Abstract
The aim of this report was to examine the safety of physical therapy for hemiplegia in a patient with neuromyelitis optica in the acute phase. A 32-year-old female with neuromyelitis optica diagnosed by magnetic resonance imaging had a T2-weighted hyperintense lesion in the central cord from C2 to T2 and was positive for serum anti-aquaporin-4 antibodies. Upon admission to the hospital, her Expanded Disability Status Scale, Medical Research Council scale for muscle strength score, and sensory score (according to the American Spinal Injury Association sensory assessment) were 9.0, 37, and 158, respectively. The patient commenced physical therapy on day 4. A locomotion exercise using an orthosis was started as soon as possible. Her left-side limb muscle strength improved to approximately grade 4 by a manual muscle test with improvements in the Expanded Disability Status Scale score (from 9.0 to 6.0), Medical Research Council scale for muscle strength score (from 37 to 54), sensory score (from 152 to 203), and all functional independence measure dominants compared with the respective values on admission. This report shows that a carefully supervised physical therapy program did not cause deterioration of the patient’s symptoms.
Introduction
Neuromyelitis optica (NMO) is an inflammatory disorders characterized by repeated exacerbation and remission of severe optic neuritis and transverse myelitis. 1 Transverse myelitis often leads to a flaccid paralysis along the area of inflammation. Physical therapy is one treatment for NMO. However, there are only few reports regarding physical therapy for NMO,2,3 and no published studies have focused on the safety of physical therapy during the acute phase. This article reports an instance of acute phase physical therapy in a patient with NMO.
Case
The patient was a 32-year-old female with NMO diagnosed by magnetic resonance imaging (MRI) who had a T2-weighted hyperintense lesion in the central cord from C2 to T2 (Figure 1) and was positive for serum anti-aquaporin-4 antibodies. Written informed consent was obtained from the patient for her anonymized data to be published in this article. The patient visited our hospital because of left hemiplegia and limb sensory disorder. Upon admission to the hospital, her Expanded Disability Status Scale (EDSS) score, 4 Medical Research Council (MRC) score for muscle strength score, 5 and sensory score (according with American Spinal Injury Association sensory assessment) 6 were 9.0, 37, and 158, respectively. She had paralysis and decrease muscle tone in the left side of her body (mainly in the lower limb, L1−S5 region), but no respiratory and pain problem. The clinical chart of the medical treatment, rehabilitation program, and clinical improvement is shown in Figure 2. The patient underwent treatment by steroid pulse therapy (methylprednisolone 1000 mg/day, days 2–4, and days 6–8). In addition, plasma exchange (plasma volume = 60 mL/kg/day, days 10, 13, 16, 18, and 20) was performed five sessions and intravenous immunoglobulin (dosage = 0.4 g/kg/day, days 24, 25, 26, 27, and 28) was administered five times (Figure 2(a)). Prednisolone was taken orally 20 mg/day as post-therapy. The patient commenced physical therapy on day 4. During the first physical therapy evaluation, her general status was lucid and her left-side limb muscle strength (i.e. paralysis side) was approximately grade 1–2 by a manual muscle test (MMT). She started a standing exercise and muscle strength training immediately. Physical therapy was performed 5 days/week for 60 min. The muscle strength training started with isotonic movements (shoulder flexion, hip flexion, and knee extension). The paralyzed muscles received electrical muscle stimulation (EMS) using an EMS device (AUTO Tens PRO Rehabili Unit; Homer Ion Co. Ltd., Tokyo, Japan). We performed EMS at 20 Hz with a monophasic square-wave pulse with a duration of 250 µs, an on time of 5 s, an off time of 2 s, no rise time, and decay using the EMS device. Belt electrodes were used for skeletal muscle stimulation. The electrodes were wrapped around the waist, distal parts of the thigh, and the crus of the paralyzed side (Figure 3). Each exercise was performed without exceeding 17 on the 15-point Borg scale. Furthermore, she was carried out gait practice step by step as the spasticity improved. She performed a locomotion exercise using a walker and knee-ankle-foot orthosis from day 14. She also started locomotion using an elbow crutch and foot orthosis without assistance from day 24. She was subsequently transferred to a rehabilitation hospital on day 34 because her overall condition had improved and she had completed treatment in the acute phase. Her left-side limb muscle strength improved to approximately grade 4 by the MMT, with improvements in the EDSS score (from 9.0 to 6.0), MRC muscle strength score (from 37 to 54), sensory score (from 152 to 203), and all functional independence measure dominants compared with the respective values on admission (Tables 1–3). She did not experience worsening of the symptoms (e.g. pain and spasticity) during hospitalization.

Spinal cord MRI in the acute phase. The sagittal plane (a) and axial plane (b) of T2-weight hyperintense lesion in the central cord.

The clinical chart in a patient with NMO. (a) Medical treatment, (b) rehabilitation program, and (c) clinical improvement.

Belt electrodes were wrapped around the waist, distal parts of the thigh, and the crus of the paralyzed side. The waist electrode had a width of 5.5 cm and a length of 40 cm twice (a), the thigh electrode had a width 5.5 cm and a length 54 cm (b), and the crus electrode had a width 5.5 cm and a length 31 cm (c).
The functional independence measurement score.
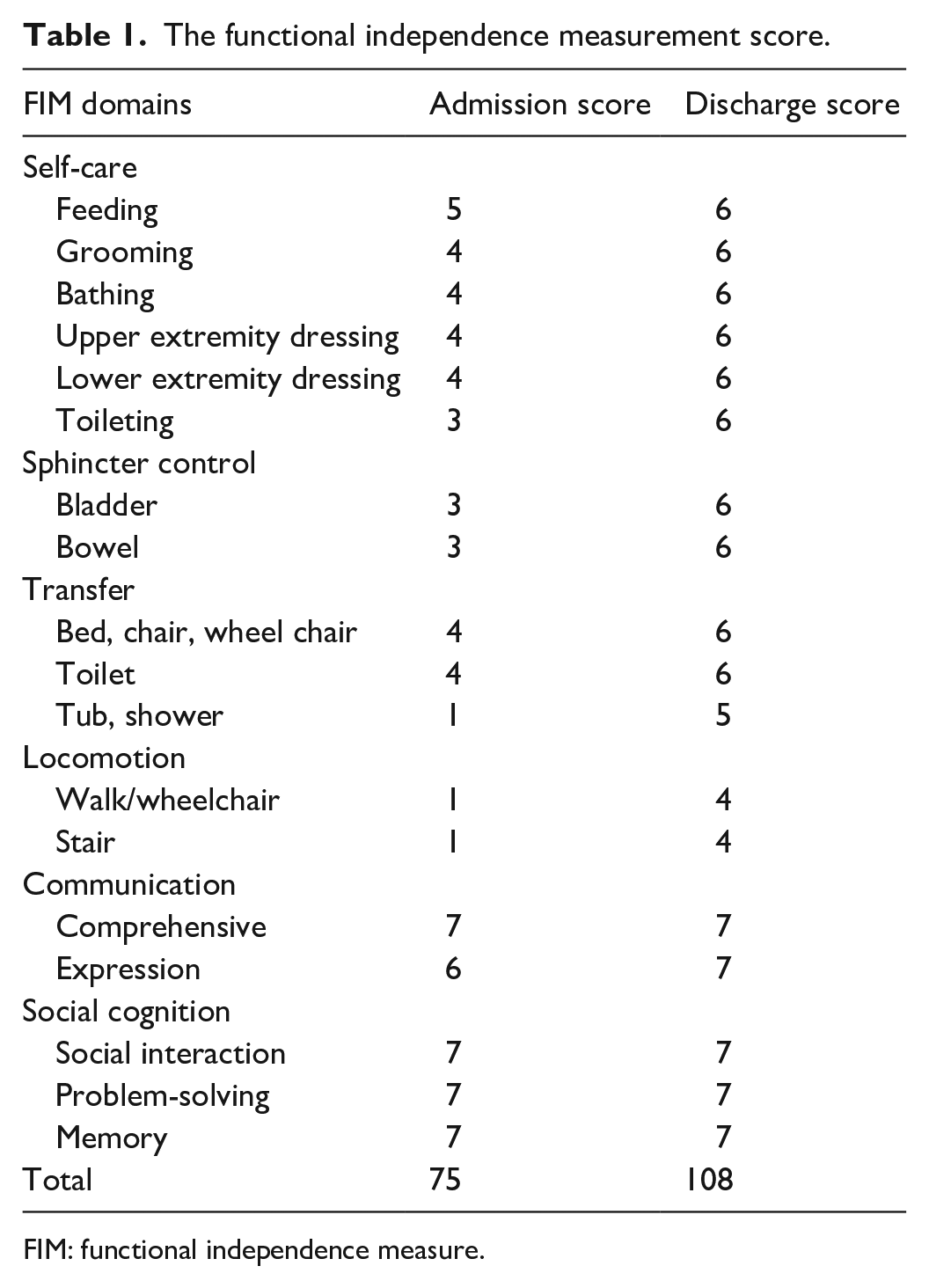
FIM: functional independence measure.
Medical Research Council scale for muscle strength score.

Sensory assessment.
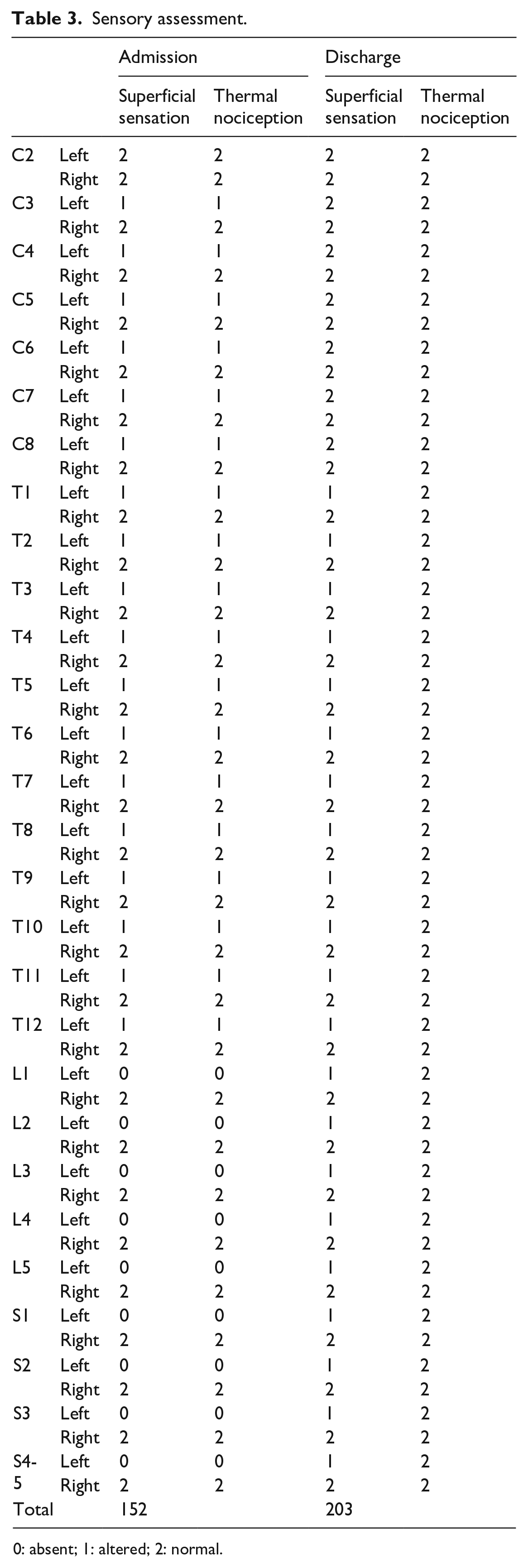
0: absent; 1: altered; 2: normal.
Discussion
In this study, we performed intensive rehabilitation from the inflammatory phase in a patient with NMO. To our knowledge, there are no previous reports on the safety of physical therapy in patients with NMO during the acute phase. However, it has been considered that the performance of exercise during the acute phase of the disease can lead to deterioration of the patient’s symptoms. Nevertheless, intensive physical therapy has recently been shown to have positive effects in patients with inflammatory myopathies.7,8 This study showed that a physical therapy program did not exacerbate symptoms in a patient with NMO when performed cautiously based on the patient’s degree of subjective fatigue and muscle pain. Therefore, we consider that a carefully supervised physical therapy program can be applied safely and effectively in patients with NMO, as well as in patients with inflammatory myopathies7,8 and neuromuscular disorders. 9
In most cases of NMO, treatment in the acute phase does not involve physical therapy until medical treatment has been completed. In this situation, many patients with NMO must stay in bed for 1–2 weeks. Previous studies have revealed that the bed rest leads to muscle disuse atrophy.10,11 It has further been reported that muscle atrophy can even occur in healthy people, and may be apparent after only 5 to 10 days.10–12 In recent studies, early physical therapy with medication resulted in effective physical outcomes in patients with several diseases, as long as the therapy was performed cautiously and was supervised by a medical doctor and/or physical therapist.7,13 Therefore, starting physical therapy during the acute phase may also be important in patients with NMO. However, there are no reports on physical therapy in patients with NMO in the acute phase. In future research, it is necessary to increase the number of patients and consider the safety of physical therapy in the acute phase.
Conclusion
We described a physical therapy program in a case of hemiplegia and sensory disorder caused by NMO. This study showed that a carefully supervised physical therapy program did not cause deterioration of the patient’s symptoms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from the Hiroshima University of Ethics Committee (approval no./ID: E-1171-1, Pg. 6, lines 106–108).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article (Pg. 7, lines 110–112).
