Abstract
Lumbo-pelvic pain is a common and non-specific problem during pregnancy and postpartum. Although perinatal pyogenic sacroiliitis is uncommon during this time, it might be difficult to distinguish from reactive sacroiliitis in women who are experiencing significant lower back and pelvic girdle pain, as the symptoms and signs are not clear and the tests are not definitive. A 34-year-old primigravida went to the emergency department with severe lower back pain radiating to the right gluteal region and down to the back of the right thigh. This pain began 12 days prior to her presentation and eventually worsened to the point that she could not stand or walk. Her vital signs were within normal ranges, and she was experiencing a fever. Apart from a slight widening of the symphysis pubis, her pelvic and lumbo-sacral pain X-rays revealed no important findings. With the clinical impression of right lumbo-pelvic pain, the patient was admitted for pain management and further inquiries. Despite the fact that the antibiotherapy was prescribed to treat a urinary tract infection, the significant recovery of the patient’s symptoms, even in the absence of a definitive culture of aspirate from the right sacroiliac joint, supported the diagnosis of pyogenic sacroiliitis.
Introduction
The pelvis and sacroiliac joints (SIJs) undergo significant alterations during pregnancy, which have a deleterious impact on dynamic stability and may cause pelvic girdle and lower back pain.1,2 While pregnancy-related pyogenic sacroiliitis (PSI) is uncommon, with fewer than 20 cases reported in the literature, 3 it is tough to diagnose because it necessitates a high level of clinical suspicion and diagnostic imaging confirmation. 4 Medical care should not be postponed because it risks not only joint and bone damage but also septicemia in both the mother and the child. 5
Case presentation
A grossly obese 26-year-old lady with a body mass index (BMI) of 43, gravida-1, para-1 + 0, type 2 diabetes mellitus, and hypothyroidism. She came to the causality with 12 days history of severe continuous right-sided lower back pain, right side pelvic pain increased with ambulation. Her pain started in a mild way prior to her recent admission for elective cesarean section for twin babies, using spinal epidural anesthesia, got worse and began to radiate to the back of the right thigh, increased with ambulation. There was no history of trauma, fever, chills, urinary, gynecological, or other systemic symptoms. Her physical examination revealed a temperature of 37.1°C, severe tenderness over the lower back, and right side of the pelvic region with intact neurology. Her cesarean section wound in the abdomen has healed. Her lower back and pelvis were stiff with severe pain on movements, standing, or sitting. The straight leg raising test was positive (30–40°), and the FABER Patrick’s test was difficult to evaluate due to intense pain despite the strong analgesia.
The lumbo-sacral spine X-ray was unremarkable, but the pelvis X-ray revealed a 12 mm widening of the symphysis pubis (Figure 1).
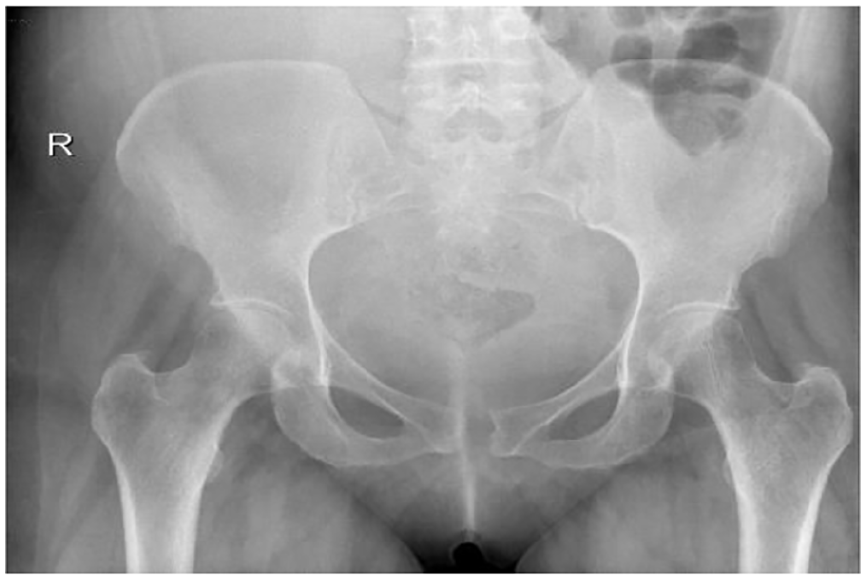
AP plain radiographic of the pelvis and sacroiliac joints revealed minor widening of the symphysis pubis and no other abnormalities.
She was hospitalized for further examinations and pain control after a clinical diagnosis of perinatal right-sided lumbo-pelvic pain (LPP). Laboratory testing found 6.85 white blood cells (WCC) G/l with 69.2% neutrophils, a 48 mm/h elevated erythrocyte sedimentation rate (ESR) (Figure 2), normal procalcitonin (0.02), and a 68.7 mg/L elevated C- reactive protein (Figure 3).

ESR mm/h.

CRP mg/L.
The lumbar spine magnetic resonance imaging (MRI) was negative, but the pelvic MRI found minor fluid signal amplitude in the right SIJ, which was associated with myositis (Figure 4).

Pelvic MRI on admission showing bone marrow edema (arrowheads), small intraarticular fluid (arrow) and muscle edema-like change, myositis (asterisk).
The patient was accused of developing unilateral sacroiliac arthritis and piriformis muscle syndrome. In the context of pain modality and incremental mobilization, we started her treatment with narcotic and non-narcotic analgesics, as well as non-steroidal anti-inflammatory medicines and physiotherapy. Her pain could not be controlled, and she became worse, unable to ambulate or step in or out of bed. Neurosurgery, neurology, rheumatology, anesthesia, obstetrics, and gynecology teams were consulted, but nothing was added to the preliminary diagnosis or the treatment plan. Despite the absence of any symptoms or signs indicating an acute or chronic infection, urine and blood cultures, protein purified derivative (PPD) for tuberculosis, and serology for brucella were required on Day 8. Because it was impossible to place the patient in the prone position on the same day, CT-guided aspiration of the right SIJ was performed in the left lateral position (Figure 5), but no aspirated fluid was obtained; however, SIJ block was performed using 20 mL of 0.25% bupivacaine after which she briefly noticed some relief of her shooting pain.

CT-guided right SI joint needle aspiration in the left lateral position.
Although the blood culture, brucella serology, and PPD all came back negative, the urine culture revealed asymptomatic
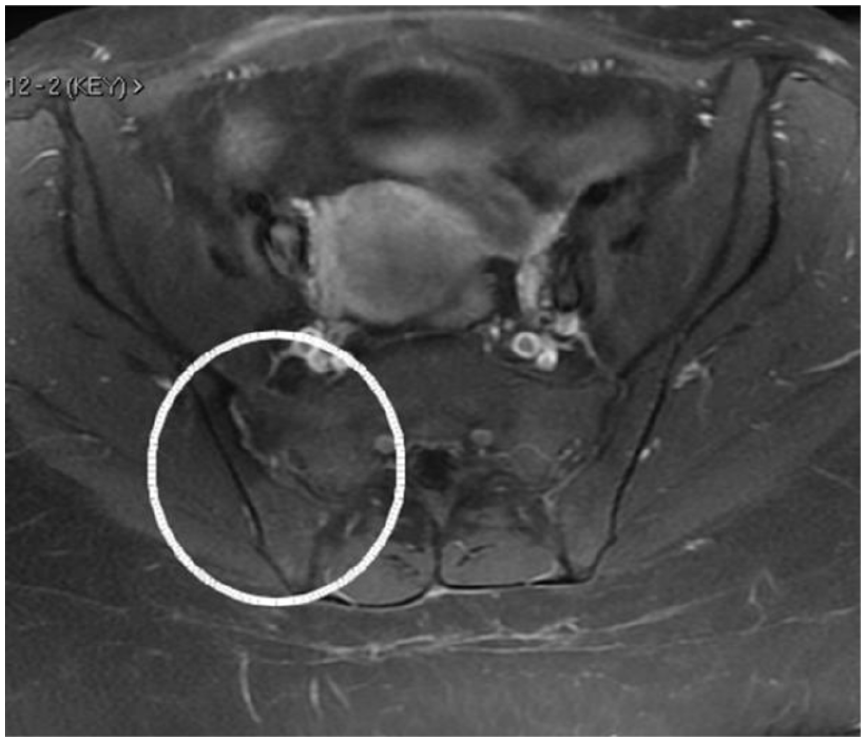
A pelvic MRI a year later revealed normal bone marrow signal and normal surrounding muscle. There are no fluids within the joints.
Discussion
Pain in the lower back and buttocks is widespread and vague in 20% of pregnant women, making SIJ diseases difficult to diagnose. 6 Despite the fact that PSI accounts for just 1%–1.5% of all instances of septic arthritis, it is estimated that 10% or more of these infections occur in women during pregnancy, postpartum, or following an abortion.7,8
In PSI, the SIJ is always involved unilaterally, with the left side predominating in 59% of cases. On admission, the diagnosis is infrequently suspected (12.85%), and the clinical picture can be misunderstood as sciatica or spondylodiscitis. This unique clinical presentation looked to be the reason for the long period between diagnosis and treatment. 9 The pelvic ligaments loosen with increasing pelvic motions as a result of increased weight and hormone-induced alterations in the pelvis, which can influence the microvasculature of joint surfaces, rendering the periosteum more susceptible to transient bacteremia and bacterial invasion.10–12 Furthermore, the venous plexus system, which drains the paravertebral and pelvic areas and the subchondral circulation in the ilium have a sluggish blood flow, which is thought to increase the risk of blood-borne bacteria forming a host site in the SIJs.13,14
PSI can be diagnosed if bacteriological confirmation of sacroiliitis is achieved, or if clinical, biological, and radiological evidence are consistent with this diagnosis in the absence of pathogenic agents, and if the patient’s evolution was favorable on antibiotic therapy, as in our case. 9 Despite the fact that urinary tract infection is one of the most common causes of reactive arthritis and may be a risk factor for infectious sacroiliitis (ISI), yet it was extremely difficult to distinguish between reactive and infectious arthritis in our case. 15
The explosive onset of the disease is seen in two-thirds of patients and typically includes high- or low-grade fever and severe continuous pain, originating from the affected SIJ and felt most often in the buttock, low back, and/or hip area. 16 Physical examinations and particular tests, such as pain provocation tests, P4/thigh thrust, Patrick’s FABER, and the active straight leg raise (ASLR) test, are suggested but not definitive. 17
Due to their non-specific nature, laboratory testing initially made diagnosis more challenging. Although the amount of C-reactive protein (CRP) and ESR may be relatively sensitive indices of PSI, the leukocyte count has not been found to be a sensitive marker of PSI, and one-third of patients have a normal WCC.3,8,9 Only one-third to two-thirds of patients have positive blood cultures.
18
Gram-positive cocci, primarily
At the time of presentation, a plain pelvic roentgenogram is usually normal.1,8 Because considerable bone loss is required before changes can be noticed on plain films, the earliest changes on plain films include blurring of joint borders, an increased joint space, or periarticular erosion, which emerge 2 weeks after onset of symptoms.
7
As a result, false negative radiographs are usual in acute cases, and caution should be exercised when ruling out a recent infection based on initially normal-looking photos.21–23 In the perinatal phase, MRI is most likely the imaging diagnostic tool of choice for detecting sacroiliitis. It provides a detailed evaluation of the joint and surrounding soft tissues, demonstrating prominent bone marrow edema adjacent to the SIJ surfaces, synovitis of SIJ itself, and edema in the neighborhood soft tissues, which may help to distinguish mimickers of spondyloarthritis, such as crystal arthropathies, reactive arthritis, insufficiency fractures, septic arthritis, IgG4-related disease, sarcoidosis, hematologic conditions, and neoplastic disorders.24–26 PSI might be detected when low signal intensity on T1 and high signal intensity on T2 were observed on orientated MRI slices.
9
This test also allows physicians to see whether the infection has spread to nearby muscle tissues, which was seen in 48.1% of cases.
27
Because MRI signal anomalies can last for months, even though there is no fever or clinical and biological progress appears to be on the way, it should be done in a systematic manner.
28
The most sensitive imaging modality for infection is 99mTc radionuclide scanning. Increased radionuclide uptake in the sacroiliac region can happen as early as 2 to 7 days after the illness begins.6,22 As a result, a positive bone scan will help to avoid delays in diagnosis and care. Although highly sensitive for infection, the specificity of bone scanning is low. A Technetium scan should be accompanied by a Gallium-67 citrate (67Ga) scan in cases of suspected sacroiliac infection. This radiopharmaceutical is useful for detecting infections because it has a preference for polymorphonuclear leukocytes (PMNs).
27
Although radionuclide scans can be useful for monitoring post-delivery care, they should not be used during pregnancy.29–31 CT can reliably direct joint aspiration of joint fluid in cases of suspected PSI. If an aspirate is not obtained, flushing the joint with normal saline and aspirating the saline will boost the chances of a positive culture. A para-articular bone biopsy may be used to confirm or rule out infection if SIJ fluid cannot be collected despite saline flushing, as in tuberculous sacroiliitis. Pigtail insertion into PSI abscesses can also be guided by CT.
32
The treatment for pregnancy-related PSI is similar to that for non-pregnancy instances.
4
Delaying medical intervention can result in joint and bone damage, as well as maternal and newborn septicemia.
5
Although there is no unanimity on how long antibiotic treatment should last in PSI, it seems reasonable to propose 2 weeks of parenteral antibiotics followed by 6 weeks of oral antibiotics. Prolonging care beyond 6 weeks does not seem to be justified, as it does not reduce the likelihood of relapse.
33
In suspected PSI or in the absence of any known microorganism, empirical antibiotic treatment active against
Conclusion
Perinatal sacroiliitis is a difficult diagnosis to make since the symptoms and signs are non-specific, and investigations are inconclusive, thus delaying proper care. The extreme intensity of the patient’s lumbogluteal pain on presentation, which is increased by weight-bearing or some effort to displace the SIJ, may aid in distinguishing PSI from other musculoskeletal pain sources. Pain aversion to opioids and/or non-narcotic analgesics, a dramatic clinical reaction to parenteral antibiotics, and the existence of a defined source of infection are all additional diagnostic clues. In the absence of a fever or a positive blood culture, and standard biological parameters, such as ESR, CRP, and WCC, MRI is the most effective imaging technique for determining early and subsequent joint changes.
Footnotes
Acknowledgements
The authors thank Dr. rer. nat. Areeg Abdelrahman for editing a draft of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
