Abstract
Chronic eosinophilic leukemia, not otherwise specified can be challenging to differentiate from hypereosinophilic syndrome and myelodysplastic syndromes with elevated eosinophilia. We present a diagnostically challenging case of chronic eosinophilic leukemia, not otherwise specified that initially seemed like a myelodysplastic syndrome but progressed to eosinophilic tissue infiltration and overt eosinophilic dyspoiesis. In addition, we discuss the morphologic and molecular findings that can overlap among these entities that made the diagnosis difficult in the case presented.
Keywords
Introduction
Chronic eosinophilic leukemia, not otherwise specified (CEL, NOS) is a rare myeloproliferative neoplasm that can be difficult to diagnose even for the experienced hematopathologist. The diagnostic criteria for CEL, NOS requires a circulating eosinophil count of greater than or equal to 1.5k/µL without rearrangements of PDGFRA, PDGFRB, FGFR1, or JAK2, exclusion of other myeloproliferative neoplasms (MPN) or core binding factor leukemias, less than 20% blasts in the peripheral blood and bone marrow, and a clonal cytogenetic or molecular genetic abnormality must be present or ⩾2% blasts in the peripheral blood or ⩽5% blasts in the bone marrow. 1 It is important to note that those with PDGFRA, PDGFRB, and FGFR1 rearrangements have been re-classified under myeloid neoplasms with these specific rearrangements as there are targeted therapies for these entities. 2 Those within the CEL, NOS category can be difficult to diagnose because there is significant overlap in the morphological features of CEL, NOS; idiopathic hypereosinophilic syndrome (HES); and myelodysplastic syndrome (MDS) with eosinophilia. Recent studies of CEL and HES have shown that these patients can show mutations in one or more genes that are commonly identified in MPN or MDS, further blurring the distinction between these entities and increasing the difficulty in diagnosis.3,4 Herein, we describe a diagnostically challenging case of an elderly female presenting with eosinophilia.
Case presentation
An 80-year-old Hispanic female with a past medical history of diabetes mellitus, hypertension, and hyperlipidemia was referred to the hematology-oncology clinic for evaluation of anemia, leukocytosis, and eosinophilia. Her original complete blood count (CBC) showed a white blood cell (WBC) count of 15k/µL with 4.8k/µL absolute eosinophil count (AEC), hemoglobin (Hgb) of 11.3 g/dL, hematocrit (Hct) of 35.1%, mean corpuscular volume (MCV) of 95.5 FL, red blood cells (RBCs) of 3.68 million/µL, and platelets of 301k/µL. During her examination in the oncology clinic, she denied weight loss, night sweats, and fevers and had no physical exam signs of hepatosplenomegaly or rashes.
A bone marrow biopsy was performed. At the time of bone marrow biopsy, her blood cell counts showed a Hgb of 11.1 g/dL, 222k/µL, a WBC count of 13.6k/µL with a manual differential of 42% eosinophils (AEC of 6.3k/µL; image of peripheral blood eosinophils in Figure 1(a)). Bone marrow biopsy showed a hypercellular marrow for age (60% cellularity) with trilineage hematopoiesis and eosinophilia (19%) (Figure 1(b)). Iron stain on the aspirate smear showed increased ringed sideroblasts (Figure 1(c)). No evidence of reticulin fibrosis was identified by silver stain. Mild dysmegakaryopoeisis was identified. Mild megaloblastoid changes in some erythroid precursors were present.

(a) Peripheral blood smear at 50× magnification showing multiple peripheral eosinophils showing relatively normal morphology. (b) Hypercellular bone marrow core biopsy H&E at 20× magnification showing elevated eosinophil count and slight dysmegakaryopoiesis seen. (c) Iron stain at 100× magnification showing ringed sideroblasts present. (d) Colon biopsy H&E at 20× magnification showing tissue infiltration of eosinophils and eosinophilic cryptitis. (e) Giemsa stain of bone marrow aspirate 2 years after initial bone marrow biopsy showing increased eosinophils. (f) Bone marrow core needle biopsy H&E at 10× magnification showing increased marrow cellularity from previous biopsy, with increased megakaryocytes and eosinophils.
Molecular studies were performed using a targeted myeloid panel by next generation sequencing (NGS) technology analyzing gene mutations including ASXL1, BCOR, BRAF, CALR, CBL, CEBPA, CSF3R, DDX41, DNMT3A, ETNK1, ETV6, EZH2, GATA2, GNAS, GNB1, IDH1, IDH2, JAK2, KIT, KRAS, MPL, NF1, NPM1, NRAS, PDGFRA, PHF6, PPM1D, PTPN11, RAD21, RUNX1, SETBP1, SF3B1, SH2B3, SMC1A, SMC3, SRSF2, STAG2, STAT3, STAT5B, TET2, TP53, U2AF1, WT1, and ZRSR2. Results of sequencing revealed an ASXL1 nonsense mutation, a SRSF2 missense mutation, a TET2 frameshift mutation, and a TP53 missense mutation (see Table 1). Cytogenetic analysis performed showed a normal female karyotype [46,XX]. Fluorescent in situ hybridization (FISH) was performed for PDGFRA/B and FGFR1 rearrangements and were negative. In addition to the sequencing of JAK2 in the NGS panel, polymerase chain reaction (PCR) for JAK2 (V617F) was also performed and was negative for the mutation. At this time, the differential diagnosis included CEL, NOS, and MDS with increased ringed sideroblasts associated with eosinophilia. The patient was closely followed—without medical treatment.
Pathogenic variants detected in our case.

The patient presented with diarrhea 9 months after initial work-up. The diarrhea occured approximately two times a week containing loose stools and typically followed a meal. Stool studies for infectious etiologies were performed and were negative. Colonoscopy with random site biopsy showed microscopic colitis with lymphocytic and eosinophilic infiltration and eosinophilic cryptitis (see Figure 1(d)). The diarrhea symptoms resolved after 3 weeks of oral 9 mg budesonide treatment. No additional chemotherapy treatment was given at this time for the patient’s eosinophilia.
Two years after initial presentation, the patient’s WBC count increased as well as the AEC. A second bone marrow biopsy was performed to rule out disease progression and showed markedly hypercellular marrow for age (95%) with marked eosinophilia (Figure 1(e) and (f)). Given the patient’s clinical course, colonic tissue infiltration by eosinophils, bone marrow morphologic findings and gene mutations identified by NGS, a diagnosis of chronic eosinophilic leukemia, NOS was made. A cardiac magnetic resonance imaging (MRI) was performed due to a new second-degree heart block to rule out eosinophilic infiltration. The cardiac MRI was negative for eosinophilic infiltration and the heart block was ruled idiopathic as her ventricle sizes were normal with an ejection fraction of 63%. No further work-up was performed for the new onset of heart block. The patient continued to be closely monitored without interferon, hydroxyurea, or any chemotherapy treatment.
A third bone marrow biopsy was performed 39 months after initial presentation. The peripheral smear associated with this bone marrow showed 2% circulating blasts with a leukocytosis consisting of eosinophilia (11%, absolute count 4.7k/µL), neutrophilia (absolute count 24.8k/µL), and monocytosis (absolute count 3.9k/µL). Macrocytic anemia (Hgb 9.5 g/dL and MCV 106.4 fL H) and mild thrombocytopenia was also present (143k/µL). Bone marrow aspirate smears show a blast count of 6% with increased eosinophils (15%). There was decreased storage iron present with 40% ringed sideroblasts. Trilineage dyspoeisis was present. Cytogenetics of the most recent bone marrow showed Trisomy 8, 13, and 14, suggesting disease progression (please refer Table 2 for information about disease course). The patient has not taken any chemotherapy agents during her follow-up care for her CEL, NOS to date.
Laboratory studies over the course of this patient’s treatment.
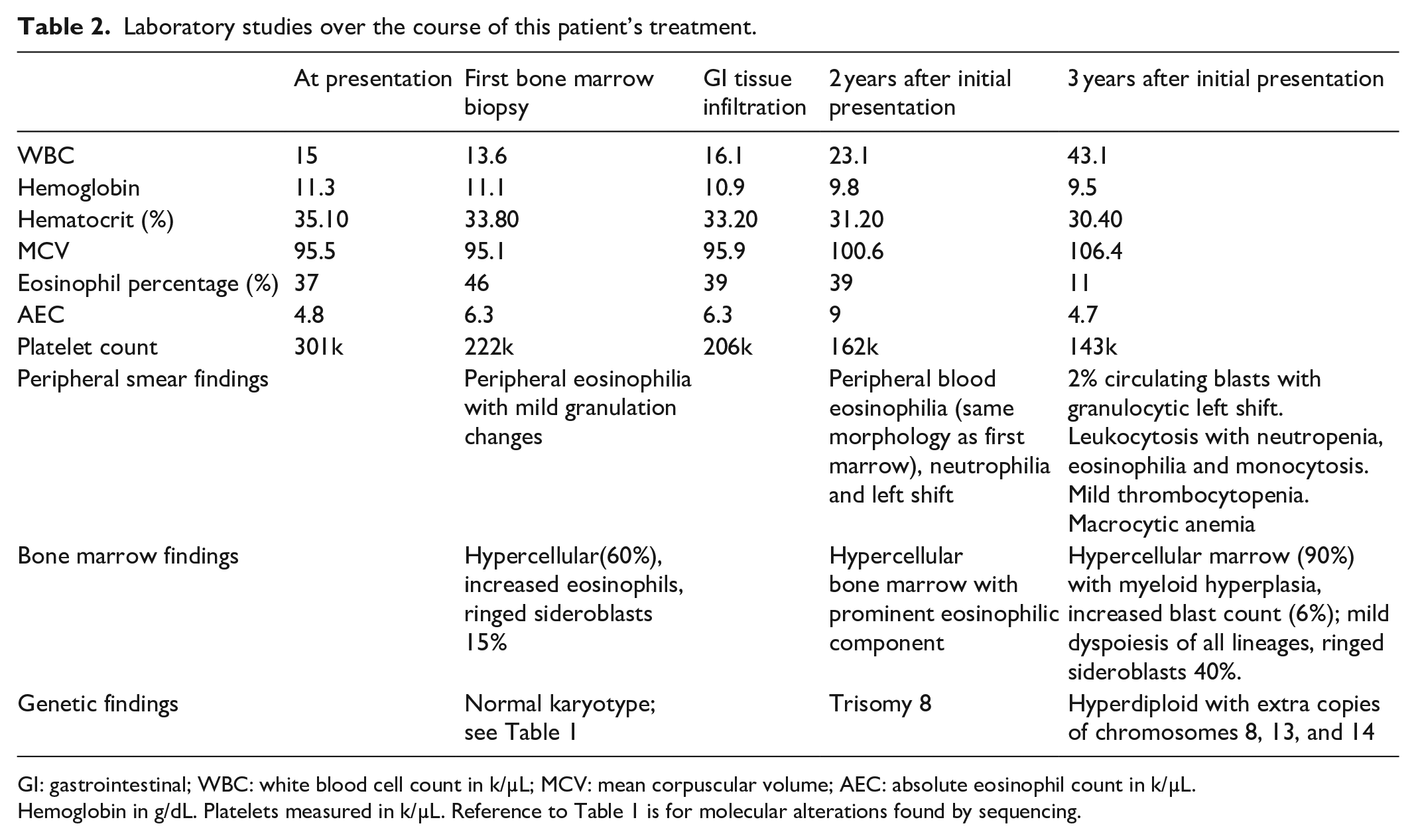
GI: gastrointestinal; WBC: white blood cell count in k/µL; MCV: mean corpuscular volume; AEC: absolute eosinophil count in k/µL.
Hemoglobin in g/dL. Platelets measured in k/µL. Reference to Table 1 is for molecular alterations found by sequencing.
Discussion
This is a case of an elderly female who initially presented with mild anemia, eosinophilia and was found on bone marrow biopsy to have eosinophilia, mild dysmegakaryopoiesis, mild dyserythropoiesis and increased ring sideroblasts. After the initial work-up with these bone marrow findings, negative FISH, normal karyotype, and multiple mutations detected by NGS, MDS with eosinophilia was a considered diagnosis in this case prior to the end organ involvement by the colon and the progressive eosinophilia. This is an important differential as patients with MDS-associated eosinophilia have a poorer prognosis than matched MDS patients without eosinophilia.4,5 This differential can be difficult as many cases of CEL have been recently described with various levels of dyspoiesis involving different cell lineages including dysplastic megakaryocytes of MDS or MPN morphology, dyserythropoiesis, and dysgranulopoeisis. 6 Distinguishing between MDS with eosinophilia and CEL, NOS can be difficult as in this patient presentation with mild anemia with ring sideroblasts, megakaryocyte dysplasia, and an eosinophilia. Given the patient’s clinical course of progressive eosinophilia in peripheral blood and bone marrow along with colonic tissue infiltration by eosinophils, a diagnosis of CEL, NOS was made.
In more classic presentations of CEL, NOS, the main differential diagnosis is idiopathic hyereosinophilic syndrome which can also have end organ involvement and eosinophilia. 1 The difference in these diagnoses is the blast cells present in the peripheral blood (>2%) or bone marrow (>5%) or molecularly defined clonality in CEL, NOS. 1 Clonality is classically described as karyotype abnormalities or MLL, CLER, or JAK2 mutations, but with the widespread use of larger NGS panels, this definition is ever expanding. 1 In a study by Wang et al., most HES cases that showed mutations had one or two alterations with the most common genes involved being ASXL1 and TET2. These cases did not show TP53 mutations and none had more than three mutations identified. The CEL, NOS cases commonly had multiple mutations, even without karyotype alterations. 3
An important point to make in our case is the mutations seen (most notably ASXL1, TET2, and TP53) have been identified by NGS in elderly patients with no evidence of myeloid neoplasms, referred to as age-related clonal hematopoiesis (ARCH).7,8 Most commonly, these alterations are a point mutation consisting of cytosine-to-thymine transition. Allele frequency can be helpful in determining if this is a somatic mutation of a clonal population causing malignancy or a potential pre-malignant clone. It is important to also mention that these patients, although not having overt malignancy at the time of variant detection, can progress to overt malignancy or may have higher risk for factors contributing to morbidity and mortality. 8 In one study of 10 patients, half progressed to acute transformation within 20 months and all but two patients died from disease-related causes during the follow-up period. 9 Median survival in this cohort was 22 months. It has been shown that mutations associated with aging have a low allele frequency. We determined the mutations found in our case to be an indication of a myeloid neoplasm rather than ARCH because of a higher variant allele frequency, multiple genetic abnormalities, and morphologically significant dysplasia as evidenced by ring sideroblasts, megakaryocyte lineage changes in the context of eosinophilia and organ infiltration.
Unique to our case is the presentation of eosinophilia with ring sideroblasts on bone marrow biopsy. Within our literature search to date, we did not find mention of ring sideroblasts as a feature of CEL, NOS despite multiple articles identifying dyserythropoeisis.3,6 Ring sideroblasts have been associated with mutations in splicing proteins, most notably SF3B1 which is the most common mutation seen in MDS with ringed sideroblasts (MDS-RS). 10 Although other mutations can be seen in other splicing genes in MDS-RS, all cases lacking the SRSF2 mutation have less favorable outcome. 11 SRSF2 is one of the proteins involved in recognizing the 3ʹ splice site within the mRNA, although it is not known whether the mutation of this gene resulted in increased ring sideroblasts in this patient. 12 These alterations in mRNA splicing lead to abnormal deposition of iron in mitochondria of early erythrocyte precursors in a pathway that has yet to be determined.
Conclusion
Our patient has been followed by our clinic for 3 years before disease-related symptoms requiring medical intervention surfaced. Previous publications have shown that CEL, NOS is usually associated with a poor prognosis with high risk of acute transformation to acute myeloid leukemia (AML). Although the most recent studies show disease progression, our patient has yet to progress to outright AML. An atypical presentation is presented here as our patient is 39 months from initial diagnosis with evidence of disease progression without any prior treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
