Abstract
Vasoplegic syndrome, a possible complication of cardiopulmonary bypass, is a critical state of unregulated systemic vasodilation with decreased vascular resistance and a pathological insensitivity to conventional inotropes and vasoconstrictors. This case demonstrates the use of methylene blue and hydroxocobalamin as medications in the treatment of refractory vasoplegic syndrome in the context of cardiac surgery due to their differences in mechanism of action. A 24-year-old female with history of intravenous drug abuse and hepatitis C infection underwent mitral valve repair for infective endocarditis. Preoperative transesophageal echocardiography showed normal right ventricular function, left ventricular ejection fraction of 65%–75%, and severe mitral regurgitation with vegetation. In order to maintain a mean arterial pressure over 60 mmHg during cardiopulmonary bypass, norepinephrine, epinephrine, and vasopressin infusions were required. Given the patient’s minimal response to these medications, a 1.5 mg/kg bolus of intravenous methylene blue was also given intraoperatively; vasoplegic syndrome remained refractory in the post-cardiopulmonary bypass period. A 5 g dose of intravenous hydroxocobalamin was administered in the intensive care unit postoperatively. Postoperative liver function tests were abnormal, and post-cardiopulmonary bypass transesophageal echocardiography revealed mildly decreased right ventricular function. While in the intensive care unit, the patient was placed on venoarterial extracorporeal membrane oxygenation and underwent therapeutic plasma exchange. Vasopressors were weaned over the course of the next 24 h. The patient was able to be transferred out of the intensive care unit on postoperative day 5. Traditional vasoconstrictors activate signal transduction pathways that lead to myosin phosphorylation. Vasodilatory molecules such as nitric oxide (NO) activate the enzyme soluble guanylyl cyclase (sGC), ultimately leading to the dephosphorylation of myosin. Nitric Oxide Synthase (NOS) can potentially increase NO levels 1000-fold when activated by inflammatory cytokines. Methylene blue is a direct inhibitor of NOS. It also binds and inhibits sGC. Hydroxocobalamin is a direct inhibitor of NO, likely inhibits NOS and may also act through additional mechanisms.
Introduction
Recognized as a possible complication of cardiopulmonary bypass (CPB), vasoplegic syndrome (VS) is a critical state of unregulated systemic vasodilation and decreased vascular resistance with normal or increased cardiac output, and a pathological insensitivity to conventional catecholamine vasoconstrictors. 1 VS is associated with longer hospitalization, longer intensive care unit (ICU) duration, more blood transfusions, and mortality rates as high as 25% in patients with catecholamine-resistant VS lasting longer than 36 h.2,3 Due to their mechanistic differences from standard inotropes and vasoconstrictors, methylene blue and hydroxocobalamin have been previously described as rescue medications to treat refractory VS. 4 In an effort to describe the use of these agents as part of our armamentarium in refractory VS in the context of cardiac surgery, we present a case displaying the use of intraoperative methylene blue and postoperative high-dose hydroxocobalamin, followed by therapeutic plasma exchange (TPE) and venoarterial extracorporeal membrane oxygenation (VA-ECMO), after onset of VS symptoms during a mitral valve repair in a patient with infective endocarditis in the setting of intravenous drug abuse.
Case description
A 24-year-old female with a past medical history of intravenous drug abuse and hepatitis C infection underwent mitral valve repair due to infective endocarditis. Preoperative transesophageal echocardiography (TEE) showed a left ventricular ejection fraction (EF) of 65%–75%, normal right ventricular systolic function, and severe mitral regurgitation with vegetation (Figure 1). Preoperative laboratory testing was significant for hypoalbuminemia of 1.7 g/deciliter (g/dL), alkaline phosphatase of 320 international units/liter (U/L), and a platelet count of 122 × 103 µL−1; liver, renal, and coagulation studies were otherwise normal. While the patient was on CPB, an increase in dosage of norepinephrine was required, with the addition of epinephrine and vasopressin infusions and boluses, to maintain a mean arterial pressure above 60 millimeters of mercury. Decreasing blood glucose levels requiring dextrose administration raised concerns for liver dysfunction. Postoperative laboratory and liver function tests were abnormal with aspartate transaminase (AST) of 452 U/L, alanine transaminase (ALT) of 181 U/L, albumin of 1.1 g/dL, and decreased total protein of 3.4 g/dL. Postoperative INR was increased at 1.65 and platelet count decreased to 76 × 103 µL−1 over the course of the next 48 h. Postoperative creatinine level was 1.04 mg/dL. Post-bypass TEE revealed a left ventricular EF consistent with pre-CPB findings, with mildly reduced right ventricular function and no tricuspid regurgitation. Florid pulmonary edema required multiple ventilator circuit changes throughout the operation.
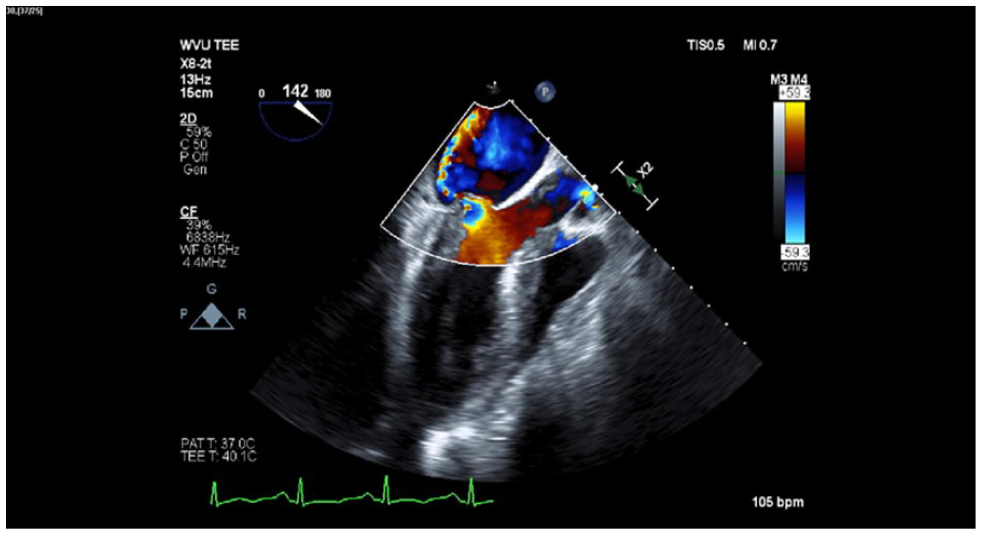
Midesophageal long-axis view with color-flow Doppler demonstrating anterior mitral leaflet flail, a posteriorly directed mitral regurgitant jet exhibiting the Coanda effect, and a subvalvular vegetation in the mitral chordal apparatus.
In addition to the vasopressors and inotropes, the patient received an approximately 1.5 mg/kg bolus of intravenous (IV) methylene blue intraoperatively, and a 5 g dose of IV hydroxocobalamin administered as an infusion over 15 min in the ICU postoperatively. While in the ICU, the patient was placed on VA-ECMO and underwent TPE. Vasopressors were weaned over the course of the next 24 h. After 2 days of VA-ECMO, and 3 days of veno-venous ECMO, the patient was decannulated and extubated, allowing her to be transferred to the ward for recovery.
Discussion
The mechanism of vasoconstriction ultimately relies on the phosphorylation of the myosin molecule in vascular smooth muscle cells, leading to contraction and constriction of the blood vessel. Vasoconstrictors such as angiotensin II and norepinephrine activate signal transduction pathways, leading to an increase in intracellular calcium. 5 This calcium forms a complex with calmodulin, which leads to myosin phosphorylation. 4 Vasodilatory molecules such as atrial natriuretic peptide (ANP) and nitric oxide (NO) trigger an increase in cyclic guanylate monophosphate (cGMP) production by the enzyme soluble guanylyl cyclase (sGC), ultimately leading to activation of myosin phosphatase, and the dephosphorylation of myosin 5 (Figure 2).
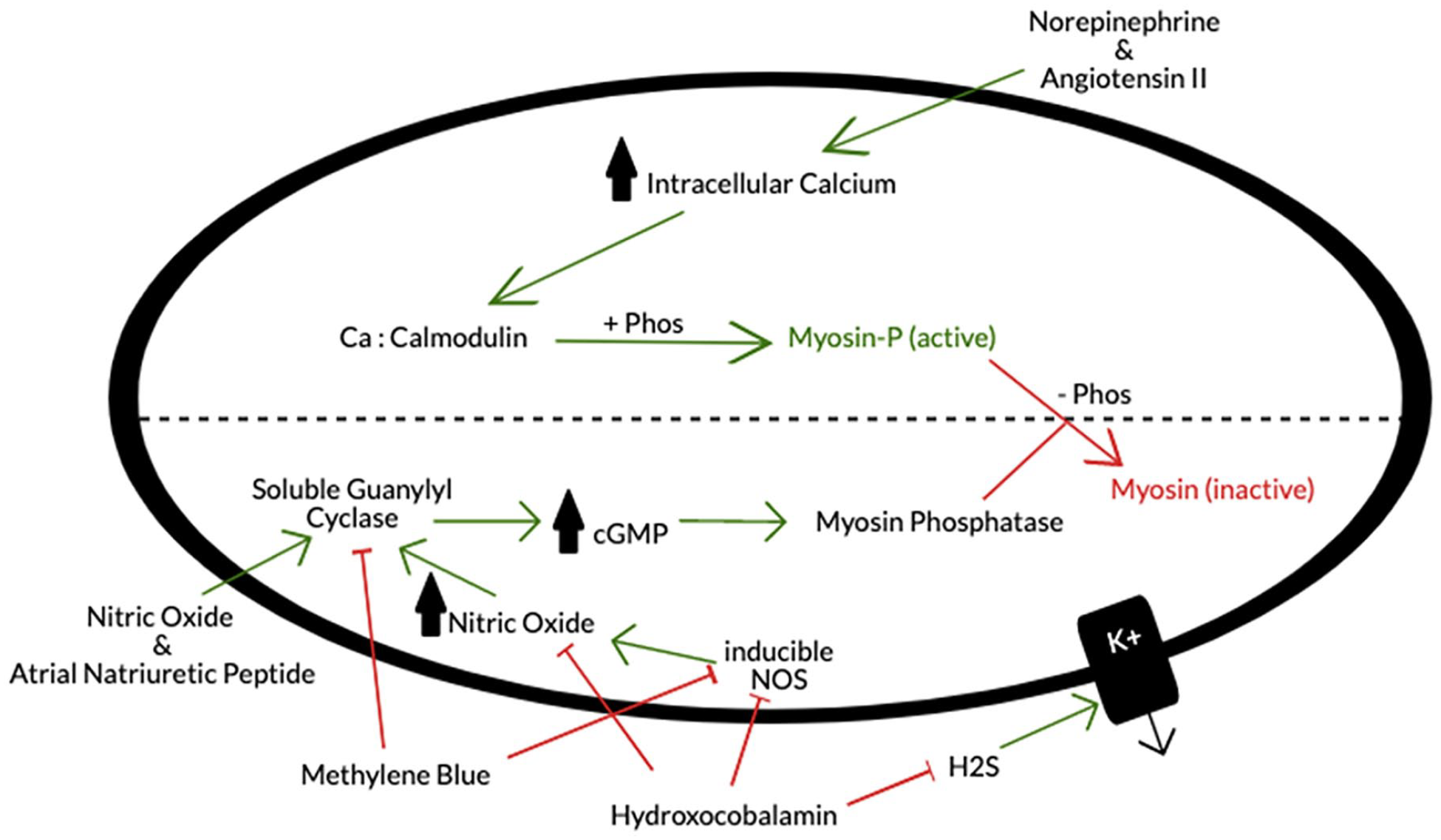
Depiction of effects of norepinephrine, angiotensin II, methylene blue, hydroxocobalamin, atrial natriuretic peptide, and nitric oxide on activation and deactivation of myosin molecules in vascular smooth muscle cells.
NO is produced physiologically by two forms of the enzyme nitric oxide synthase (NOS): constitutive (cNOS) and inducible (iNOS). Inflammatory cytokines, such as IL-1, IL-6, and TNF-α, activate iNOS and lead to an increase in NO levels by up to a factor of 1000. While cNOS is controlled by a negative feedback mechanism, iNOS is not. Rather, iNOS has the ability to irreversibly bind calmodulin and block the formation of the kinase complex that phosphorylates myosin, exacerbating its vasodilatory effects.4,6 Catecholamine-refractory VS in certain post-cardiotomy patients is largely thought to be the result of an increase in expression of iNOS in the presence of inflammatory cytokines, produced as a response to the insult of CPB. 4
Methylene blue is a direct inhibitor of the NOS enzyme. It also binds with inhibitory activity to sGC, leading to a decrease in overall cGMP and NO production in vascular smooth muscle cells and, therefore, an increase in vascular tone.4,6 This is important because other inflammatory mediators can activate sGC. 4 Methylene blue thus counteracts NO and cGMP pathways allowing myosin phosphorylation and systemic vasoconstriction to occur when traditional cAMP-mediated vasoconstrictors are ineffective (Figure 2). Levin et al. 7 showed in a randomized trial that use of 1.5 mg/kg of intravenous methylene blue significantly reduced mortality rates and led to faster recovery in cardiac surgery patients meeting criteria for VS. Currently published randomized trials only have sample populations with less than 60 patients. For this reason, Hosseinian et al. 4 concluded in a 2016 review of current literature on the topic that while methylene blue clearly demonstrates that it can increase blood pressure, it should not be used as a first-line agent and should be reserved for use in unresponsive vasoplegia until further studies have been conducted.
The administration of high-dose hydroxocobalamin in VS acts through multiple proposed mechanisms. The hydroxocobalamin molecule is a direct inhibitor of NO and also likely binds directly to NOS. 8 An alternate mechanism proposed for the increase in vasodilation associated with VS is through the indirect inhibition of potassium channels. Potassium efflux leads to membrane hyperpolarization, which closes voltage-gated calcium channels. Hydrogen sulfide chemically modifies potassium channels, producing this vasodilatory effect. It has been proposed that hydroxocobalamin decreases membrane hyperpolarization by blocking the action of hydrogen sulfide. Therefore, inhibition of hydrogen sulfide by hydroxocobalamin prompts vasoconstriction (Figure 2). 9 The dosage of hydroxocobalamin for its conventional use as a cyanide antidote is a 5 g infusion over 15 min, with the option to increase to 10 g with severe poisoning. Shapeton et al., 8 therefore, proposed that this dosage should be utilized as a rescue therapy for refractory VS until further studies have been done to determine a minimum effective dose.
TPE is a procedure in which blood is separated into its plasma and cellular components. The plasma is typically replaced by a mixture of colloid and crystalloid fluids and returned to the body with the cellular components. In addition, a procedure deemed double filtration plasmapheresis involves the separation of plasma and cells with an additional step to further filter the plasma, before returning it to the body. TPE has been utilized for the removal of inflammatory mediators, such as pro-inflammatory cytokines, with some success in various pathologies, including antibody-mediated disorders and various complications associated with cardiac surgery. 10 A 2014 literature review by Rimmer et al. ruled that sufficient evidence does not exist, in the form of a randomized controlled trial, to recommend TPE for sepsis or septic shock, but a 2018 trial did not see any adverse effects in 23 patients treated with TPE for inflammatory cytokine reduction in septic shock.11,12 Since there is evidence suggesting certain inflammatory cytokines and proteins contribute to the refractory vasodilation of VS, the use of TPE in VS may be reasonable despite a lack of controlled trials supporting its efficacy.5,10
Angiotensin II administration is a potential alternate therapy that was not utilized in this case but could have been considered if the patient continued to deteriorate clinically. Angiotensin II is a hormone that occurs naturally in the body as a part of the renin-angiotensin-aldosterone system. It is a potent vasoconstrictor of both arteries and veins. It also causes many indirect effects leading to vasoconstriction, including increased vasopressin, increased adrenocorticotropic hormone, increased aldosterone release from the adrenal cortex and may potentiate sympathetic effects by direct action on post-ganglionic sympathetic neurons. Chawla et al. 13 demonstrated a significant and substantial decrease in the use of norepinephrine in treatment of high-output shock with concomitant angiotensin II administration as a continuous infusion at a dose of 20 ng/kg/min. In addition, in a 2017 follow-up trial, Khanna et al. 14 demonstrated an effective increase in blood pressure and reduction in catecholamine dosage with the use of angiotensin II in cases of catecholamine-refractory vasodilatory shock. This trial prompted United States Food and Drug Administration approval for intravenous angiotensin II in patients with distributive shock who do not respond to conventional therapies. 15
Conclusion
This case is an addition to the growing body of literature describing the use of methylene blue and hydroxocobalamin, in addition to TPE, as potential therapies for catecholamine-refractory VS. We present this case in the context of VS associated with cardiac surgery requiring ECMO as a bridge to recovery. In this case, a critically ill patient with sequelae of intravenous drug abuse-related endocarditis made a complete recovery after mitral valve repair performed in the setting of refractory VS. More trials need to be conducted to truly elucidate the efficacy and safety of each intervention, as well as proper dosing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
