Abstract
Gastroesophageal variceal hemorrhage is a substantial cause of death in patients with portal hypertension. Cyanoacrylate injection is a widely used endoscopic treatment for variceal hemorrhage. We report herein the case of a 49-year-old male with decompensated alcoholic cirrhosis, who received endoscopic sclerotherapy to stop gastroesophageal variceal hemorrhage during hospitalization. The following day, he developed acute progressive dyspnea, and computed tomogram of pulmonary artery revealed acute pulmonary embolism at the right lower pulmonary artery. A final diagnosis of sclerotherapy-associated pulmonary embolism was made, and he gradually improved conservatively without anticoagulant treatment 2 weeks after hospitalization.
Introduction
Sclerotherapy with N-butyl-cyanoacrylate, a watery solution that polymerizes immediately when in contact with blood, is widely used for the treatment of gastric variceal bleeding. Most complications associated with cyanoacrylate injection treatment include transient fever, tissue necrosis at injection site probably leading to perforation, post-sclerotherapy ulcer, portal and splenic vein thrombosis with and without splenic infarction, and most importantly pulmonary embolism. Risk factor includes volumes and speed of injection and the size of gastric varices.1,2 We present herein a case of pulmonary embolism following.
Case report
A 49-year-old male patient with type 2 diabetes mellitus and Child-Pugh class B alcoholic cirrhosis (MELD score 19, PT 18.5, APTT 34.1, INR 1.69) was hospitalized due to primary Streptococcus constellatus bacteremia, and had received penicillin G sodium treatment. Two days after hospitalizaition, he developed massive hematemesis, and emergent endoscopy was performed which revealed large gastroesophageal varices (GOVs) type 2 with blood spurting at the cardia of stomach. The patient then recieved a total of 15.6 mL of cyanoacrylate injection until bleeding was stopped (Figure 1). The following day, he developed acute progressive dyspnea with partial pressure of oxygen (PaO2) from arterial blood gas of 80 mmHg at room air. Acute pulmonary embolism was suspected, chest X-ray (Figure 2) and computed tomogram of pulmonary artery (Figure 3) was emergently carried out and exhibited a concentric intraluminal hyperdense filling defect within a lumen of anterior and lateral basal segmental branches of right lower pulmonary artery. Echocardiogram showed normal pulmonary pressure without cardiac abnormalities. A final diagnosis of sclerotherapy-associated pulmonary embolism was made. He was managed conservatively without anticoagulant treatment due to high risk of bleeding tendency from decompensated cirrhosis, and gradually improved and discharged home 2 weeks after hospitalization.

Endoscopy revealed large gastroesophageal varices (GOVs) type 2 with blood spurting at the cardia of stomach, which was stopped by cyanoacrylate glue injection.
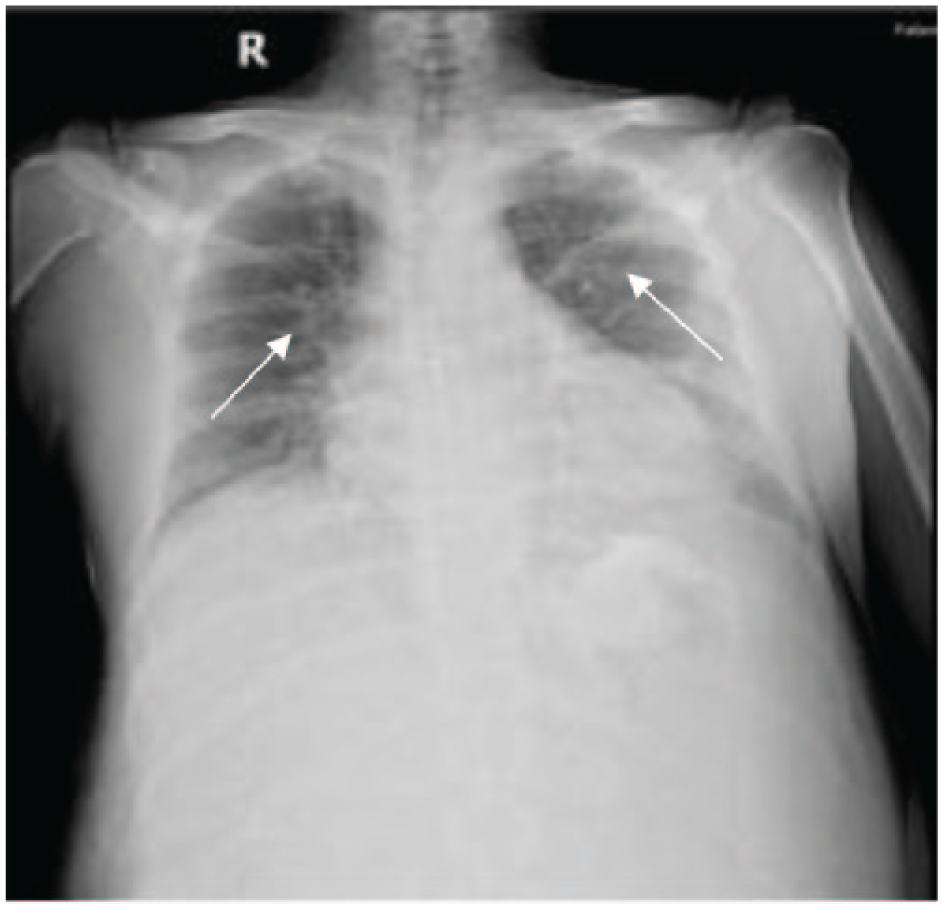
Chest X-ray revealed bilateral patchy opacity suspected pulmonary venous congestion.

CTPA showed concentric intraluminal hyperdense filling defect within anterior and lateral basal segmental branches of right lower pulmonary artery, and suspected sclerotherapy-related pulmonary embolism.
Discussion
Pulmonary embolism is a rare but life-threatening complication of sclerotherapy. In a review article, Saraswat and Verma 3 concluded that the incidence of embolization ranged from 0.5% to 4.3%. Alexander et al. 4 described that the risk of embolism included the volume of glue, injection rate, and the size of gastric varices. Another study showed that the volume of glue was associated with the risk of embolization. 5 Our patient illustrates that the risk of pulmonary embolism is associated with the large volume of cyanoacrylate.
The pathophysiology of pulmonary embolism during endoscopic sclerotherapy is associated with a portosystemic vascular shunt (Figure 4), espectially a gastrosplenorenal shunt which reopens from embryonic vascular pathways. This shunt is a high flow shunt portal circulation to systemic circulation and may carry clots or embolic material from injected varices to the systemic venous flow, right heart, and then pulmonary circulation. 6
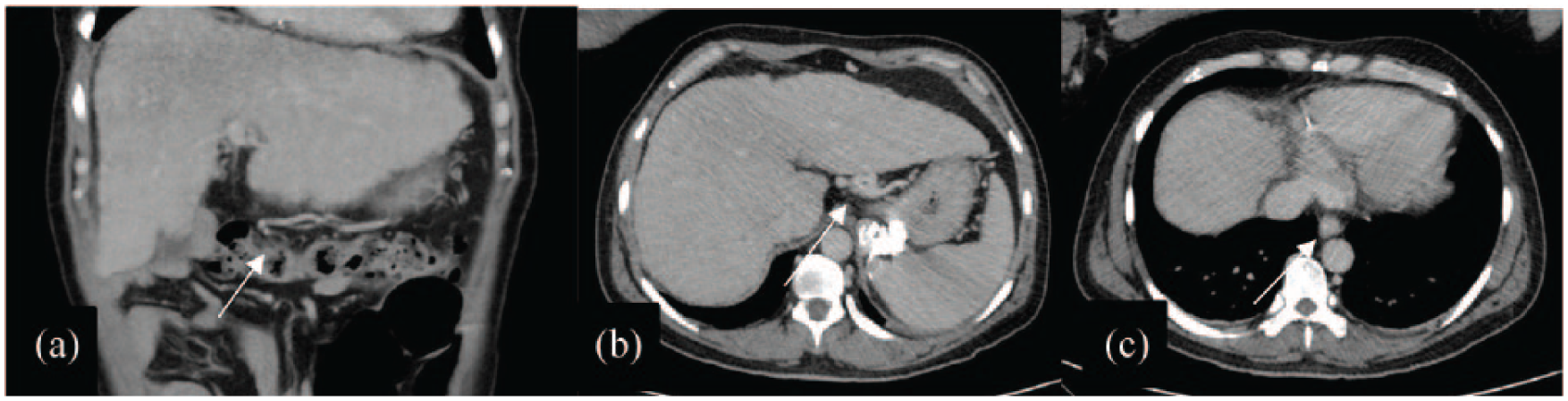
Preoperative contrast-enhanced abdominal CT scan from this patient shows portosystemic shunt. (a) Dilated gastroepipoic vein. (b) Dilated left gastric vein. (c) Esophageal varice.
There were 17 cases of sclerotherapy-associated pulmonary embolism, including our patient (Table 1).5–15 The median age of the patients was 56 years (range 11–77 years, IQR 41 and 60 years), and 58.8% were male. The etiology of cirrhosis includes alcohol (3, 17.6%), hepatitis B (3, 17%), hepatitis C (2, 11.7%), others (3, 17.6%), and unknown cause (6, 35.2%). Cyanoacrylate was used as sclerosing agent in all patients and the mean volume of the injected cyanoacrylate mixed with lipiodol was 5.5 ml (range 1–12 mL). Clinical manifestations of sclerotherapy-related pulmonary embolism is similar to pulmonary embolism, which varies from asymptomatic to severe hypoxia with cardiac arrest. Onset of symptoms also varies from immediate after procedure to late onset up to 10 days. The location of pulmonary embolism is mostly the bilateral pulmonary arteries (9, 53%). Of 17 patients with available information, 14 (82%) received only supportive care and 2 recieved cardiopulmonary resuscitation due to variceal bleeding and massive pulmonary embolism. One patient (6%) died from massive pulmonary embolism.
Previous report of sclerotherapy-related pulmonary embolism.

GV: gastric varix; GOV: gastroesophageal varix; IGV: isolated gastric varix; HBV: hepatitis B virus; HCV: hepatitis C virus; PA: pulmonary artery; CPR: cardiopulmonary resuscitation; UGIB: upper gastrointertinal bleeding.
Conclusion
This case report shows that pulmonary embolism may occur during endoscopic sclerotherapy of gastric varices and is usually related with the volume of cyanoacrylate glue injection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient’s legally authorized representative for their anonymized information to be published in this article.
