Abstract
We herein described a case of a 45-year-old male recently detected with HIV and Hepatitis C who presented with signs of sepsis, developed delirium, and received haloperidol. Surprisingly, 10 min after intravenous administration, the patient developed Torsades de pointes and required immediate resuscitation. The report discusses the limited instances of cardiac arrest linked to haloperidol use, despite its generally perceived safety. The patient had no apparent risk factors, emphasizing the need for increased caution when administering haloperidol, particularly in critically ill patients with HIV and Hepatitis C.
Introduction
Haloperidol is a typical antipsychotic drug. A multinational inception-cohort study published in 2018 showed that it continues to be the most frequently used agent to treat delirium in Intensive care Unit (ICU) patients regardless of delirium subtype, followed by benzodiazepines and dexmedetomidine. 1 Most psychotropic medications are well tolerated with minimal side effects. However, haloperidol has been associated with prolonged QT interval leading to lethal ventricular arrhythmias, such as Torsades de pointes (TdP), especially in medically ill patients. 2 The severe repercussions of the treatment are most probably attributed to the primary inhibition of cardiac potassium channels. 3
Very few reports of a cardiopulmonary arrest related to the use of haloperidol in the literature have been published. We, herein this report, describe a case of TdP after the use of a single dose of haloperidol in a patient with HIV and Hepatitis C.
Case report
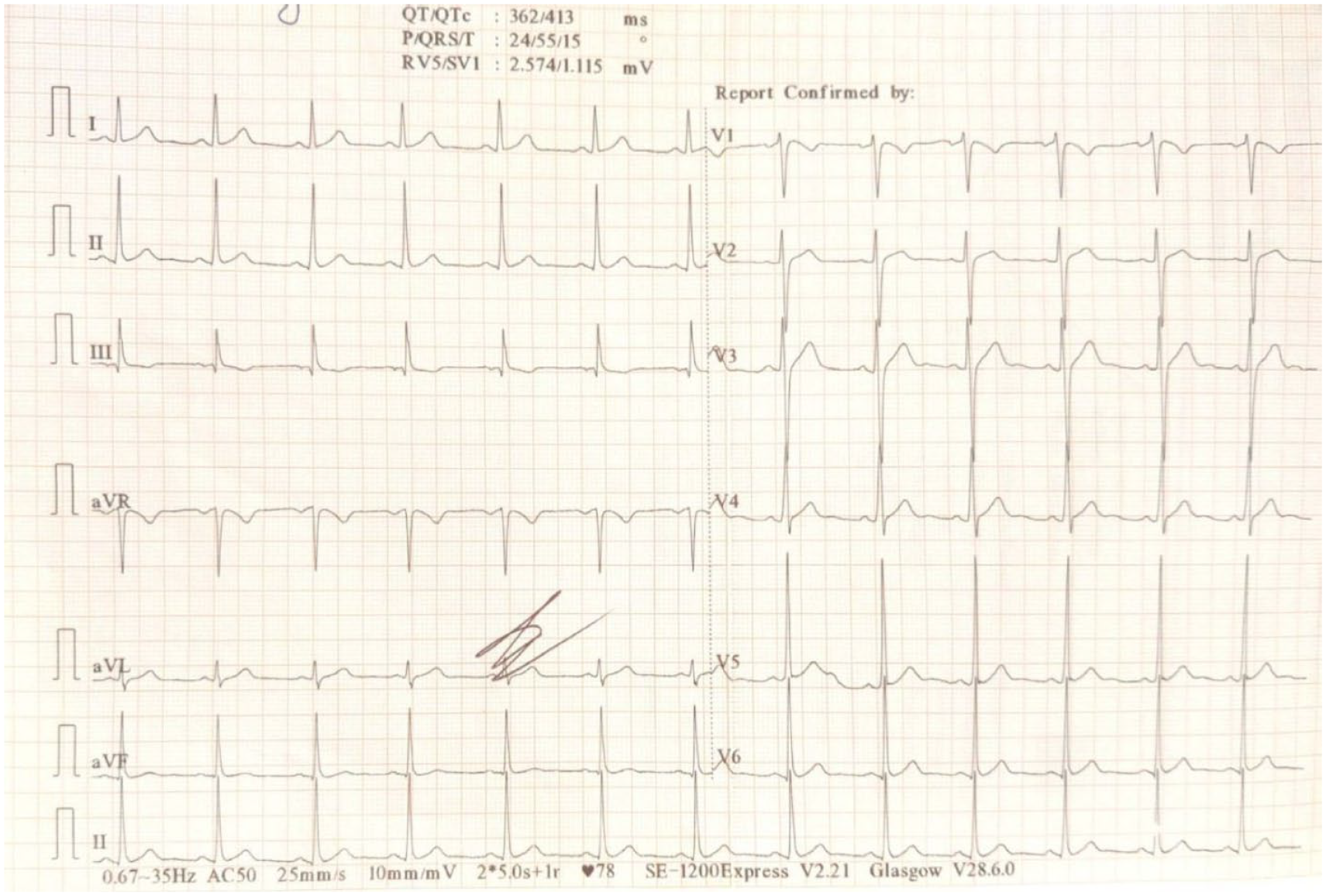
A 45-year-old male with newly detected HIV and Hepatitis C, presented to us after being referred with a diagnosis of pneumonia with severe sepsis concomitant acute kidney injury. Upon arrival, the patient presented with tachycardia, with a heart rate of 110 beats per minute, and was hypoxic, as indicated by a blood oxygen saturation level (SpO2) of 88% while breathing room air. Other vitals were stable with a Glasgow Coma Scale score of 15/15 (Eye-opening E4. Verbal V5, and Motor M6).4,5 The patient was oxygenated with a nasal cannula at 2 liters per minute. SpO2 rose to 96%, and subsequently, the patient reported a notable increase in overall comfort and well-being. All the relevant investigations were sent. The patient’s treatment regimen during admission included the following intravenous medications: (i) Piperacillin and Tazobactam (2.25 gm IV TDS), (ii) Acetaminophen, (iii) pantoprazole, (iv) fentanyl (25 µg single dose), and (v) Ringer lactate. The patient had no known comorbid conditions such as hypertension, coronary artery disease, or psychiatric illnesses. However, the patient did consume alcohol, cigarettes, and marijuana occasionally. There was no history of intravenous drug abuse or blood transfusion. Antivirals were not started because confirmatory tests were due. Arterial blood gas (ABG) analysis, chest X-ray, blood investigations, culture, and procalcitonin were normal. Toxicology screening results were negative. The patient’s serum electrolyte levels were also normal, with potassium 4 mmol/L, magnesium 1.9 mg/dL, and calcium 9 mg/dL. Liver enzymes showed ALT 50 U/L, AST 35 U/L, ALP 134 U/L, total serum albumin 3.6 g/dl and total bilirubin 0.8 mg/dl. HIV viral load was 52,000 copies/ml, and CD4 level was 350 cells/mm3. The baseline electrocardiogram (ECG) (Image 1) obtained from the referring hospital was normal (HR: 78 bpm, QTc interval: 413 ms). No structural abnormalities were noted on echocardiography.

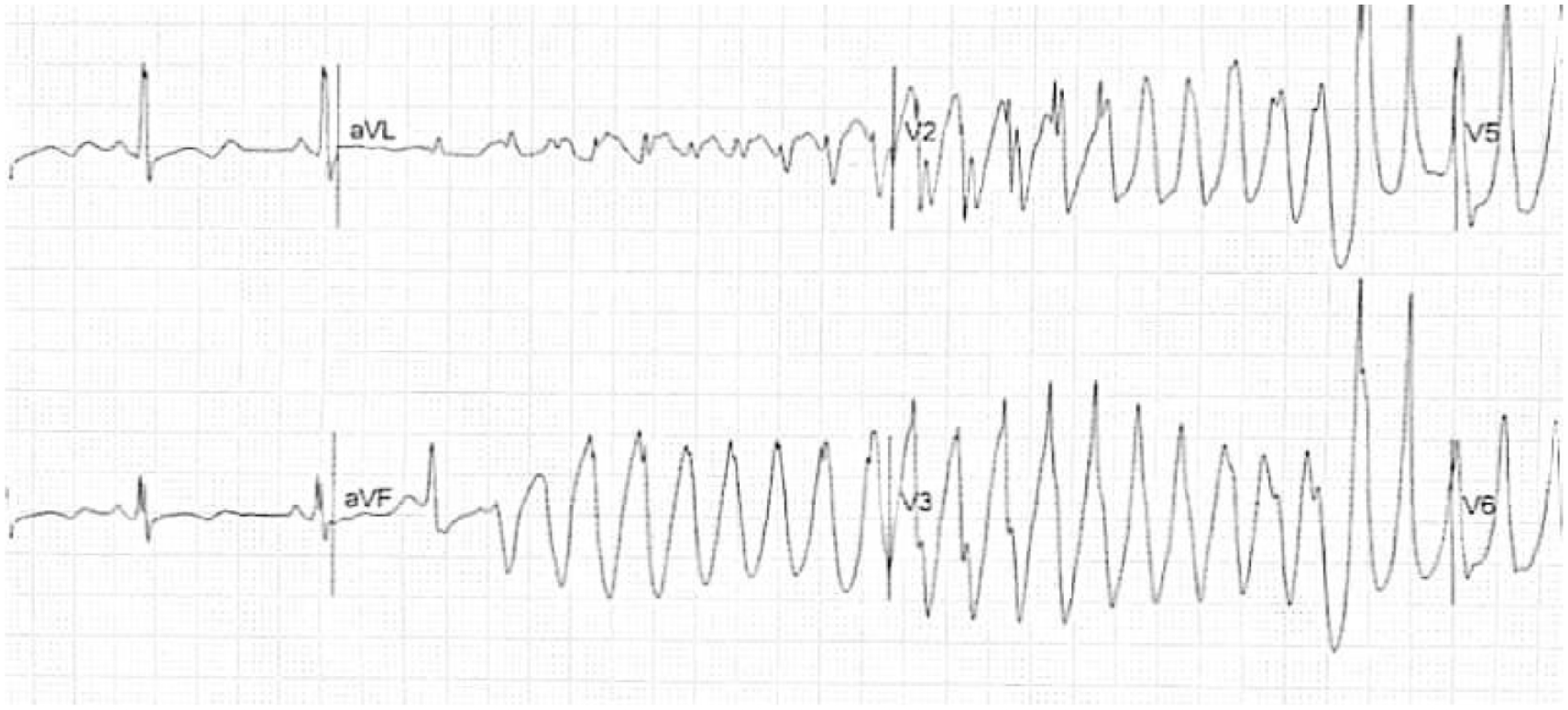
ECG showing Torsades de pointes.
The patient was clinically and hemodynamically stable on the first and second day of ICU admission with normal lab values. On the third day of admission, he suddenly presented with abnormal behaviour, which involved pulling his intravenous cannula out and shouting violently. He had normal vitals and normal ABG. The serum electrolytes measured potassium at 4.8 mmol/L, magnesium at 1.8 mg/dl, and calcium at 8.5 mg/dl. Then, after injection, Haloperidol 2.5 mg was given intravenously. The patient stopped exhibiting abnormal behaviour and became calm. Ten minutes later, he suddenly developed TdP (QTc 650: ms), as shown in (Image 2).
Baseline ECG showing normal sinus rhythm.
Cardiopulmonary resuscitation (CPR) was started as per Advanced Cardiovascular Life Support (ACLS) guidelines. Unsynchronized shock was given three times at 200 Joules, and adrenaline 1 mg was given two times. The patient was intubated and kept on the ventilator after the return of spontaneous circulation. ECG obtained after CPR is shown in (Image 3) (HR: 83 bpm, QTc: 438 ms).
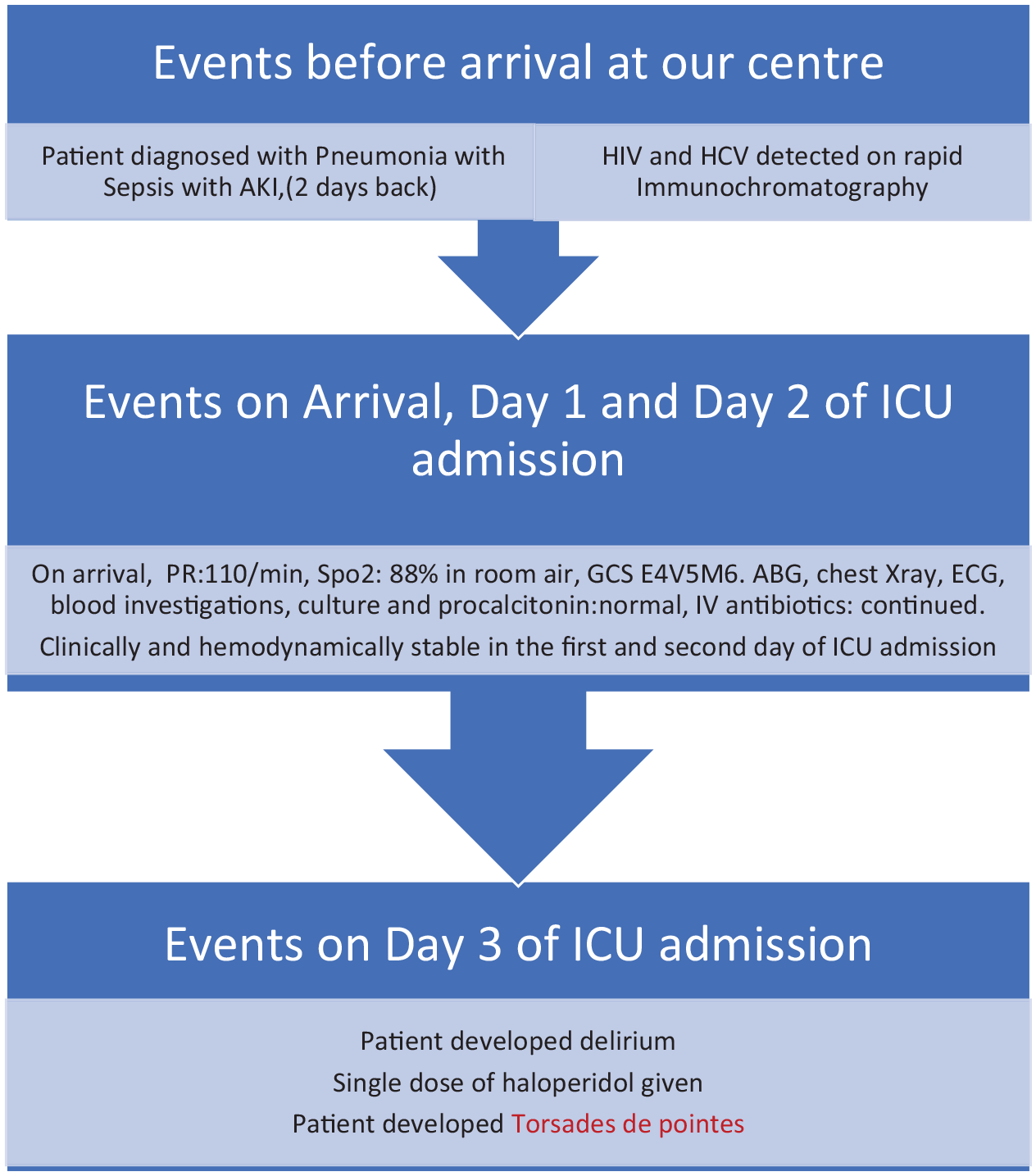
Timeline showing major clinical events in different time frames.
Magnesium 2 gm was given slowly via IV push. The reversible causes for the cardiac arrest, including hypovolemia, hypoxia, hypothermia, hypo/hyperkalemia, cardiac tamponade, exposure to toxins, tension pneumothorax, and thrombosis, were diligently assessed and managed. ABG was done, which showed elevated lactate levels, and metabolic acidosis, and the patient was treated accordingly. Magnesium infusion was started at 2 gm/h. Nor adrenaline was started via continuous intravenous infusion for hypotension. Central venous catheterization and an arterial line were kept. Serial electrolyte monitoring was done with ABG. The cardiology department was informed regarding the case. Eight hours later, the patient went into asystole again, and the patient could not be revived despite resuscitation. The postmortem evaluation was not performed due to lack of consent. A timeline showing major clinical events in different time frames is shown in Figure 1.
ECG showing sinus rhythm after CPR.
Discussion
We herein described a case of a 45-year-old male recently detected with HIV and Hepatitis C who presented with signs of sepsis, developed delirium, and received haloperidol. The patient’s delirium may be linked to their ICU stay, which can disrupt sleep patterns and contribute to cognitive impairment. Additionally, hypoxia and alcohol deprivation could have also played a role in the development of delirium. The patient developed cardiac arrest after 10 min of administration of intravenous haloperidol. We suspect the cause of cardiac arrest to be IV haloperidol given nonsignificant risk factors for other causes of cardiac arrest. Reversible factors were sought but were not found; thus, we labelled the cause of arrest to be the administration of haloperidol. Some published reports support the cause that haloperidol could be behind these instances of arrest.6–9 Dar et al. 8 reported a similar case as ours involving a patient who had experienced a road traffic accident, undergone multiple surgeries, and had an extended ICU stay received 2.5 mg of intravenous haloperidol and subsequently developed cardiac arrest. The authors concluded the patient’s preexisting incident to be the cause for worsening the situation, leading to fatal cardiovascular arrhythmia. In a study by Mehtonen et al. 10 in Finland, antipsychotic or antidepressant drugs were involved in 49 sudden deaths. Of these, 28 cases involved thioridazine 15, perphenazine 12, levopromazine 10, and chlorpromazine 10, while only 6 cases were linked with haloperidol. Notably, none of such death cases occurred from solitary haloperidol use. Conversely, 15 thioridazine cases, along with 1 perphenazine case, 1 levopromazine case, and 2 chlorpromazine cases, did involve single-drug usage.
Meyer et al. 11 reviewed the Food and Drug Administration (FDA)’s guidance on continuous ECG monitoring for intravenous haloperidol use where 70 cases of IV haloperidol- related QT Prolongation (QTP) and/or Torsades de pointes (TdP) were identified. There were 54 TdP cases of which 42 were preceded by QTP. Post-event data showed QTc was prolonged in 96% of cases, and 97% had additional TdP risk factors. Based on their finding they concluded <2 mg cumulative IV haloperidol could be safely administered without constant ECG monitoring in cases without additional risks.
Lonergan et al. 12 compared haloperidol with other drugs like risperidone, olanzapine, and placebo for delirium management. A higher dose of haloperidol, more than 5 mg, was found to be associated with significant extrapyramidal side effects, while low dose had negligible side effects as other drugs. Metzger et al. 9 reported three cases that developed TdP or QT prolongation with intravenous haloperidol use. Dilated cardiomyopathy and alcohol abuse could have been the associated risk factors, and cardiac monitoring is required for safety in such cases.
Haloperidol is recognized for blocking the rapidly activating delayed-rectifier potassium current, which plays a significant role in cardiac repolarization. As a result, individuals taking haloperidol may experience mild to prominent QT prolongation, although in some cases, the QT interval may remain within normal limits. Likewise, ventricular arrhythmias associated with haloperidol use are mainly TdP. 13 Hypokalemia, old age, female gender, drug interactions, genetics, low magnesium, heart failure, and bradycardia are potential risk factors found to be involved with drug-induced TdP. 14 Leonard et al. 15 conducted a study in 459,614 incident antipsychotic users where they found the incidences of sudden death/ventricular arrhythmia and death were 3.4 and 35.1 per 1000 person-years, respectively. Haloperidol and chlorpromazine demonstrated a less desirable cardiac record than olanzapine, while risperidone exhibited a similar profile. Notably, quetiapine is associated with a lower risk when compared to olanzapine.
Patients who are HIV positive and have high viral loads, low CD4 counts, chronic inflammation, and autonomic neuropathy may be prone to experiencing QT interval prolongation. Exposure to the HIV transactivator protein is another factor that can extend the QT interval. 16 It is important to highlight that both HIV and Hepatitis C infections can individually raise the risk of QTc interval prolongation. According to a study, individuals coinfected with both Hepatitis C and HIV had an almost twofold increase in the likelihood of experiencing QTc interval prolongation compared to those with either infection alone (29.6% vs 15.8%, p < 0.001). This indicates that having both infections concurrently may significantly heighten the risk of QTc interval prolongation. 17 Long QT syndrome (LQTS) is a rare inherited arrhythmia disorder that can lead to life-threatening TdP and is also a leading cause of sudden cardiac death. KCNQ1, SCN5A, and KCNH2 are the genes most associated with LQTS. 18
In our case, despite having no additional apparent risk factors except for HIV and Hepatitis C positive status, the patient developed cardiac arrest. Thus, greater precautions need to be considered due to the risk of pulseless ventricular tachycardia, prolongation of QT, and cardiac arrest. Vigilant cardiac monitoring with serial ECG and ABG must be done in patients receiving more than 2 mg IV cumulative dose of haloperidol with concomitant risk factors 11 and any abnormal findings should be sought out immediately. Serum electrolytes, potassium, and magnesium should be checked at regular intervals. Indeed, avoiding all QT-prolonging medications in conjunction with haloperidol for critically ill patients is an exaggeration. Cautious discretion is crucial, but a comprehensive assessment of potential benefits and risks is essential. Tailored patient evaluations and meticulous monitoring are integral elements of safe clinical care. Additional, comprehensive studies are necessary to enhance our understanding of the underlying mechanisms responsible for prolonged QTc intervals in HIV-positive patients.
Conclusion
Unexpectedly, cardiac arrest was preceded by the administration of haloperidol, a drug generally regarded as safe for the management of delirium. Although haloperidol is typically well tolerated, there are several situations where its potential arrhythmogenic effects should be taken into account. This event highlights the need for more caution, especially around HIV and Hepatitis C-infected patients who are critically ill. Additional comprehensive studies are necessary to enhance our understanding of the underlying mechanisms responsible for prolonged QTc intervals in such patients.
Footnotes
Acknowledgements
We would like to thank all the nursing staff and supporting staff in our infectious and tropical disease hospital ICU who contributed to the management and provided compassionate care for this patient.
Author contribution
K.T. and S.K. were responsible for designing the study and developing the conceptual framework. They also managed the cases and prepared the article. B.K.C. and S.T. played a crucial role in data collection and were actively involved in patient management. Finally, all authors participated in the review process and gave their approval for the final article.
Data availability
All the data are presented in the article itself.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical clearance for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the legally authorized representative of the deceased subject for the anonymized patient information to be published in this article.
