year-old male patient with a rapidly growing single tumor in the right liver lobe, initially suspected as a different hepatic tumor due to an unclear preoperative diagnosis. Following a multidisciplinary treatment (MDT) discussion and histopathological examination, the diagnosis of HEAML was confirmed, and the patient successfully underwent the operation. Immunohistochemistry analysis further validated the HEAML diagnosis. This case report highlights the MDT approach’s significant role in managing HEAML. It contributes to developing diagnostic and treatment protocols for this tumor, aiming to deepen understanding of its presentation and management.
Case presentation
Chief complaint
A 61-year-old man presented with an interstitial liver lesion noted for over a year.
History of illness
During an abdominal ultrasound (US) performed 1 year earlier, a solitary hypoechoic mass measuring approximately 27 mm × 24 mm was detected in the right liver lobe, with a defined boundary and detectable blood flow within and around the lesion, initially suggestive of hepatic hemangioma (Figure 1(a)). Due to the patient’s poor compliance, regular follow-up was not conducted. Recently, while undergoing evaluation for an upper respiratory infection, a chest computed tomography (CT) scan incidentally revealed a sizable liver lesion within the right liver lobe (Figure 1(b)). The initial US and recent CT scans of the patient indicated a substantial increase in the liver mass size, suggesting significant disease progression. The patient reported no consistent symptoms such as liver pain, bloating, fever, or jaundice and had no history of chronic hepatitis. Physical examination revealed a flat abdomen without muscle tension, tenderness, or rebound pain. The liver and spleen were not palpable below the rib cage; percussion tests of the liver were negative, abdominal percussion yielded tympanic sounds, and there was no shifting dullness. Bowel sounds were regular at 5 per min. Laboratory evaluations showed average results across all parameters, including complete blood count, electrolytes, liver and kidney function tests, and coagulation profiles. Screening for hepatitis and other infections revealed no abnormalities. Tumor biomarkers such as alpha-fetoprotein (AFP), alpha-fetoprotein-L3 (AFP-L3), prothrombin induced by vitamin K absence II, carbohydrate antigen 19-9, and carbohydrate antigen 125 were all within normal limits. However, the carcinoembryonic antigen was slightly elevated at 6.00 μg/L (range 0–4.5 μg/L).
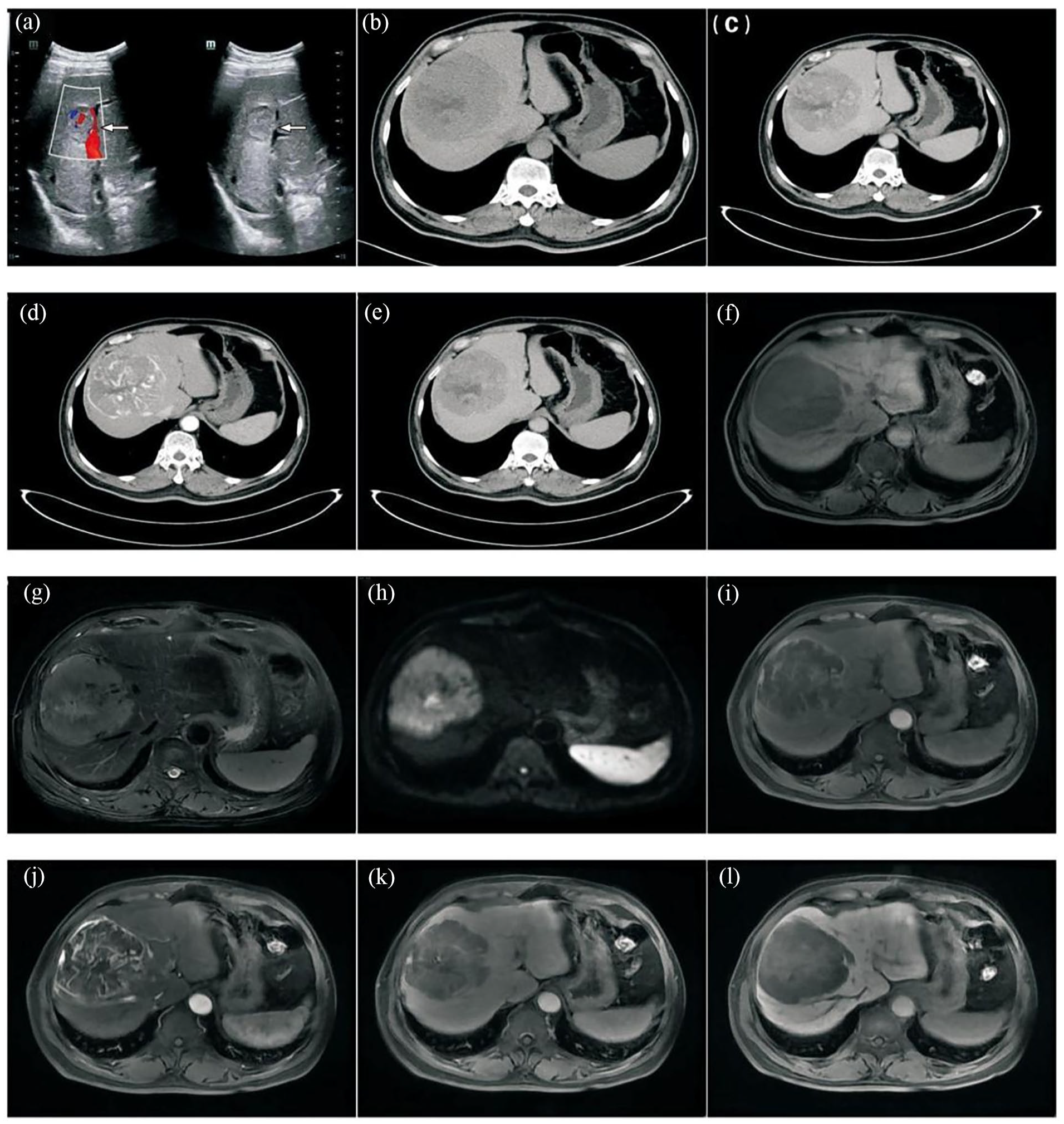
A 61-year-old man with HEAML. (a) Initial ultrasound showing a hypoechoic mass, measuring approximately 27 mm × 24 mm (arrows). (b–e) CT and CECT imaging. (b) Hypoattenuating lesion in the right lobe of the liver, measuring approximately 101 mm × 99 mm with heterogeneous density. (c) Heterogeneous hyperenhancement in the arterial phase with visible vascular shadows. (d–e) Hypoenhancement in the portal and delayed phases. (f–l) MRI imaging. (f) Hypointensity in T1-weighted images. (g–h) Mixed-hyperintensity in T2-weighted and DWI. (i) Inhomogeneous enhancement in the arterial phase with thickened blood vessels in the lesion. (j–k) Decreased enhancement in the portal and delayed phases. (l) Hypointensity in the hepatobiliary phase.
Imaging examinations
Contrast-enhanced computed tomography (CECT) of the liver showed a well-defined, hypoattenuated lesion in the right lobe, measuring approximately 101 mm × 99 mm, with heterogeneous density. During the arterial phase, the lesion demonstrated heterogeneous hyperenhancement with visible vascular structures, while the portal and delayed phases showed hypoenhancement (Figure 1(c)–(e)). Abdominal magnetic resonance imaging (MRI) indicated a sizeable, well-defined tumor in the right liver lobe, which appeared hypointense on T1-weighted images and mixed-hyperintense on T2-weighted and diffusion-weighted imaging (DWI). The arterial phase showed inhomogeneous enhancement with multiple thickened blood vessels supplying the lesion, which decreased in the portal and delayed phases. The hepatobiliary phase showed a hypointense signal (Figure 1(f)–(l)). These imaging characteristics suggested a substantial likelihood of HCC.
Final diagnosis
Based on the patient’s clinical characteristics, laboratory findings, and imaging results, the hepatic surgery team determined that there was insufficient evidence to confirm a diagnosis of HCC, leaving the preoperative diagnosis inconclusive. After an MDT discussion, it was unanimously agreed that while the liver tumor showed progression, the characteristics were not entirely consistent with HCC. It was recommended to perform a pathological examination to ascertain the tumor’s nature before formulating a treatment plan. Subsequently, a US-guided percutaneous transhepatic biopsy was performed, revealing numerous epithelioid cells with eosinophilic cytoplasm, heterogeneous cells, scattered mature adipocytes, and a vascular interstitium. The biopsy suggested HEAML (Figure 2(m)–(n)).

(m) Pathological biopsy showing numerous epithelioid cells with eosinophilic cytoplasm, heterogeneous cells, mature adipocytes in focal areas, and a vascular interstitium (×100). (n) Highlight the different components of HEAML: adipocytic (*), vascular (**), and numerous epithelioid cells (***), (×100). (o) Macroscopic view showing a grayish-yellow, moderately soft, well-defined tumor with hemorrhages and necrosis. (p–s) Histopathological examination reveals a network of fibrillar vessels separating tumor cells in a nest-like pattern. Epithelioid hyaline cells with eosinophilic cytoplasm, multinucleated giant cells with distinct nucleoli, rare nuclear fragmentation, and significant hemorrhagic necrosis are present (p × 100, q × 200, r × 400, s × 200). (t–u) Immunohistochemical staining showing strong and diffuse positivity for HMB-45 and Melan-A (t–u × 200).
Treatment
HEAML, a rare mesenchymal liver tumor with uncertain malignant potential, typically demonstrates aggressive behavior and may require surgical intervention. With informed consent from the patient and family, a laparotomic hepatectomy was adopted. Intraoperative US was employed to trace the course of the middle and right hepatic veins, ensuring a safe incision margin more significant than 10 mm. The right anterior Glissonean pedicle was ligated and divided, and the tumor was resected entirely without complications. Macroscopically, the excised tumor appeared grayish-yellow, moderately soft, well-circumscribed, with areas of hemorrhages and necrosis. Histopathological examination revealed a network of fibrous vessels surrounding tumor cells arranged in a nest-like pattern. Epithelioid cells with eosinophilic cytoplasm, multinucleated giant cells with distinct nucleoli, rare nuclear atypia, and significant hemorrhagic necrosis were observed. Immunohistochemical staining showed positivity for HMB-45, Melan-A, vimentin (Vim), and Transcription Factor E3 (TFE-3), while cytokeratin (CK), SMA, and S-100 were negative. The Ki-67 proliferation index was <5% (Figure 2(o)–(u)). The final diagnosis was confirmed as HEAML based on histopathological and immunohistochemistry findings.
Outcome and follow-up
The patient made a full postoperative recovery and was discharged. No recurrence or metastasis has been observed during routine outpatient visits and follow-up calls. This case report adheres to the Declaration of Helsinki, with written informed consent obtained from the patient and approval granted by the Institutional Ethics Committee of the Affiliated Chuzhou Hospital of Anhui Medical University (approval code: 2024 Ethical Review [Bio] no.31; date of approval: October 9, 2024).
Discussion
AMLs are primarily found in the kidneys; however, they can also affect the liver, with a higher incidence in women. AML is composed of a complex mixture of histological constituents, and it can be classified based on the relative proportions of these constituents. 8 HEAML, a rarer form within the PEComa family, shares histological features with AML and is distinguished by epithelioid cells. 9 The origin of HEAML cells remains unclear, and it was once considered a benign tumor. However, increasing evidence suggests its potential for malignant behavior, 10 with reports of local recurrence and distant metastasis. 11
HEAML lacks specific clinical manifestations; most patients do not experience abdominal discomfort upon initial presentation. Liver masses are frequently discovered incidentally during imaging. While large masses can cause liver pain or even rupture and bleeding, 12 our patient did not present these symptoms. Furthermore, laboratory tests, especially tumor biomarkers, remained within the normal range and did not aid in diagnosing HEAML. Although some cases report mild elevation of tumor biomarkers, the findings lack clinical diagnostic value, 13 as seen in our case.
Imaging plays a critical role in diagnosing liver lesions. However, the rarity of HEAML complicates its identification. 14 Moreover, due to the variety of components in PEComa family tumors (fat, smooth muscle, blood vessels, and epithelioid cells), their diverse imaging features contribute to the diagnostic challenge. 15 HEAML typically lacks or has minimal fat components, distinguishing it from other tumors in the PEComa family, which frequently contain varying proportions of adipose tissue. In the abdominal US, tumors with significant fat or smooth muscle components usually exhibit intense echogenicity, while those with more vascular components tend to show low echogenicity. Vessel signals within the tumor can be detected using color Doppler flow imaging. Contrast-enhanced US (CEUS) has been reported to enhance the diagnostic accuracy for HAML, improving the detection rate from 24% with the standard US to 52% with CEUS. 16 The findings from CEUS typically show hyperenhancement during the arterial phase, followed by wash-out in the portal and delayed phases. In some cases, a continuous enhancement pattern (mild over-enhancement or equivalent enhancement) may be observed. 17 CT scans reveal low-density masses that appear homogeneous or heterogeneous, depending on the fat content. Lesions frequently contain soft tissue components, such as deformed blood vessels and spindle-shaped smooth muscle cells. On CECT, HEAML typically presents with excessive vascular proliferation, showing hyperenhancement in the arterial phase and hypoenhancement or delayed enhancement in the portal or delayed phases, based on vascular distribution. 18 MRI findings usually show hypointensity in the T1-weighted (T1-WI) phase, with hyperintensity or a mixed-signal in the T2-weighted (T2-WI) phase and hyperintensity in the DWI phase. Postcontrast imaging shows hyperenhancement in the arterial phase, and a reduction in enhancement during the portal and delayed phases. 19 These imaging features were observed in this case. However, the preoperative imaging characteristics of HEAML can resemble those of other vascular-rich liver tumors, such as HCC, focal nodular hyperplasia (FNH), and hepatocellular adenoma (HCA), complicating the differential diagnosis. 20 In this case, the diagnosis was initially misinterpreted as HCC, which typically shows the classic wash-in and wash-out imaging pattern, and frequently presents with a capsule. 21 FNH generally exhibits centrifugal enhancement with a radial vessel distribution on CEUS. MRI may reveal a central scar with rapid and uniform enhancement in the arterial phase. 22 HCA typically appears as a homogeneously enhancing lesion with high density and potential necrosis or internal bleeding. 23 Although the typical imaging manifestations of HEAML remain undefined, it should be considered in patients without a hepatitis history, exhibiting no obvious clinical symptoms, standard tumor biomarkers, and imaging findings that indicate clear mass boundaries, absence of a capsule, lack or minimal fat content, and hyperenhancement in the arterial phase.
The clinical manifestations, laboratory findings, and imaging characteristics of HEAML lack specificity, complicating diagnosis. Histopathological examination and immunohistochemical analyses are essential for confirming the diagnosis. 24 In this case, the MDT team concluded that a pathological examination was necessary for definitive diagnosis due to variations in imaging results and interpretations from hepatobiliary surgeons. AML is histologically categorized into classical and epithelial variants, with the epithelioid component representing 10%–100% of the epithelial variant. 6 The predominance of the epithelioid element is a critical histological factor in diagnosing HEAML. Research suggests that at least 10% of epithelioid cells are pivotal for diagnosis. 25 These cells can form sheets, nests, or vesicles, with occasional atypical epithelioid and tumor giant cells. The tumor cells typically exhibit abundant cytoplasm and rare nuclear pleomorphism; they are associated with a stroma rich in thin-walled blood vessels, some of which may show signs of necrosis. Immunohistochemical positivity for melanocytic markers (HMB-45 or Melan-A or both) and smooth muscle markers (SMA or desmin or both) is characteristic of HEAML, 26 while CK and S-100 are generally negative. 27 Histopathological examination necessitates differentiation from other liver neoplasms, such as HCC and metastatic tumors. Special attention is warranted to distinguish melanoma, as it strongly expresses melanocyte markers, and frequently shows robust S-100 protein positivity and B-Raf proto-oncogene, serine/threonine kinase V600E (BRAFV600E) mutations. The differential diagnosis must consider epithelioid extragastrointestinal mesenchymal tumors, smooth muscle tumors, smooth muscle sarcomas, and rhabdomyosarcomas.
The MDT approach plays a pivotal role in the management of HEAML. 14 The importance of collaboration among hepatic surgery, imaging, laboratory, and pathology departments for comprehensive patient care is emphasized. Regular follow-up may be recommended for asymptomatic or small-volume HEAML confirmed histopathologically. However, adverse events, including tumor progression leading to fatal outcomes, have been reported, 28 highlighting the necessity of assessing the malignant potential of HEAML. Histological findings of HEAML include sheets of large polygonal cells with abundant granular eosinophilic cytoplasm arranged in a radial pattern around perivascular areas, suggesting aggressive behavior. 29 In their review of real-world studies, Folpe et al. 30 proposed six high-risk histological features for assessing PEComas, determining that tumors with a size ⩾5 cm and no additional high-risk features or nuclear pleomorphism/multinucleated giant cells were classified as having uncertain malignant potential. Tumors with two or more high-risk features were considered malignant. Bleeker et al. 31 concluded that a primary tumor size ⩾5 cm and a high (1/50 high power field (HPF)) mitotic rate were significant factors of recurrence following surgical resection. Calame et al. 32 suggested that a tumor size >5 cm, infiltrative growth patterns, high nuclear grade, necrosis, and mitotic activity >1/50 HPF might be associated with recurrence and metastasis risk. Despite the rarity of PEComas, there remains limited evidence-based data on tumor progression, recurrence, and metastasis. However, clinical symptoms, imaging indicative of disease progression, tumor diameter >5 cm, or with cytological manifestations of tumor abnormalities and necrosis, are a strong evidence in support of malignant features, so a diagnostic biopsy is recommended.32,33 There is a paucity of consensus regarding the optimal treatment strategy for HEAML, and a dearth of high-quality, evidence-based medical evidence to demonstrate which treatment modality is the most efficacious. The majority of patients undergo treatment via surgical intervention. In this case, the patient’s tumor exhibited rapid growth and reached a considerable size, with histopathology and immunohistochemistry confirming a diagnosis of HEAML, supporting the decision for surgical removal. For patients with minor or multiple scattered lesions, or in cases of spontaneous rupture and internal bleeding associated with large tumors, some studies recommend primary transcatheter arterial embolization, followed by regular follow-up, with secondary tumor resection as needed. 7 In light of recent advances in targeted therapies, there is a growing body of evidence suggesting that mammalian target of rapamycin (mTOR) inhibitors may offer a promising avenue for treating malignant PEComa. 31 It is recommended that HEAML patients be followed up over the long term in order to facilitate the timely administration of treatment and the monitoring of recurrence based on changes in the disease.
In future clinical practice, the development of novel diagnostic and prognostic markers for HEAML is a necessity. A systematic study of a broader molecular spectrum is required to identify mutant genes and clarify the heterogeneity of different cell populations to identify specific targets for targeted therapies. Quantitative analysis of imaging features through artificial intelligence and deep learning is necessary to enhance discrimination from other tumors. Finally, a growing understanding of HEAML is essential to objectively assess the efficacy of surgical, local, and targeted therapies and develop individualized treatment and management plans. These measures will deepen the understanding of HEAML and provide a solid basis for clinical decision-making.
Conclusion
A rare case of HEAML is reported in a male patient, which progressed rapidly over a short period without significant clinical symptoms. Laboratory tests lacked diagnostic value, and imaging revealed hyperenhancement in the arterial phase with enlarged blood supply arteries. Histopathological and immunohistochemical analysis confirmed the diagnosis of HEAML. The patient achieved full recovery following operational resection. The current difficulties in diagnosing and treating HEAML are reflected in the fact that it has nonspecific clinical manifestations, laboratory findings, and imaging characteristics, HEAML is frequently misdiagnosed as other liver tumors. Accurate diagnosis is challenging but crucial, with histopathology and immunohistochemistry being the gold standards. There is a lack of consensus on treatment strategies and the difficulty of prognostic assessment, which highlights the importance of long-term monitoring and follow-up. Given HEAML’s uncertain malignant potential and aggressive behavior, operational resection is recommended when clinical symptoms and imaging suggest disease progression or tumor diameter ⩾5 cm, even if histopathology remains inconclusive. This case report underscores the diagnostic and therapeutic challenges of HEAML, highlighting the importance of the MDT approach in its management.
Footnotes
Acknowledgements
We are thankful to the patient and all the physicians and technicians who participated in this case.
Author contributions
D.T., Y.X., Q.M., and Q.Y.: concept and design; D.T., Y.X., Q.M., and Q.Y.: acquisition, analysis, or interpretation of data; D.T. and Y.X.: drafting of the manuscript; D.T., Y.X., and Q.Y.: critical review of the manuscript for important intellectual content; Q.Y.: supervision.
Availability of data and materials
The data support that the current study is available from the contact author upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Youth Natural Science Foundation of Anhui Medical University (NO. 2021xkj084) and Health Research Project of Anhui Province (NO. AHWJ2022b120)
Ethics approval
This case report was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Ethics Committee of the Affiliated Chuzhou Hospital of Anhui Medical University (Chuzhou, China). The specimen were obtained from the Affiliated Chuzhou Hospital of Anhui Medical University (Chuzhou, China; approval code: reference 2024 Ethical Review [Bio] no.31) and written informed consent was obtained from the patient in this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Consent for publication
All authors contributed to the design and interpretation of the study and to further drafts and approved the final version to be published. Qingsong Yang is the guarantor.
