Abstract
Demons syndrome is defined by hydrothorax and ascites associated with a benign genital tumor that resolves after resection of the tumor. However, Demons syndrome with pericardial effusion has never been reported. Intensive care unit–acquired weakness is a neurological sequela to sepsis/systemic inflammatory response syndrome, or multi-organ failure. A 47-year-old, nulligravid, Japanese woman, was transferred to our hospital for refractory heart failure and a ruptured ovarian tumor. She had an 11-cm left ovarian tumor with ascites, hydrothorax, and pericardial effusion; she was intubated for pulmonary hypertension and admitted to the intensive care unit for septic shock. Four days later, a left salpingo-oophorectomy was performed for Demons syndrome with pericardial effusion. The histological diagnosis indicated a serous cystadenoma with fibrotic changes. Following surgery, ventilator weaning was delayed due to intensive care unit–acquired weakness. The association between Demons syndrome and pericardial effusion should be recognized to ensure early treatment and for preventing sequalae from the disease.
Keywords
Introduction
Demons syndrome (DS) was first reported as the association of a benign genital tumor with hydrothorax and ascites, which resolved after tumor removal. 1 Later, Meigs 2 described fibroma-like tumors (fibroma, thecoma, granulosa cell tumor, or Brenner tumor) in DS as Meigs syndrome (MS) (also known as Demons-Meigs syndrome); cases with other tumors in non-genital locations, such as a pancreatic location, were termed pseudo-MS. Recently, MS with pericardial effusion that disappeared after tumor removal has been termed Meigs-like syndrome. 3 However, DS other than fibroma-like tumors with pericardial effusion has never been reported.
Intensive care unit (ICU)-acquired weakness is neuromuscular and occurs in patients recovering from critical illnesses such as sepsis, systemic inflammatory response syndrome (SIRS), and multiorgan failure. 4 It manifests clinically as difficulty in withdrawing ventilator support, and flaccid generalized weakness in ICU patients. Although a variety of primary diseases can cause critical illness, data regarding gynecological diseases and ICU-acquired weakness are lacking. 5
We report a case of DS with pericardial effusion, followed by ICU-acquired weakness due to sepsis and multi-organ failure, secondary to a ruptured ovarian tumor.
Case
A 47-year-old Japanese nulligravid woman was transferred to our hospital for heart failure and a ruptured ovarian tumor. She had a history of myomectomy for fibroids and right salpingo-oophorectomy for an ovarian cyst. Two weeks previously, she was admitted to a secondary-care hospital due to dyspnea and generalized edema persisting for 4 months. She was diagnosed with chronic heart failure associated with a benign ovarian tumor, based on echocardiography and computed tomography (CT); her symptoms remained unchanged despite diuretic (furosemide 40 mg once daily) and vasodepressor (enalapril maleate 2.5 mg once daily) administration. Three days prior to referral, the tumor capsule had ruptured spontaneously.
In the emergency room, she presented with shock-related vital signs: tachycardia (117 beats/min), hypotension (86/67 mmHg), tachypnea (23 breaths/min) despite administration of 2 L/min oxygen (SpO2 97%), and a body temperature of 36.6°C. Arterial blood gas analysis showed respiratory acidosis (pH 7.274, PCO2 74.7 mmHg, HCO3 33.5 mmol/L); chest radiography demonstrated an increased cardiothoracic ratio (72%) and absent bilateral costophrenic angles. Blood tests showed elevated white cell counts (22,590 per mm3), C-reactive protein (27.61 mg/dL), and procalcitonin (3.69 ng/mL), with anemia (hemoglobin: 9.6 g/dL). N-terminal pro-brain natriuretic peptide was elevated, at 7074 pg/mL; levels of creatinine, urea nitrogen, and carbohydrate antigen 125 were 1.14 mg/dL, 31.7 mg/dL, and 830.8 U/mL, respectively. Contrast-enhanced CT revealed massive pericardial effusion and hydrothorax (Figure 1(a)). Echocardiography demonstrated a D-shaped left ventricle with a 50% ejection fraction; right heart catheterization showed pulmonary hypertension with mean pulmonary artery and pulmonary capillary wedge pressures of 32 and 12 mmHg, respectively. We suspected DS with pericardial effusion associated with pulmonary arterial hypertension. She was placed on ventilator support, underwent pericardial drainage catheter placement and was supported with continuous renal replacement therapy, prophylactic antibiotics (Meropenem 1 g IV q8 h), adrenergic reagents, and blood transfusions; blood cultures isolated
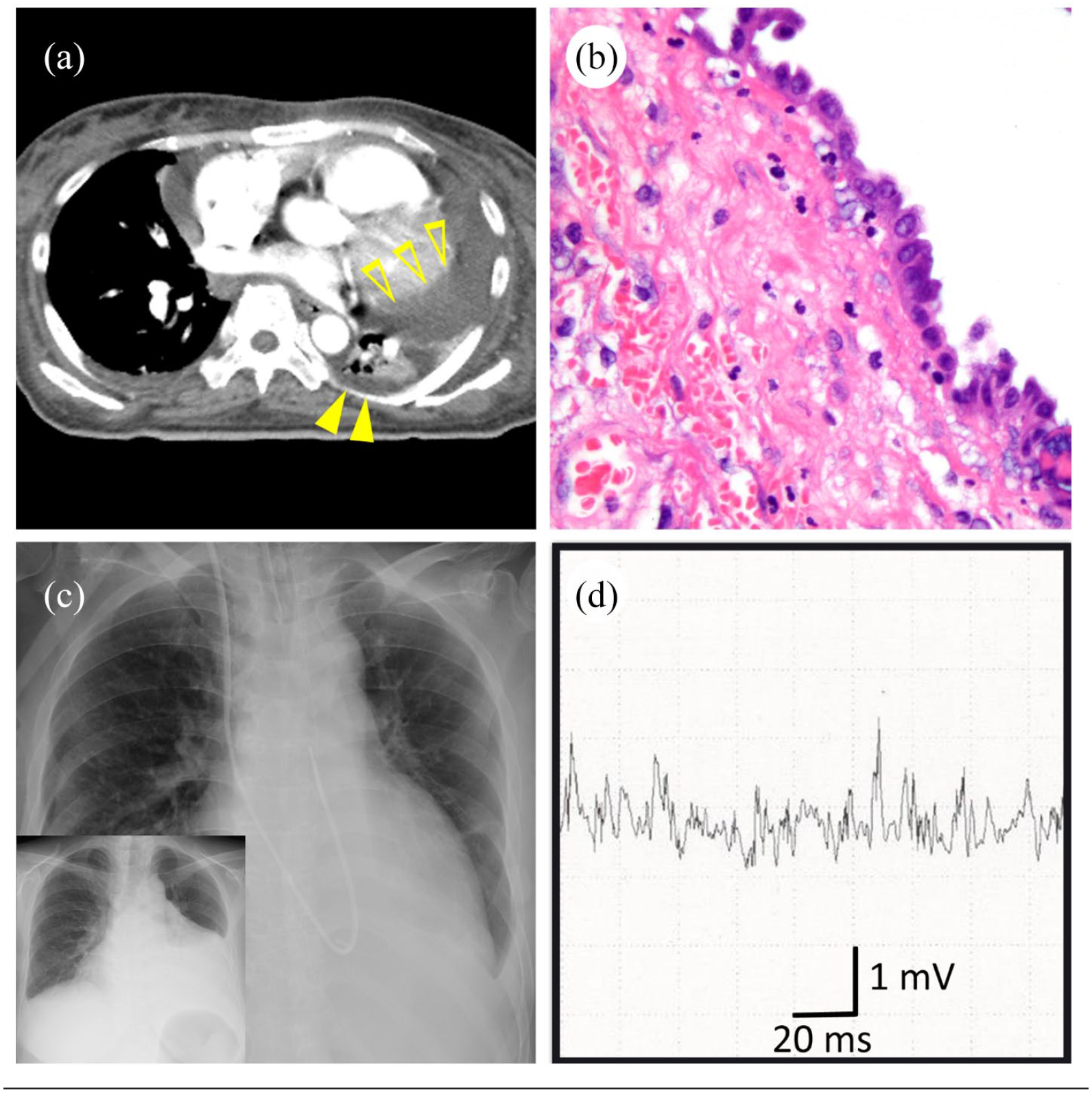
(a) Chest computed tomography (CT) showing massive pericardial effusion (closed arrows) and pleural effusion (open arrow). (b) Microscopic findings showing a single layer ciliated columnar epithelium and fibrous thickening. Hematoxylin and eosin stain. Original magnification ×40. (c) Chest radiograph showing resolution of pericardial effusion after the definitive surgery. Insert: pre-operative chest radiograph. (d) Needle electromyography of the biceps showing low motor unit potentials and normal interference pattern.
Chest radiography on postoperative day (POD) 1 showed no pathological findings (Figure 1(c)). She was weaned from ventilatory support on POD 2, needing reintubation for refractory hypercapnia on POD 6. She had difficulty breathing without assistance, requiring prolonged intubation and subsequent tracheostomy on POD 11. Further neurological investigations were performed for respiratory muscle weakness, and rehabilitation was recommended. Motor nerve conduction studies showed low amplitudes of compound muscle action potential, with normal motor conduction velocity and F-waves. Sensory nerve conduction studies in the distal median and ulnar nerves showed normal latency, amplitudes, and conduction velocity. Normal patellar and Achilles’ tendon reflexes with or without strong contraction enabled differentiation from Lambert-Eaton myasthenic syndrome. Needle electromyography showed low and short motor unit potentials with normal interference and early recruitment (Figure 1(d)). Based on all findings, she was diagnosed with ICU-acquired weakness. Respiratory muscle weakness gradually improved; ventilator support was withdrawn on POD 36, and a speaking valve was placed. She was transferred to a rehabilitation-specialty hospital, and discharged on POD 135.
Written informed consent was obtained from the patient and her sister for publication of anonymized data in this article.
Discussion
This case demonstrates two important clinical issues. First, DS may be complicated by pericardial effusion in addition to ascites and hydrothorax. Second, severe DS with pericardial effusion may prolong ICU stay, resulting in ICU-acquired weakness.
We conducted a literature review regarding DS and MS in PubMed/MEDLINE and Google Scholar. We used the terms “Demons syndrome,” “Meigs syndrome,” and “pericardial effusion” to search for literature in English, without publication date filters. The proposed schematic diagram representing overlap and distinction between these syndromes is shown in Figure 2. Demons was the first to report a paraneoplastic syndrome resulting from benign genital tumors in 1887. Similar syndromes associated with uterine fibroids were later reported and designated as pseudo-DS. 1 In 1957, Meigs 2 defined a “true” MS, as fibroma-like tumors inducing hydrothorax, pericardial effusion, and ascites; Meigs defined similar syndromes and labeled them “pseudo”-MS. DS overlaps with MS among patients with benign ovarian fibroma-like tumors, resulting in confusion regarding the terminology in literature.

A schematic representation of Demons and Meigs syndromes.
DS involving types other than fibroma-like tumors with pericardial effusion, which resolves following tumor resection, has not been reported. However, MS with pericardial effusion has been reported by Qaisar et al. 6 and was subsequently termed Meigs-like syndrome. Similarly, we termed DS with pericardial effusion as “Demons-like syndrome.” Table 1 summarizes four cases of DS with pericardial effusion in English scientific literature, with histopathologic diagnoses. The pathophysiology of fluid collection in DS with pericardial effusion remains unknown and unresolved. Jayasree et al. 7 found a uterine fibroid with degenerative changes, while we observed a serous cystadenoma with fibrotic changes. The fact that a fibrotic component is common among these four cases supports Meigs’ 8 theory that fluid collection may be caused by leakage or pressure on the surface lymphatic vessels.
Reported cases of Demons syndrome with pericardial effusion in literature.

ICU: intensive care unit; NA: not available; SO: salpingo-oophorectomy; TAH: total abdominal hysterectomy.
ICU-acquired weakness includes both, background risk factors including underlying disease severity, ICU-stay duration, and mechanical ventilation, and treatment-related risk factors including parenteral nutrition, corticosteroids, and aminoglycoside use.
9
These necessitated inevitable ICU care, and subsequent mechanical ventilation. DS with pericardial effusion usually recovers without any sequelae following tumor removal. In the present case, ovarian tumor rupture prior to its excision resulted in reactive chemical peritonitis. Investigations for pericardial effusion and risk-reducing pericardial drainage for general anesthesia delayed surgery. We speculate that these resulted in septic shock owing to disseminated
Conclusion
We report the first case of DS with pericardial effusion, followed by ICU-acquired weakness due to SIRS/sepsis caused by a ruptured ovarian tumor. Recognition of this condition may facilitate early diagnosis and therapy. Further studies are warranted, as the pathophysiology of DS with pericardial effusion is largely unknown.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient and her sister for their anonymized information to be published in this article.
